Abstract
The effect of liposomes, constituted from soy lecithin and cholesterol, on hydrogen peroxide bleaching of wool fabrics was investigated. A thin lipid layer method was used for liposome production. Use of commercial liposomes was also investigated. Whiteness index (Stensby), breaking strength, solubility in alkali, and several instrumental analysis methods for determining morphology were carried out. Bleached samples were dyed with a 1:2 metal complex dye and color strength values were analyzed. The effect of liposome type on hydrogen peroxide consumption in the bleaching bath was also analyzed using UV-Vis spectroscopy. Elemental analysis was carried out to determine the ratio of elements in the bleached wool samples. Liposomes acted as a stabilizer in the bleaching bath and improved the physical, bleaching, and dyeing properties of wool.
Introduction
Liposomes are closed vesicles with an aqueous interior region. They are spherical phospholipid bilayers that have diameters ranging from 10 nm to 10 μm. Liposomes are not substances, they are carrier systems.1–3 Liposomes consist of two different parts: hydrophobic and hydrophilic. The hydrophilic portion consists of phosphate groups and choline, whereas the hydrophobic part consists two hydrocarbon chains. 4
The ability of liposomes to encapsulate different materials and advances in liposome technology have led to increased use of liposomes for the intake of active agents. The liposo-mal transport system (LDS) has made remarkable progress in the last decade. 5 In recent years, liposomes have become the focus of many applications in the pharmaceutical, cosmetic, food, detergent, and textile industries.
One new application area for liposomes is textile dyeing, primarily in dyeing wool. This clean and efficient technology is used to increase levelness and allows dyeing at a lower temperature to facilitate the dye uptake of various fibers and fiber blends. Liposomes release the encapsulated dye slowly while promoting a retarding effect comparable with retarding agents. This effect makes them a good alternative to commercial levelling products.6–12
Liposomes are also highly effective in aiding the peroxide bleaching of fabrics made of cellulosic fibers and its blends with polyester or wool. Liposomes containing oxidant can act as a reservoir from which the bleaching agent is gradually released during the bleaching process. Sequestration of polyvalent metal ions, thus removing of catalysts of radical hydrogen peroxide decomposition from the reaction medium, could be another mechanism of action. In both situations, liposomes in the bleaching bath can play the role of hydrogen peroxide stabilizers. 13
In recent research, the effectiveness of liposomes in the peroxide bleaching of different textile materials was evaluated. Wetting agents can be eliminated by the addition of liposomes to the bleaching bath. The liposome content in the peroxide bath increased the brightness and breaking load of the samples, thereby improving the quality of the bleached fabric.4,13 Liposome technology was also used to eliminate wetting agent in conventional cotton bleaching baths, resulting in improved mechanical properties, brightness, and softness of cotton fabric. 4
The aim of this research is to examine the effect of lipo-somes in the hydrogen peroxide bleaching of woolen fabrics. Soybean lecithin and cholesterol were used to form the liposome membrane used in the bleaching bath. Liposome production was done according to the thin lipid layer method (Bangham Method) using a rotary evaporator. The effect of commercial liposomes was also investigated. Whiteness index (Stensby), breaking strength, solubility in alkali, elemental analysis, Fourier transform infrared (FTIR) spectroscopy, scanning electron microscopy (SEM), and fluorescence microscopy morphological studies of bleached woolen samples were carried out. Hydrogen peroxide consumption in the bleaching bath was also analyzed. Bleached samples were dyed with 1:2 metal complex dyes and dyeing color strength values were analyzed.
Experimental
Materials
In this research, 100% wool fabric provided by Bahariye Mensucat (Turkey) was used (240 g/m2, warp and weft yarn density of 29 and 25 yarns/cm respectively). Lecithin (phos-photidylcholine), wetting agent (Setawet MNG), stabilizer (Setabicol A-4), ammonia, anti-creasing agent (Setalub ACA), levelling agent (Setalan PM71), pH adjuster (Setacid VSN) and 1:2 metal complex dye (Nyloset Scarlet N-2G, C.I. Acid Red 315) were provided by Setaş Kimya A.Ş. Cholesterol, commercial liposome, ammonia, hydrogen peroxide, and sodium hydrosulfite were purchased from Sigma Aldrich Co. Chloroform and ethanol were supplied by Merck.
Bleaching and Dyeing
Trials were performed according to the previously determined optimal lecithin/cholesterol ratio (Table I). 14 Liposomes were prepared by the Bangham Method. 15 Lecithin and cholesterol were homogenously dissolved in a 2:1 v/v chloroform/ethanol mixture. Afterwards, liposomes were obtained by evaporating the solvent mixture using a rotary evaporator (IKA-Germany) at atmospheric pressure and 30 °C. After solvent evaporation, the liposomes were collected in water.
Liposome Contents
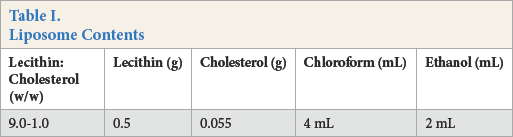
Conventional bleaching was performed using the following recipe: 15 g/L hydrogen peroxide (50%), 1 g/L wetting agent, and 1 g/L stabilizer were used to treat the fabric according to the bleaching diagram (Fig. 1). The bleaching bath pH was adjusted with ammonia. Stabilizing agent was used instead of liposomes in the conventional procedure. At the end of the process, 4 g/L of hydrosulfite was added at the temperature used for bleaching. After bleaching, washing was performed by rinsing at room temperature (RT), rinsing at 50 °C for 10 min, and neutralizing at RT with acetic acid for 10 min. Each experiment was repeated three times.
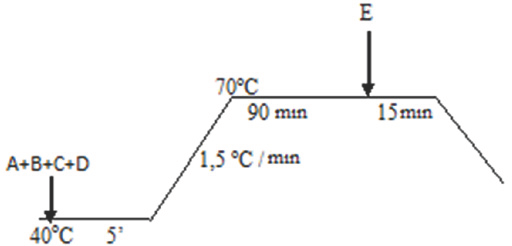
Wool bleaching process. A: Wetting agent, B: stabilizer, C: ammonia, D: hydrogen peroxide, and E: hydrosulfite.
The dyeing procedure is depicted in Fig. 2. The dyeing recipe was 1% owf Acid Red 315, 1 g/L anti-creasing agent, 1 g/L leveling agent, and 2 g/L pH adjuster. The washing procedure after dyeing, recommended by SETAŞ, was rinsing at RT for 10 min, rinsing at 50 °C for 10 min, and rinsing at RT for 10 min. The liquor ratio (LR) was 10:1 for both bleaching and dyeing.
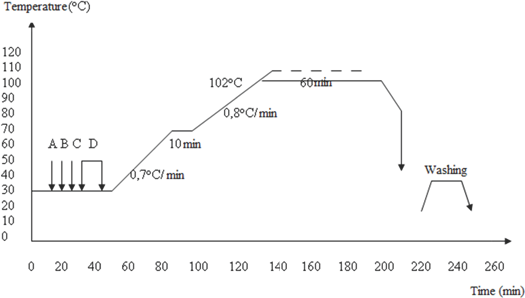
Wool dyeing process. A: Anti-creasing agent, B: levelling agent, C: pH adjuster, and D: C.I. Acid Red 315.
Analysis
Morphological features of samples were analyzed by SEM using a Quanta 250 FEG microscope and a fluorescence microscope Olympus BX43 (BW filter). Before SEM evaluation, the samples were coated with Au-Pd. The samples were dyed with Benzo Purpurin 10 B before fluorescence microscopy. 16
FTIR with attenuated total reflectance (FTIR-ATR) was performed using a Perkin Elmer Spectrum BX FTIR (range from 600–4000 cm−1) at RT.
The whiteness index of bleached samples and color strength values of dyed samples were determined using a Minolta Spectrophotometer CM-3600d (D65, specular inclusion, 10°). All samples were conditioned at 20 °C and 65% relative humidity (RH) for 24 h. The whiteness index was determined according to the Stensby Whiteness index and color strength values were determined the Kubelka-Munk formula (Eq. 1).
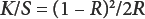
C (carbon), H (hydrogen), N (nitrogen), and S (sulfur) were analyzed using a Vario Elemental C, H, N, S analyzer. The sample (5.0 mg) was placed in a tin capsule for determining the percentage composition of C, H, N and S.
ASTM D1283-05 “Standard Test Method for Alkali-Solubility of Wools” 17 was applied to bleached samples to determine the degree of damage to wool caused by bleaching.
After bleaching, wool moisture absorption values were determined. The treated and untreated wool fabrics were weighed (20 g). Each test fabric was heated in a drying oven at 150 °C for 1 h. After drying, the samples were weighed again. The amount of absorbed moisture was determined by weight difference.
Tensile properties of the fabrics were measured according to TS EN ISO 13934-1 “Textiles- Tensile Properties of Fabrics-Part 1: Determination of maximum force and elongation at maximum force using the strip method” 18 using an Instron 4411 tensile strength tester.
The effect of liposomes on decomposition of hydrogen peroxide in bleaching solution was analyzed by observing the absorbance of hydrogen peroxide at 250 nm wavelength using a Perkin Elmer Lambda 35 UV–Vis Spectrophotom-eter (USA) at different time intervals.
Results and Discussion
Morphological and Chemical Effects
Chemical treatments, such as hydrogen peroxide bleaching, affect intercellular cohesion in the wool cortex as well as in the cuticle. This can be manifest in long step fractures that show poor adhesion between cortex and cuticle, resulting in a hollow cuticular sheath. This lack of adhesion can be more clearly seen at higher magnification, where a loose, undulating cuticle is observed. Failure in the cuticle also occurs in a stepwise manner with individual cuticle cell failure, reflecting poor intercuticular adhesion. 19
In general, hydrogen peroxide and peroxy compounds damage wool fibers due to progressive oxidation of the disulfide bond of cystine (CYS). This chemical damage can lead to adverse effects on the fibers’ mechanical properties. 20 The scale structure of conventionally-bleached wool had some roughness, wool fiber breakages, and deterioration in the cuticle layer (Fig. 3a). By contrast, the scale structure of the liposo-mal-assisted bleached wool fiber was clearly visible (Fig. 3b).

SEM Images of wool fabrics. a) Non-liposomal and conventional peroxide bleached wool fabric, and b) liposomes on wool fabric.
Fluorescence microscopy can be helpful in observing treated fiber damage. Figs. 4a and b show fluorescence microscopy images of liposome-assisted bleached and conventionally-bleached fibers respectively. Fiber color was approximately the same throughout the entire fiber after liposome-assisted bleaching, however, after conventional bleaching, there was some yellowing. This can be attributed to oxidative damage.
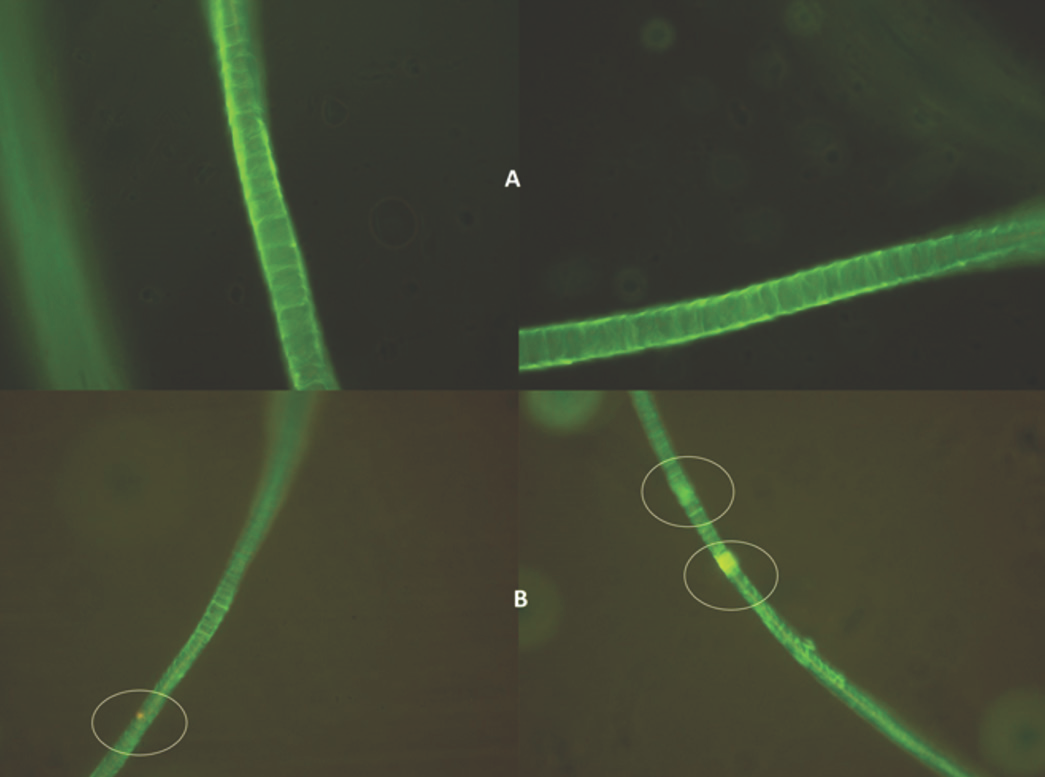
a) Liposomal-bleached wool fabric, and b) non-liposomal bleached and damaged wool fabric.
Fig. 5 shows the FTIR spectra of bleached samples with liposomes in comparison with conventional bleaching. No change in the main characteristic R–C=O absorption band at 1713 cm−1 was observed in any of the spectra. The FTIR spectra of liposome-assisted bleached wool showed an elastic C=O vibration peak at 1620–1630 cm−1, which was different than the peak observed from conventionally-bleached and commercial liposome-assisted bleached wool. Helical bands that are assignable to the α-helix of the keratin protein at ∼3300 cm−1are associated with ordered regions in the protein structure and quaternary ammonium salts. 21 More intense peaks were observed at 3287 cm−1 in the FTIR spectra of liposome-assisted bleached wool samples. Otherwise, there were no differences between FTIR spectra of liposome-assisted bleached fibers and conventionally-bleached fibers.
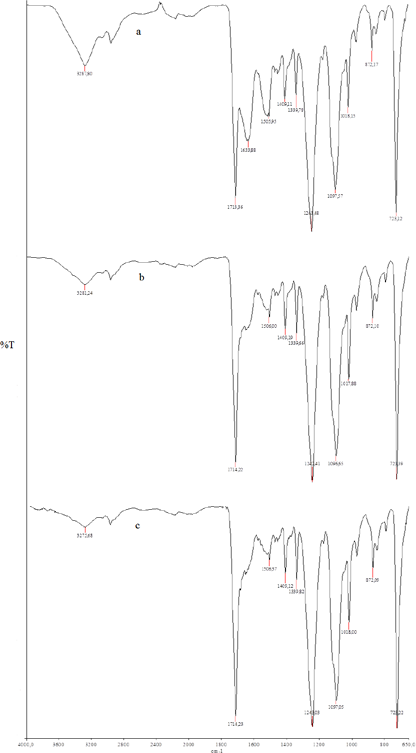
FTIR spectra of a) Conventional-bleached wool, b) liposome-bleached wool, and c) commercial-liposome bleached wool.
Hydrogen peroxide is commonly used for wool bleaching under both acidic and alkaline conditions, which can lead to damage in wool due to gradual oxidation of disulfide bonds and production of cysteine. 22 Bleaching can be achieved using hydrogen peroxide formulations at the expense of increased damage, particularly to CYS.
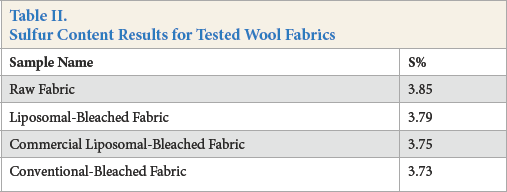
CHNS elemental analyses of bleached samples were carried out to examine the sulfur contents of the wool fabrics (Table II). Elemental analysis test results showed that cystine disul-fide bonds were broken during the conventional-bleaching process, but protected during the liposome-assisted bleaching process. As expected, the highest sulfur content was achieved for the raw wool fabric. When bleached samples were compared, the highest sulfur content was observed after liposome-assisted bleaching, while it decreased slightly after conventional bleaching.
Sulfur Content Results for Tested Wool Fabrics
Physical and Mechanical Effects
Breaking strength, solubility in alkali, changes in whiteness degree of bleached wool fabrics, and color measurement results for bleached and dyed wool samples, were examined. Bleached wool fabric test results are shown in Table III.
Physical and Mechanical Properties of Wool Fabrics Tested
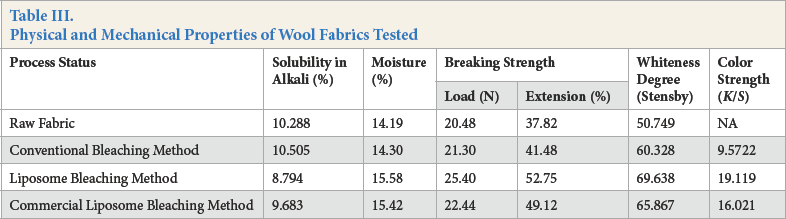
The natural color of animal fibers is very dependent on their environmental living conditions, although the selected animals had a pale-yellow color. Wool bleached in the presence of liposomes gave higher whiteness values than wool bleached by conventional bleaching. This result shows the higher efficiency of liposome-assisted bleaching when compared with conventional bleaching.
There were no significant differences between untreated and bleached wool fabric in moisture values. However, liposome-assisted bleached wool fabrics had the greatest moisture values. Absorption of liposomes, which is rich in polar lipids containing -OH and -NH groups, could facilitate the formation of hydrogen bonds, thereby increasing the moisture content of the wool. 23
Solubility in alkali indicated that considerable wool damage occurred due to peroxide bleaching when compared to untreated wool. Hydrogen peroxide and peroxy compounds damage wool fibers due to progressive oxidation of disulfide bonds, especially in alkaline medium. 19 The liposomal preparation had greater resistance to alkali damage. The presence of cholesterol in the membrane probably gives liposomes greater strength. Liposomes contain hydroxyl and amino groups capable of forming hydrogen bonds, which compact the structure of liposomes. Consequently, the resistance of liposomes to alkali depends on the chemical structure of the liposome membranes.
The presence of liposomes in the peroxide bleaching bath also improved the mechanical properties of the wool fabrics. The improvement of the physicomechanical characteristics of the wool indicated a significant reduction in the destructive effect of the oxidant. In the presence of liposomes, the breaking load reached 25 N, whereas without liposomes, it was 21 N. These liposome-assisted bleached fabrics had both greater elongation capabilities and greater strength. When comparing the warp/weft yarn density of wool fabrics after bleaching, the fabric density was 32/27 yarns/cm for conventionally-bleached wool (due to shrinkage) as compared to 36/30 yarns/ cm for bleaching with liposomes. The fabric weight of the liposome-assisted bleached wool also increased.
The bleached fabrics were then dyed with a 1:2 metal complex dye. From the maximum absorption wavelength of the dyed fabrics,
Liposomes in the peroxide bleaching bath increased the breaking load, whiteness, and resistance to alkali of the wool samples. The simultaneous increase in the strength characteristics and whiteness indicates the creation of more favorable conditions for peroxide bleaching. The results obtained cannot be explained by the high wetting power of liposomal preparations alone. The kinetic characteristics of oxidant decomposition, in the presence and absence of the proposed preparations, must be studied to understand the mechanism of liposome action.
Liposome Effect on Peroxide Decomposition
The effect of liposomes on decomposition of hydrogen peroxide in bleaching solutions was analyzed by observing the UV-Vis absorbance of hydrogen peroxide at 250 nm during different time intervals. 22 The results are given in Fig. 6. The decomposition of hydrogen peroxide with liposomes was similar to conventional bleaching for up to 5 h. When this method was analyzed as an alternative to the pad-batch method, hydrogen peroxide decomposed rapidly in the bath after 6 h for conventional bleaching; however, the peroxide concentration remained approximately the same for liposome-assisted bleaching. This indicates that liposomes can act as a stabilizer.
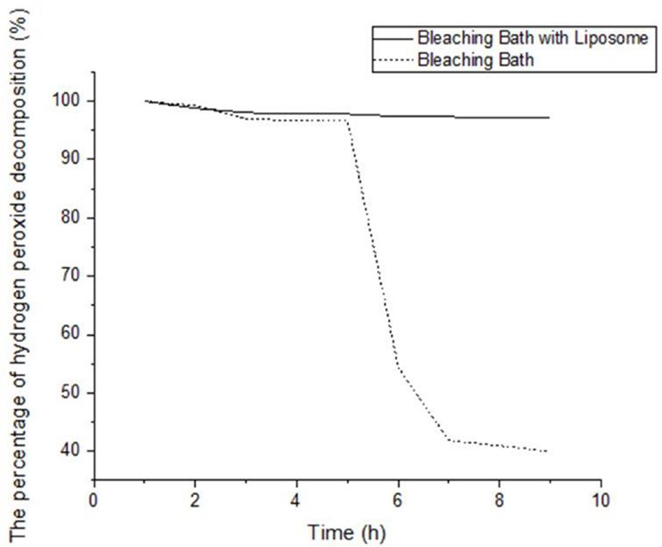
Peroxide decomposition curves. a) Bleaching bath with liposomes, and b) bleaching bath without liposomes.
Sheveleva at al. describe the mode of action of liposomal bleaching. Hydrogen peroxide decomposes rapidly when a conventional bleaching bath is used. Liposomes contain oxidant present in the bleaching solution that act as a reservoir and release the used bleaching agent gradually into the bleaching bath. Moreover, encapsulation of catalysts used for radical decomposition of hydrogen peroxide could retard the rate of decomposition. In this way, liposomes can act as a peroxide stabilizer, which is more efficient than the use of commercial peroxide stabilizers in the bleaching system. 13
Conclusion
In this work, the effect of liposomes on the wool hydrogen peroxide bleaching process was investigated. Soybean lecithin and cholesterol were used to form the liposome membranes.
Hydrogen peroxide and peroxy compounds damage wool fibers. The scale structure of the liposomal-bleached wool fibers was very clear by SEM analyses. The sulfur contents of the wool fabrics showed that cystine bond losses occurred in the conventional bleaching process, but were protected by the liposomal bleaching process.
Liposome-assisted wool bleaching gave a greater resistance to alkaline solutions when compared with conventional bleaching, with a breaking load of 25 N for the former and 21 N for the latter. The whiteness degree of bleached wool increased from 60 Stensby for conventional bleaching to 69 Stensby for liposome-assisted bleaching.
Hydrogen peroxide was observed to decompose more rapidly in conventional bleaching, whereas the peroxide concentration remained approximately the same during liposome-assisted bleaching.
This work shows that liposomes, which can be easily prepared from commercial lecithin and cholesterol, can be a good alternative to common bleaching stabilizers due to the resulting improvement in the mechanical properties and appearance of bleached wool fabrics.
Footnotes
Acknowledgment
We especially thank the Ministry of Science, Industry and Technology (01265.STZ.2012-1) and the Dokuz Eylül University Scientific Research Projects Unit (2012.KB.FEN.032) for support of our project.
