Abstract
The dispersion property of modified nano-TiO2 particles and its antibacterial properties on cotton fabrics were studied. The nano-TiO2 particles were modified with reactive groups (such as hydroxyl groups, amino groups and silica groups). The modified nano-TiO2 particles were characterized by particle size analyzer, Fourier transform-infrared spectroscopy and transmission electron microscopy. Besides, the chemical load technology of optimizing the nano-TiO2 on the textile fiber combined with fiber by grafting and cross-linking was studied. The results showed that the stability of nano-TiO2 sol was related to the concentration of the modifier and the pH values of the system. The washing fastness and the antibacterial property of the fabric treated by modified nano-TiO2 had been increased and the antibacterial property of treated fabric was also related to the concentration of the modifier. By determining the fiber’s crystallinity, as well as the fabric’s breaking strength and tear strength, the influence of the modified nano-TiO2 photocatalyst on the structural properties of cotton fiber was discussed.
Keywords
Introduction
In recent years, nanotechnology has been flourishing. The development of nanomaterials is a foundation for the development of nanotechnology. Nanomaterials refer to materials with special properties, whose geometric dimension reaches nanoscale [1,2]. Among nanomaterials, great importance has been attached to nano-oxide owing to its promising application prospect in textiles due to its outstanding characteristics [3,4]. A typical example is that the photocatalysis and ultraviolet absorption properties of nano-TiO2 can be utilized for antibacterial finishing and anti-ultraviolet finishing of textiles to meet the demand of multi-functional finishing of textiles [5–9]. However, when nano-TiO2 particles are distributed in the medium, multi-phase thermodynamically unstable system will be formed, where nanoparticles will agglomerate and precipitation occurs. Therefore, the stability of nano-oxide particles in medium needs to be improved in order to facilitate the application of nano-oxide in the field of finishing of textile [10,11].
Nano-oxide powder particles have much smaller particulate size and bigger surface ratio, which regularly lead to mutation resulting in the sudden change in properties. For example, silver nanometer particles have extreme broad-spectrum sterilization effect and are non-hazardous to human body [12,13]. Thereby experts on material science and textile industry all over the world are trying to fix nano-oxide particles dispersed onto textiles to keep their functions, for example, anti-ultraviolet, sterilization, antibacterial and deodorization [14–20].
Currently, nanometer particles or membranes are mostly coated onto fibers or textile surfaces by an adhesive to fabricate functional textiles. This method is very simple and some functional indexes can be obtained. But this leads to less degree of washability and non-durability of textiles and even affects the softness of the fabric because no chemical bond exists between inorganic nanometer particle and textile fiber [10,21]. Otherwise, textile surfaces coated by nano-TiO2 have normal photocatalyst function, but fiber material may deteriorate. Because of the strong oxidizability of the photocatalyst, the fiber macromolecules can be easily broken so that the end group number of molecular chain of amorphous part of fiber increases and the supermolecular structure of the fiber weakens. This finally results in a decrease in macro-mechanical property of the fibers. Thus, making the nanometer particle evenly disperse onto textile and tightly combining it with nanometer material and fiber shall be crucial during development and application for nanometer composite multifunctional textile.
Nano-oxide is a kind of inorganic compound lacking functional groups, which can react with textile materials directly. During the after-finishing processing of the fabrics, adhesive are used to fix nano-oxide onto the surface of the fabrics. This leads to an inferior touch feel as well as inferior wash fastness. In order to improve the stability of nano-oxide in various media and facilitate the application of nanomaterials, one of the most effective ways is to modify the surface of nanoparticles [22,23]. In this article, some chemical methods were applied to modify nano-TiO2 sol and then hydroxyl, amino, and silica groups were introduced to form nanometer particle with reactive groups or simple organic compound ‘cluster’. Reactivity between nanometer oxide particle and natural cellulose was improved, and the dispersion stability of nano-TiO2 sol in water was also improved. Nanometer oxide particles were coated onto natural cellulose by grafting or cross-linking method. As a result the treated textiles had durable antibacterial and anti-ultraviolet effects [24,25].
Product studied in this article shall be widely used in garment material, household textile industry and made into new-design textile characterized by air purification, deodorization and sterilization.
Experimental
Materials and reagents
Cotton fabrics, 100% dyed (172.2 g/m2) 30 s × 30 s (density 68 × 68), were used as the substrate. Tetrabutylorthotitanate (Ti(C4O9)4), diethanolamine, sodium hydroxide, ethanol, hydrochloric acid, glacial acetic acid, and ethylene glycol used were of analytical grade. Surface modifiers were silicon compounds with multi-active group, such as γ-aminopropyl triethoxysilane (98% of KH-550, dosage 20–60 g/L). Grafting or cross-linking agents with double-active group were hydroxy silicone oil, hydrogen silicone oil and polyurethane (dosage 20–60 g/L).
Surface modification of nano-TiO2 particles
Measured amounts of tetrabutylorthotitanate (Ti(C4O9)4) and diethanolamine were mixed with ethanol by thorough stirring at room temperature for 1 h. The molar ratio of Ti(C4O9)4 (68.1 mL), diethanolamine (19.3 mL) and ethanol (46.7 mL) was 1:4:1, respectively. Hydrochloric acid (37%) (0.5 mL), distilled water (6.8 mL), and ethanol (134.1 mL) were added into the mixture by burette and stirred vigorously. After 3 h the TiO2 sol was obtained.
A certain amount of nano-TiO2 was distributed in ethylene glycol using the high-shearing mixer and ultrasonoscope, and proper surface modifier such as hydroxy silicone oil or hydrogen silicone oil or KH-550 (dosage 20–60 g/L) was added to the solution. After certain reaction time, modified nano-TiO2 was prepared.
Antibacterial finishing of the fabrics
Antibacterial finishing bath was prepared with modified nano-TiO2, crosslinking or grafting agent (KH-550), dispersant (ethylene glycol), pH regulator (sodium carbonate), etc. The fabric was treated by dip-pad-cure process. The cotton fabrics were dipped in the antibacterial finishing bath, and then padded using an automatic padder (Rapid Labortex Co. Ltd., Taipei, Taiwan) with pick-up value of 80%. This process was repeated twice. The fabrics were then dried at 100℃ for 4 min, and cured at 150℃ for 3 min in an oven.
Test methods
Infrared spectrum analysis
Fourier transform infrared (FT-IR) spectroscopy (NICOLET AVATAR-370, Thermo Fisher, USA) was used to characterize the structures of unmodified nano-TiO2 and modified nano-TiO2 by accumulating 32 scans at a resolution of 4 cm−1.
Stability test of nano-TiO2 sol
Still-setting method: The modified nano-TiO2 was distributed in ethylene glycol using high-shearing mixer and ultrasonoscope. About 200 g/L of modified nano-TiO2 sol was prepared and was poured into a capped cylinder. The layer heights of the sol at different time points were measured.
Sedimentation method: The above-mentioned modified nano-TiO2 sol was poured into the measuring cylinder. The sedimentation quantities of nanoparticles at different time points were determined using the self-refitted sedimentation balance, and the sedimentation rate was calculated according to equation (1)
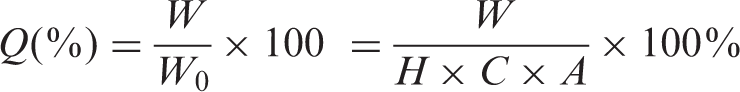
Determination of Zeta potential
The nano-TiO2 sol was poured into the electrophoresis tube, and the electrophoretic velocity of the sol at certain voltage was measured. The Zeta potential was calculated according to the following equation [26].

Here
Determination of washing fastness
The fabrics were treated with a finishing solution of modified nano-TiO2 (5 g/L) as well as with a finishing solution of unmodified nano-TiO2 (5 g/L) concentration. The treated fabrics were immersed in the washing bath at 42℃. Determination of washing fastness was carried out according to the JIS217-103 standard. The bath ratio used was 1:40, and the powdered detergent (phosphor-free detergent consisting of linear alkyl benzene sulphonic acid sodium and solid sodium aluminum silicate) content used was 6 g/L (soap content was 3 g/L). Washing was carried out once every 5 min, followed by rinsing in cold water for a few minutes. And then the samples were flat-dried without any additional stretching or twisting. The test was repeated for more than 10 times.
Antibacterial performance test
The bacteria liquid (containing gram-positive golden yellow
The number of the colonies was counted. Control experiments were carried out at the same time. Percent decrease of bacteria was calculated using the following equation

Determination of the crystallinity of the cotton fiber
The fiber crystallinity was determined by D/MAX-2000 X-ray diffractometer (Japan Rigaku Company). An X-ray tube was used as copper target. It was used to radiate copper Kα, where the voltage was 40 kV, the current was 40 mA, and the wave length λ was 0.15406 nm. The fiber crystallinity was calculated using the Segal empirical method [30]

Breaking strength and tearing strength
The breaking strength of the fabric was determined by the YG (B) 026D-250 electronic fabric strength testing instrument. The tearing strength of fabric was determined by YG033 fabric tearing testing instrument.
Results and discussion
Modification of the nano-oxide surface with siloxane active group
The modified nano-TiO2 had the following structural formula:
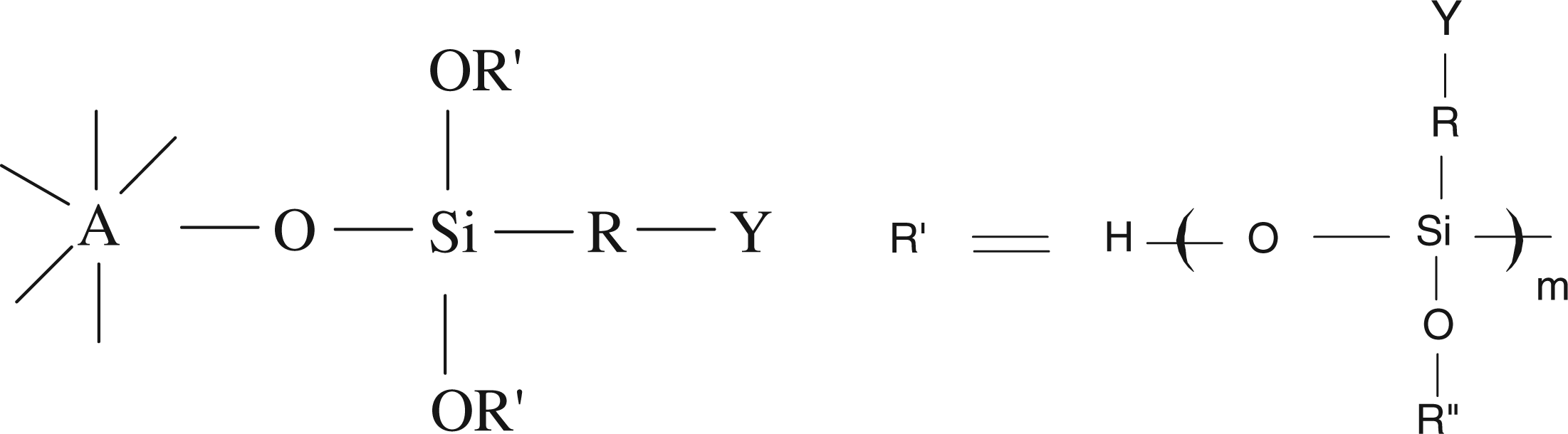
The above structural formula showed that the surface modifier can dehydrate and condense on nanometer dioxide to form oligomer. By changing the molar ratio of surface modifier and nanometer dioxide, the polymerization degree of oligomer was found to be 1–20.
The nano-TiO2 particle surface is rich in hydroxyl groups (Ti–OH) with high hydrophilic and reactive properties, which is easy to react with siloxane and other active groups, forming stable chemical bonds (such as titanium oxygen bond). In this article, the prepared nano-TiO2 precursor particle (A) surface contained a large number of titanium-hydroxyl groups, which could react with the modifier (B) that contained functional active groups such as siloxane under certain conditions. The titanium oxygen bond was then generated, and finally the surface modified nano-TiO2 particles were obtained. The modified reaction mechanism was as follows [31].

Here X represented –OH, –OCH3, OC2H5, etc., R represented –NH2, –OH, epoxy, siloxane, etc., A represented nano-TiO2 precursor particle, and B represented the modifier.
As for the above reaction, if the modifier is KH-550, Si–OCH3 in modified agent (B) was hydrolyzed in the presence of water, generating Si–OH.

The hydrolysate Si–OH was then made to react with Ti–OH in (A), generating Ti–O–Si and liberating water. On the other hand, the interaction and condensation of Si–OH groups generated by modifier hydrolysis creates condensation polymerization under some condition leading to the formation of dimer and polymer, which could react with Ti–OH of nano-TiO2, finally producing the modified nano-TiO2.
Due to the introduction of the functional group R on the surface of modified nano-TiO2 particle, if R is the active group (such as epoxy, siloxane, etc.), the modified nano-TiO2 could directly react with some groups (such as hydroxyl, amine, amide, etc.) in the fiber under certain conditions, thus forming covalent graft combination. If R is NH2 or OH, the cross-linking agent could contribute to the covalent bond between the modified nano-TiO2 and the fiber. Moreover, Si–O bonds on the surface of modified nano-TiO2 could prevent the direct contact between nano-TiO2 and the fiber. Thereby its photcatalytic activity is decreased, leading to less degradation of textile fibers.
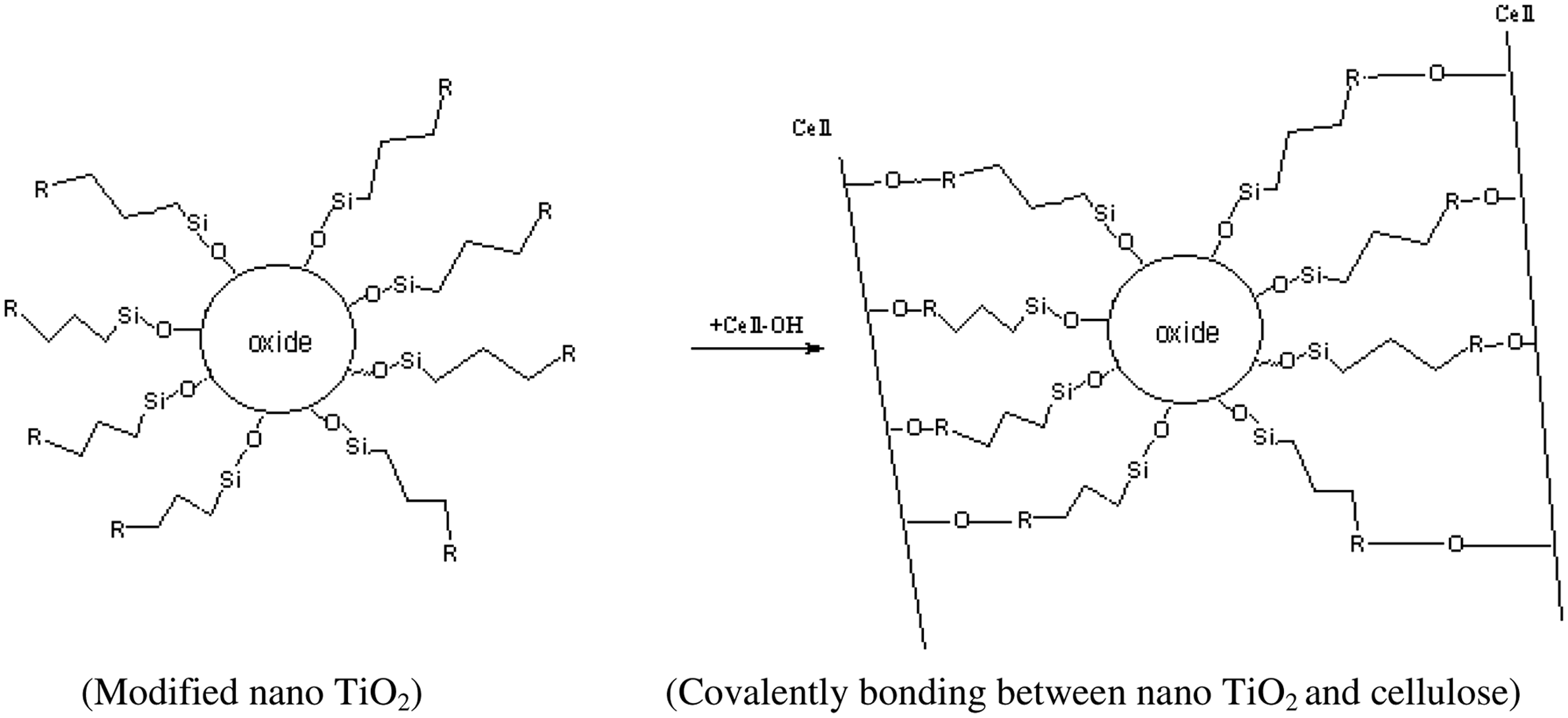
The modified nano-TiO2 were covalently bonded with cellulose, as shown in the following schematic. The nano-TiO2 modified by KH-550 could directly react with some groups in the fiber under certain conditions, thus forming covalent graft combination. The binding capacity between nano-TiO2 particles and cellulose was improved. Therefore the treated fabrics had durable photocatalytic effect [31].
Particle diameter of modified nano-TiO2
Figures 1 and 2 show the particle size distribution of modified nano-TiO2 and transmission electron microscopy (TEM).
(a) Particle size distribution of: (a) unmodified nano-TiO2; (b) modified nano-TiO2. Transmission electron microscopic photos of modified nano-TiO2: (a) at 200,000 × magnification; (b) at 500,000 × magnification.

Figure 1(a) shows the particle size distribution of unmodified nano-TiO2 sample, where the particle size distribution was relatively wide from 50 nm to 80 nm. Figure 1(b) shows the particle size distribution of modified nano-TiO2 samples. It was shown that after adding silicon compound modifier containing multi-active groups and a certain amount of dispersing agent, modified TiO2 had better dispersibility. There was no obvious agglomeration, and the particles were clearer. The particle size distribution was narrow with the average size smaller than 100 nm ranging from 70 nm to 80 nm, confirming that the modified TiO2 had reached nanometer level.
From the above TEM photos, we could find much less-sized crystal spherical particle from 60 nm to 80 nm. Such crystal particles integrated into TiO2 particle with their sizes of tens of nanometer. The integration added up specific surface area of single nanometer TiO2 sphere and effectively improved its photocatalysis.
Figure 3 shows TEM photos of unmodified nano-TiO2. It indicated that the prepared TiO2 particle was spherical and TiO2 particulate diameter ranged from 60 nm to 80 nm. The particles were obvious and had narrow size distribution. Compared to TEM photos of modified nano-TiO2, there was no considerable variation except for larger particulate diameter for modified nano-TiO2, indicating that chemical modification had negligible effects on the structure of nano-TiO2.
Transmission electron microscopic photos of unmodified nano-TiO2.
Stability of modified nano-TiO2 sol
Effect of modifier dosage on stability of nano-TiO2 sol
The nano-TiO2 particles were modified with different amounts of surface modifier, and were distributed in ethylene glycol with the help of high-shearing mixer and ultrasonoscope to form 200 g/L of modified nano-TiO2 sol. The sol was poured into the capped measuring cylinder with a total height of 80 mm. After letting it stand still for 3 days, the height of supernatant fluid was measured, as shown in Figure 4.
Effect of the modifier dosage on stability of modified nano-TiO2 sol.
Figure 4 shows that when nano-TiO2 was modified, the stability of sol was significantly improved. Before the addition of modifier (unmodified nano-TiO2), sedimentation rate of sol was much higher. Thereby collosol was hard to be stable due to the agglomeration of the particles. When the pH value was 3, the modified nano-TiO2 sol was relatively unstable. With an increase in pH values, the stability of sol increased. At some time, with the increase of modifier dosage, the stability of modified nano-TiO2 sol was improved. The sol had much better stability when the modifier dosage was up to 40 g/L. In general, the modifier dosage should be selected as 40 g/L as its stability had no obvious improvement when the modifier dosage was 60 g/L.
During the modification, the modifier molecules were grafted onto the surface of nano-TiO2 particles by reaction with hydroxyl group on the nano-TiO2 particle surface. The particles were surrounded by the modifier dosage, forming core-shell structure. When the particles came close to each other, the shells adhered to each other, and the shell layer was compressed. As a result, the number of the shell configurations possibly decreased, and the configuration entropy reduced, causing the increase of the free energy and producing repulsive potential energy. So the effect of the steric hindrance prevented mutual contact of particles. The steric hindrance is depicted as follows.

Meanwhile, large quantity of epoxy groups were introduced onto nano-TiO2 particle surface and hydrogen bonds with the formation of medium molecule. As a result, the stability of the nano-TiO2 particles in the medium was improved. The stabilizing function of modifier correlated with its dosage. When dosage of the modifier reached 40 g/L, the sol exhibited greatly improved stability. As for nano-TiO2 sol, hydroxyl, amino and silica groups were introduced to form nanometer particle with reactive groups or simple organic compound ‘cluster’. So the reactivity between nanometer oxide particle and natural cellulose was improved, and dispersion stability in water was also improved. With the addition of modifier, particle diameter becomes less and prevents it from reuniting. Besides, from Figure 4, when the dosage of modifier was 40–60 g/L, its stability was greatly improved.
Effect of pH value of disperse system on the stability of nano-TiO2 sol
Nano-TiO2 particles treated by 40 g/L modifier were used to prepare 200 g/L modified nano-TiO2 sol. Zeta potentials of the modified nano-TiO2 particles at different pH values were also determined. The results are shown in Figure 5.
(a) Zeta potentials of (a) unmodified nano-TiO2 particles and (b) modified nano-TiO2 particles at different pH values.
Electrostatic repulsion between two particles is minimized when pH of solution is equipotential as per DLVO theory. Particles can easily become the conglomerate under such condition. In general, isoelectric point of nanometer TiO2 particle stayed at about pH = 6.7, as shown in Figure 5(a).
In particular, non-modified nanometer TiO2 collosol is characterized by instability and is easy to reunite within the pH range 3–7.
Figure 5(b) shows the modified nano-TiO2 reaching its isoelectric point at pH = 3, and isoelectric point is decreased. The absolute value of Zeta potential increased with the increase of the pH value. As the particle surfaces are charged, the electric double layers are formed and the particles are surrounded with ionic atmosphere. When the particles come close to each other, if the repulsion of ionic atmosphere is larger than the attraction between the particles, the agglomeration of the particles could be prevented, and the stability of the sol could be improved. The repulsive potential energy of electric double layer is related to its thickness, and the thickness of electric double layer correlates with the Zeta potential on surface of nanoparticles. The increase of the Zeta potential of nanoparticles would cause an increase in the repulsion of electric double layer, which consequently improves the stability of the sol.
FT-IR spectra of modified nano-TiO2
The nano-TiO2 was modified by siloxane coupling agent KH-550 (γ-aminopropyl triethoxysilane, NH2(CH2)3Si(OC2H5)3). FT-IR spectra of the modified nano-TiO2 characterized by the Thermo Nicolet infrared spectrometer are shown in Figures 6 and 7.
Fourier transform infrared spectra of the unmodified nano-TiO2. Fourier transform infrared spectra of the modified nano-TiO2.
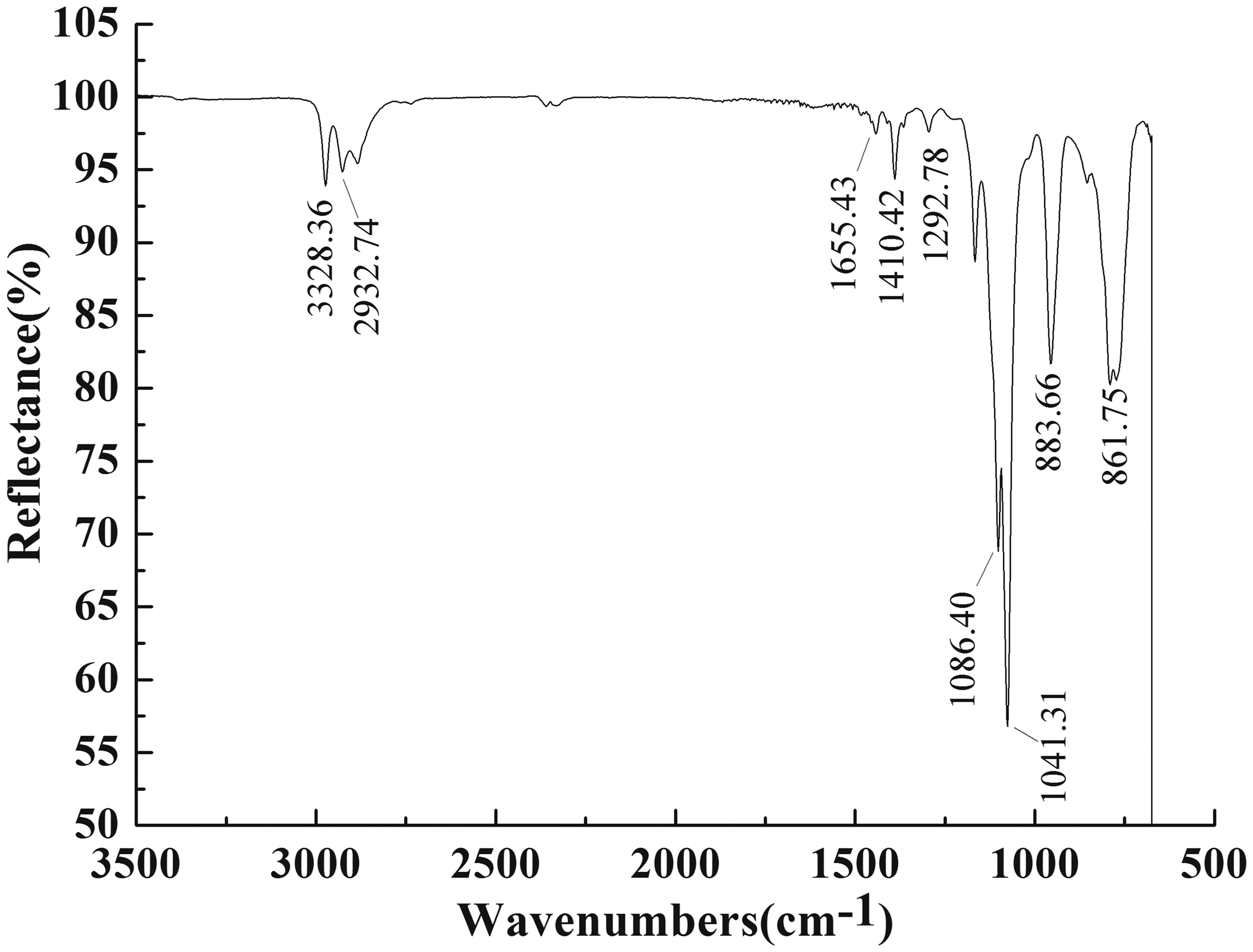
From Figures 6 and 7, it can be seen that there are many strong absorption peaks in infrared spectrum of nano-TiO2 after modification compared with unmodified nano-TiO2. The absorption peaks at 1090–1020 cm−1 are the stretching vibration of Si–O groups characteristic, corresponding to Si–O–Si and Si–O–C. The peaks at 883.66 cm−1 are corresponded to unsymmetrical stretching vibration of C–O–C groups. It indicated that the epoxide group was introduced on the TiO2 particle surface. The flexible vibration absorption peak or bending vibration absorption peaks of −OH groups are respectively available at 3328.4 cm−1 or 1655.4 cm−1. However, absorption peak at 1086.4 cm−1 is stronger than that of unmodified nano-TiO2, which is caused by superposition of Si–O flexible vibration peaks of linear siloxane. Siloxane coupling agent KH-550 was hydrolyzed in the presence of hydrogen ion, and then dehydrated and condensed into polymer, which could be bonded with hydroxyl group on inorganic surface via hydrogen bond. The dehydration and condensation between OH groups at nano-TiO2 surface and the hydrolyzed KH-550 produced Ti–O–Ti bonds. And KH-550 was finally linked onto nano-TiO2 surface. It was shown that certain new active groups such as Si–OH groups and epoxide groups were introduced onto modified TiO2 particle surface.
The surface morphology of cotton fabrics treated with the nano-TiO2 sol
Figure 8 shows SEM pictures of cotton cellulose surface treated by modified nanometer TiO2 sol at different magnifications. From Figure 8(a), large number of TiO2 nanometer particles was evenly distributed on the cotton cellulose surface. The treated cotton had dense and low-porosity characteristics. In addition, from Figure 8(b), most of near-spherical TiO2 particles’ diameter was about 80 nm, which was in accordance with the result tested by particle size analyzer. While the unmodified TiO2 particles size was about 70 nm.
Scanning electron microscopic images of the cotton fiber coated with TiO2 nanoparticles at different magnifications: (a) low magnification with the scale bar 1 µm; (b) high magnification with the scale bar 100 nm.
Application of modified nano-TiO2 for antibacterial finishing of fabrics
Antibacterial property comparison of fabrics before and after treatment
Antibacterial performance of modified nano-TiO2 after treating the fabric.

From Table 1 it can be concluded that photocatalysis modified nano-TiO2 had strong antibacterial effect on gram-positive golden yellow
Effect of nano-TiO2 concentration on antibacterial property
Effect of modified nano-TiO2 concentration on antibacterial effect.

It can be concluded from Table 2 that the antibacterial effect highly correlates with the concentration of modified nano-TiO2. Only when the modified nano-TiO2 in finishing solution reached certain concentration, the finished fabrics exhibited obvious antibacterial property. Therefore, when the concentration of modified nano-TiO2 in the finishing solution exceeded 5 g/L, the antibacterial efficiency was higher than 90%.
Wash fastness test of the antibacterial fabric
The treated fabrics were washed according to standard method, and the antibacterial effect was determined. The results are shown in Figure 9.
Wash fastness of the treated fabrics.
In Figure 9, modified A and modified B represented the fabrics treated by modified nano-TiO2 for
Effect of photocatalyst on cotton crystallinity under different light irradiation conditions
The sample used for testing is cut and squeezed into a slim piece under indoor temperature, then the intensity curve angle of 2θ is attained, with the sample scan angle ranging from 10° to 60° (2θ). On such scanning curve, peak of 002 face diffraction is available around 2θ = 22°, and minimum peak is available around 2θ = 18°. Hereby, relative degree of crystallinity of cotton cellulose could be calculated. Figure 10 shows X-ray diffraction strength distribution of cotton fiber treated by 4% modified nano-TiO2 photocatalyst after sunlight irradiation for 49 days and ultraviolet irradiation for 120 h, respectively.
Comparison of X-ray diffraction strength of treated and untreated cotton fibers after sunlight irradiation and ultraviolet irradiation: (a) the untreated reference sample; (b) the treated sample irradiated by sunlight for 49 days; (c) the untreated sample irradiated by ultraviolet for 120 h; (d) the treated sample irradiated by ultraviolet for 120 h.
Figure 10 shows the typical distribution of X-ray diffraction strength of cellulose I [32]. The test data showed that the relative crystallinity values of the modified nano-TiO2 photocatalyst untreated reference sample (a) and the modified nano-TiO2 photocatalyst untreated reference sample irradiated with UV light for 120 h (c) are 73.76% and 74.95%, respectively. There is minor difference between the two. Therefore, it can be concluded that UV irradiation does not destroy the crystalline structure of cotton cellulose [33]. Figure 10(b) and (d) indicates that the relative crystallinity of cotton fabrics treated by nano-TiO2 photocatalyst under sunlight irradiation for 49 days and UV light for 120 h were 72.36% and 71.67%, respectively. Thus the oxidation of cotton fiber macromolecule was caused by modified nano-TiO2 photocatalysis, leading to the chain fragmentation of the molecules. Meanwhile, this article also tested degree of polymerization variation of textile under same condition. Test results indicated that its degree of polymerization reduced by 7.4% under exposure to sunlight for 49 days. However, it also only reduced by 9.5% under exposure to ultraviolet radiation for 120 h.
However, the chain fragmentation of the molecules was only confined to the surface and amorphous region of fiber, and photocatalytic effect on crystalline structure of cotton cellulose was very limited.
Effect of photocatalyst on mechanical property of cotton fabric at different light irradiation time points
Because nanometer TiO2 particles have stronger photocatalysis and oxidation, macromolecule link partially break under sunlight or especially under ultraviolet radiation. This added up terminal group of fibrous formless molecular chain and deteriorated fibrous super-molecular structure, finally reducing fibrous macroscopic mechanical property and deteriorating the fiber.
So, to stop fiber from obvious deterioration, the liquid-phase deposition technique was generally applied to cover inorganic film onto TiO2 particle surface (such as SiO2 nanometer film). However, the continuous and dense nanometer wrappage film onto TiO2 particle surface was formed, and property of photocatalyst (degradation of VOC) also greatly reduced.
In this article nanometer catalyzer was studied, and KH-550 was added to nanometer TiO2. Silicon oxygen bond groups were formed on TiO2 surface, which could prevent direct contact of nanometer TiO2 with fiber and offered some protection for fiber. So modified TiO2 reduced photo-oxidation of fiber and prevented or reduced deterioration of fibers.
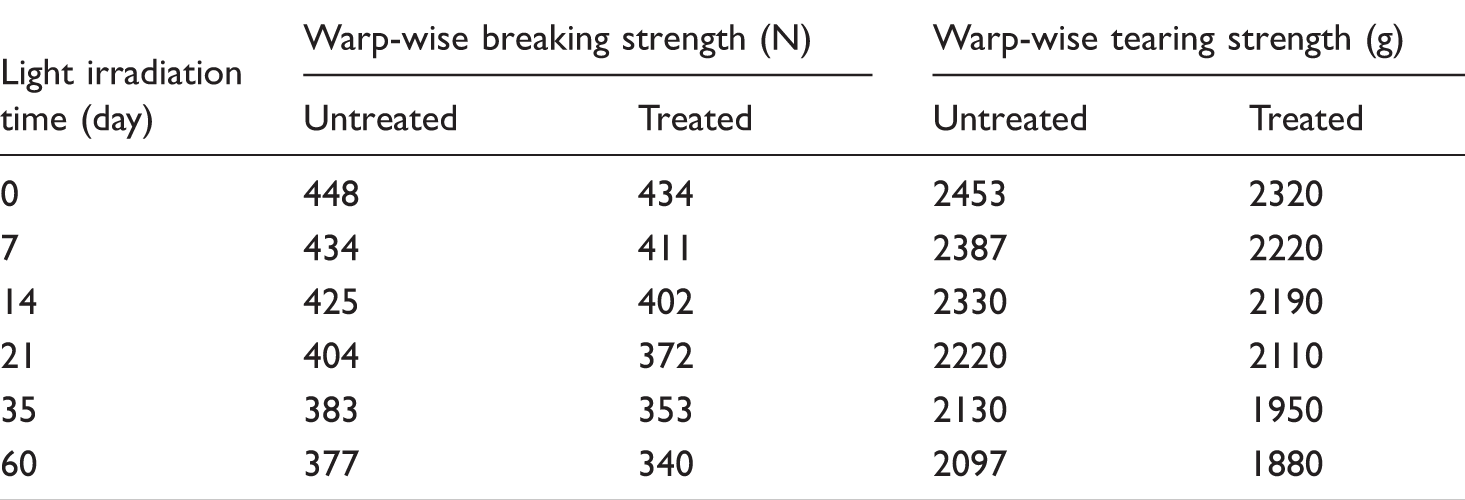
Effect of different sunlight irradiation time on mechanical property of cotton fabric.
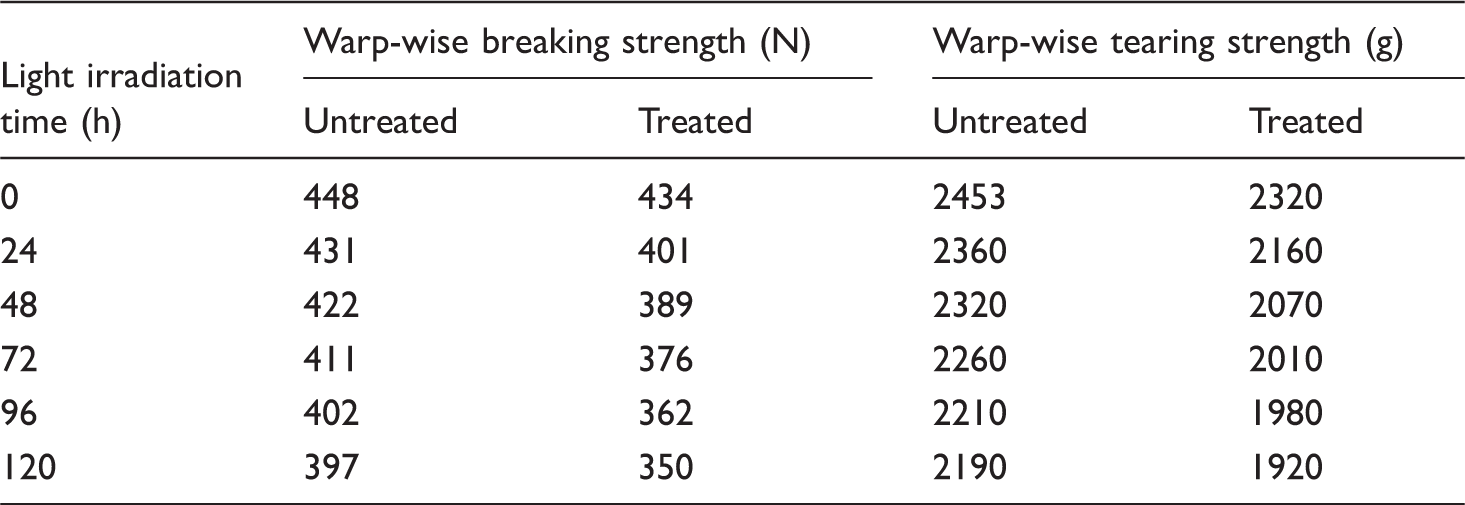
Effect of different ultraviolet irradiation time on mechanical property of cotton fabric.
It was concluded from Table 3 that the warp-wise breaking strength and warp-wise tearing strength of cotton fabric treated by modified nano-TiO2 photocatalyst decreased as the light irradiation time increased, and this decrease became slower. After 35 days of sunlight irradiation, the warp-wise breaking strength and warp-wise tearing strength of the fabric treated by photocatalyst decreased by 7.8% and 8.5% respectively compared with that of the reference sample, and this decrease became much slower.
Table 4 shows that covalent bond in macromolecule structure of untreated reference cotton fabric under ultraviolet irradiation was broken, leading to the decrease of the breaking strength and tearing strength. However, the warp-wise breaking strength and warp-wise tearing strength of cotton fabric treated by modified nano-TiO2 photocatalyst under ultraviolet irradiation for 120 h decreased by 11.8% and 12.2%, respectively, compared with that of the reference sample treated under UV light for the same irradiation time. It can be concluded that modified nano-TiO2 photocatalyst causes damage to cotton fiber under UV irradiation. The descent rate is controlled at about 12% and the longer the irradiation time, the greater the damage.
Conclusions
The stability of the modified nano-TiO2 sol was significantly improved. The stabilizing property of the modifier correlated with its dosage. When the dosage of the modifier was higher than or equal to 40 g/L, the modified nano-TiO2 sol had superior disperse stability. The disperse stability of nano-TiO2 sol was fully dependent on pH. The optimum isoelectric point was 3.0, and if the level was exceeded the stability of nano-TiO2 sol increased. The concentration of modified nano-TiO2 in the finishing solution had a great impact on the antibacterial effect of the finished fabrics. When the modified nano-TiO2 concentration in the finishing solution was higher than 5 g/L, the finished fabric exhibited better antibacterial effect with over 90% antibacterial efficiency. The applicability of nano-TiO2 was greatly improved after surface modification, and the wash fastness of the finished fabric was significantly enhanced as well. There were no obvious changes of crystallinity and crystalline structure of cotton fiber treated by modified nano-TiO2 photocatalyst under sunlight irradiation and UV light irradiation. In addition, the breaking strength and the tearing strength of the treated fabric decreased to some extent. The higher dosage of photocatalyst and the longer irradiation time of sunlight and UV light caused more damages to the treated cotton fiber.
Although some modifier was added to nanometer TiO2, there was still no obvious rise in the cost of textile treated by modified nanometer TiO2 and hence is still accepted by the consumers.
Footnotes
Funding
This research was supported by Innovation Program of Shanghai Municipal Education Commission (11YZ213).
