Abstract
This study aimed to increase the photocatalytic efficiency and durability of functional fabrics coated with TiO2/SiO2 nanoparticles. Tetrabutyl titanate and tetraethyl orthosilicate were used as precursor and ultrasonic irradiation was utilized as a tool for synthesis of TiO2/SiO2 in low temperature and loading nanoparticles onto the polyester-cotton fabric. The multifunctionality of such nanocomposite material was evaluated by analyzing its hydrophilicity, photocatalytic, and antibacterial activities. The hydrophilicity and photocatalytic activity were analyzed based on water droplet contact angle measurements and removal of methylene blue stain under UV, respectively. On the other hand, antibacterial activities were examined against the Gram-positive S. aureus and the Gram-negative E. Coli. TiO2/SiO2-coated fabrics exhibited outstanding self-cleaning, antibacterial, and superhydrophilic properties. Moreover, particle size analysis, X-ray photoelectron spectroscopy, Fourier-transform infrared spectroscopy, and UV–Visible spectrophotometry confirmed that TiO2/SiO2 nanoparticles produced higher photocatalytic activities on the treated fabrics than pure TiO2 nanoparticles. Furthermore, the self-cleaning properties remained after 30 launderings, indicating excellent washing durability. Therefore, this process had no significant negative effect on the wearability including tensile strength, whiteness index, ventilation property, and surface roughness of sonotreated fabrics.
Introduction
The field of self-cleaning coatings on fabrics is divided into hydrophilic and hydrophobic. Both types clean themselves through the action of water; the former by sheeting water and the latter by rolling droplets to remove dirt [1]. However, hydrophilic coatings based on titanium dioxide (TiO2) under ultraviolet (UV) irradiation have an additional ability to chemically break down organic dirt under sunlight [2,3]. Numerous studies deal with self-cleaning surfaces, but the majority of them still involve materials used in the construction industry [4–7]. Accordingly, enhancing a fabric’s functional characteristics through nanotechnology modification (TiO2, silica (SiO2), iron oxide, zinc oxide, silver, etc.) has become an attractive research field that provides new insights for scientists to develop high value-added products over the recent years. Among the numerous nanoparticles, TiO2 is widely used for the functionalization of textile materials because of its excellent optical properties, long-term stability, nontoxicity, and relatively low cost [8]. Under UV-light irradiation, TiO2 catalyst can produce oxygen species such as •OH, H2O2, and O2•−, which are considered important in increasing photocatalytic activity [8]. However, TiO2 mainly absorbs UV with <380 nm wavelengths, which covers only 5% of the solar spectrum. Therefore, a new visible-light photocatalyst that extends the absorption-wavelength range into the visible-light region is of great interest to develop. Many studies have reported that doping TiO2 with metals or nonmetals can extend the light-absorption region into the visible region [9–16]. Among such dopants, SiO2 has been integrated into a surface-coating formulation to augment the efficacy of TiO2 photocatalyst. The presence of SiO2 plays dual roles of increasing the surface area in the vicinity of TiO2 and the surface acidity of photocatalyst [17]. Yu et al. [18] prepared monodispersed anatase TiO2/SiO2 composite microspheres by calcining at 400℃ and 600℃. However, the temperatures used during the calcinations were too high for any organic substrate. Matsuda et al. [19] prepared TiO2/SiO2 nanocomposite films on organic polymer substrates using a sol–gel process followed by a boiling treatment. However, the films were easily leached out from the substrates. Meanwhile, sonochemical irradiation has been proven as an effective and environmental method for TiO2 synthesis [20,21]. Prasad et al. [20] also synthesized nano-TiO2 through an ultrasound-assisted sol–gel method and found that crystallite size decreased with ultrasonic irradiation. Neppolian et al. [21] prepared TiO2 photocatalysts by sol–gel and ultrasonic-assisted sol–gel methods; they demonstrated the advantages of ultrasonication in improving the photocatalytic properties of mesoporous nano-TiO2. In addition, many researchers have attempted to improve wash fastness of textile materials coated with TiO2 nanoparticles by using some binders or other methods [22–24], conforming to textile-application requirements. However, the use of binders may have negative effects on the photocatalytic activity of TiO2. Fabric pretreatment such as plasma and grafting treatments can lead to overprocesses and high cost. In this study, TiO2/SiO2 nanocomposites were sonosynthesized through hydrolysis of tetrabutyl titanate (TBT) and tetraethyl orthosilicate (TEOS) and then loaded onto cotton/polyester fabrics through a one-step process. This method was used to improve the photocatalytic efficiency and washing durability of cotton/polyester fabrics modified by doped TiO2 nanoparticles. The samples loaded onto TiO2/SiO2 and single TiO2 nanoparticles were compared in terms particle size, surface groups, and light-absorption region. Furthermore, the morphology, self-cleaning property, durability, antibacterial activities, and mechanical properties of the treated fabrics were characterized and investigated.
Experimental
Materials
Bleached plain-weave 35/65 cotton–polyester fabric with a warp density of 30 yarn/cm (warp count, 15.5 tex), weft density of 40 yarn/cm (weft count, 15.5 tex), and fabric weight of 108 g/m2 was supplied by Pengyu textile Co., Ltd. The reagent-grade chemicals used included TBT was obtained from Sinopharm Chemical Reagent Co., TEOS was supplied by Shanghai Wulian Chemical Plant for Chemical Reagent Purchasing, hydrochloric acid (37%) and ethyl alcohol was purchased from Pinghu Chemical Reagent Plant, and methylene blue (MB) were obtained from Shanghai Lingfeng Chemical Reagent Co., Ltd. Deionized water was used throughout this study.
Methods
Scouring
The cotton–polyester samples were washed in a bath containing 2 g/L nonionic detergent with liquor-to-good ratio = 50:1 at 35℃ for 20 min to remove impurities, such as wax and grease. The samples were then rinsed with distilled water and dried at 50℃ for 10 min.
Preparation of TiO2-coated fabric
TiO2 was synthesized by TBT hydrolysis in the presence of ethanol, distilled water, and hydrochloric acid as dispersant and then loaded onto the cotton–polyester fabric with simultaneous ultrasonic irradiation (50 kHz, 50 W). In typical synthesis, 100 mL specified ethanol/distilled water (volume ratio = 3:4) was mixed with 0.5 mL HCl (37%) under vigorous stirring (250 r/min). A piece of cotton-polyester fabric (10 × 10 cm2) was then immersed in prepared solution (pH = 3.0) and sonicated in an ultrasonic bath. 5 mL TBT was then added dropwise into the working solution during sonication under ambient condition (20 ± 2℃).And then the temperature was increased to 65℃ during 2 h and hold to the end of the procedure. The treated samples were dried at 80℃ for 5 min and then cured at 120℃ for 3 min.
Preparation of TiO2/SiO2-coated fabric
TiO2/SiO2-coated fabric was prepared in a similar manner. SiO2 sol was poured dropwise to the above-mentioned mixed liquor with the cotton–polyester fabric (10 × 10 cm2) dispersed in an ultrasonic bath after 2 h of sonication under 65℃. SiO2 sol was prepared following the method used to fabricate TiO2-coated fabrics. In a typical procedure, 10 mL of TEOS in ethanol was hydrolyzed with water containing HCl at 60℃ for 2 h under vigorous stirring (800 r/min). The volume ratio of TEOS, ethanol, H2O, and HCl was 15:40:5:0.1, respectively. The mixture was sonicated for another 2 h and the fabric color had not changed at the end of the procedure. The treated samples were dried at 80℃ for 5 min and then cured at 120℃ for 3 min.
Test methods
Characterization
To examine dispersion, the sizes of TiO2 and SiO2 particles in hydrosol were measured using a particle size and zeta-potential analyzer (Zetasizer Nano ZS, England, UK). Chemical compositions were studied using a PHI 5000 C ESCA System X-ray photoelectron spectroscopy (XPS) system with a 300 W Al Kα X-ray source. The local composition and chemical characteristics of the sample surface were evaluated using a Fourier-transform infrared (FTIR) spectroscopy system (Nicolet 8700, USA). UV absorption properties of TiO2 and TiO2/SiO2 on fabrics were measured using a diffused UV–Visible (UV–Vis) spectrophotometer (Lanbda A 35, USA) at 200–800 nm wavelength, 0.1 step scan speed, 0.1 sampling interval, and 1.0 slit width. Surface morphologies of the samples were observed using Hitachi TM-3000 scanning electron microscopy (SEM) in secondary electron imaging mode at 1000× and 2000× magnifications.
Water-droplet adsorption
The hydrophilicity of samples was evaluated by measuring the time required for a water droplet to be adsorbed. Accordingly, water (5 µL) was dropped 1 cm from the fabric surface, and the time of complete adsorption was recorded using a Water Contact Angle Metre (KSV CAM101).
Self-cleaning property
Self-cleaning performance of the samples was assessed by methylene blue (MB) discoloration. The treated samples were placed on a flat surface, and then one drop (50 µL) of 0.01% MB was vertically dripped using a burette from 1 cm above the fabric. After 3 min of MB absorption, the padding was irradiated with a 24 W lamp that produces 365 nm UV at a distance of 10 cm. The light intensities were adjusted at 0.7–0.8 mW/cm2 for UV irradiation. Color changes of MB absorbed by padding were monitored by comparing images taken after every 0.5 h of irradiation using a Samsung ST700 digital camera.
Washing durability
Washing durability test of the treated samples was conducted according to AATCC Test Method 61-2007. The washed fabric was dried at room temperature prior to further investigation. The MB-stained cotton–polyester fabrics were irradiated under UV light for 2 h. The color changes of MB on the unwashed fabrics were then compared with those observed on fabrics washed after 5, 15, and 30 times.
In addition, a quantitative method of evaluating the washing durability of treated fabrics was also carried out by monitoring the photodegradation of MB solution (50 mg/L) on immersed fabrics under UV irradiation. After 1 h of stirring in darkness, the concentration of MB solution was measured as C0. The concentration of MB solution was also measured as C after different periods of UV illumination. C/C0 was obtained to compare the trend of MB photodegradation in the presence of treated fabrics.
Antibacterial activity
Antibacterial activities of TiO2- and TiO2/SiO2-treated fabrics were examined against the Gram-positive S. aureus and the Gram-negative E. coli according to AATCC 100-2004. The irradiation device used after inoculation was a UVA lamp (365 nm, 0.1 mW/cm2), and then the samples began to elute after stabilization for 0.5 h. The antibacterial properties of the treated fabrics were calculated based on the ratio of surviving bacteria on the sample to the initial number of bacteria on a control sample, i.e. survival (%) = N/N0 × 100%, where N0 and N are the number of colony forming units before and after illumination, respectively.
Tensile properties
The tensile properties of samples were tested on a YG(B)026 fabric-strength tester, with constant rate of extension according to ASTM D5035-2011. The clamp speed and gage length were 300 mm/min and 150 mm, respectively. And the width of samples was 50 mm. Moreover, the number of every kind sample that tested is 20.
Whiteness index
The impact of treading process on the natural color of fabrics was assessed through measuring the whiteness indices of samples using a datacolor (Spectraflash, SF 600-PSUS, USA). Next, two UVA lamps (Philips 12 W) were used to illuminate the cotton-polyester samples. The specimens were irradiated for 10 h in a light box.
Ventilation property
Ventilation property was tested using air permeability tester (YG461E, Wenzhou) according to GB/T 5453-1997. Air permeability is an important feature in the performance of textile materials used to provide an indication of the breathability of the coated fabrics, in this test a 20 × 20 cm2 fabric was fixed into the tester and by using 100 Pa air pressure through the fabric, then the rate of air flow was used to determine the air permeability of the tested fabric.
Surface roughness
Surface roughness was monitored according to FZ/T 01054-2012 standard, using KES-FB fabric style instrument. The samples were cut in 20 × 20 cm2 and tested under 20℃. It’s well known that surface roughness directly affects the handle property of fabric.
Results and discussion
Particle size distribution of titanium and silicon hydrosols
Particle dimension dispersion in various hydrosols is shown in Figure 1, where the x-axis is the diameter of particles in hydrosol, and the y-axis is the scattered light intensity. As shown in Figure 1(a), the particle size distribution in TiO2 hydrosol was relatively wide, i.e. from 10 nm to 30 nm. This finding indicated that the hydrolysis of precursors was complete and uniform, helping form more even and thinner film on the surface of fabrics. After adding SiO2, the composite particle size distribution was narrow (from 6 nm to 15 nm), as shown in Figure 1(b). Thus, the addition of SiO2 hindered TiO2 grain growth. Meanwhile, the average size of composite particles was smaller than that of pure TiO2 particles, which indicated a lower agglomeration tendency in solution than the pure TiO2 dispersions. At the same time, they were almost entirely nanoscale, and the sol formulation was reasonable for coating quality and sol stability.
Particle size distribution of TiO2 (a) and TiO2/SiO2 (b) sol.
XPS analysis
The surface chemical composition of samples coated with TiO2/SiO2 was investigated using XPS. As shown in Figure 2(a), XPS survey spectra indicated the presence of strong O 1s, C 1s, Ti 2p, and Si 2p peaks, which confirmed that TiO2/SiO2 particles had been successfully coated and self-assembled onto the fabric surface. The high resolution C 1s peak exhibited three distinct subpeaks at 284.6 eV of C–C bonds, 285.0 eV of –CHX bonds, and 288.9 eV of O = C–OH bonds (Figure 2(b)). The O 1s spectrum containing the Ti–O–Ti species (530 eV) and Ti–O–Si species (531.8 eV) can be obviously observed in Figure 2(c), in accordance with FTIR analysis results. Figure 3(d) shows the Ti 2p XPS core level spectrum, in which two distinct peaks at 458.9 and 464.6 eV can be assigned to Ti 2p3/2 and 2p1/2, respectively. The splitting energy between the Ti 2p3/2 and 2p1/2 core levels was 5.7 eV (Figure 2(d)), which suggested the existence of a normal state of Ti4+. Another obvious indications of the presence of Si were in the peak at 102.4 eV (Figure 2(e)) and in the peaks of the Si–O–Si (532.9 eV) and Ti–O–Si (531.8 eV) groups (Figure 2(c)) [30].
(a) XPS survey spectra of the fabric coated with TiO2/SiO2. Corresponding high-resolution XPS spectra of C 1s (b), O1s (c) Ti 2p (d) and Si 2p (e). FTIR spectra of TiO2 and TiO2/SiO2 treated samples.

FTIR analysis
Figure 3 shows the FTIR spectra of fabrics coated with TiO2 particles and composite particles. The spectral peaks near 3300 and 1640 cm−1 present in both samples were attributed to the stretching mode of water hydroxyl bonds [25–27]. The peak at around 1640 cm−1 was due to the bending vibration of the O–H bond of chemisorbed water, and the peak at around 3300 cm−1 was due to the stretching mode of the O–H bond of free water. Additionally, the bands at 792 and 1062 cm−1 were for symmetric and asymmetric Si–O–Si stretching vibrations, respectively. Thus, Si existed as both segregated amorphous SiO2 and some Ti–O–Si bonds [28–31]. The stretching vibration of Si–O–Ti species was responsible for the band at 946 cm−1, indicating the existence of stable linkages between Ti and Si [32,33]. The FTIR spectra at 500–1000 cm−1 showed only one absorption peak for TiO2 at 640 cm−1, which was ascribed to the stretching vibrations of Ti–O bond [34,35].
UV absorption properties
To understand the optical absorption property and the UV-light utilization efficiency of as-prepared TiO2 and TiO2/SiO2-coated fabrics, UV–vis absorption data of the two samples were obtained. As shown in Figure 4, TiO2/SiO2-coated fabric was seen to have higher absorption values than the TiO2-coated one. The absorption edge of TiO2/SiO2-coated fabrics shifted towards longer wavelength, i.e. the so-called red shift. Spectral data of TiO2/SiO2-treated fabric showed a cutoff at around 500 nm, in which the absorbance value almost reached the minimum, whereas TiO2-treated fabric exhibited absorption bands at 200–390 nm. Then, the band-gap energy was calculated according to the formula (αhv)1
/n
= k(hv − Eg), where α is the absorption coefficient, k is a parameter, n is equal to 1/2 for direct transition, hv is the energy of light absorbed, and Eg is the band-gap energy. The calculated absorption edge of TiO2-coated sample was 3.18 eV (390 nm) and this result corresponded well with the semiconductor band gap energy of crystalline bulk anatase (3.20 eV, 387.5 nm) UV–Vis absorption of pure TiO2 and TiO2–SiO2 composites coated fabrics.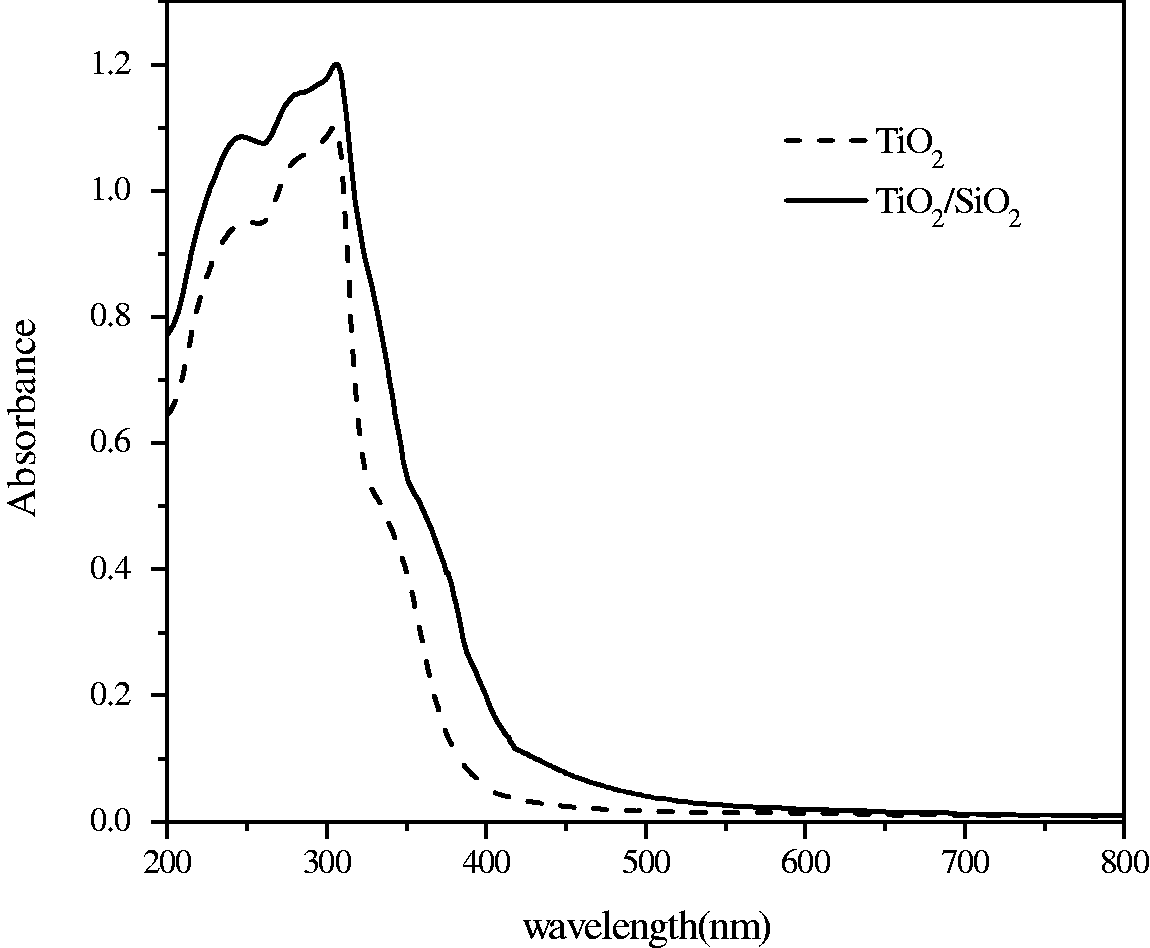
Mechanism of in situ synthesis of TiO2–SiO2 nanoparticles on fabrics
The synthesis of TiO2/SiO2 composite nanoparticles and their subsequent loading on fibers in a one-step process involved simultaneous synthesis and loading processes through ultrasonication. Nanoparticles were formed during sonochemical irradiation according to the hydrolysis and polycondensation reactions in Scheme 1. In the sonosynthesis of nano-TiO2/SiO2 composites, an acoustic cavitation process can generate an ephemeral localized hot zone with extremely high temperature and pressure. These swift changes may lead to the sonolysis of water forming OH• and H• radicals [36]. Overall, the exact mechanism of sonosynthesis and the sonocrystalization of TiO2/SiO2 composite nanoparticles were not completely recognized, but the generation of nano-TiO2/SiO2 composites at low temperature (65℃) may be mainly due to transient localized hot zone, microturbulence, and shock waves.
Mechanism of TiO2/SiO2 composite nanoparticles sonosynthesized on fabric.
In a system including water and fabric, cavitation can occur at three possible places: (1) in bulk liquid; (2) in the boundary layer between fabric and bulk liquid; and (3) in the interior of the yarns of fabric [37]. Cotton–polyester woven fabric is mainly a porous medium with relatively large inter-yarn pores between yarns and comparatively small intra-yarn pores between fibers [38]. Most of the water flows through the inter-yarn pores. Bubbles formed in the fabric were due to air pockets trapped inside yarns [39]. Thus, strong convection induced by transient bubble motion in the close vicinity of fabric enhances mass transfer through the fabric, leading to higher absorption of sonosynthesized nano-TiO2/SiO2 composites.
Except for ultrasound effects, TiO2/SiO2 composite nanoparticles had significant affinity towards hydroxyl group; hence, the dehydration reaction between the hydroxyl groups of fabric and the hydroxyl groups of TiO2/SiO2 resulted in interfacial bonding, which enabled good adhesion of nanoparticles onto the fabric surface [40].
SEM images
To investigate the morphology of the sonotreated and untreated samples, comparison between the SEM images of blank (pristine cotton–polyester fabric), TiO2-treated samples, and TiO2/SiO2-treated samples are shown in Figures 5(a) to (f). The results in Figure 5(a) and (b) illustrated that the surface morphology of blank sample was clean and smooth. Figure 5(c) and (d) depicted that fibers of TiO2-coated sample were homogeneously coated with nanoparticles resulting from sonication. The surfaces were obviously covered completely by TiO2 nanoparticles condensed as layer. In view of the results obtained from Figure 5(e) and (f) of sonotreated sample with TiO2/SiO2, some small spherical nanocomposites were observed. Overall, TiO2 and TiO2/SiO2 nanoparticles were well distributed on the fiber surface because of ultrasound irradiation. However, some agglomerated nanoparticles were observed on both TiO2- and TiO2/SiO2-treated surfaces. This finding may be due to the small dimension and large specific surface area of nanoparticles [41]. Although some small aggregations were observed, the presence of SiO2 still enhanced the self-cleaning function of fabric.
SEM images of cotton–polyester samples. (a, b) raw; (c, d) TiO2-coated fabric and (e, f) TiO2/SiO2-coated fabric.
Water-droplet absorption time
Figure 6 shows the shorter time of water-droplet absorption on TiO2/SiO2-coated fabric compared with pure TiO2-coated fabric. The water contact angle on untreated fabrics was 62.3° for 0.5 s after locating the water droplet. Compared with uncoated fabrics, water droplets took 225 and 135 ms to spread on the surfaces of TiO2- and TiO2/SiO2-coated fabrics, respectively. Applying the sols to fabrics induced an increase in the surface energy of fabrics, resulting in higher wettability [42]. Essentially, the surface energy of fabrics largely depended on the intermolecular forces existing on the surface and on the specific surface area [42]. Superhydrophilicity on TiO2/SiO2-coated samples in the presence of SiO2 could be justified by the higher surface acidity that provided a higher concentration of hydroxyl groups because of the presence of Ti–O–Si linkages [43]. Meanwhile, a charge imbalance that provided Lewis acid sites ensued, leading to the increased absorption of hydroxyl groups on the surface of TiO2/SiO2 composite nanoparticles [43].
Water droplets on samples: (a) pristine fabrics; fabrics treated with (b) TiO2 and (c) TiO2/SiO2.
Self-cleaning property
Self-cleaning property was compared by monitoring the removal of MB stain on fabrics at different UV-irradiation intervals. UV was used to trigger the photocatalytic decomposition of stains by TiO2 and TiO2/SiO2 composites [44,45]. Given that TiO2 nanoparticles had a relatively large surface area per unit mass and volume under UV rays with higher energy than their band gaps (3.18 eV), pairs of negative electrons (e−) and positive holes (h+) were produced [25]. These holes reacted with water molecules and generated powerful oxidative intermediates, such as hydroxyl radicals. These free radicals further participated in various chemical reactions with the latent capability of decomposing or degrading numerous organic pigments [46]. Figure 7 depicts less fading on the MB spotted blank during UV irradiation; nevertheless, all stained sonoprepared samples were decolorized either completely or partially. Consequently, the self-cleaning feature of TiO2/SiO2-treated samples was obviously higher than that of TiO2-treated samples. Based on the results, 2 h of UV irradiation on samples was sufficient to remove MB stains. During UV irradiation, cationic dye was transferred into the conduction band of excited TiO2 and SiO2, changing the configuration of stains and producing the degraded products that were mainly colorless [47].
Degradation of MB on pristine fabrics, TiO2-coated fabric, and TiO2/SiO2-coated fabric after 0, 1, 1.5, and 2 h of UV irradiation.
Washing durability
The self-cleaning durability of as-treated substrates can have important potential applications. The implication of self-cleaning was the localization of the reaction center on the semiconducting surface durably attached onto the substrate. To our best knowledge, self-cleaning durability is seldom reported. In this work, washing-durability tests of the treated fabrics as an ideal substrate were conducted, and results are presented in Figure 8. TiO2/SiO2-treated samples retained significant decoloration even after 30 times of washing, showing great washing durability compared with TiO2-treated samples.
Wash-durability effect on degradation results of MB stain on pristine fabric, TiO2-coated fabrics and TiO2/SiO2-coated fabrics before and after 2 h of UV light irradiation.
Durability improvement may be due to the low polycondensation levels of TiO2/SiO2 nanosols and the chemical bonding of hydroxyl groups between fabrics and nanosols. The origin of the UV-light-driven activity of TiO2/SiO2 can be attributed to the strengthened adsorption in the near UV-A (320–420 nm), the improvement of UV–Vis vacancies, and the red shift in adsorption edge, as discussed in sections “FTIR analysis” and “UV absorption properties”. The significance of SiO2 can be related to the improvement of the rate of electron transfer to oxygen, thereby decreasing the rate of recombination between excited electron and hole pairs.
In addition to the examination of MB-stain degradation on fabrics by imaging, the washing durability of treated fabrics was also quantitatively evaluated, and results are shown in Figure 9. The trends of MB photodegradation after UV 2 h in the presence of different treated samples were compared by plotting C/C0. The concentration of MB solution with pristine fabric did not significantly change, and the negligible decline in MB concentration can be attributed to slight water evaporation from the solutions during UV illumination. For TiO2-coated samples, MB was partially decomposed within 2 h, and C/C0 decreased from 1 to 0.62. TiO2/SiO2-coated fabric showed the highest MB degradation rate, with C/C0 decreasing from 1 to 0.16. At the same time, after 30 times of washing, these samples did not show significant change in photocatalytic activity towards MB degradation. The rate constants (k = −ln(C/C0)/t) of blank samples, TiO2 samples and TiO2/SiO2 samples before washing for the degradation of MB were 0.0204/h, 0.1438/h, and 0.4023/h respectively. Similarly, the K-value did not show significant variation after washing, which turned to 0.0202/h, 0.1232/h, and 0.3902/h respectively [48]. The good stability towards washing was a result of chemical bonding between fabric and TiO2/SiO2. The covalent linkages between the hydroxyl groups of fabric and TiO2 or TiO2/SiO2 systems played the main role in producing a durable loading of TiO2 or TiO2/SiO2 nanoparticles on fabric surfaces. Accordingly, the covalent bonding between fabric surfaces and TiO2 or TiO2/SiO2 could be established during sonication.
MB degradation in the presence of fabrics (unwashed (1) and washed 30 times (2)) under UV for 2 h. (a) Pristine fabric, (b) TiO2-coated fabric, and (c) TiO2/SiO2-coated fabric.
Antibacterial properties
Antibacterial activities of the raw and sonotreated fabrics were evaluated against both E. coli and S. aureus bacteria after a specified contact time. S. aureus is a pathogenic microorganism causing many diseases, such as toxic hock, cross-infection in hospitals, purulence, abscess, fibrin coagulation, and endothermic. Moreover, it is resistant to common antimicrobial agents [49]. Meanwhile, E. coli causes urinary tract and wound infections and is a popular test organism. To examine the efficiency of the antibacterial properties of sonotreated fabric, the percentage of bacteria killed in various samples are reported in Figure 10.
Antibacterial activities of untreated and treated samples against E. coli and S. aureus bacteria. (a) Blank, (b) TiO2-treated fabric, and (c) TiO2/SiO2-treated fabric. 1 – in dark, 2 – under UV illumination (365 nm, 12 W).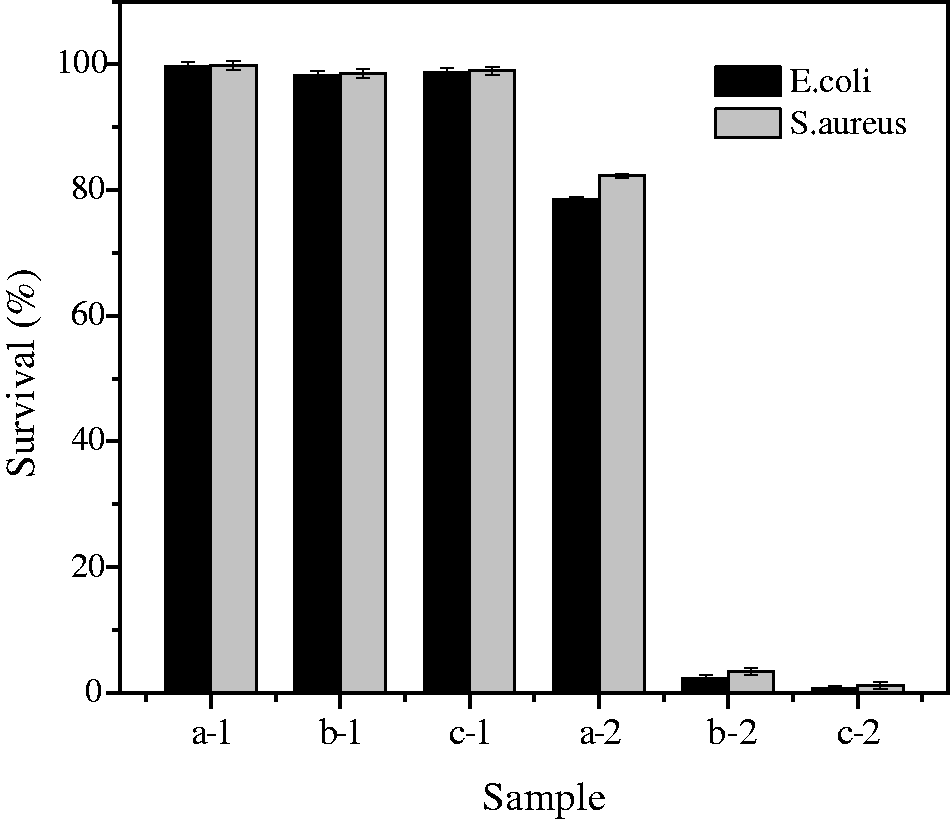
As shown in Figure 10, samples in darkness almost did not show antibacterial effect. However, TiO2 and TiO2/SiO2-treated samples showed highly significant antibacterial properties compared with the blank samples under UV illumination. In tests with E. coli and S. aureus, TiO2-fabric achieved 2.3% and 3.5% survival of bacteria, and TiO2/SiO2-treated fabric samples achieved 0.8% and 1.2% survival of bacteria, respectively. The possible mechanism of the bactericidal activity of TiO2 and TiO2/SiO2 nanoparticles was through an attack on reactive oxygen species such as hydroxyl radicals (OH•) and superoxide anions (O2•−) when photocatalysts were in direct contact with cells [50–52]. TiO2/SiO2 samples showed a slight improvement in antibacterial activities, which can be attributed to the high surface activity and porous structure of SiO2 nanoparticles. Moreover, the smaller size and more uniform distribution of TiO2/SiO2 nanoparticles than TiO2 nanoparticles on the fabric surface increased the antibacterial activity.
Figure 11 shows the results of S. aureus colony assay on an agar plate before and after UV illumination.
Images of S. aureus colonies on an agar plate before (1) and after (2) illumination. (a) Blank, (b) TiO2-treated sample, and (c) TiO2/SiO2-treated sample.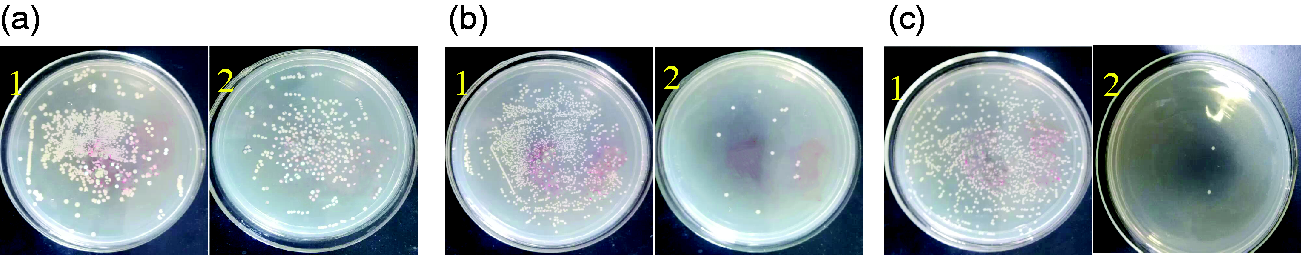
Tensile strength
Tensile strength and elongation of different samples.

Whiteness index
The whiteness indexes of all samples before and after exposure UVA irradiation are presented in Figure 12. The whiteness of the treated samples increased compared with the blank samples. This could due to nano-TiO2 is a white pigment coated the fabric surface producing a white color. However, integrating SiO2 into the system did not impact on the color of TiO2-coated fabrics dramatically before irradiation. For blank samples, the whiteness decreased after irradiation, which may due to impurities such as pectin or pesticide in cotton decomposed and turned yellow through UV irradiation. However, nano-TiO2 and TiO2/SiO2-treated samples after exposure to 10 h UVA irradiation generate oxidizing radicals and degrade the natural pigments such as pectin create whiter fabrics. Also, TiO2/SiO2-treated samples showed more obvious. This may because the linkages of Ti–O–Si results in a charge imbalance producing positive charge in photocatalyst. To offset the positive charge, a great deal of oxidizing radicals is attracted to the surface of photocatalyst. The decomposition rate of pigments is accelerated for the higher amount of oxidizing radicals attached to the photocatalyst.
Whiteness indexes of different samples before and after irradiation.
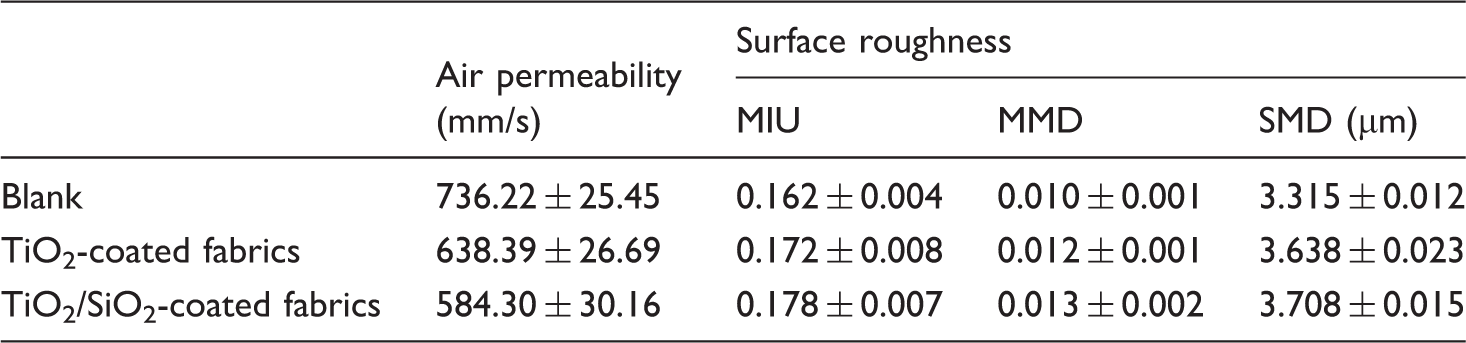
Ventilation property and surface roughness
Air permeability and surface roughness of untreated fabrics and fabrics coated with TiO2, TiO2/SiO2.
Conclusions
TiO2/SiO2 and pure TiO2 nanoparticles were sonosynthesized and loaded simultaneously onto cotton–polyester fabric. MB-decomposition measurements showed that TiO2/SiO2-treated fabrics possessed higher photocatalytic activity than pure TiO2-treated fabrics. This enhanced photoactivity was related to the smaller size of nanoparticles, the existence of Si–O–Ti, the superior hydrophilicity, and the wide absorption edge of UV irradiation of TiO2/SiO2 based on particle size, XPS, FTIR, and UV–Vis analyses. SEM images confirmed the presence of TiO2 and TiO2/SiO2 nanoparticles on the sonotreated fabric surface. Moreover, the washing durability confirmed the role of sonochemical coating and covalent bonding between fabric and nanoparticles. In addition, this treatment exhibited excellent antibacterial property against the Gram-negative E. coli and the Gram-positive S. aureus for both TiO2/SiO2 and pure TiO2. Finally, the synthesis method had no notable influence on the tensile strength, whiteness and air permeability of the treated fabric, as well as surface roughness.
The main conclusion was that ultrasonic irradiation played a synergistic role in the simultaneous synthesis and loading of TiO2/SiO2 on cotton–polyester fabric at a low temperature and was able to enhance the washing durability of this fabric.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Science and Technology Achievements Transformation and Industrialization Project in Shanghai (No. 14441901600), the Science and Technology Support Program of Jiangsu Province (No. BE2014036), the Fundamental Research Funds for the Central Universities, and the Donghua University Innovation Fund of Graduate Project (No. EG 2015001).
