Abstract
Enzymatically-generated hydrogen peroxide (H2O2) using glucose oxidase (GOX) is a sustainable alternative for cotton textile bleaching. GOX catalyzes the oxidation of glucose to gluconic acid with generation of H2O2 as a side product for bleaching. This work involves the study of cotton fabric bleaching with enzymatically-generated H2O2. Enzyme and glucose concentrations significantly affect peroxide generation as well as bleaching and were optimized using statistical experimental design in reference to maximum whiteness. Furthermore, the sustainability of this bleaching system was premeditated by GOX immobilization. GOX was covalently immobilized onto a natural and inexpensive support (chitosan) based on statistical experimental design results. The immobilization improved whitening results as well as allowing repeated application of enzyme for H2O2 generation, which was successively implemented for cotton textile bleaching.
Introduction
Textile bleaching removes natural pigments present in fibers that discolor the substrate. Traditionally, whitening was achieved with chlorine- and oxygen-containing oxidizing agents. In the last few decades, hydrogen peroxide (H2O2) has been accepted as a universal bleaching agent because it is a more environmentally-friendly (e.g., it can decompose into oxygen and water) and is a versatile application processes. 1 Enzymatically, the discoloration of natural impurities can be achieved using laccase/mediator systems, peroxidases, and in situ generated peroxide or peracids by glucose oxidases (GOX) for cotton-based textiles. 2 The use of GOX for producing H2O2 can be a sustainable method for bio-bleaching. GOX enzymes are electron-transfer oxidases, which catalyze the oxidation of glucose to glucolactone in the presence of oxygen, and in turn spontaneously yield gluconic acid as well as H2O2 as a byproduct for bleaching purposes.3-5
Generation of peroxide with GOX requires slightly acidic to neutral conditions at low temperatures; the bleaching effect of H2O2 under these conditions is insignificant. High temperature (80-90 °C) and alkaline (pH 11) conditions for bleaching provide efficient results,6-10 and the assistance of ultrasound energy also improves fabric whiteness due to increased enzyme reaction. 11 Several researchers investigated the activation of generated H2O2 with different activators such as Prestogen SP, 3 peroxidases, 12 and peracid precursors9,13-16 to enable bleaching at low temperature in neutral media. Integration of pre-treatment with GOX enzyme and dyeing was also studied by researchers to minimize energy consumption.17,18
The important parameters for H2O2 generation with GOX are pH, temperature, and sufficient aeration.19-21 A considerable increase in H2O2 generation was observed by aeration rather than by mechanical agitation.7,10 In preliminary trials, we also observed that the concentration of GOX, amount of glucose, and the incubation time were the prime factors for peroxide generation,7,8,21 while maximum H2O2 generation did not ensure maximum whiteness from the bleaching bath. In 2017, Reis and coworkers optimized the peroxide generation and bleaching processes separately for one-step pre-treatment of knitted cotton textiles using GOX enzyme. 22 The focus on parameter optimization, however, should be directed towards the bleaching process. Therefore, in the present study, we adopted experimental design to optimize parameters for whiteness instead of H2O2 generation.
The industrial application of this system was quite limited due to the enzyme expense. In addition, a higher quantity of enzymatically-produced peroxide was needed to accomplish a bleaching equivalent to that of conventional bleaching. Immobilization of the GOX enzyme, however, can overcome the problems associated with use of the native enzyme and can ensure long-term application due to easy recovery and recycling. The first textile application using immobilized GOX was reported in 1999 by Opwis, with cyanuric chloride-mediated attachment to cotton—with subsequent release of H2O2 at levels high enough to support adequate bleaching. 23 In other research, GOX was covalently immobilized on two inexpensive and commercially-available supports, viz. alumina and glass. High protein immobilization yields, as well as sufficient bleaching levels were attained. Glass permitted a higher generation rate of H2O2 (0.35 g/L) due to its porous morphology and thus bound more protein; however, alumina produced a higher operational stability and permitted reuse. 24
The support material plays a very significant role in the performance of immobilized enzymes. Stability under working conditions, availability at low cost, and adequately large surface area, together with the least diffusion limitations, are the important criteria for selecting support materials for industrial applications. 25 In previous studies,23,24,26 the supports used were hydrophobic and poorly diffuse, whereas, in the present work, GOX was immobilized on a natural, inexpensive, biocompatible, and hydrophilic support material: chitosan. Experimental design was used to optimize the process of immobilization for maximum conjugate activity, with Fourier transform infrared (FTIR) spectroscopy and enzyme assays confirming bond formation. The H2O2 generated with native, as well as immobilized, GOX were successively used for cotton fabric bleaching. The reusability of immobilized enzyme was studied for five consecutive cycles. The purpose of this study was to establish GOX- H2O2 cotton fabric bleaching for industrial application.
Experimental
Materials
Cotton fabric (plain weave, 172 g/m2, epi×ppi: 65 × 56, warp and weft count: 30 Ne), Gluzyme Mono 10000 BG (10,000 glucose oxidase units (GODU)/g, Novozymes), D(+)-Glucose (Merck), water soluble chitosan (chitosan chloride, India Sea Food Ltd.), and glutaraldehyde (25%, Thomas Baker) were used. All other chemicals used were of analytical grade and were procured from SD Fine Chemicals Ltd., India. Gluzyme Mono 10000 BG (Aspergillus oryzae) is a commercially-available product of glucose oxidase (GOX) and a cost-effective alternative to GOX from Aspergillus niger.
H2O2 Generation Using GOX
H2O2 was generated by means of the GOX enzyme using glucose as the substrate. The enzyme was added in small doses to counteract the time dependent deactivation of the enzyme and a buffer was used for pH control to avoid enzyme deactivation caused by decreased pH.5,23 The medium (50 mL) was prepared in Erlenmeyer flasks using a buffer. The experiments were carried out with constant aeration at 5 L/min from a pressurized oxygen cylinder through a glass tube introduced into the solution and mechanical agitation of 100 rpm.
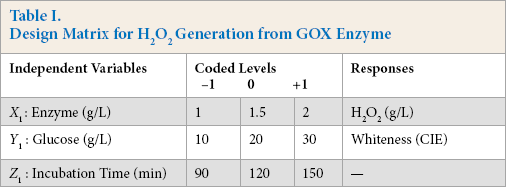
Preliminary experiments were performed at optimal temperature and pH for maximum generation of H2O2 from enzyme. The final optimization to achieve maximum whiteness on cotton fabric was determined using statistical experimental design. The design variables were chosen that potentially affected H2O2 generation as well as whiteness, and the range of each variable was selected based on primary optimization. The experimental combination and the levels of these variables in coded and actual values are illustrated in Table I.
Design Matrix for H2O2 Generation from GOX Enzyme
Determination of H2O2 Concentration
The concentration of H2O2 was determined by potassium permanganate titration in acidic medium using AATCC TM102-2002. 27
Bleaching Process
For conventional bleaching, the desized and scoured samples were bleached at a material to liquor ratio (LR) of 1:20 with varying H2O2 concentrations (2, 4, 6, 8, and 10% owf), 5 g/L of sodium hydroxide, and 2 g/L of sodium silicate at pH 11, various temperatures (75, 85, 95, and 105 °C), and various times (30, 60, 90, and 120 min).
For enzymatic bleaching, samples were placed in the GOX- H2O2 bath and the bath was activated by raising the temperature to 85 °C and pH 11 using NaOH. The gluconic acid produced with the oxidation of glucose acted as a chelating agent for metal ions, thus, the use of additional stabilizers was not required in the bleaching bath.7,24,28 The bleaching experiments were carried out in an Innolab water bath shaker. Continuous stirring was performed with a glass rod to ensure uniform bleaching. After the requisite time, the samples were washed with hot and cold water, and finally dried.
Fabric Whiteness
The whiteness of the bleached samples was measured using a Premier Colorscan SS 5100A according to AATCC TM110-2005 and represented in CIE. 27 All determinations were performed in triplicate and the result represented as the mean value.
Immobilization of GOX Enzyme on Chitosan Beads
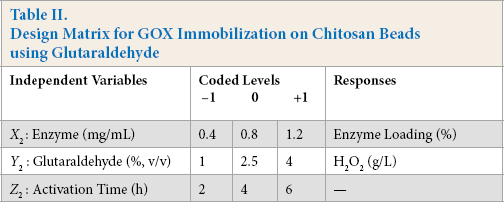
Chitosan beads were prepared from water soluble chito-san.29-31 These beads (5 g) were activated with aqueous glutaraldehyde solution (% v/v) at room temperature (RT) under mild stirring. GOX immobilization was performed in two steps using glutaraldehyde as a cross-linking agent (Scheme 1). 32 Following the activation for given time, the beads were washed with sodium acetate buffer to remove unbound glutaraldehyde. The activated chitosan beads were then immobilized with different concentrations of GOX enzyme at RT for 12 h under mild stirring. The effect of immobilization parameters were studied using experimental design (Table II).
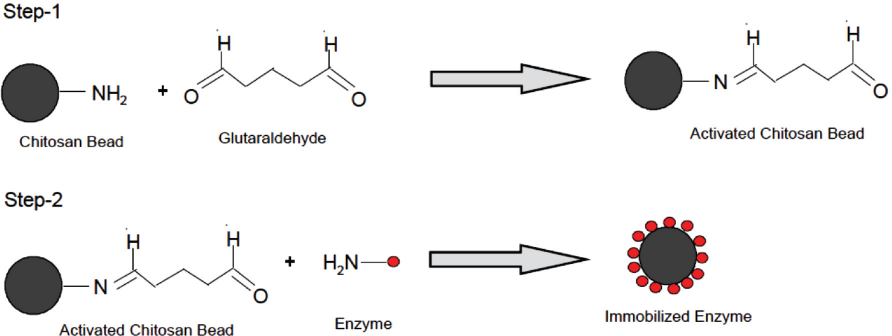
Immobilization of GOX on chitosan beads using glutaraldehyde.
Design Matrix for GOX Immobilization on Chitosan Beads using Glutaraldehyde
The immobilized beads were filtered and washed to eliminate unbound protein and the enzyme loading percentage was measured for each run. The conjugate activity of immobilized GOX was evaluated in terms of H2O2 generation with reaction of glucose as described earlier.
The Fourier transform infrared-attenuated total reflection (FTIR-ATR) spectra of chitosan beads, glutaraldehyde activated beads, and immobilized beads were recorded between 4000 and 400 cm−1 using a Brucker Eco Alpha FTIR spectrophotometer.
Enzyme Loading (Protein Assay)
The amount of protein in the solution was determined according to the Bradford method, using bovine serum albumin (BSA) as the protein standard. The amount of immobilized enzyme was calculated from the protein content in the solution before and after immobilization.
Results and Discussion
Conventional H2O2 Bleaching
In initial trials, it was observed that the high concentration of oxidant and elevated temperatures resulted in a significant fabric strength loss. Keeping the strength loss to 10-15% as a reference, 33-35 the conventional bleaching parameters were set to 4% (owf) or 0.5 g/L H2O2 at 85 °C for 60 min to obtain a sufficient CIE Whiteness Index value of 60.
H2O2 Generation using GOX
Initial Parameter Selection
GOX enzymes from different strains are active under different conditions. The recommended range for the provided GOX was 30 to 50 °C within a pH of 4 to 6. Initial experiments were carried out using 2 g/L of enzyme and 20 g/L glucose at 25, 40, and 55 °C at pH 4.5, 5, 5.5, and 6 to obtain maximum generation of H2O2 (Fig. 1).
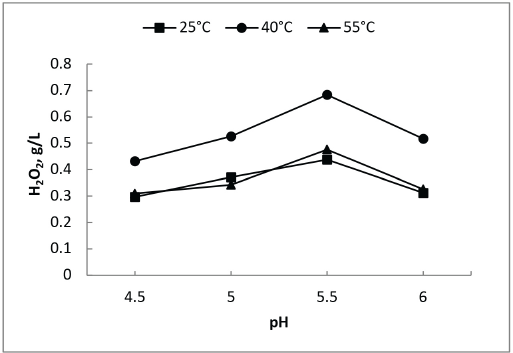
Optimum pH and temperature for H2O2 generation with GOX enzyme.
GOX generates H2O2 efficiently at all temperatures. A temperature of 40 °C was chosen for further investigation because it gave the greatest H2O2 generation. Reduction of H2O2 production at higher temperature may be due to enzyme denaturation.25,26 Irrespective of temperature, maximum generation of H2O2 was observed at pH 5.5 in the above experiments. For all further experiments, the temperature and pH were kept constant at these values.
Primary Optimization
For preliminary studies, H2O2 was generated by varying concentrations of GOX enzyme using different glucose amounts and bleaching experiments were performed using the peroxide. Peroxide generation increased with enzyme concentration (0.5 to 2 g/L) as shown in Fig. 2a, which consecutively resulted in increased whiteness (Fig. 2b). The difference in peroxide generation and whiteness was very insignificant for 2 g/L and 2.5 g/L GOX concentrations. For the GOX concentration of 3 g/L, decreased whiteness was observed even with greater peroxide generation. The whiteness obtained with 0.5 g/L of GOX was insufficient, therefore, GOX was used at concentrations ranging from 1 to 2.5 g/L during further experiments.
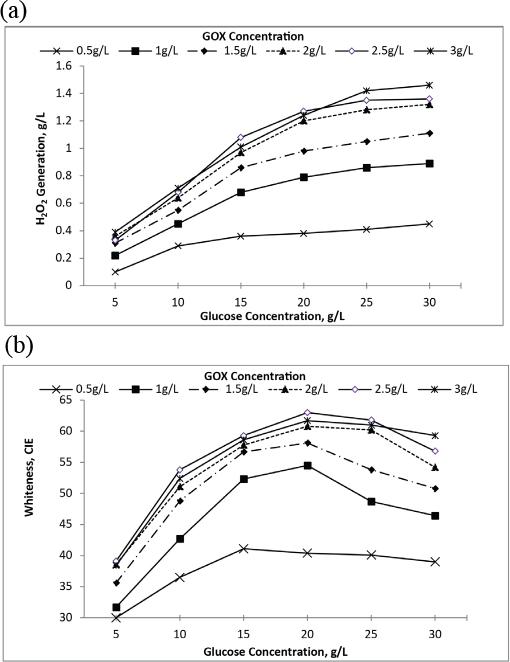
(a) H2O2 generation at various GOX concentrations using varying amounts of glucose and (b) whiteness obtained at various GOX concentrations using varying amounts of glucose.
Invariably, H2O2 generation increases with glucose content at all enzyme concentrations. For each enzyme concentration, after a particular concentration of glucose was reached, an insignificant change in H2O2 generation was observed (Fig. 2a). Whiteness also showed a similar trend with the glucose concentration. Hence, an optimal amount of enzyme and glucose was required for bleaching, beyond which no additional improvement was observed.
Final Optimization
Finally, to optimize enzymatic bleaching using GOX enzyme, experimental design was used (Table III). Statistical analysis of design was performed using a trial version of Design Expert-10.0.3. Results show that the enzyme and glucose concentrations, and time, favored H2O2 generation, with enzyme concentration having the most significant effect, followed by time of incubation and glucose concentration (Table IV).
Results of Cotton Fabric Bleaching using H2O2 Generated from GOX Enzyme
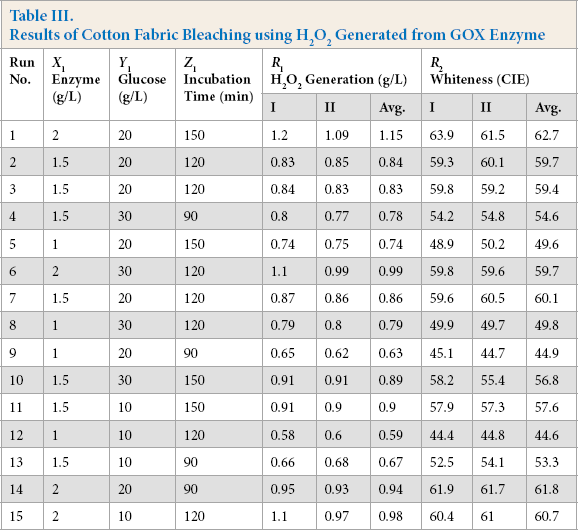
Statistical Analysis of Results for Cotton Fabric Bleaching
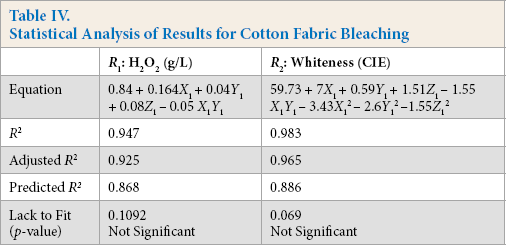
The response surface obtained for significant interaction factors is shown in Fig. 3. H2O2 generation increased with increased GOX and glucose concentrations. The higher concentration of reactants favored the reaction, resulting in more peroxide generation, while the higher concentration of GOX in the bleaching bath necessitated high concentrations of glucose. The increase in incubation time also favored H2O2 generation and the higher GOX and glucose concentrations required prolonged incubation times for reaction completion.
Effect of GOX enzyme and glucose concentration on (a) peroxide generation and (b) cotton fabric whiteness.
Similarly, statistical analysis for whiteness shows that the coefficients for quadratic terms of enzyme, glucose, and time were very significant (i.e., these factors had very large effects on the bleaching). The linear effect of enzyme and time was also significant, whereas for glucose it was very small (Table IV). For interaction effects, only the interaction between enzyme and glucose was significant.
The response surface for whiteness (Fig. 3) indicates that bleaching increased with GOX enzyme and glucose concentrations. Whiteness values reached 60 (CIE) when increasing the enzyme concentration from 1 to 2 g/L with 10-15 g/L of glucose. Similar levels of whiteness were obtained easily with lower enzyme concentrations by increasing the glucose amount. But, at high concentrations of glucose (30 g/L) and enzyme (2.5 g/L), whiteness decreased even with increased H2O2 generation. This may be due to increased gluconic acid formation or residual glucose in the bath, which may significantly affect bleaching.
For enzymatic bleaching, a higher quantity of enzymatically-produced H2O2 was required to achieve the bleaching effect equivalent to that of conventional methods. In conventional bleaching, a minimum concentration of 0.5 g/L of H2O2 was required to achieve a whiteness (CIE) value of 60 while ∼0.8-0.85 g/L of enzymatically-produced hydrogen peroxide provided equivalent bleaching results. This might be due to the presence of glucose and enzyme in the bleaching bath, resulting in lower whiteness values. To study the impact of their presence in the bleaching bath, two separate series of experiments were performed.
In one series, 2-10 g/L of glucose was added in the bleaching bath containing 2 g/L of GOX enzyme and 0.6 g/L of H2O2. Each bath was used for bleaching cotton samples and the consumption of H2O2 was measured (Fig. 4). In absence of glucose, the maximum consumption of H2O2 was observed with lower bleaching results due to rapid decomposition of H2O2 (as no gluconic acid was produced). The graph indicates that the presence of glucose had a stabilizing effect on H2O2 and resulted in improved whiteness up to a certain concentration of glucose. Higher concentrations of glucose over-stabilized the bleaching bath and resulted in decreased H2O2 consumption. Therefore, the reduction of the bleaching power of peroxide was reflected in decreased whiteness.
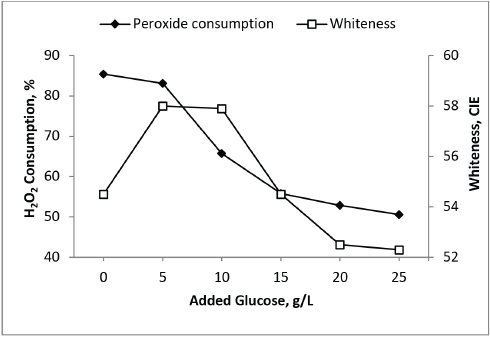
Impact of glucose in cotton fabric bleaching bath.
In the second series, the interaction of protein in the bleach bath was studied with addition of 0.5-2.5 g/L GOX in the bath containing 0.6 g/L H2O2. The enzyme also had a stabilizing effect on the generated peroxide, resulting in reduced whiteness values. As the protein concentration increased in the bleaching bath, the H2O2 consumption decreased and resulted in lower whiteness values (Fig. 5). Both of these experiments confirm that the presence of excess glucose and enzyme in the bleaching bath resulted in reduced whiteness values due to over-stabilization of H2O2 (Fig. 6). The higher concentrations of both glucose and GOX favored H2O2 generation, but were unfavorable for whiteness.
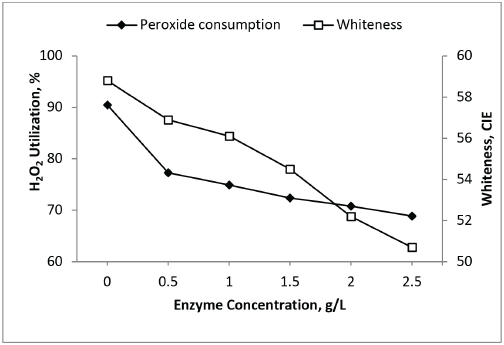
Impact of enzyme in cotton fabric bleaching bath.
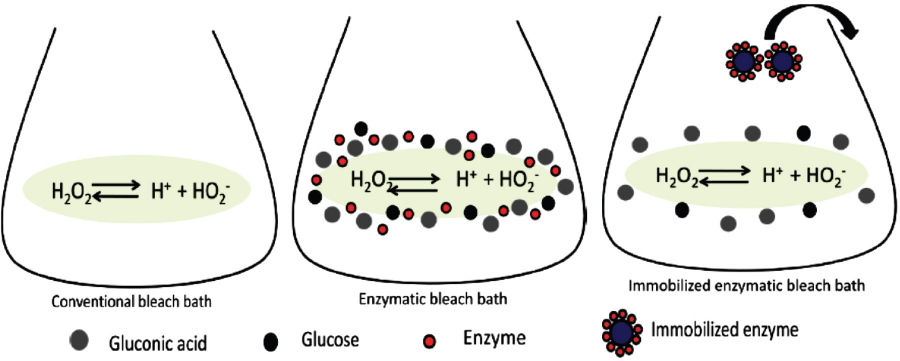
Comparative bleaching bath composition depicting over-stabilization of peroxide.
Optimal Bleaching Conditions using GOX Enzyme
To obtain the optimal values of parameters with respect to achieving the maximum whiteness value R2, numerical analysis was performed with respect to the desirability function (DF1) in Eq. 1.
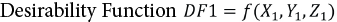
X1 is the enzyme concentration (g/L) set in range, Y1 is the glucose concentration (g/L) set to the minimal value, Z1 is the incubation time (min) set in the 90-150 min range, and R1 is the H2O2 concentration set in the 0.8-0.9 g/L range.
The H2O2 generation range was set based on the preliminary trials that indicated the concentrations of enzymatically-generated H2O2 required to achieve sufficient whiteness values (60) when compared to conventional bleaching. Since controlling the cost of chemicals is paramount for industrial applications, these were kept to a minimum. Numerical optimization of the design gave the following results: 1.8 g/L of GOX with 12 g/L of glucose with 110 min of incubation time satisfied the function DF1 at a desirability of 1. The results obtained experimentally for H2O2 generation and whiteness values also validated the model at a significance level of 95%. Finally, these optimized reaction conditions were used for bleaching using H2O2 generated with free and immobilized GOX.
GOX Immobilization
GOX was covalently immobilized on chitosan beads using glutaraldehyde as a crosslinking agent. In initial trials, it was observed that the enzyme amount, concentration of glutaral-dehyde, and time of incubation were influencing factors for immobilization. Higher enzyme loading did not give higher conjugate activity. Hence to optimize these parameters, statistical design was employed as per experiments in Table V and the loading percentage was evaluated for each run along with the GOX conjugate activity.
Results of GOX Immobilization at Varying Parameters
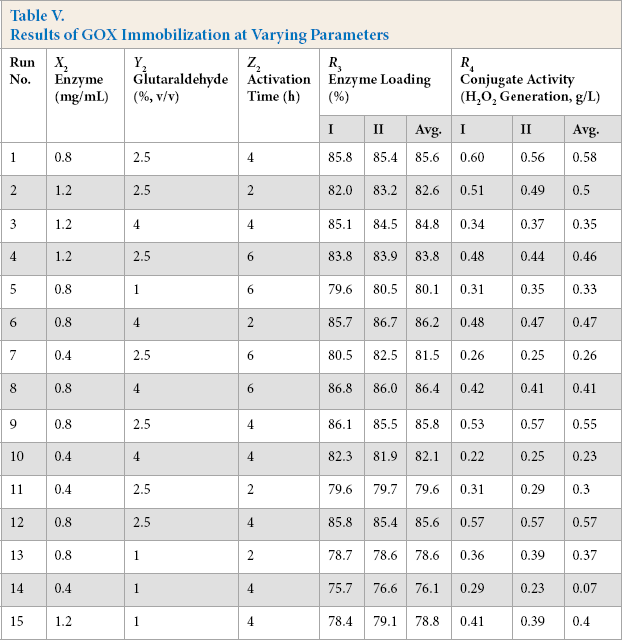
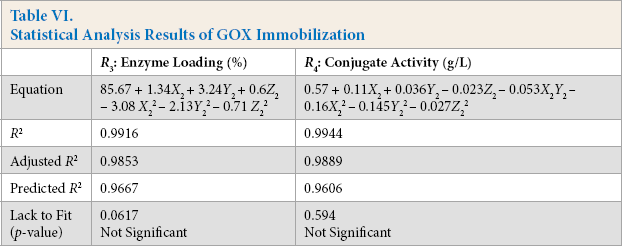
Furthermore, the design was analyzed to obtain a statistical model for maximal GOX conjugate activity. The analysis shows that the concentration of enzyme, glutaraldehyde, and time significantly affect GOX immobilization for a statistical model at a 95% confidence limit. According to estimated coefficients, the proposed model is shown in Table VI. It is apparent from the coefficients that the GOX enzyme and glutaraldehyde concentrations were acting in favor of conjugate activity, while their quadratic terms negatively influenced the activity. The interaction factors were also significant, but they negatively influenced the activity. The linear enzyme concentration was the major influencing factor for conjugate activity. The value of correlation coefficient R 2 and the non-significant lack to ft the p-value validated the predicted model. Moreover, a small difference between the predicted R 2 and the adjusted R 2 further supported the regression model (Table VI).
Statistical Analysis Results of GOX Immobilization
The response surfaces (Figs. 7a and b) indicate that conjugate activity increased with GOX enzyme concentration from 0.4 to 0.9 mg/mL while, for concentrations higher than 0.9 mg/mL, it became constant. Initially, the activity increased in accordance with the amount of protein bound, while higher enzyme loadings on the support did not give higher activity. This could be explained by the increase in enzyme concentrations favoring bond formation and by the increase of enzyme loading over the support. The higher concentrations of enzyme can cause aggregation/clustering over the support, which result in steric hindrance and reduced conjugate activity even after high enzyme loadings. 29 Maximal conjugate activity was observed for glutaraldehyde concentrations ranging from 2.2 to 3.4%. Increased glutaraldehyde concentrations undoubtedly increased protein loading, but resulted in multipoint attachment, reducing the activity. 32 Similarly, longer cross-linking times increased the conjugate activity, with 4 h being sufficient for maximum activity.
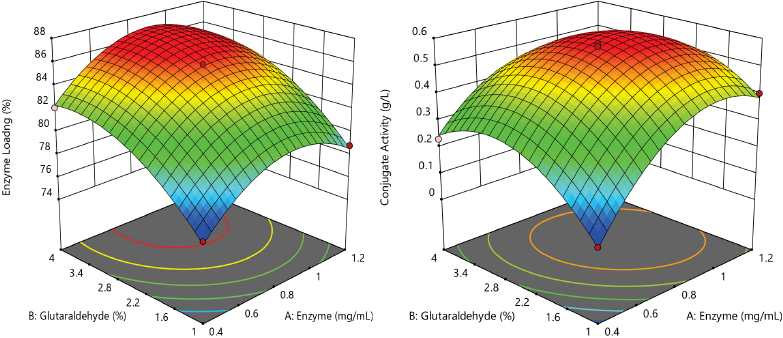
Effect of GOX enzyme and glucose concentration on (a) loading percent and (b) conjugate activity.
Optimal GOX Immobilization Conditions
To obtain the optimal parameter values for immobilization with respect to the maximum conjugate activity R4, numerical analysis was performed with respect to the desirability function (DF2) in Eq. 2.
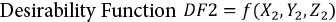
X2 is the GOX concentration (mg/mL) set in the 0.4 to 1.2 mg/mL range, Y2 is the glutaraldehyde (%, v/v) concentration set in the 1 to 4% range, Z2 is the crosslinking time (h) set in the 2 to 6 h range, and R3 is the enzyme loading (%).
Numerical analysis was used for parameter optimization and the values of 0.95 mg/mL GOX enzyme with 3% glutaralde-hyde and 4 h of crosslinking satisfied the function DF2 with a desirability of 1. The values obtained experimentally for enzyme loading and conjugate activity with these optimized parameters validated the model at a 95% significance level.
Immobilized GOX Characterization
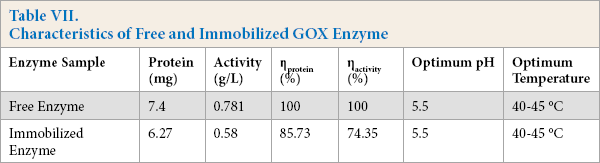
The defined optimal conditions characteristics of free and immobilized GOX were evaluated and are presented in Table VII. GOX activity was reduced after immobilization, while no change in optimum pH and temperature was observed for the immobilized GOX enzyme. This was expected because immobilization normally results in structural rearrangements in enzyme conformation and microenvironment due to the attachment on the polymeric support. Many studies with GOX also showed that the conjugate activity decreased after immobilization while the enzyme stability increased.4,26,36
Characteristics of Free and Immobilized GOX Enzyme
The FTIR spectra of chitosan beads (Fig. 8) show the characteristic absorption peaks at 1588 (-NH2) and 1380 cm−1 (-CH2) bending. The absorption bands at 1150 (anti-symmetric stretching of the C-O-C bridge), 1040, and 985 cm−1 (skeletal vibrations involving C-O stretching) were characteristic of its saccharide structure. The activated and immobilized chitosan beads show a peak at 1644 cm−1 attributed to imine bond (-C=N-) formation from the interaction of glutaral-dehyde and amine groups present in chitosan and enzyme crosslinked with glutaraldehyde. This region of the spectrum contained overlapped peaks corresponding to -NH and -C=N- stretching of the newly-formed Schif's base. The peak corresponding to 1150 cm−1 (-C-O-C bridge) was reduced in intensity due to formation of new bonds. (Fig. 8). The peaks corresponding to 1588 (amine) and 2362 cm−1 (amine stretching) were also reduced after bond formation in this spectrum. Thus, the FTIR results confirmed the covalent immobilization of GOX enzyme on chitosan beads using glutaraldehyde as a crosslinking agent.37-40
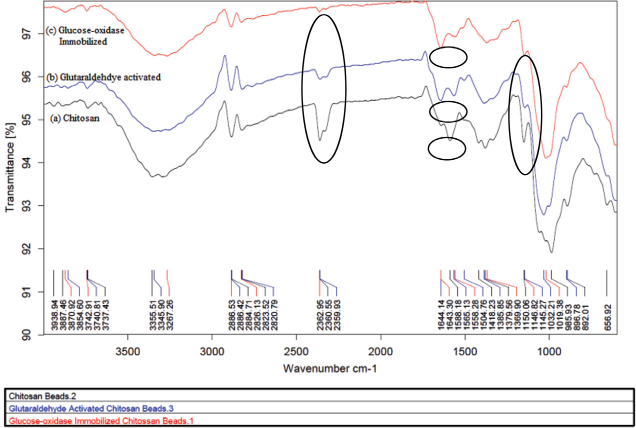
FTIR spectra of (a) chitosan beads, (b) glutaraldehyde activated chitos immobilized GOX on chitosan beads.
H2O2 Generation using Immobilized GOX
H2O2 generation for bleaching was evaluated and compared for both immobilized and free GOX using the same equivalent amount of each. Aliquots of reaction mixtures were separated at various time intervals and H2O2 generation was measured (Fig. 9). The amount of H2O2 generation using immobilized GOX was less than that of free GOX with the equivalent enzyme amount. It was also observed that H2O2 generation also increased with increased incubation time and became constant beyond 180 min with immobilized GOX. For free GOX, an incubation time of 120 min was sufficient for maximal peroxide generation. Immobilization restricted enzyme mobility and reduced its interaction with the reaction media. An additional concentration (20-25%) of immobilized GOX enzyme was required to generate H2O2 equivalent to that of the free GOX enzyme.
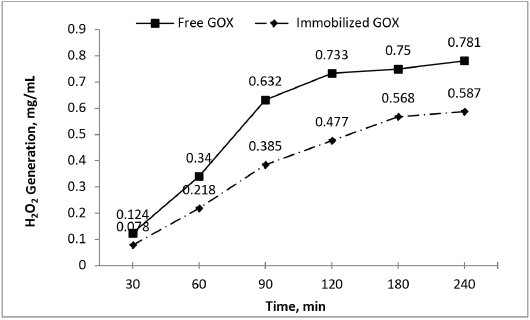
Comparative peroxide generation using free and immobilized GOX enzyme.
Cotton Fabric Bleaching Results
The performance of free and immobilized GOX enzymes for bleaching cotton fabric was compared at equivalent H2O2 concentrations. Results from conventional H2O2 bleaching of cotton fabric were also obtained for benchmarking purposes.
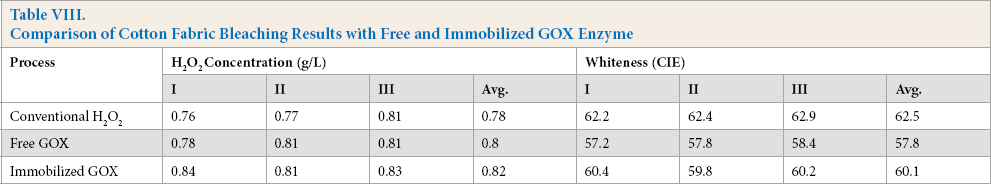
The immobilized GOX was successfully used for in situ H2O2 generation to bleach cotton textiles. Comparable whiteness values were obtained with enzymatically-generated and conventional peroxide (Table VIII). Application of the immobilized GOX enzyme slightly increased whiteness values compared to the free GOX enzyme. This observation was also reported by Tzanov in 2002 with GOX immobilized on alumina. 24 This may due to the reduced interaction of protein with the bleaching liquor as the enzyme is eliminated after H2O2 generation. The comparative bleaching bath compositions of these bleaching methods suggest the overstabilization of H2O2 in the presence of glucose and GOX enzyme as explained earlier. It was concluded that only an additional 10% of the enzymatically-produced H2O2 was required with use of the immobilized GOX, while for the free GOX enzyme, 30% additional H2O2 provided whiteness values equivalent to that of the conventional H2O2 bleaching.
Comparison of Cotton Fabric Bleaching Results with Free and Immobilized GOX Enzyme
Immobilized GOX Enzyme Reuse
As enzyme immobilization allows recovery, the immobilized GOX (7-g beads) was used for H2O2 generation for five consecutive cycles and conjugate activity was measured after each cycle. The activity of immobilized GOX was reduced after each cycle and reached less than 50% for 4th and 5th cycles (Fig. 10a). The peroxide generated from each cycle was used for cotton bleaching and a good whiteness value (>50 CIE) was obtained for up to three consecutive cycles (Fig. 10b). Insufficient peroxide generation due to reduced activity restricted its further application.
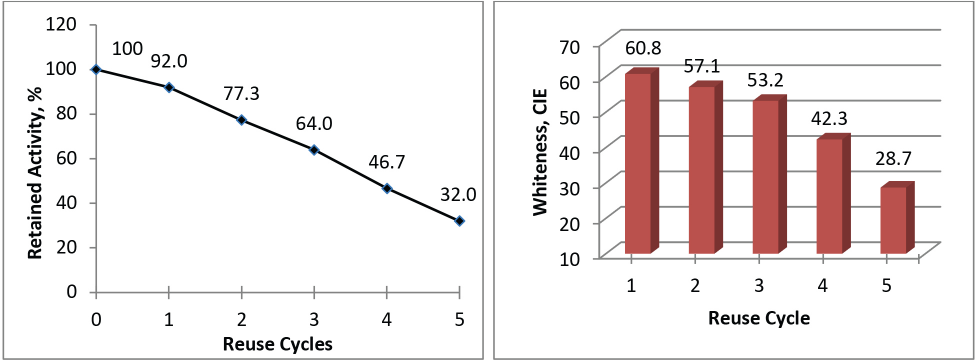
(a) Activity retained and (b) cotton fabric whiteness obtained with immobilized GOX after each use cycle.
Conclusions
Hydrogen peroxide generation using glucose oxidase (GOX) depended significantly on enzyme and glucose concentrations, and time of incubation. Higher concentrations of enzyme and glucose favored hydrogen peroxide (H2O2) generation, but were unsuitable for cotton fabric bleaching due to over-stabilization of the bleaching bath. Statistical experimental design showed that the 1.8 g/L of GOX with 12 g/L of glucose were optimal for H2O2 generation to obtain sufficient whiteness. In conventional bleaching, 0.5 g/L of H2O2 provided a whiteness value of CIE 60 to cotton fabric, while in the enzymatically-generated bleaching bath, due to interactions of glucose and GOX, a higher concentration (0.8-0.85 g/L) of H2O2 was required to achieve the equivalent bleaching results.
The use of immobilized GOX enzyme for H2O2 generation improved the whiteness value and 0.55-0.6 g/L H2O2 provided cotton fabric bleaching results equivalent to that of conventional H2O2 bleaching. Concentrations of enzyme and glutaraldehyde and time of activation were the significant factors for immobilization of GOX on chitosan. Statistical experimental design was used and these results showed that the loading percent increased with the enzyme and glutaral-dehyde concentrations, but the conjugate activity decreased. Use of 0.9 mg/mL of GOX enzyme with 3% glutaraldehyde solution followed by 4 h of activation gave the maximal conjugate activity.
In addition, the immobilized GOX enzyme can be reused for H2O2 generation, whereas the free GOX enzyme was lost after a single use. In this study, appropriate bleaching results were obtained for up to three consecutive uses with immobilized enzyme, although the reduced activity (40% residual activity) restricted their further application. Thus, the industrial feasibility of this process is worth further study, with recycling of immobilized enzyme providing a potential economic benefit, as well as improved cotton fabric bleaching results due to the absence of protein in the bleaching bath.
