Abstract
Trials were conducted to examine the efficiency of photocatalytic afterclearing on disperse dyed polyester. The photocatalytic afterclearing was performed by ultraviolet (UV) irradiation and hydrogen peroxide (H2O2). Polyester fabric samples were dyed with three disperse dyes at 4% depth of shade, individually. The control afterclearing treatment was a conventional reductive clearing by thiourea dioxide and caustic soda. The H2O2 impregnated samples, using various concentrations of H2O2 at various pH values, were treated under 254 nm UV irradiation (5, 10, 20, and 30 min) at room temperature. Color, color difference, wash fastness, and tensile strength values of the samples were compared. Photocatalytic clearing of disperse dyed polyester was successful in giving comparable wash fastness results as compared to conventional reduction clearing without significant color differences.
Introduction
Polyester (polyethylene terephthalate or PET) fibers are mostly dyed with disperse dyes, which have high substantivity for polyester fibers. Tree methods exist for polyester dyeing: the carrier method, the high-temperature method, and the dry-heat or thermosol method. A 5% to 25% portion of disperse dye in the dyebath was not fixed to the polyester fiber according to the US Environmental Protection Agency (US EPA). These unfixed disperse dyes deposit on the surface of the polyester fibers and decrease fastness properties (wash fastness, sublimation fastness, and rubbing fastness). Therefore, especially for dark shades, an afterclearing treatment subsequent to dyeing is a common practice. The unfixed disperse dyes on polyester are broken down into smaller, often colorless, and more readily water-soluble fragments by reduction clearing.1-10
Reduction clearing is the conventional afterclearing process where disperse dyed polyester is treated in a strongly reductive bath. The reduction clearing bath is prepared using an appropriate reducing agent (sodium dithionite (Na2S2O4) or thiourea dioxide (CH4N2O2S, TUDO)) and caustic soda (NaOH). Reduction clearing is usually followed by a neutralization step. Not only the wastewater generated, but also the content (sulfur) of reduction clearing has environmental impact.11-14
Since the basis of reduction clearing is breaking down the unfixed disperse dyes on polyester into smaller fragments, any process that breaks down the highly-substantive unfixed disperse dyes on polyester may be used as an afterclearing method for disperse dyed polyester, if the material strength and color is preserved. Therefore, oxidative clearing of disperse dyed polyester is reported in the literature. Hydrogen peroxide, 15 and especially ozone, is reported to yield good fastness properties for oxidative clearing of polyester.9,14,16-18 Ozone treatment is an advanced oxidation process (AOP), as is UV/H2O2 treatment. The basis of the UV/H2O2 process is the production of hydroxyl radicals (•OH) by the direct photolysis of H2O2 via UV irradiation (Eq. 1). Hydroxyl radicals have a high redox potential of 2.80 V, which is much greater than most oxidizing agents such as H2O2 (1.77 V). Hydroxyl radicals attack organic compounds relatively non-selectively (Eq. 1).19,20
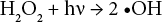
Photocatalytic UV/H2O2 treatment has been reported for decolorization of dyeing effluent. Decolorization occurs by breaking down the disperse dyes in the colored dyeing effluent. The successful decolorization reports indicated the potential for UV/H2O2 treatment to break down the disperse dye. Afterclearing of disperse dyed polyester also depends on breaking down the unfixed disperse dye on the dyed polyester. Therefore, UV/H2O2 treatment has the potential for afterclearing disperse dyed polyester in continuous open-width production lines.21-24 However, UV/H2O2 clearing of disperse dyed polyester has not yet been studied.
In this study, UV/H2O2 treatment was tested as a novel method for afterclearing of disperse dyed polyester.
Experimental
Materials
The 100% polyester woven fabric used in the experiments was a plain weave, with a warp density of 16 ends/cm and weft density of 15 picks/cm, weighing 108 g/m2.
Tree disperse dyes of yellow, red, and blue colors were used in this study. C.I. Disperse Yellow 211, C.I. Disperse Red 82, and C.I. Disperse Blue 337 were supplied by Huntsman.
Methods
Dyeing
Polyester fabric samples were dyed with the disperse dyes at 4% omf (on mass of fabric) depth of shade. The dyeings were performed in a laboratory scale dyeing machine (Termal Model 410, Termal Lab.) The recipe consisted of 4% omf disperse dye, 1 mL/L dispersing agent (Dispergator XHT, CHT Turkey), and acetic acid (Aykim) to adjust the pH to 4. Dyeings were performed at 130 °C for 45 min at a liquor to goods ratio (LR) of 15:1.
Conventional Reduction Clearing
Conventional reduction clearing was the control afterclearing process for this study. For this purpose, 3 g/L TUDO (Bursa Teknik Kimya) and 2 g/L sodium hydroxide (Bursa Teknik Kimya) were used at 85 °C for 20 min with an LR of 15:1. The fabrics were rinsed with cold tap water for 2 min after the treatment.
Photocatalytic Clearing
The photocatalytic UV/H2O2 process was performed in a custom-designed UV cabinet, equipped with 254 nm UV light sources (Philips) of various lamp powers (Fig. 1). The total UV lamp power used was 254 W during the experiments. The UV cabinet has a total installed UV lamp power of 470 W (127 W on each side (left and right) and 216 W at the top). However, only the left and right UV lamps were creating a total UV lamp power of 254 W.
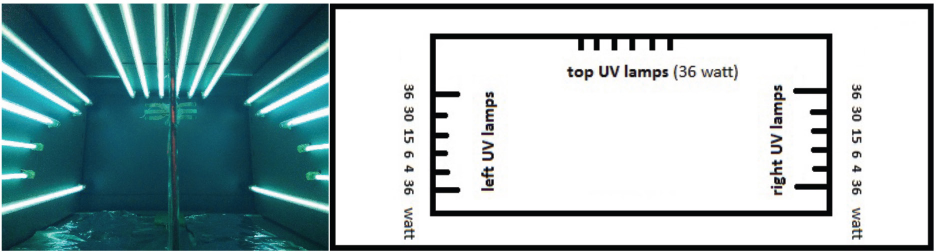
UV cabinet used during experiments.
The dyed polyester samples were padded with H2O2 solution at room temperature (RT) and a wet pickup rate of 80%. Ten, the impregnated samples were hung vertically in the middle of the UV cabinet for UV irradiation exposure. H2O2 (35%) was added to deionized water to prepare the H2O2 solutions (at 0.5, 1, 2, 3, 4, 5, and 10 mL/L H2O2 concentrations). The tested UV irradiation times were 5, 10, 20, and 30 min. H2O2 solutions were prepared at various pH values; pH 7 (neutral), pH 2 (by hydrochloric acid), pH 4 (by acetic acid), and pH 10 (by caustic soda). The UV-irradiated samples were first rinsed with 0.1 mL/L catalase enzyme Rucolex HTK (Rudolf Duraner) at 50 °C for 3 min to remove any residual H2O2, then rinsed under cold tap water for 2 min.
Color Difference
The color differences (DE) values of the samples were determined using a Konica Minolta CM 3600d spectrophotometer. Each sample was measured from three different areas on each side of the fabric and the average values were calculated.
Wash Fastness
Wash fastness was determined according to the ISO 105-C06 C2S test method. Tests were carried out at 60 °C for 30 min with 25 steel balls. 25 The stainings of each fiber component on the multifiber adjacent fabric (SDC Multifiber) were read via spectrophotometer for fastness ratings.
Tensile Strength
Tensile strength tests were performed in accordance with ISO 13934-1999, using a Shimadzu Model AG-X- Plus tensile tester with a gauge-length of 100 mm and at a speed of 100 mm/min. 26
Results and Discussion
Color Differences of Dyed Fabrics
The initial trials were performed using the blue dyed samples. Various treatments that affect color or fastness properties were tested at this stage of the trials.
UV alone on dry or wet (water impregnated sample at a wet pickup rate of 80%)
Using 0.5, 1, and 2 mL/L H2O2 impregnated (wet pickup of 80%) without UV and under UV for 10 min
Using 2 mL/L H2O2 impregnated (wet pickup of 80%) without UV and under UV for 10, 20, and 30 min
Using 2 mL/L H2O2 impregnated (wet pickup of 80%) without UV and under UV for 10 min at pH 2, 4, 7, and 10
Front and Back Side
Color differences on the front and back sides of the samples were investigated first; samples were hung in the UV cabinet and irradiated by UV light sources on both sides. Therefore, any uneven clearing effect on different sides (front and back) of the samples were measured, and the results are given in Fig. 2.
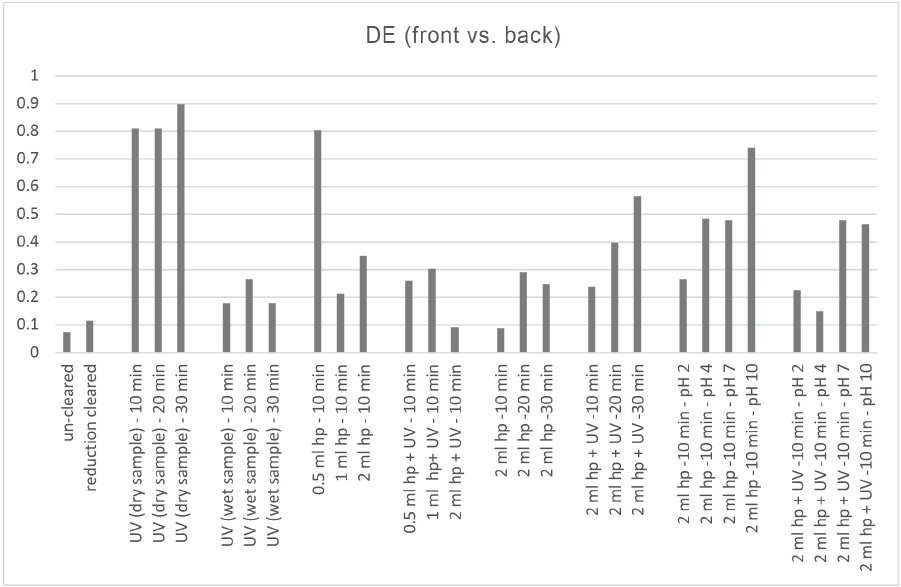
Color difference of the front and back sides of the blue dyed samples (reference: front side of each sample; UV: ultraviolet irradiated; wet sample: sample impregnated by water at a wet pick up of 80%; the treatment pH was neutral unless mentioned).
In general, DE = 1.0 is a useful limit to rate the color of a sample pass/fail compared to the reference. As seen in Fig. 2., the color differences (DE) between front and back sides of all samples were less than DE = 1.0, indicating that the photocatalytic treatment in the UV cabinet did not cause a significant color differences between front and back sides of the samples.
Samples Measurements and Wash Fastness Evaluation
After confirming that the photocatalytic treatment applied did not cause color differences between the front and back sides of the samples, the color differences were measured using the reduction cleared sample as reference. The results are given in Table I. A color difference value of DE < 1.0 was considered acceptable. The wash fastness results of the samples are also given in Table I.
Color Difference of the Blue Dyed Samples and Wash Fastness Ratings a
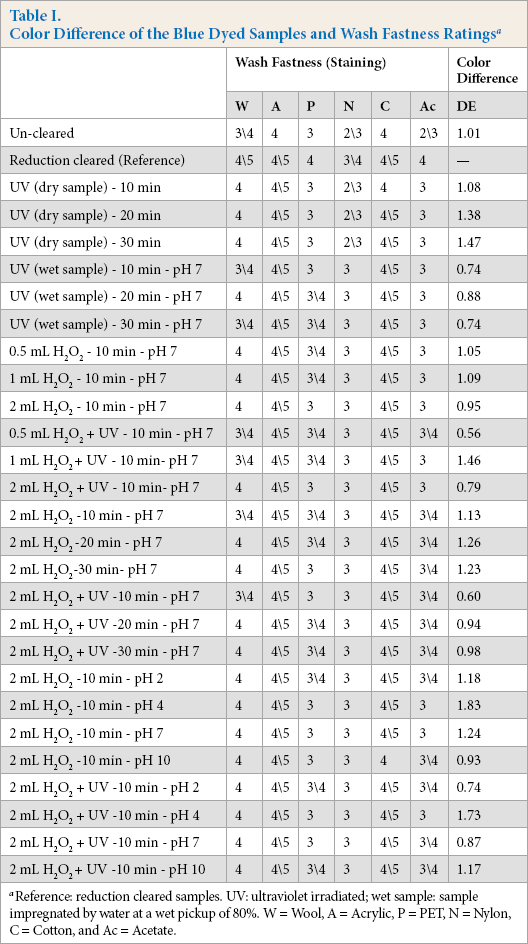
Reference: reduction cleared samples. UV: ultraviolet irradiated; wet sample: sample impregnated by water at a wet pickup of 80%. W = Wool, A = Acrylic, P = PET, N = Nylon, C = Cotton, and Ac = Acetate.
Table I was used to select the treatment types for the next set of experiments. Two aspects were considered during this selection: fastness and color differences.
First, the highest wash fastness ratings were reviewed for each fiber component on the multifiber adjacent fabric for all fiber components. Ten, the DE values of the treatments were reviewed; DE < 1.0 was accepted as the limit to rate the color difference of a sample pass/fail compared to the reference. Consequently, three types of treatments were selected as promising treatment types at this first level of trials. These selected treatment types were UV/H2O2 treated samples at pH 7 for 20 and 30 min and UV/H2O2 treated samples at pH 2 for 10 min.
UV/H2O2 treatments at pH 7 (neutral) and pH 2 were selected to be applied in the second stage of the experiments according to these results. The wash fastness ratings improved by respective treatments, but none of the treatments could reach the wash fastness ratings of the reduction cleared samples. Therefore, higher H2O2 concentrations were used in the next stage of experiments.
The second set of trials were performed on the blue dyed samples with 3, 4, and 5 mL/L H2O2 at pH 7 (neutral) and pH 2 for 5, 10, and 20 min. Color difference and wash fastness results are presented in Table II.
Color Difference of Blue Dyed Samples after Respective Treatments a
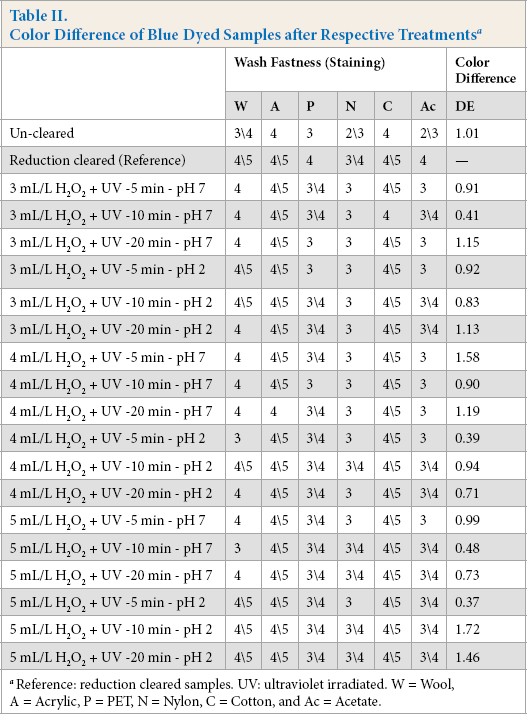
Reference: reduction cleared samples. UV: ultraviolet irradiated. W = Wool, A = Acrylic, P = PET, N = Nylon, C = Cotton, and Ac = Acetate.
Table II was evaluated the same way as Table I: the highest wash fastness ratings were reviewed, and then the color difference values of DE < 1.0. Tree of the 18 treatments passed as seen in Table II. The promising treatment types were the UV/ H2O2 treatment by 5 mL/L H2O2 for 10 min at pH 7, 4 mL/L H2O2 for 10 min at pH 2, and 5 mL/L H2O2 for 5 min at pH 2.
The wash fastness ratings presented in Tables I and II show that the wash fastness improved as the amount of H2O2 increased. The wash fastness ratings for the 5 mL/L H2O2 concentration are 0.5 gray scale degrees higher for wool, nylon, and acetate fiber components compared to the 3 mL/L H2O2 concentration in Table II.
The wash fastness ratings for the 5 mL/L H2O2 concentration at pH 2 were almost the same as the reduction cleared samples as seen in Table II. These results were quite satisfactory, therefore trials were continued using 5 mL/L H2O2 for the other colors (yellow and red).
The color differences and wash fastness ratings for all three colors tested are given in Table III. These results were encouraging for the proposed novel photocatalytic clearing process. Wash fastness improvement was observed in all cases, as well as limited color differences.
Color Difference and Wash Fastness of the Dyed Samples after Respective Treatments a
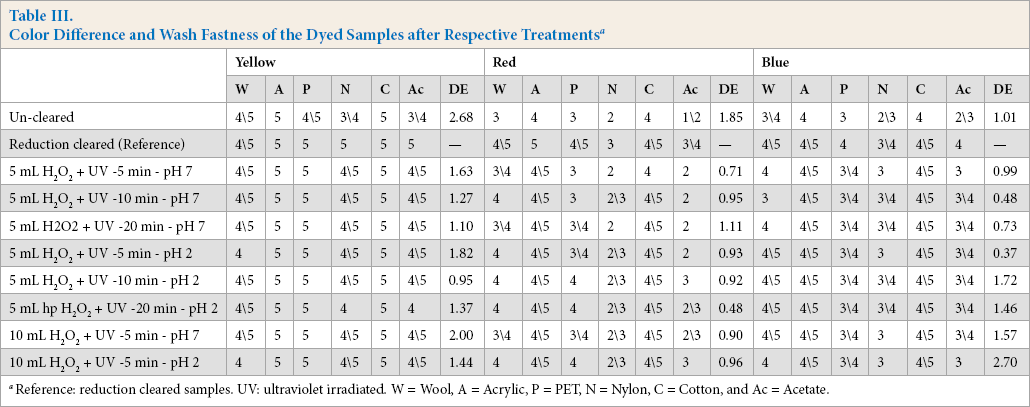
Reference: reduction cleared samples. UV: ultraviolet irradiated. W = Wool, A = Acrylic, P = PET, N = Nylon, C = Cotton, and Ac = Acetate.
Tensile Strength Properties of Dyed Fabrics
The measured tensile strength values of the samples after respective treatments are presented in Fig. 3. The test results did not show significant sample strength losses, indicating that the photocatalytic clearing process can be safely applied to polyester under the conditions used here.
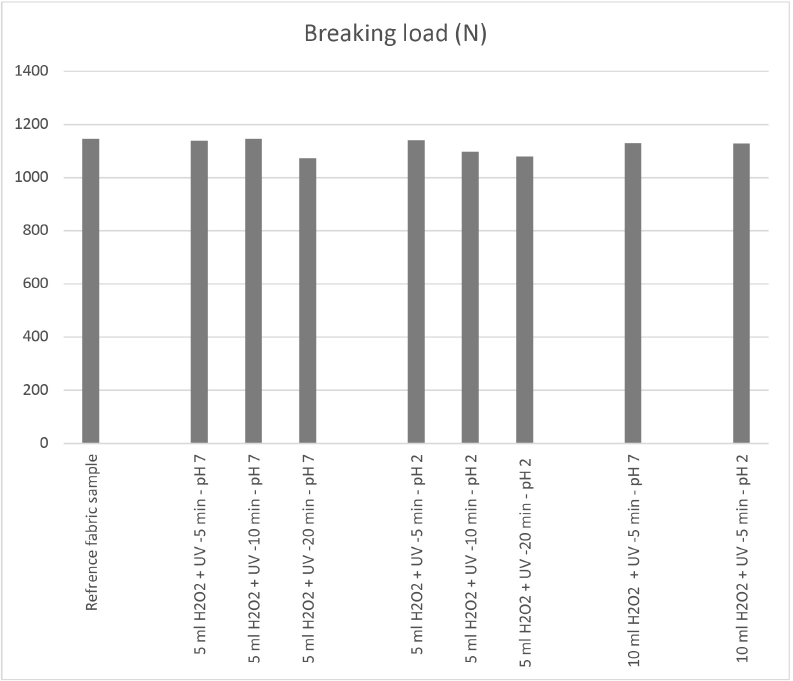
Tensile strength of the samples (UV: ultraviolet irradiated).
Conclusions
In this study, the photocatalytic clearing of disperse dyed polyester was demonstrated. This is a novel method, since this application has not been reported previously in the surveyed literature.
Several H2O2 concentrations, treatment times, and pH values were tested for yellow, red, and blue dyed fabric samples. Results indicated that photocatalytic clearing of disperse dyed polyester was successful in achieving comparable wash fastness results compared to conventional reduction clearing without significant color differences.
This novel approach may substitute for or reduce the use of alkali for clearing of disperse dyed polyester in continuous open-width production lines. Also, it can be evaluated as an alternative method, especially for pad-thermosol dyed goods. This method may also be tested for clearing dyed blends. More studies of this novel method are needed before practical use in industry can occur.
