Abstract
A novel chlorine-resistant fixing agent DE-2 was designed, synthesized, and mixed for application to yarn-dyed cotton fabrics by the pad-dry-cure process. The properties of fabrics treated with DE-2, AR-200, and CF-2 were studied. The results showed that the chlorine fastness of the fabrics treated with DE-2 reached 3.5. The chlorine fastness was better than that of AR-200 and CF-2. Moreover, formaldehyde emissions from the treated fabrics were lower than 75 mg/L. However, formaldehyde emissions from CF-2 treated fabrics were greater than the standard content. Except for color change in a few CF-2 treated fabrics, other fixing agents had no effect on the fabric. Among them, although the DE-2 presented good comprehensive properties, the washing durability was relatively poor.
Keywords
Introduction
Yarn-dyed cotton fabric 1 that was woven with colored cotton yarn has the advantages of being breathable, comfortable, and beautiful. 2 However, dyes used for cotton yarns, including vat dyes, direct dyes, and reactive dyes, present certain chlorine fastness problems.3,4 When the resulting fabric is washed, the dye on the fabric can react with the active chlorine (generally 0.7 mg/L) in tap water.5,6 Even a small amount of chlorine will make dyes, including direct dyes, cationic dyes, and especially reactive dyes, degrade, which changes its colorfastness, in particular, its chlorine fastness.7,8
Chinese finishing, dyeing, and weaving factories received an increasing number of foreign orders with strict requirements since China's accession to the World Trade Organization (WTO). 9 The United States, the European Union, Japan, and other countries have developed strict standards for color-fastness to chlorine. These standards affect the enterprise's production operations significantly. 10 Therefore, research to improve colorfastness to chlorine of yarn-dyed cotton fabrics is important. In this paper, a novel chlorine-resistant fixing agent DE-2 was synthesized, mixed, and then applied to yarn-dyed cotton fabric. The properties of several commonly-used chlorine fixing agents were examined by comparison.
Experimental Section
Materials
Various specifications of yarn-dyed cotton fabric (dyed warp and dyed weft—warp and weft may be different colors) were purchased from Jialida Textile Co. Ltd. Diethylenetri-amine (DETA), epichlorohydrin (ECH), hydrazine hydrate, potassium dihydrogen phosphate, and glacial acetic acid-were obtained from Shanghai Medicines Group, and the reagents were used as received without further purification. Chlorine-resistant fixing agent AR-200, CF-2, and Emulga-tor OP were obtained from Guangzhou Rihua Chemical Co. Ltd. Cationic film CAM-100 was purchased from Taiwan Juyi Chemical Industry Co. Ltd. Nonionic penetrant WXA was purchased from Huntsman Chemical Co. Ltd. HT333 medium temperature amylase was purchased from Wuhan Xinhua Yang Biological Co. Ltd.
Instrumentation
A Matex 6000 Standard Monforts Fong setting stenter was used (Lixin Dyeing Machinery Co. Ltd). A Type pull-cylin-der machine was obtained from Shaoyang Textile Machinery Co. Ltd. Measurement of fabric color change was carried out using a Datacolor 600 reflection spectrophotometer. A KL-T200 yarn dyeing proofer was purchased from Foshan Kelun Electromechanical Equipment Co. Ltd.
Synthesis of Chlorine Fixing Agent
DETA (30 mL) and the same volume of water were added to a four-necked (100 mL) flask equipped with a magnetic stirrer and a thermometer. The mixture was heated to 80 °C, 30 mL of ECH was added to the flask slowly, and then refluxed and stirred at 80 °C for 3 h. A light yellow clear liquid was obtained. Thereafter, 20 mL of hydrazine hydrate was dropped into the reactor slowly, and then the mixture was kept at 80 °C for 3 h. The reaction mixture was naturally cooled and purified by extraction and rotary evaporation, resulting in isolation of a pale yellow solid (DE). The synthetic reactions to produce DE are shown in Scheme 1.
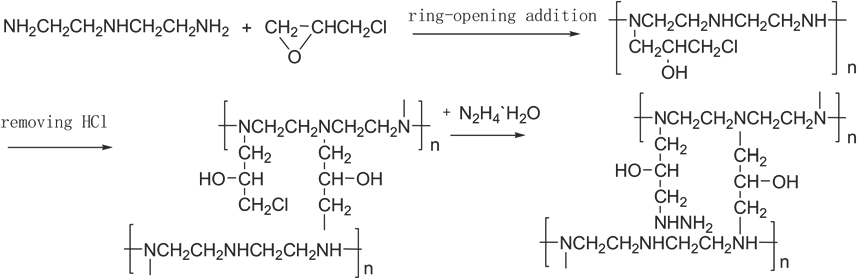
Reaction equations for synthesis of DE.
A 1:1 molar ratio of stearic acid and triethanolamine was added to a four-neck flask containing 30 mL of toluene as solvent and water-carrying agent under a nitrogen atmo-sphere. The mixture was refluxed, 10 mg of p-toluene sulfonic acid was added as catalyst after all the reactants dissolved, and esterification/dehydration at 130∼175 °C was performed for 3 h, then sodium chloroacetate was added with further refluxing for 1 h. The progress of the reaction was monitored by consumption of stearic acid using a titration method. When all of the stearic acid had been consumed, the mixture was allowed to cool. Toluene was removed by rotary evaporation. After that, a milky white or light yellow solid substance R was obtained. The synthesis reactions are shown in Scheme 2.
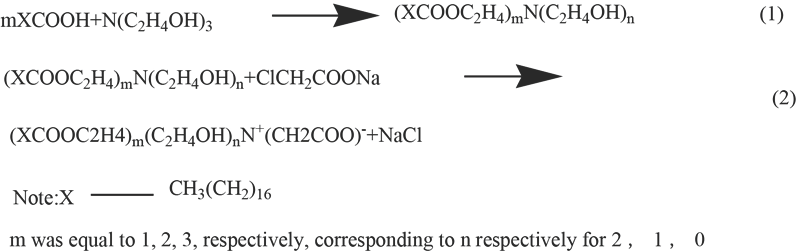
Reaction equation for synthesis of R.
The pale yellow stable emulsion DE-2 was obtained from DE, R, Emulgator OP, and water by high shearing emulsifi-cation 2 h at 40 to 50 °C.
Treatment of Pre-Treated Fabric with Chlorine-Resistant Fixing Agent
Desized cotton yarn-dyed fabric (118 g/m2, 10 × 10 cm) was washed twice with HT333 medium temperature amylase (2 g/L) about 30 min at 60 °C in a pull-cylinder machine. The bath solution was replaced with water and heated to 80 °C for 15 min. The water was drained and the fabric was washed twice again. Subsequently CAM-100 (2 mL/L), the chlorine-resistant fixing agent (1 mL/L), and acetic acid (0.5 mL/L) were added to the fabric sequentially, and the reaction solution was kept at 60 °C for 2 h. Finally, the pull-cylinder machine was drained, dehydrated twice by centrifugation, and opened. The moisture content of the treated fabrics was kept at about 7.5%.
Performance Test
Colorfastness to Chlorine
The colorfastness to chlorine test was performed according to the JIS Test Method L0844-1996, and JIS gray card was used for rating.
Evaluation of Formaldehyde Content
Formaldehyde content was determined by subjecting treated fabrics to JIS Test Method L1041-2000, Test Methods for Resin Finished Textiles.
Color Difference
Color differences of the textile fabrics were evaluated by making color measurements of treated fabrics using a Data-color 600 spectrophotometer.
Washing Durability
According to JIS L0217-1979 103, samples were washed using a household automatic laundering machine at 40 °C using standard reference detergent (KAO, 2 g/L). One wash cycle (10 min) was considered equivalent to one commercial laundering. The chlorine fastness tests were performed after washing and drying.
Results and Discussion
Performance of Chlorine-Resistant Fixing Agents
Brands YPCT24323, YPCT13786, YPCT25421, and YPCT29546 yarn-dyed cotton fabrics were selected and treated with various chlorine-resistant fixing agents. The results are shown in Table I. Chlorine fastness of the fabrics were improved at least half a grade after chlorine-resistant fixing, with one exception. For the same types of treated fabrics, the finishing results of the DE-2 treated fabrics were better than the other fixing agents. For different kinds of treated fabrics, the chlorine resistance of the treated blue (YPCT24323 and YPCT13786) and green fabrics (YPCT29546) were the best, while it was poor on the red fabric (YPCT25421).
Finishing Effects of Various Chlorine-Resistant Fixing Agent Treated Fabrics
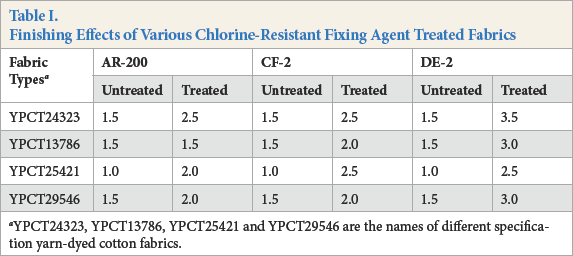
YPCT24323, YPCT13786, YPCT25421 and YPCT29546 are the names of different specification yarn-dyed cotton fabrics.
The DE-2 cationic groups interacted with anionic groups in the dye, forming ionic bonds, and with the remaining cellulose hydroxyls, thus reducing dye destruction by free chlorine. Secondly, the hydrazine hydrate groups in the fixing agents reduced the presence of hypochlorite ions, effectively reducing the presence of active chlorine in water. Additionally, stearic acid and trimethyl ammonium B lactone had good dye shielding effects, effectively prevented the free chlorine from destroying the dye, and improved the chlorine resistance performance of fabric.
Formaldehyde Emissions
Although the fixation effect of early fixing agents based on bisnitrile amine formaldehyde resins were preferable, the treated fabrics would release formaldehyde in storage and use. Non-formaldehyde fixing agents were developed, but the finishing results using these were worse than formaldehyde-containing fixing agents. Moreover, they were mixed with formaldehyde-containing fixing agents to obtain optimal finishing results. Therefore, the fabrics still released formaldehyde after dye fixing.
Formaldehyde has some adverse effects on human health. Thus, the formaldehyde contents of textiles are strictly limited by the European Union and the United States. Therefore, formaldehyde content tests of finished fabrics must be performed. Results in Table II suggest that the formaldehyde emissions of fabrics, treated with various fixing agents and washed, met the JIS L1041-2000 standard. However, the formaldehyde test of CF-2-treated fabrics was unqualified. Fabrics treated with AR-200 and DE-2 met the JIS L1041-2000 standard. The formaldehyde emissions of fabrics treated with DE-2 were quite low. Because of the absence of the hydroxymethyl group in DE-2, formaldehyde could not be produced at high temperature or in a complex environment.
Formaldehyde Content of Variously Treated Samples
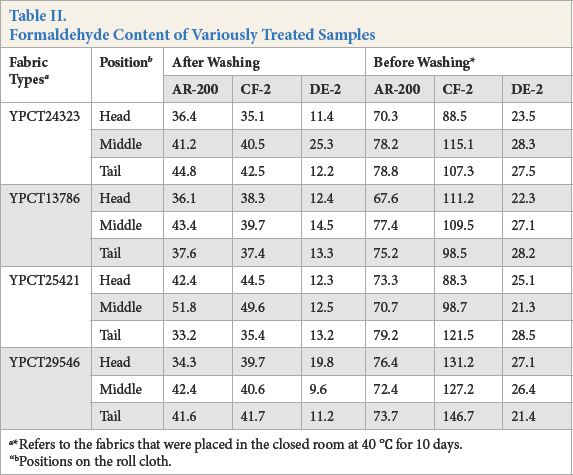
Refers to the fabrics that were placed in the closed room at 40 °C for 10 days.
Positions on the roll cloth.
Effects on Fabric Colors
Many factors influenced the fabric shades during treatment with chlorine-resistant fixing agents, such as agent type, the type of reactive dye(s) used, and the presence of other additives. Quaternary amino, imidazoline-based cationic chlorine-resistant fixing agents are now commonly used. These groups may interact with light, heat, and metal ions, which will change the fabric color. Therefore, to eliminate these adverse effects, the fabric should be treated with softener before fixing. Fabrics were pretreated with CMA-100 softener for chlorine-resistant fixing. The treatment effect of various chlorine-resistant fixing agents on the fabrics is described in Fig. 1. The color shade (CMC ΔEvalue) of untreated and treated fabrics is shown in Table III. Fig. 1 and Table III suggest that the chromatic aberration of fabrics before and after finishing was mostly less than 1, except the YPCT25421 treated with CF-2. However, the chromatic aberration caused by synthetic fixing agent DE-2 was minor. The results show that the color change caused by fixing was effectively avoided by use of the chlorine-resistant fixing agent DE-2.

Color change of fabrics before and after finishing
Color Change of Fabrics Treated with Various Chlorine-Resistant Fixing Agents
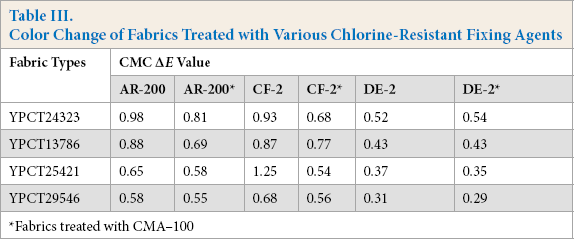
Fabrics treated with CMA–100
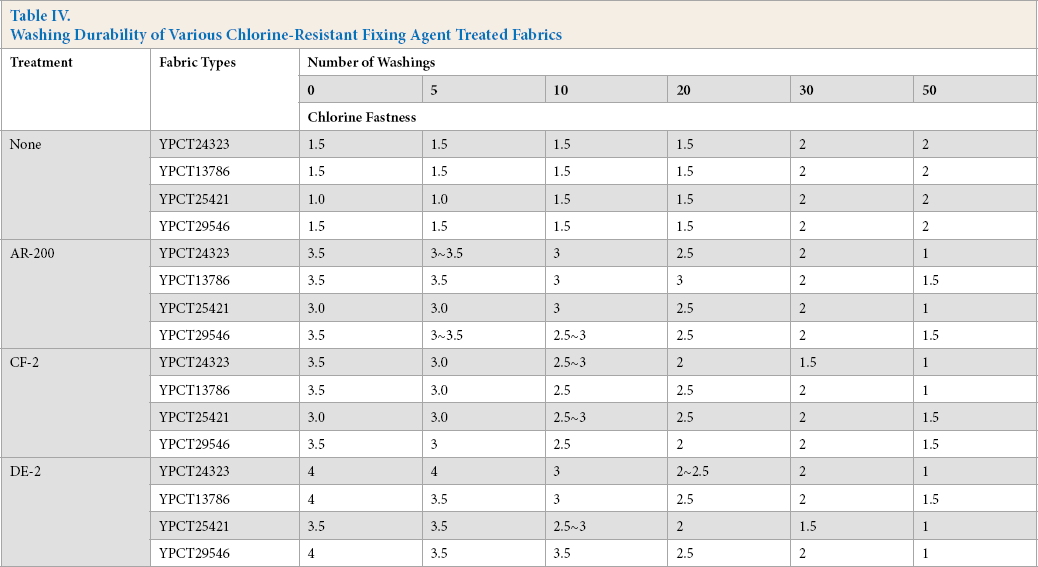
Washing Durability
Washfastness results of chlorine-resistant fixing agent treated fabrics are shown in Table IV. The treated fabric chlorine resistant performance decreased with increased wash cycles. The washing durability of DE-2 treated fabrics was relatively poor. After 30 wash cycles, the chlorine resistance was close to that of the untreated fabrics. This result indicates that the bonding of DE-2 to the fiber and dye was weaker than the other two fixing agents. Moreover, the reticular membrane formed on the fiber surface was not resistant to washing.
Washing Durability of Various Chlorine-Resistant Fixing Agent Treated Fabrics
Conclusion
Yarn-dyed cotton fabrics treated with DE-2 gave chlorine fastness results that improved up to 3.5, better than that of AR-200 and CF-2. This was due to the presence of a strongly reducing hydrazine group in DE-2 that could effectively capture the available chlorine in aqueous solution, thus protecting the dye on the fiber. The formaldehyde test of CF-2-treated fabrics was unqualified. However, treatments with AR-200 and DE-2 met the JIS L1041-2000 standard. Moreover, the R component in DE-2 has a betaine structure and is an environmentally-friendly additive. The formaldehyde test of CF-2- treated fabrics was unqualified; only a few CF-2-treated fabrics color were changed, and other fixing agents had no effect on the yarn-dyed fabric. In conclusion, the washing durability of DE-2-treated fabrics was relatively poor, and the chlorine durability decreased dramatically after 30 washes. Further study is needed.
Footnotes
Acknowledgements
The project was funded by the Innovation Fund Project in Hunan Province colleges and universities (13K107 and 15K030).
