Abstract
The micro-nano-sized electrospun fibers with carboxyl groups were prepared by a high-voltage electrospinning technology. The PbS-ZnS heterojunction/electrospun fiber composites were firstly synthesized by low-temperature hydrothermal method which directly loaded PbS-ZnS heterojunctions on the surface of the electrospun fibers. The composites were characterized by scanning electron microscope, X-ray diffraction, X-ray photoelectron spectroscopy, thermo gravimetric analysis, and UV-Vis reflectance spectra. The results showed that PbS-ZnS heterojunctions were evenly distributed on the electrospun fibers. The unique structure afforded PbS-ZnS/electrospun fiber desirable photocatalytic activity and stability for the hydrogen production under solar ultraviolet light irradiation. The average hydrogen production capacity was 10.45 mL every 2 h.
Introduction
Photocatalysts which convert solar energy into useful energy have been extensively investigated in recent years (Chen et al., 2018; Fouquet and Pearson, 2012; Yu et al., 2016). The application of semiconductor materials in photocatalytic hydrogen production has become a hot spot of scholars around the world (Liu et al., 2012; Meng and Zhang, 2016). A host of scientists and scholars have done a lot of researches and achieved better photocatalytic effect in order to find an excellent photocatalyst (Leary and Westwood, 2011; Wang et al., 2013; Zhong et al., 2016; Zhu and Zäch, 2009). However, there are still several practical problems in powder photocatalysts, such as the easy recombination of the photo-generated electron-hole pairs, the little surface area, the poor dispersion, the easy floating and sticking to the wall, and incomplete to recycle. The dispersion can be improved, the specific surface area will be increased, and the recovery of the photocatalyst can be promoted while the semiconductor loaded on a specific carrier. Electrospinning is a simple and low-cost method for making the nano-fibers with ultra-small diameters. Many materials had been prepared using this method since the first patent on this process issued in 1934 (Dejin et al., 2014; Li and Zhang, 2014; Wang et al., 2006).
PbS is an important direct bandgap semiconductor whose width is 0.41 eV and has unique electrical and optical properties. Thus, it has been extensively studied in some important fields such as infrared detection, biological fluorescent probe, and solar photocatalysis (Ankah et al., 2016; Golabi et al., 2015; Lee et al., 2009; Mamiyev and Balayeva, 2015). ZnS is an important semiconductor in groups II–VI, which is widely used in optical and electrical fields. The conduction band of ZnS is −1.04 eV, and valence band is 2.56 eV (Fang et al., 2011; Lee and Wu, 2017; Wang et al., 2009). ZnS attracts more focus because of the unique advantages in photocatalytic water hydrogen production such as good hydrogen production performance and high catalytic efficiency (Chang and Chu, 2016; Hernández-Gordillo et al., 2012; Kimi et al., 2016; Li et al., 2013).
In this paper, PbS-ZnS/electrospun fiber composites were prepared by supporting PbS-ZnS heterojunction on the polyvinylidene fluoride (PVDF)/styrene-maleic anhydride (SMA) polymer fibers in the way of hydrothermal synthesis. This composite was the first studied and used as photocatalyst of hydrogen energy production. The photocatalytic performance of the composites was also studied by the mercury lamp.
Experiments
Materials
N, N-dimethylacetamide (DMAC) was purchased from Shanghai Pilot Chemical Co., Ltd. SMA was supplied from Shanghai SpecTek petrochemical high tech Co., Ltd. PVDF was purchased from Shanghai 3F New Material Co., Ltd. Acetone was obtained from Shanghai Su Yi Chemical Reagent Co., Ltd. Lead nitrate, zinc nitrate and thiourea were brought from Sinopharm Shanghai Chemical Reagent Co., Ltd. All chemicals were used as received.
A high-voltage electrostatic generator was bought from Jinan Loufang Co., Ltd. The morphologies of the products were characterized by scanning electron microscopy (JSM-6490LV), which was bought from JEOL Co., Ltd. ESCALAB 250 X-ray photoelectron spectroscopy bought from Thermo Electron Co., Ltd was applied to study the structure of composites. Thermo-gravimetric analysis (TG-209-F3) purchased from NETZSCH-Gerätebau GmbH was utilized to estimate the weight loss of composites. The crystal structure was detected through the X-ray diffraction (D/MAX2500V) bought from Rigaku Co., Ltd. The amount of hydrogen production was measured by gas chromatography (SP-6801), which was purchased from Shandong Lunan Ruihong Chemical Instrument Co., Ltd. Photocatalysis experiment was conducted by Multifunctional chemical reaction instrument (SGY-IB) bought from Nanjing Hoop Technologies Co., Ltd. Mercury lamp with the power of 300 W was purchased from Changsha Kexing Co., Ltd.
Electrospinning of PVDF/SMA
SMA (0.41 g) and PVDF (3.6 g) were weighed into a stirred three-necked flask with N2 protection, and then they were dissolved into the mixture of acetone and DMAC to prepare a spinning solution. PVDF/SMA was prepared by a high-voltage electrospinning device.
Preparation of PbS-ZnS /electrospun fiber composites
The as-prepared electrospun fiber mats were immersed in 25 mL, 0.25 mol L−1 zinc nitrate solution for 24 h at room temperature, making zinc ions fully contact the carboxyl group on the surface of the electrospun fiber. The ZnS/electrospun fibers were synthesized via hydrothermal method, and the fiber mats containing Zn2+ and 25 mL, 0.25 mol L−1 thiourea solutions were added into a 50 mL Teflon-lined stainless steel autoclave and maintained at 120°C for 12 h. After it was cooled to room temperature, the fibers were taken out and ultrasonic cleaned by deionized water for 30 min in order to remove the byproducts and unreacted precursor and then dried in vacuum for 12 h at 60°C.
The dried fibers were immersed in 0.25 mol L−1 lead nitrate aqueous solution at room temperature for 24 h. The ZnS/electrospun fiber mats were placed in a 50 mL hydrothermal reactor, and 25 mL, 0.25 mol L−1 thiourea were added, reacting at 120°C for 12 h. After being cooled at room temperature, it was washed in deionized water by ultrasonic cleaning method for three times and dried at 60°C for 12 h. The PbS-ZnS/electrospun fiber composites were prepared. Also, the PbS-ZnS powders were prepared by the same conditions.
Production of H2
Photocatalytic activities of the as-prepared samples for the generation of hydrogen were carried in SGY-IB multi-purpose chemical reactor from Nanjing Hoop Technologies Co., Ltd. The light source was a mercury lamp (300 W, 225 nm), and the system temperature was kept at 25 ± 1°C using cool water to remove the heat generated by Hg lamp. The electrospun fiber composites (0.29 g containing 0.15 g PbS-ZnS heterojunction) were added in an aqueous solution (250 mL) containing 10 g of Na2SO3 (0.32 mol L−1) and 1.95 g of Na2S (0.10 mol L−1), while the PbS-ZnS powders (0.15 g) were dispersed in the same solution with a magnetic stirrer. The system was initially purged by N2 for 20 min in the dark environment to exclude the oxygen. The generated gas was collected using water drainage method every 2 h. The amount of H2 production was measured by gas chromatography (SP-6801, MS-5A column, TCD detector, N2 carrier).
Results
Figure 1(a) showed the SEM image of PVDF/SMA fiber mats produced by the electrospinning method. From the image, it can be seen that the fiber diameter distribution is uniform. Figure 1(b) showed that the spherical shape of ZnS particles grew on the surface of electrospun fibers. From the inset of Figure 1(b), we can see that ZnS/electrospun fiber composites can be formed after the hydrothermal reaction, because fluorine fibers with carboxyl can complex zinc ions. Figure 1(c) and (d) showed typical SEM images of the fiber mats in low and high resolutions, in which it can be seen that PbS-ZnS particles with an average size of about 1 μm in diameter are uniformly spreading on the surface of electrospun fibers.

SEM images of (a) PVDF/SMA electrospun fibers, (b) ZnS/electrospun fiber composites, and PbS-ZnS/electrospun fiber composites in (c) low and (d) high resolutions.
Discussions
Characterization of PbS-ZnS/electrospun fiber composites
X-ray diffraction (XRD) analysis of the fiber composite showed three clearly strong absorption peaks corresponding to the (111), (220), and (311) planes of ZnS, indicating that the ZnS supported on the fiber was hexagonal wurtzite structure of the crystalline phase (PDF 39–1363). The diffraction peaks appearing in XRD patterns of PbS-ZnS/PVDF electrospun fiber composites of Figure 2 were almost identical with the characteristic peaks of the standard PbS pattern (JCPDS 5–0592). The strong diffraction peaks corresponded to (111), (200), (220), (311), (222), and (440) planes. All indicated that PbS nano-particles and PbS supported on PVDF/SMA fiber had good galena crystal structure. The absorption peaks of ZnS and PVDF/SMA electrospun fibers were also shown in Figure 2, which indicated that PbS-ZnS/electrospun fiber composites had been formed on the electrospun fibers.
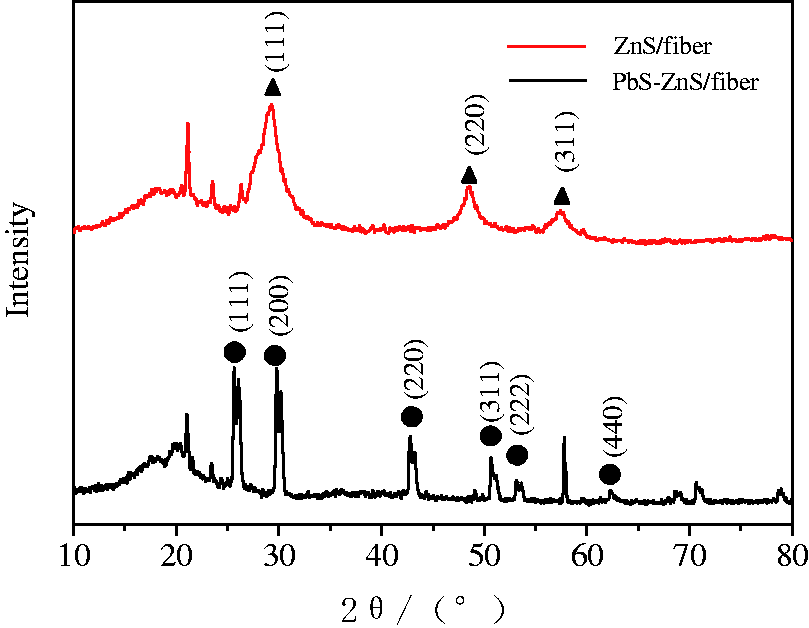
XRD patterns of ZnS/PVDF electrospun fiber composites and PbS-ZnS/PVDF electrospun fiber composites.
The XPS (X-ray photoelectron spectroscopy) analysis of PbS-ZnS heterojunction/electrospun fiber composites was shown in Figure 3. The composites were made of C, O, F, Pb, Zn, and S elements. The S2p binding energy of PbS-ZnS/electrospun fiber composites was 161.48 eV, which was close to 161.5 eV of S2− in NaS standard sample. The binding energy values at 142.18 eV and 137.38 eV were from Pb4f5/2 and Pb4f7/2. Due to the division of the Zn2p orbital, the peaks of the binding energy at 1021.68 eV and 1044.68 eV corresponded to Zn2p3/2 and Zn2p1/2, respectively.
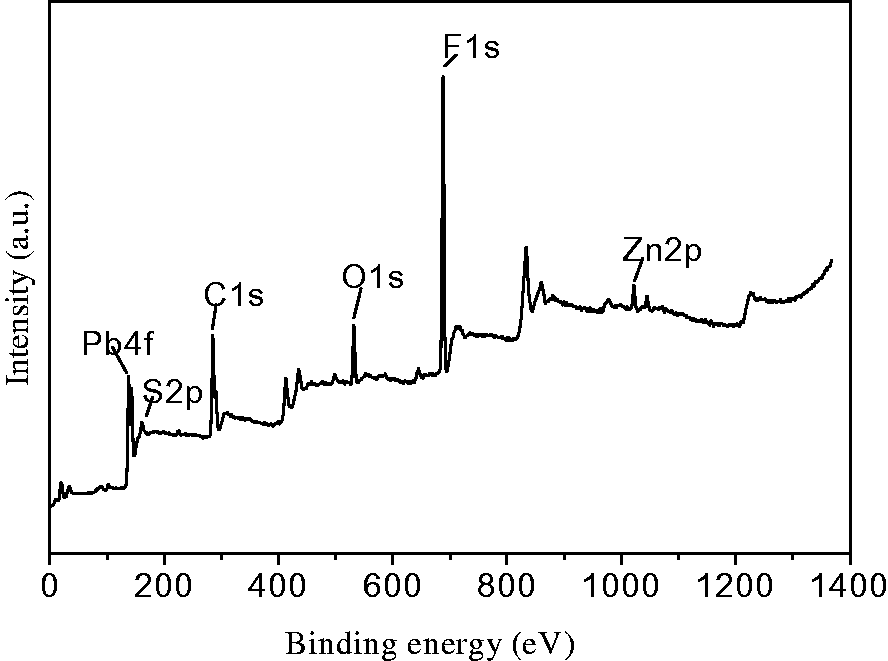
XPS spectra of PbS-ZnS/PVDF electrospun fiber composites.
Figure 4 shows that the absorption of PbS-ZnS heterojunction/electrospun fiber composites at 350 nm had a maximum absorption peak and indicates that the photocatalyst could absorb ultraviolet light with the wavelength less than 360 nm, which was caused by the quantum effect. However, the unsupported electrospun fibers had no evident absorption between 300 and 800 nm, while composites have absorption peaks at around 280 nm, which revealed that the conducting electrospun fibers did not interfere with the light absorption during the photocatalytic reaction.
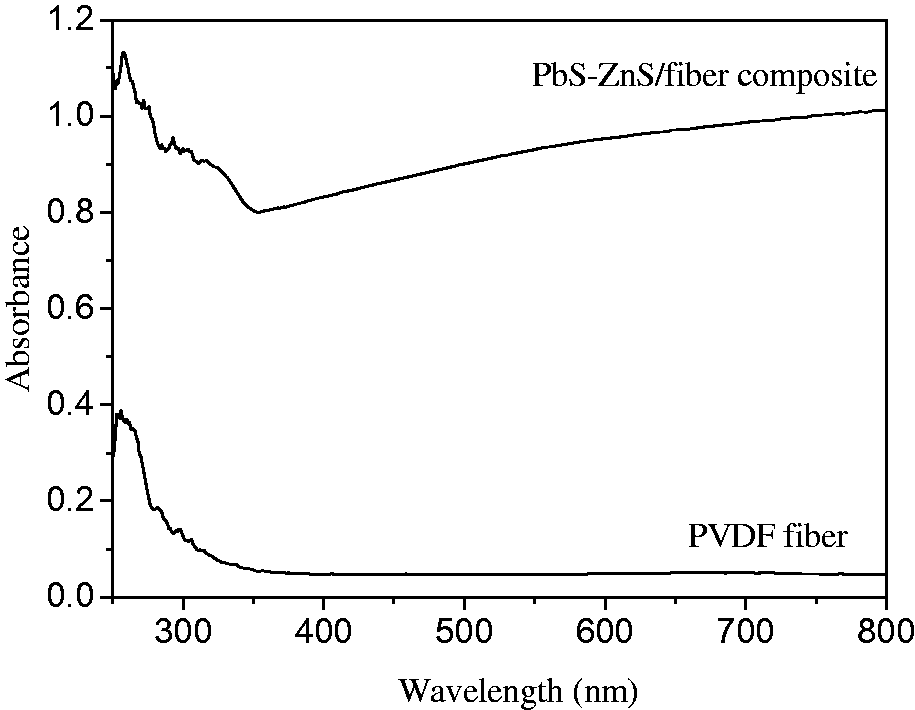
UV/VIS absorption spectra of PbS-ZnS/PVDF electrospun fiber composites and PVDF electrospun fiber. PVDF: polyvinylidene fluoride.
Figure 5 shows the thermal gravimetric analysis (TGA) curve of PbS-ZnS heterojunction/electrospun fiber composites. The final mass loss of PVDF/SMA electrospun fibers was 93% by TGA, while the final weight loss of PbS-ZnS heterojunction/electrospun fiber composites after loading was 45.23%; so, the mass fraction of the catalyst was about 51.36%.
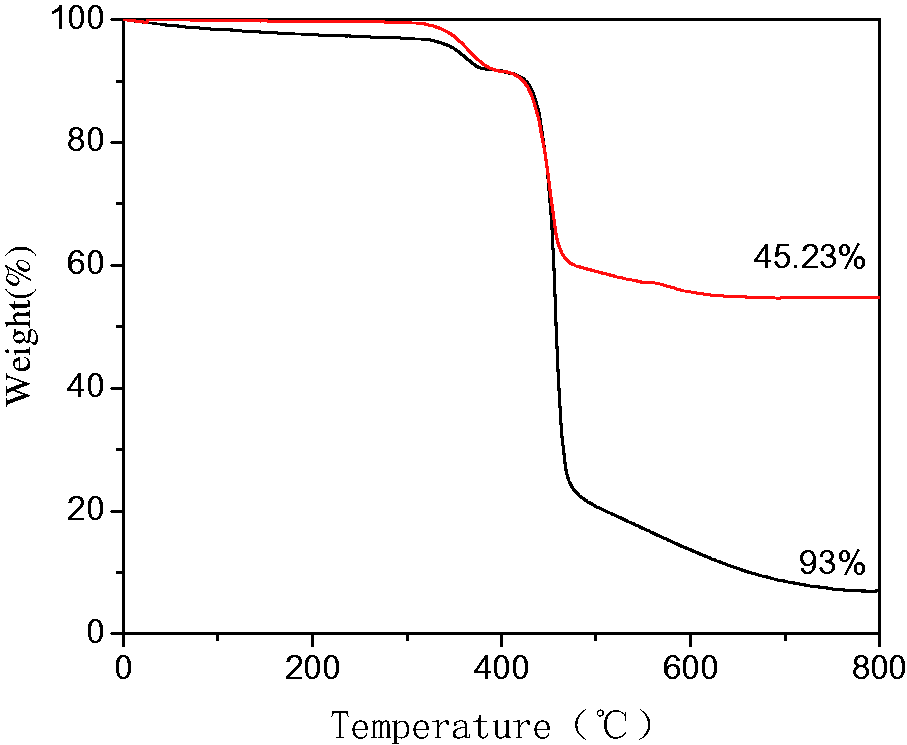
TGA spectra of catalysts.
Photocatalytic H2 evolution
Photocatalytic activities of the as-prepared samples for the generation of hydrogen were carried in SGT-IB multi-purpose chemical reactor from Nanjing Sionetech Electrical Equipment Co., Ltd; the light source was Hg lamp (300 W, 225 nm); system temperature was kept at 25 ± 1°C using cool water to remove the heat generated by Hg lamp. The electrospun fiber composites (0.29 g containing 0.15 g PbS-ZnS heterojunction) were added in an aqueous solution (250 mL) containing 10 g of Na2SO3 (0.32 mol L−1) and 1.95 g of Na2S (0.10 mol L−1), while the PbS-ZnS powders (0.15 g) were dispersed in the same solution with a magnetic stirrer. The system was initially purged by N2 in dark environment to exclude the oxygen. The amount of H2 production was measured by gas chromatography (SP-6801, N2 carrier).
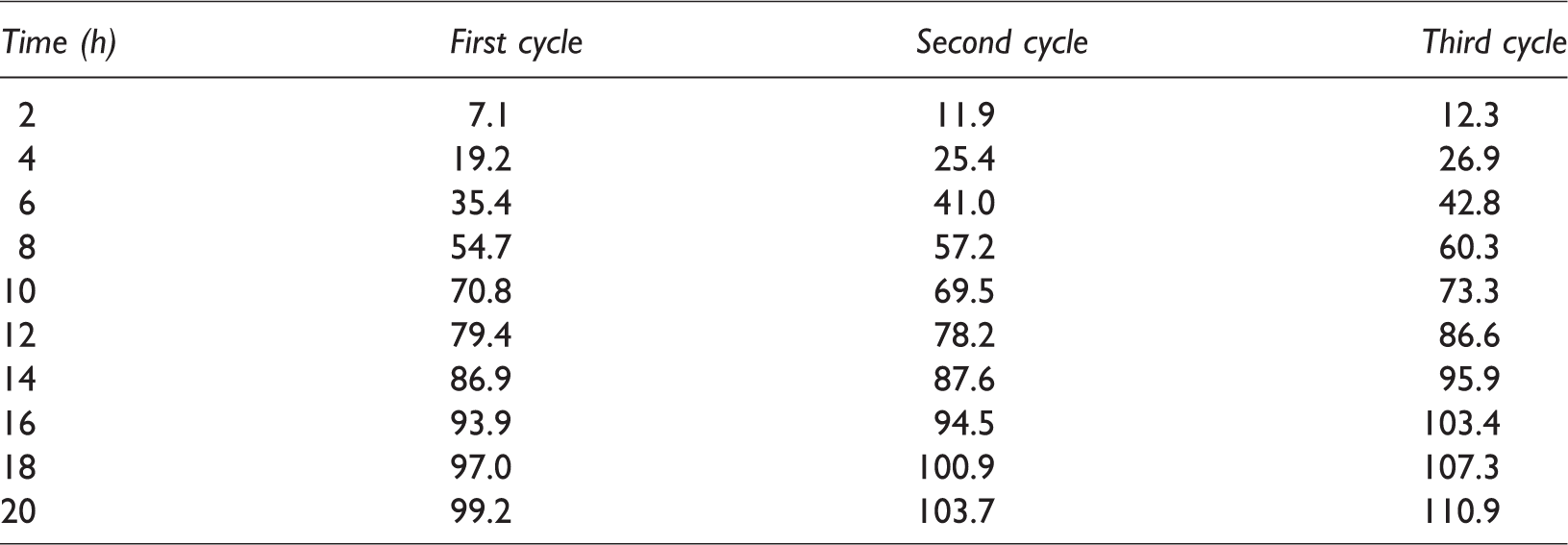
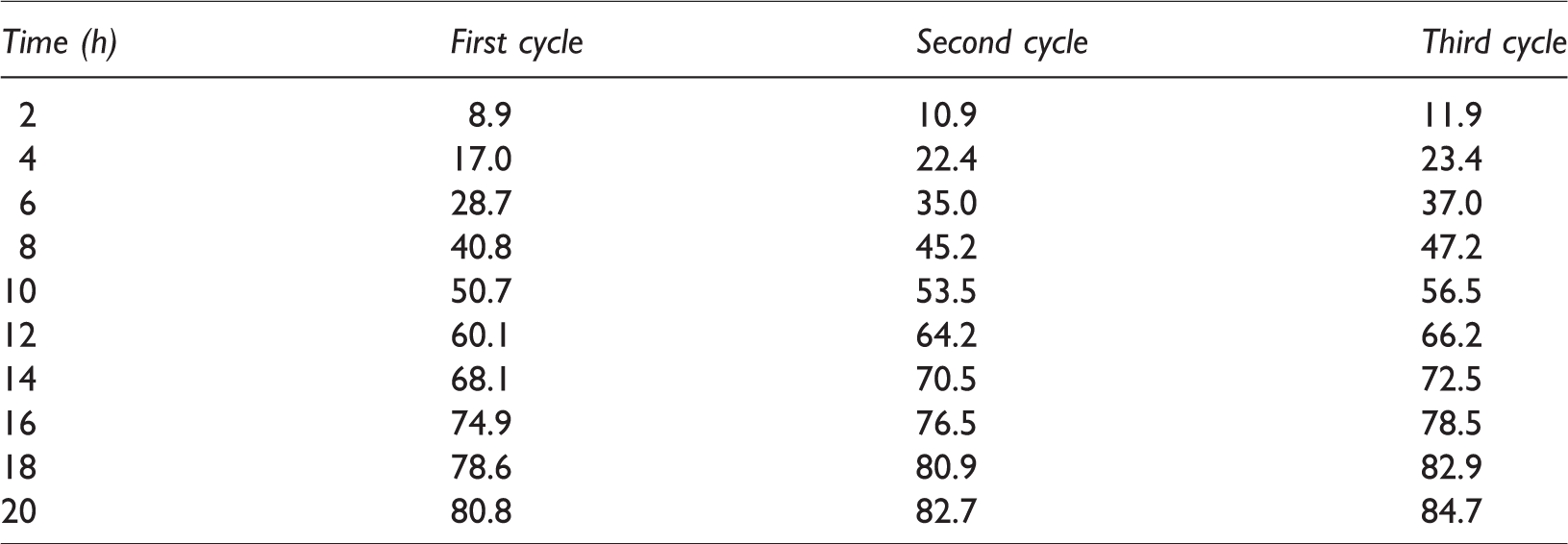
Figure 6 and Tables 1 and 2 showed the comparison of the amount of hydrogen produced by the PbS-ZnS/electrospun fiber composites and the PbS-ZnS powder for three cycles with equal sacrifices per cycle. The results showed that the hydrogen production of PbS-ZnS powders was almost unchanged from the first to the third cycle, and the hydrogen production of PbS-ZnS/electrospun fiber composite material under the same conditions increased obviously, indicating that the catalyst had good repeatability under UV light irradiation. Furthermore, in the third cycle, the amount of hydrogen produced was 1.31 times that of PbS-ZnS heterojunction powder.
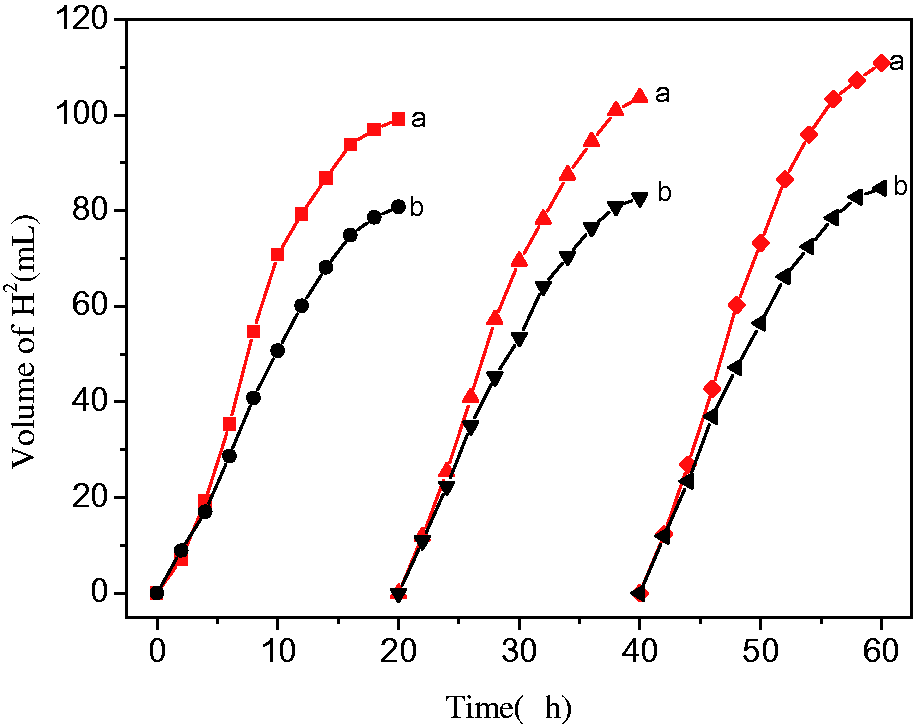
The amount of hydrogen produced by (a) the PbS-ZnS/electrospunning fiber composite; and (b) the PbS-ZnS powder.
Different cycles of photocatalytic hydrogen production of PbS-ZnS/PVDF electrospun fiber composites (mL).
Different cycles of photocatalytic hydrogen production of PbS-ZnS powders (mL).
There were three possible reasons for this phenomenon (Figure 7). First, when PbS-ZnS heterojunction particles were loaded on the surface of the fibers, they possessed large specific surface area and high-energy efficiency of UV irradiation. Second, the carrier could prevent the loss and degradation of the powder catalyst. Third, photo-generated eletron-hole pairs within the PbS-ZnS heterojunction particle were produced in the light; the electron migrated to the surface of the PbS and generated hydrogen by the reduction of water. The hole migrated to the surface of the electrospun fiber and reacted with the sacrificial agents which were adsorbed on the surface of the fiber.
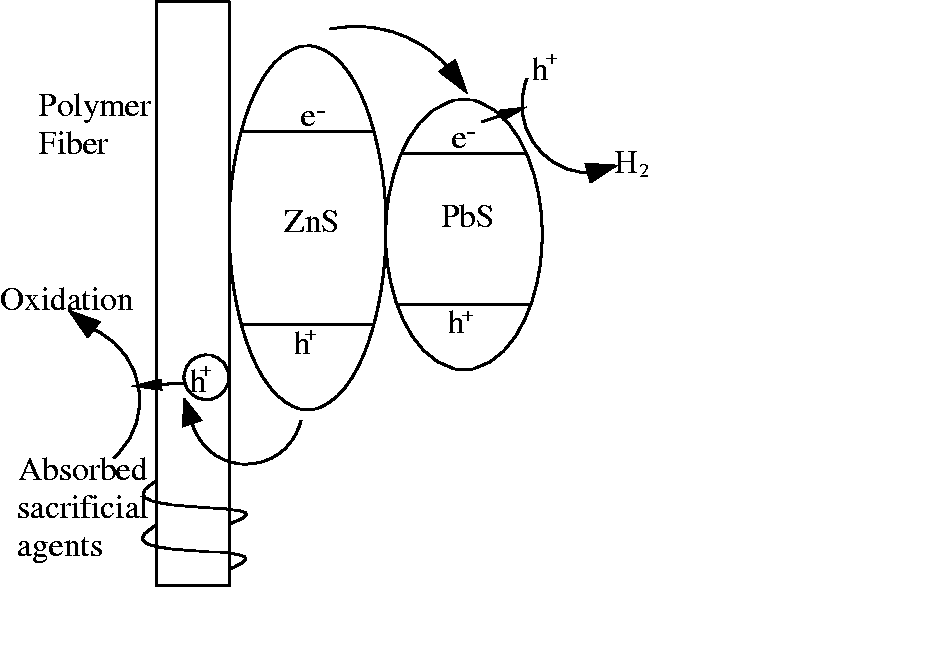
Structure and catalytic mechanism diagram of PbS-ZnS/electrospun fiber composites.
Conclusion
(1) The PbS-ZnS/electrospun fiber composites with uniform distribution are loaded on the surface of the flanged electrospun fibers by hydrothermal synthesis. (2) The UV-Vis absorption spectra of PbS-ZnS/electrospun fiber composites show that the materials have good absorption under visible light, and electrospun fiber absorption peak in the mercury lamp-simulated sunlight catalytic hydrogen from water does not affect the light absorption of the composite. (3) In the process of photocatalytic hydrogen production, PbS-ZnS/electrospun fiber composite materials have higher stability and activity relative to the powder catalyst, and the average hydrogen production capacity is 10.45 mL every 2 h.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Natural Science Foundation of Jiangsu Educational committee (no. 18KJD480001) and (no. YZGXYJS2018-KYCX-012).
