Abstract
The popular use of silver nanoparticles (Ag NPs) in commercial textile products that inhibit odor- and/or infection-causing bacteria has continuously raised concerns about their washing durability. The poor durability not only deteriorates the antibacterial performance, but also results in unwanted leaching of NPs into washing solutions. In this study, we showed how the incorporation location of Ag NPs—interior vs. exterior of cotton fiber—governs their durability against consecutive simulated home launderings. The superior washing durability of interior NPs was confirmed. The Ag losses after 50 laundering cycles for interior and exterior systems were 16% and 63% in water and 24% and 78% in detergent solution, respectively. The cotton fabric containing interior NPs predominantly released ionic Ag, whereas the fabric containing exterior NPs released particulate Ag.
Introduction
The constant emergence of new pathogens (e.g., the current outbreak of SARS-CoV-2 declared by a Public Health Emergency of International Concern by the World Health Organization (WHO) on January 30, 2020) and its worldwide spread, brings a great impact on the textile industry. With strong demand from consumers, the efforts of textile-producing companies and textile research institutes to develop products that destroy bacteria, viruses, and other pathogens are greater than ever. Particularly, effective antimicrobial functions are urged in protective textiles, such as face masks (whose wear is currently mandated by public health officials), medical gowns, and hospital curtains. According to a recent study published by Fortune Business Insights reports, the global antimicrobial textiles market value, which stood at US$9.23 billion in 2019, is forecasted to rise at a compound annual growth rate of 5.3% between 2020 and 2027. 1
The most widely used antimicrobial technologies are based on surface coatings that reduce the number of microorganisms by contact and inhibit their activity.2-4 This is because surface coatings are cost-effective and less disruptive to conventional manufacturing processes. Numerous chemicals inactivate bacteria and viruses, but many of them are impractical for coating onto textiles. Textiles are often in direct and continuous contact with human skin, as well as routinely wet and washed; therefore, appropriate antibacterial agents are limited.
With the advances of nanotechnology, some metallic nanoparticles, such as silver and copper, have emerged as a new class of reliable antimicrobial agents. In particular, silver nanoparticles (Ag NPs), due to their potent antibacterial activity, have been vastly adopted by the textile industry in the past decade. Ag NPs act as a source of Ag ions, which are known to deactivate cellular enzymes by interacting with thiol groups,5,6 as well as disable the replication ability of DNA. 7 Under oxic conditions, Ag NPs release Ag ions through desorption from NP surfaces or by oxidative disso-lution. 8 Additionally Ag NPs can physically damage the cell membrane9,10 and generate reactive oxygen species, which induce oxidative stress to bacterial cells.11,12 The popularity of Ag NPs is reflected by the wide variety of Ag NP-coated clothing and apparel available in the market (e.g., socks, T-shirts, activewear, and underwear that are odor-inhibiting and/or anti-infective).
The broad application of Ag NPs in textile products has raised concerns about their leaching during washing, leading to extensive studies on commercial Ag NP-treated textile products.13-16 Their results show that a significant amount of NPs were leached into washing solutions. For example, in the case of socks, up to 25% of the total applied Ag was lost after one machine wash in a detergent solution,17,18 and about 1% to almost 100% after four sequential 24 h gentle agitations in distilled water. 19 For shirts (including an activewear shirt) and trousers, 17%, 20%, 21%, and 18% of the applied Ag were leached by one machine washing in a detergent solution 17 and 2% loss by a 1 h agitation in tap water were reported. 20 Medical textiles (masks and cloth) lost a large amount of Ag (i.e., 23%-27% by weight), and released 11-46 μg of Ag per product mass into the tap water after one 1 h agitation. 20
Here, we show how the incorporation location of Ag NPs—in the interior vs. exterior of cotton fiber—influences Ag release behavior during consecutive washings. The interior NPs were produced by the in situ synthesis of Ag NPs inside the cotton fiber. 21 The exterior NPs were produced by coating Ag NPs on the surface of cotton fabric. The incorporation morphologies were verified by the transmission electron micrographs of the cotton fiber cross-section. To compare the washing durability, the washing test simulating home laundering conditions was conducted up to 50 laundering cycles. With incrementing the laundering cycle, the washed cotton fabrics and washing solutions were collected and analyzed using UV/Vis spectroscopy and inductively coupled plasma mass spec-troscopy (ICP-MS). Ultrafiltration was used to determine the size fractionation of Ag, and an ion-selective electrode (ISE) was used to measure Ag ions in the washing solutions to examine the transformation of Ag. The obtained results demonstrated the critical role of the incorporation method in controlling the Ag release behavior. Interior incorporation as a new effective technology to achieve NP-leach-resistant textiles was discussed.
Experimental
Materials
Bleached and desized woven cotton print cloth (98 g/m2) was purchased from Testfabrics, Inc. (Product No. 400, West Pittston, PA, USA). Mechanically, pre-cleaned raw white cotton fiber was acquired from T. J. Beall (Greenwood, MS, USA). Silver nitrate (AgNO3, 99.9%) was purchased from J. T. Baker (Phillipsburg, NJ, USA). Sodium hydroxide (NaOH, 97%), ammonium hydroxide solution (NH4OH, 28-30%), L-ascorbic acid (C6H8O6, 99%), methyl methacrylate, butyl methacrylate, and methyl ethyl ketone were purchased from Sigma-Aldrich (St. Louis, MO, USA). All materials were used as received. Deionized (DI) water was also used.
Production of Interior Ag NPs
Interior Ag NPs were produced by the in situ synthesis of Ag NPs inside cotton fiber using a previously published method 21 with modification. Briefly, a cotton fabric was immersed in an aqueous solution of NaOH (20 wt%) for 15 min and passed through a laboratory padder (Werner Mathis USA Inc.). The resulting wet pick-up was 200% ± 5%. The alkali-swollen fabric was agitated in an aqueous solution containing AgNO3 (1 mM) and NH4OH (2.5 mM) and then passed through a laboratory padder to yield the wet pick-up of 150% ± 5%. The Ag ion-impregnated cotton was immersed in an aqueous solution of L-ascorbic acid (C6H8O6, 0.1 mM), in which the fabric immediately turned yellow. The resulting interior NP-incorporated fabric was washed and air dried.
Production of Exterior Ag NPs
Exterior Ag NPs were produced by coating pre-synthesized Ag NPs onto a cotton fabric. The Ag colloidal solution was prepared by a previously published method 22 using raw cotton fiber. The unique surface chemistry of the raw cotton allowed the effective formation of Ag NPs not only inside the fiber but also on the surface of the fiber. Ag NPs formed on the fiber surface were collected by agitating the fabric in DI water at 40 ± 1 °C. The obtained dark yellow solution was filtered using Whatman No. 5 filtration paper followed by centrifugation (Ultracentrifuge, Hitachi) at 50,000 rpm for 30 min. The sedimented NPs were redispersed in DI water and diluted to a concentration of 100 mg/L. A cotton fabric was immersed in the obtained Ag colloidal solution for 15 min, passed through a laboratory padder to yield the wet pick-up of 100% ± 5%, and air dried.
Washing Experiment
The washing test was carried out using a Launder-Ometer (M228-AA, SDL Atlas LLC) based on AATCC TM61-2007, whose procedure is equivalent to five home or commercial laundering cycles, with modification. A stainless-steel canister (75 ± 5 mm in diameter, 125 ± 10 mm in height, 550 ± 50 mL) filled with 400 mL of water was preheated to 40 ± 1 °C in the Launder-Ometer. A fabric sample (5 × 5 cm) was placed in a 50-mL polypropylene bottle containing 15 mL of DI water or a detergent solution and 10 glass balls (5 mm in diameter). The detergent solution was prepared as a 0.15% w/v of a commercial laundry detergent (Tide, Procter & Gamble Co.) in DI water. The sealed bottle was placed in the preheated water in the canister and rotated at 40 ± 1 °C at a constant rate of 40 ± 2 rpm for 45 min. After completion of the washing procedure, the washing solution was collected, and the fabric was lightly rinsed with DI water and air dried. Washing in DI water without detergent was also conducted using the same procedure.
Characterization
The optical microscope images of cotton fabrics were obtained using a digital microscope (KH-8700, Hirox). The micrographs of the cotton fiber cross-sections were obtained using a transmission electron microscope (TEM) (JEM-2010, Jeol) operating at 200 kV. Cotton fibers were embedded in a mixture of methyl methacrylate and butyl methacrylate, which was polymerized in an ultraviolet (UV) cross-linker (UVP, CL-1000) for 30 min following published techniques.22,23 A block of the fiber bundle was cut into ∼100 nm-thick slices using a PowerTome Ultramicrotome (Boeckeler Instruments Inc.). The obtained sections were placed on a carbon film-coated copper grid, and the embedding medium was removed using methyl ethyl ketone. Following the acquisition of TEM images, selected area electron diffraction (SAED) analysis was performed using the same accelerating voltage (200 kV). The average size and size distribution of Ag NPs were determined by measuring more than 250 particles using Image J software. 24
The UV/Vis spectra were collected using a UV/Vis/NIR spectrometer (ISR-2600, Shimadzu). A quartz cuvette with a 1-cm optical path length was used for solution samples. An integrating sphere unit was used for cotton samples. Absorbance spectra were collected over a wavelength range of 220-800 nm.
The amount of Ag was determined using ICP-MS in the ICP-MS Metals Laboratory at the University of Utah. For solution samples, a 4-mL solution was dried down and refluxed for 1 h with 2 mL of 16 M nitric acid on a hot plate and dissolved in 1 mL of 0.8 M nitric acid with 10 ppb of indium. For cotton samples, ∼0.05 g of the sample was treated with 2 mL of 16 M nitric acid (trace metal grade) and digested in a Milestone Ethos microwave system. The digest was diluted by weight 1:10, and 10 ppb of indium as an internal standard was added. The digested solution concentrations were determined by an external calibration curve, which was obtained using a silver single element standard (Inorganic Ventures).
The concentration of Ag ions in washing solutions was determined using a silver/sulfide ion-selective electrode (ISE, ISE-8755, Omega Engineering Inc.). Before measurement, a daily calibration curve was established using silver standard solutions. Silver standard solutions were prepared from silver nitrate in the silver ion range from 0.9 to 37 μM in DI water. A total of 1 mL of an ionic strength adjuster, sodium nitrate (20 mM), was added in silver standard solutions and washing solutions. Concentrations of Ag particles less than 1 nm in size were determined using ultrafiltration/ICP-MS. The washing solution was filtrated using a centrifugal unit (Sartorius Stedim Vivaspin6, Sartorius AG) equipped with a poly(ether sulfone) membrane at a molecular weight cutoff of 3 kDa (ca. 1 nm). The filtered solution was analyzed by ICP-MS.
Results and Discussion
Interior and Exterior Ag NPs
Figs, 1a and b show the optical microscope images of cotton fabrics containing interior and exterior Ag NPs, respectively, obtained using different magnifications. The cotton fabric with interior NPs exhibits a noticeable yellow color, indicating the incorporation of a large amount of NPs by the in situ synthesis. The unique optical property of Ag NPs results from surface plasmon resonance (SPR), which is induced by the interaction of conduction electrons of the NPs with the electromagnetic wave of incoming light. It can also be seen that the fabric structure with interior NPs is denser than that with external NPs. This is because the internal synthesis involves an alkaline treatment that swells cotton fibers laterally. The increased yarn thickness is observable in its higher magnification image.

Optical microscope images of cotton fabrics containing (a) interior and (b) exterior Ag NPs taken at different magnifications.
To verify the interior and exterior incorporation morphologies, the cross-section of cotton fiber was examined by TEM. Figs. 2a and b show the TEM images obtained after the in situ synthesis of Ag NPs within the fiber and after coating of Ag NPs onto the fabric, respectively. The insets show the corresponding low-magnification TEM images and indicate the sections taken at high magnification highlighted in red circles. In line with the earlier microscope image, the internal synthesis transformed the bean-shaped cotton fiber into a rounder shape. In coating, a smooth surface is expected to be less favorable for the physical adsorption of NPs as compared with a rough surface. It is also expected that the NPs coated on a smoother surface are more easily washed of than those deposited on a rough surface. The alkaline treatment used in the internal synthesis also caused the rearrangement of cellulose chains from a parallel manner (cellulose Iβ) to an anti-parallel manner (cellulose II). 21 Such conversion increases the number of available hydroxyl groups,25,26 which may have an affinity with Ag ions present on the NP surface. However, these electrostatic interactions are expected to be too weak to alter the washing durability of NPs.
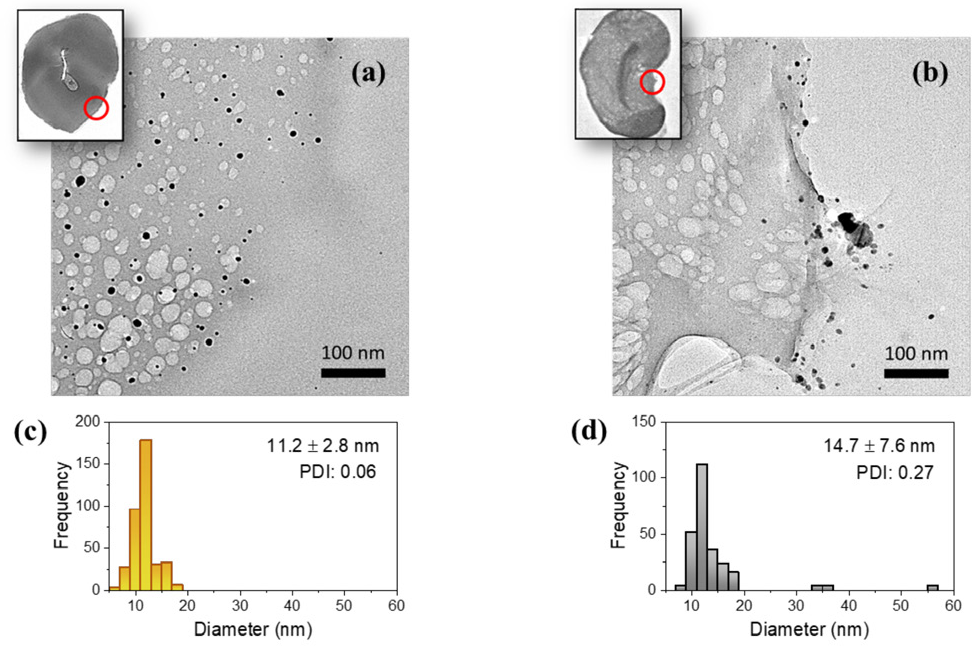
TEM micrographs of the cross-sections of cotton fibers containing (a) interior and (b) exterior Ag NPs. The insets show the low-magnification images and the sections taken at the high magnification highlighted in the red circles. Size histograms are shown for (c) the interior Ag NPs and (d) the exterior Ag NPs.
The high magnification image shows that the in situ synthesis produced Ag NPs in the interior of the fiber. The NPs were uniformly dispersed throughout the cross-section of the fiber. For the Ag NP-coated sample, most NPs were located on the edge of the cross-section of the fiber. It can be seen along the surface that some NPs were aggregated into a large particle. Both interior and exterior NPs were quasi-spherical. Figs. 2c and d show the histograms of the diameters of interior and exterior NPs, respectively. The size of interior NPs was more narrowly distributed, whereas the size of exterior NPs was right-skewed due to the presence of a few large particles. The average diameters of interior and exterior NPs were 11.2 ± 2.8 and 14.7 ± 7.6 nm, respectively. The polydispersity indices of interior and exterior NPs were 0.06 and 0.27, respectively. The larger average size and polydispersity of exterior NPs were attributed to particle aggregation during treatment. Other studies also observed inhomogeneous dispersion and particle aggregation from the bulk solution treatment of NPs. The particles observed on the surface of textiles were up to two orders of magnitude larger than the pristine particles in the colloidal solution of treatment.27-29 Figs. 3a and b show the SAED patterns of interior and exterior NPs obtained from the cross-section of cotton fibers, respectively. Both particles exhibit four concentric rings with intermediate bright dots, which correspond to the (1 1 1), (2 0 0), (2 2 0), and (3 1 1) planes of the faced-centered cubic (fcc) structure of elemental Ag.

SAED patterns of (a) interior and (b) exterior Ag NPs taken from the cross-section of cotton fibers. The concentric rings with intermediate bright dots representing the lattice planes of elemental Ag are highlighted in red.
Fig. 4a shows the UV/Vis absorbance spectra of cotton fabrics containing interior and exterior Ag NPs. These Ag NP-treated cotton fabrics developed a single absorbance peak resulting from the SPR of NPs. The untreated cotton fabric did not show any recognizable peaks over the entire wavelength range studied (not shown here). The SPR peak of interior NPs was centered at ∼427 nm (
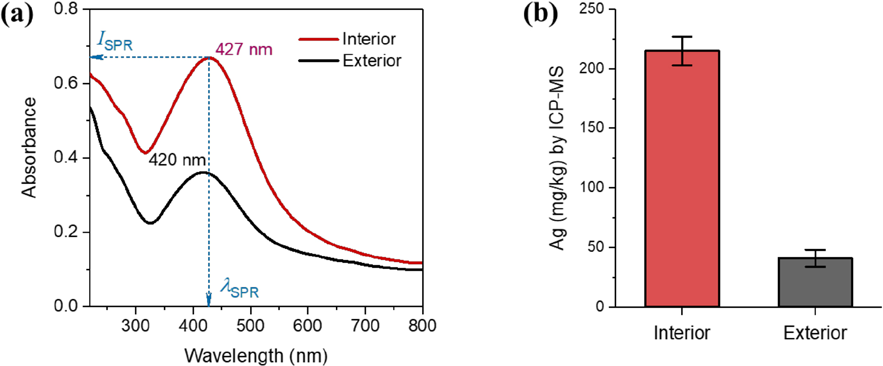
(a) UV/Vis absorbance spectra of cotton fabrics containing interior and exterior Ag NPs. (b) Concentrations of interior and exterior Ag NPs based on the dry weight of the cotton fabric measured by ICP-MS.
Analysis of Washed Cotton Fabrics
The washing durability of interior and exterior NPs was examined by monitoring the changes in the UV/Vis spectra of cotton fabrics. Figs. 5a and b show the percentage
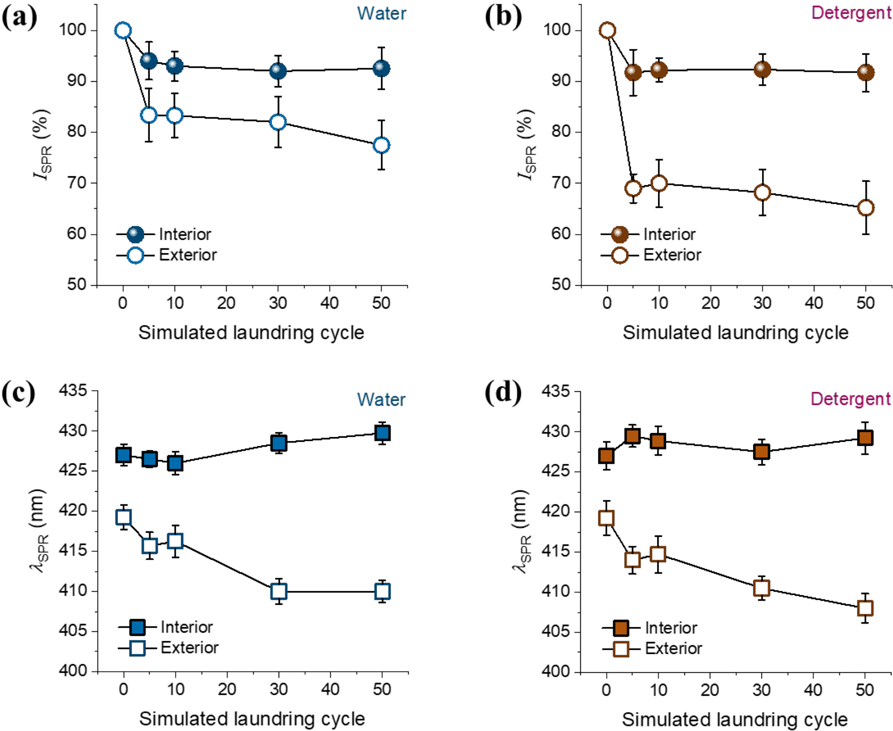
Percentage intensities of surface plasmon resonance (SPR) peaks (
It was also noted that the
The change in the concentration of Ag NPs in the cotton fabric after washing was quantified using ICP-MS. Figs. 6a and b show the percentage of Ag contents remaining (or Ag loss) in the fabrics after incremental simulated home launderings in water and detergent solution, respectively. These results were compared with the
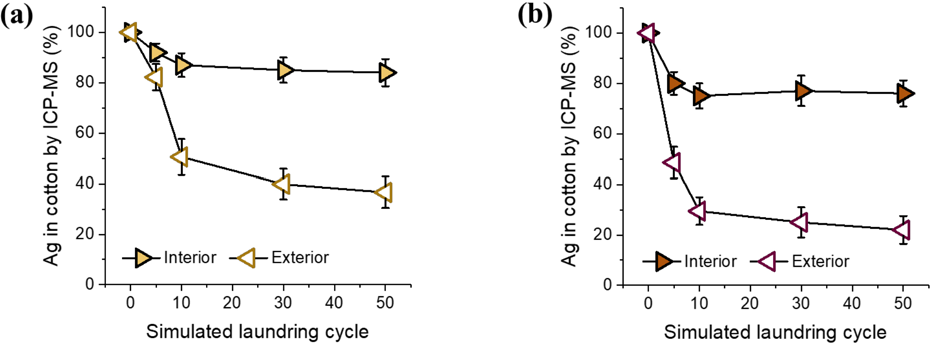
Percentage of Ag remaining in cotton fabrics containing interior and exterior Ag NPs after incremental simulated home launderings in (a) water and (b) detergent solutions.
Analysis of Washing Solutions
The analyses of the washed cotton fabrics indicate that the washing solutions may contain NPs. Washing solutions were collected after incremental laundering cycles and analyzed using various analytical methods. Figs. 7a and b show the stacked UV/Vis absorbance spectra of water obtained after washing cotton fabrics containing interior and exterior Ag NPs, respectively. The washing water for the interior system did not develop any noticeable SPR peaks for all laundering cycles studied. The cotton fabric with interior NPs released Ag NPs below the detection limit of UV/Vis spectroscopy and/or a different form of Ag (i.e., ionic Ag). On the other hand, the spectra for the exterior system clearly showed SPR peaks. The well-defined SPR peak appearing even after the initial washing indicates that exterior NPs were easily detached from the fabric. This strong SPR peak was further developed with increasing laundering cycles.
Figs. 7c and 7d show the spectra of detergent solutions for the interior and exterior incorporations, respectively. Similarly, the absorbance spectra for the interior incorporation, which were identical for all laundering cycles, exhibited no evident SPR peaks. The peak near 260 nm and a shoulder at around 320 nm were from detergents. The absorbance spectra for the exterior incorporation developed SPR peaks with higher intensities, indicating the higher concentrations of Ag NPs in the detergent solution than in water.
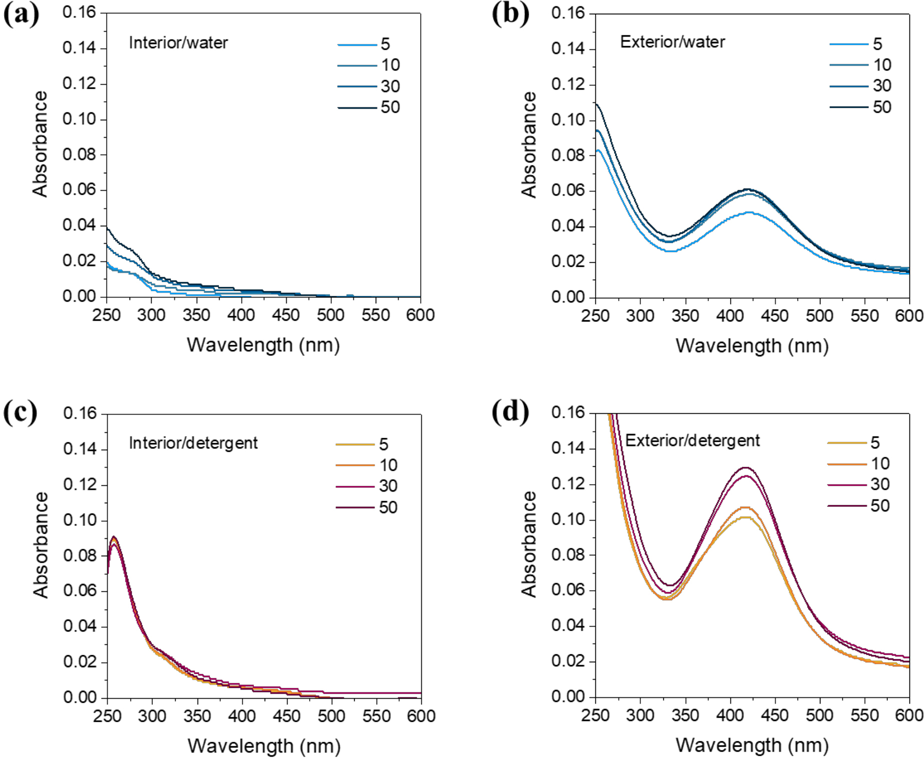
UV/Vis spectra of water collected after incremental simulated home launderings of cotton fabrics containing (a) interior and (b) exterior Ag NPs. UV/Vis spectra of detergent solutions collected after incremental simulated home launderings of cotton fabrics containing (c) interior and (d) exterior Ag NPs.
Fig. 8a shows the
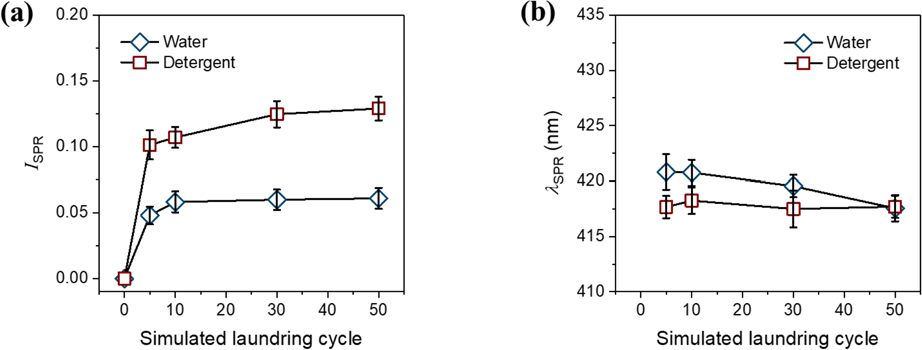
(a) Intensities of the SPR peaks (
Textiles treated with Ag NPs, if they are weakly bound, easily leach particulate Ag during laundering that causes various mechanical impacts such as centrifugal, frictional, and gravitational forces. These forces also facilitate the dissolution of Ag on the surface of NPs (either bound or free) and subsequent release of ionic Ag into washing solutions. To examine such transformation of Ag, washing solutions were further analyzed using ICP-MS and ultrafiltration. Figs. 9a and b show the concentrations of total Ag and Ag smaller than 1 nm in washing water, which were measured before and after ultra-filtration, respectively, as a function of the simulated home laundering cycle. Although the concentration of interior NPs incorporated was about five times greater than that of exterior Ag NPs coated (Fig. 4b), the amounts of total Ag detected in water were smaller for the interior incorporation. The total Ag concentration from the interior system continuously increased with subsequent laundering cycles, whereas the concentration from the exterior system reached its maximum at 10 cycles and depended less on additional cycles. For the case of Ag < 1 nm (Fig. 9b), the opposite result was observed (i.e., higher concentrations for the interior incorporation as compared with those for the exterior incorporation). When taken together, these two measurements indicate that the interior incorporation allowed the continuous, low-level Ag release, with a large portion being ionic Ag. To confirm this, the size fractions of Ag in water for the interior and exterior incorporations are compared, as shown in Figs. 9c and d, respectively. As expected, the fraction of Ag < 1 nm was greater for interior incorporation throughout the extended laundering. The majority, above 80%, of Ag released from the exterior system was in a particulate form.
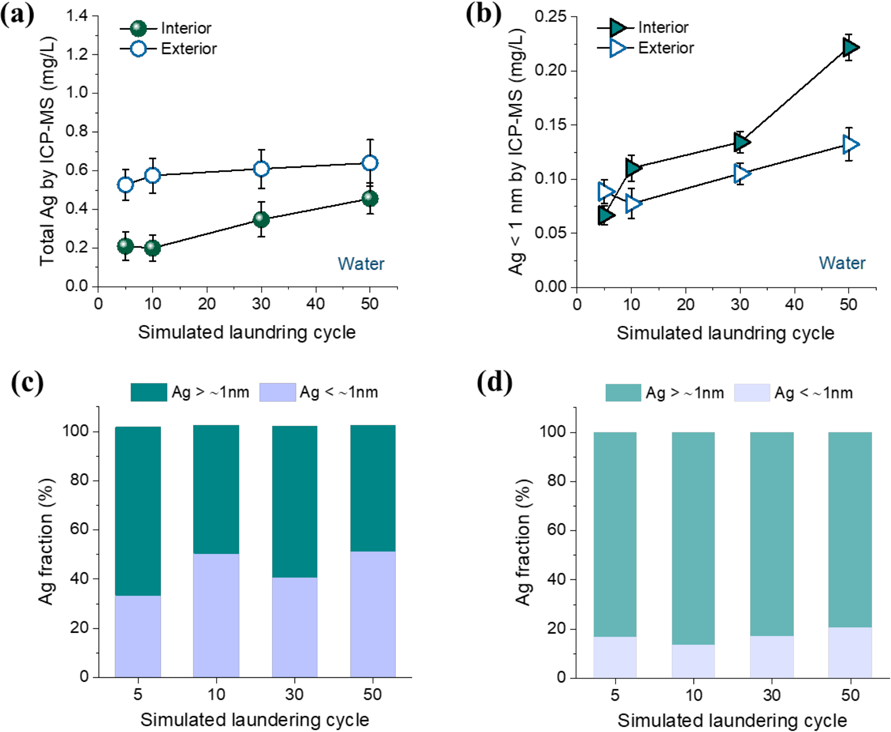
Concentrations of (a) total Ag and (b) Ag smaller than about 1 nm in water measured by ICP-MS after incremental simulated home launderings of cotton fabrics containing interior and exterior Ag NPs. Corresponding Ag size fractions for (c) interior and (d) exterior incorporations.
Figs. 10a and b show the concentrations of total Ag and Ag < 1 nm in the detergent solution, respectively. Compared with the result for water (Fig. 9a), the concentrations of total Ag for the exterior incorporation greatly increased, while those for the interior incorporation remained at similar levels. As a result, the incorporation method exerts a more pronounced effect in contaminating a detergent solution than in water. There was about a threefold reduction in the concentration of Ag of the detergent solution when switching from exterior to interior systems, even with the original amount of interior NPs being fivefold greater than the exterior NPs. On the other hand, the concentration of Ag < 1 nm appeared to be independent of the incorporation method (Fig. 10b). It was also found that the concentrations of Ag < 1 nm in the detergent solution were lower than those in water regardless of the incorporation method. These distinctive phenomena for the detergent solution were more obvious by the size fractionation of Ag, as shown in Figs. 10c and d for interior and exterior incorporations, respectively. The fractions of Ag < 1 nm for both interior and exterior incorporations greatly dropped in the detergent solution. Only less than 20% and 10% of the total Ag remained as an ionic form for interior and exterior incorporations, respectively. In combination with earlier results, this result suggests that although the interior and exterior incorporations induced different Ag release behaviors (i.e., the predominant release of ionic Ag by the interior incorporation), most of the Ag released into the detergent solution underwent the chemical transformation into a particulate form. The effect of the laundering cycle on this transformation was not noticeable.
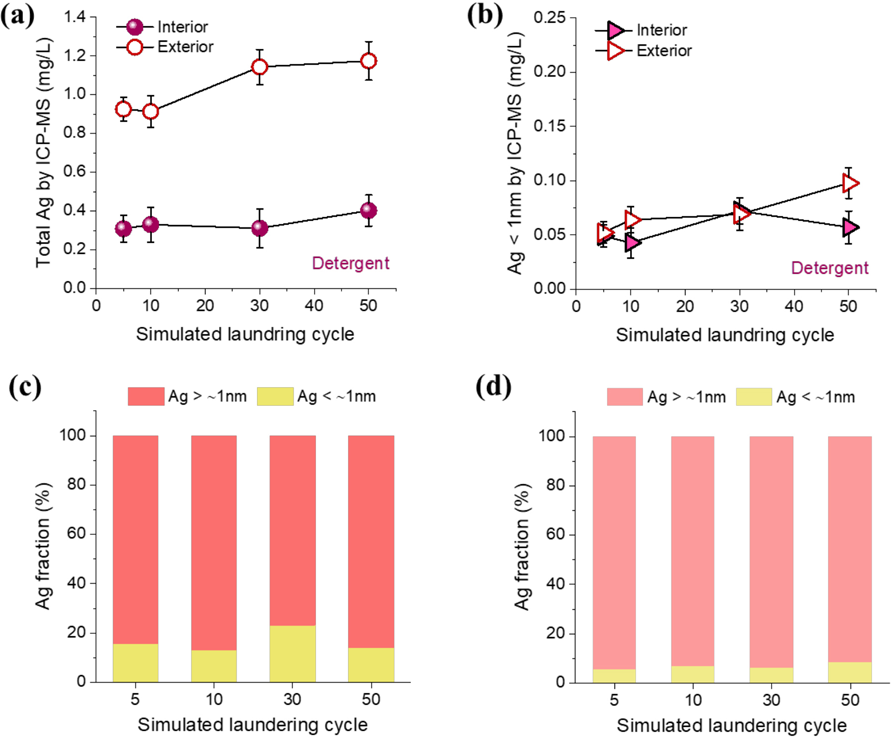
Concentrations of (a) total Ag and (b) Ag smaller than about 1 nm in detergent solution measured by ICP-MS after incremental simulated home launderings of cotton fabrics containing interior and exterior Ag NPs. Corresponding Ag size fractions for (c) interior and (d) exterior incorporations.
ICP-MS with ultrafiltration measures Ag that passed through the 3 kDa membrane, so it is assumed to measure Ag ions. Strictly, this measurement also includes sub-nanometer Ag particulates. The concentration of Ag ions alone was independently measured using an ISE. Figs. 11a and b show the concentrations of Ag ions in water and detergent solutions, respectively, as a function of the simulated home laundering cycle. The plots all showed a similar trend that the highest concentrations occurred at 5 cycles, followed by lowered concentrations with less dependence on additional cycles. This was different from the trend of increasing concentration for Ag < 1 nm observed by ICP-MS/ultrafiltration. The ionic Ag initially released could be adsorbed onto the surface of free NPs or oxidized. Such various routes of Ag fate are considered for the discrepant functions between the laundering cycle in the ICP-MS and ISE results. However, these analyses agreed that the fabric containing interior NPs released more Ag ions than the fabric containing exterior NPs. They also agreed that the Ag transformation was overtaken by the interactions with detergent components, resulting in less effect of the incorporation method on the concentration of Ag ions in the detergent solution.
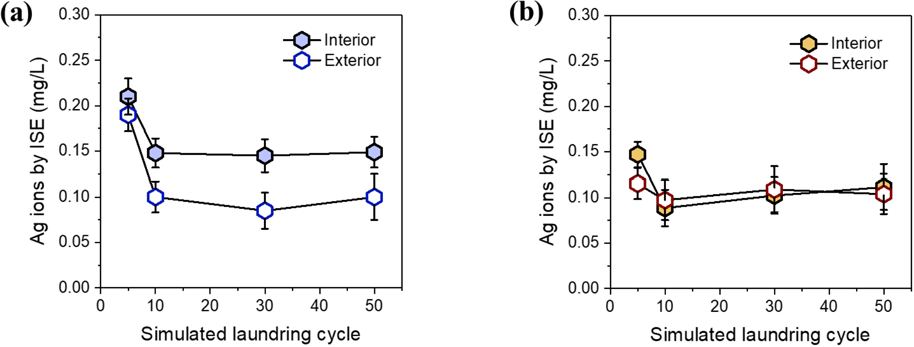
Concentrations of Ag ions in (a) water and (b) detergent solution measured by ISE after incremental simulated home launderings of cotton fabrics containing interior and exterior Ag NPs.
Conclusions
In the development of NP-functionalized textiles, the washing durability of NPs is essential to ensure their safe and reliable performance. However, many studies have reported the poor durability of commercial Ag NP-treated textile products against washing. This study shows that the incorporation location of Ag NPs for cotton fiber plays an important role in controlling the release of Ag in washing solutions. Two incorporations, locating the NPs in the interior and exterior of cotton fiber, were carried out. The interior NPs were produced by the in situ synthesis within the fiber, whereas the exterior NPs were produced by a surface coating that is generally used in the textile industry. The systematic analyses of cotton fabrics and washing solutions collected after incremental simulated home launderings up to 50 cycles agreed on the superior washing durability of interior NPs. The percentage concentrations of Ag remaining in cotton after 50 cycles for interior and exterior systems were 84% and 37% in water and 76% and 22% in detergent solution, respectively. Despite the fivefold higher concentration of interior Ag NPs incorporated into the cotton fabric, the amount of Ag detected in the washing solutions was smaller than that from the fabric with exterior Ag NPs. The cotton fabric containing exterior NPs predominantly released particulate Ag, whereas the fabric containing interior NPs released more ionic Ag. In the detergent solution, however, most Ag existed as a particulate form resulting from the interactions with detergent components. Based on these results, it can be concluded that the internal incorporation of Ag NPs within fibers is a prominent method for producing NP-leach resistant (but continuously releasing a low level of biocidal Ag ions), and thus washing durable, antibacterial textile products.
Footnotes
Acknowledgments
This work was supported by the National Science Foundation (CMMI-1562907).
Trade names are used solely to provide specific information. Mention of a trade name does not constitute a warranty or an endorsement of the product by US Department of Agriculture to the exclusion of other products not mentioned.
