Abstract
The adsorption kinetics of amide softeners on cotton fabrics in decamethyl cyclopentasiloxane (D5) medium is investigated. Pseudo-first-order and pseudo-second-order kinetic models are utilized to analyze the kinetic data. Simultaneously, the kinetic parameters are analyzed to explore the adsorption rate of amide softeners on cotton fabrics. The adsorption process is believed to follow pseudo-second-order kinetics. Based on SEM images, the adsorption of amide softeners on cotton fabrics in D5 medium occurs on the cotton surface. There is no observed diffusion of softeners into the cotton fibers. The adsorption rate of amide softeners on cotton fabrics in D5 medium is much faster than the traditional aqueous system, which may significantly improve the efficiency of softener finishing.
Keywords
Introduction
Decamethyl cyclopentasiloxane (D5) is a colorless, odor-less, transparent, and oily cyclic siloxane fluid, which is an eco-friendly medium and can be recycled after use.1–3 It is harmless to the human body, 4 thus it is widely used in cosmetics and human care products.3–5 In addition, D5 can be used as a carrier.6–8 Recently, D5 was used in the textile industry as a non-aqueous medium for eco-dyeing and washing, which represents an outstanding application.9-10 For instance, the dye uptake was nearly 100%. In addition, after use, the D5 contained hardly any dye and was readily recycled.11–19
Using D5 as a non-aqueous medium for cotton fabric softener finishing was investigated in our previous study. 20 It was observed that the high adsorption ratio (nearly 100%) of amide softeners was easily obtained at low temperature (25 °C), and the whole finishing process was wastewater free. Softener finishing in the D5 medium not only significantly increased the adsorption ratio of amide softeners, but also remarkably eliminated wastewater pollution, which fulfilled the goal of energy savings and emissions reduction.
Pseudo-first-order and pseudo-second-order kinetic models are applied to describe the adsorption of substances on solid surface in many fields. For instance, they are used in the adsorption kinetics of methyl violet onto perlite, 21 the kinetics of metal ions on peat,22,23 and the sorption of dye from aqueous solution by peat. 24 However, in the textile industry, the two kinetic models are generally applied to describe the absorption of dyes on different fabrics9,11,25,26 or other compounds.27–29 These models were used to investigate the adsorption kinetics of amide softeners on cotton fabrics for the softener/D5 suspension system and the traditional aqueous system.
Experimental
Materials
Decamethyl cyclopentasiloxane (D5) (99.4%) was provided by GE Toshiba Silicone Ltd. (Tech). The amide softener, TF449B (> 98%, Fig. 1), was obtained from a local chemical company (Tech). TF449B was cationic and dissolved in water at 60 °C, yielding a pH value of 5–6 for a 1% concentration. The fabric used in the study was a 100% cotton towel (464.21 g/m2) provided by a local textile company. The cotton towel was precut into small pieces (10 ± 0.01 g) and washed in water (40 °C) for 6 h to remove any trace of surfactant and dust.
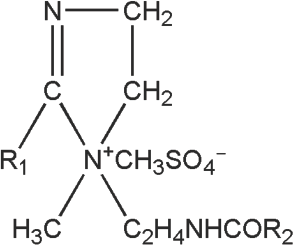
Structure of TF449B softener.
Preparation of the Softener/D5 Suspension System
TF449B (2 g) was added into D5 (20 mL) to form a mixture. The mixture was then moved into a tube in QM-3SP4J planetary grinding machine (Nanjing University Instrument Factory). Zirconia grinding beads (50 g) were also placed into the tube. The mixture was ground for 3 h to obtain the softener and D5 suspension paste in high concentration. During the finishing process, the high-concentration suspension paste was diluted wth D5 to the required concentration (1% owf).
Preparation of the Traditional Aqueous System
TF449B (2 g) was added into cold water (100 mL, 25 °C). The aqueous solution of TF449B was heated to 60 °C by stirring the mixture continuously in a Eurostar 60 digital agitator (IKA) until the TF449B was completely dissolved in water. The traditional aqueous system of softener was obtained in high concentration. When used, it was diluted to 1% owf.
Softener Finishing Process
The basic softener finishing processes for the softener/D5 suspension system and the traditional aqueous system are presented in Figs. 2a and b, respectively. For the softener/D5 suspension system, no water was used in the whole finishing process. The dry cotton fabrics were immersed into the suspension system containing 1% owf softener, which was maintained at 25 °C. During the finishing process, the cotton fabrics were treated with different finishing times. The raffinates at different finishing times were collected. The fabrics:D5 ratio was 1:10.
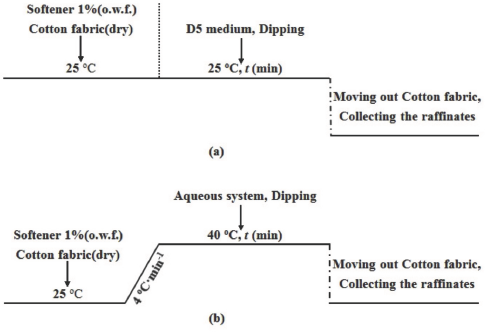
The finishing processes (a) in the TF449B-D5 suspension system and (b) in the aqueous system.
For the aqueous system, the finishing process was similar to that of softener/D5 suspension system. The only difference was that the finishing process was performed at 40 °C instead of 25 °C. For this reason, after immersion of dry cotton fabrics into the aqueous system maintained at 25 °C, the system was slowly increased to 40 °C at a rate of 4 °C/min.
Scanning Electron Microscopy (SEM)
The cotton yarns were directly removed from the cotton towel, straightened, and then placed together to form a square with a size of 2 × 5 cm. To simulate the adsorption of TF449B on the surface of cotton fabrics, the softener/D5 suspension system at 1% owf concentration was dropped on these cotton yarns to homogeneously cover them. Ten the cotton yarns were dried naturally under ambient conditions at 20 °C. Finally, the cotton yarns, with and without soft-ener, were measured after gold plating by scanning electron microscopy (SEM) using a JSM-5610LV instrument (JEOL Ltd.) with a magnification of 2000×.
Adsorption Rate Measurements
Transmittance values of the softener/D5 suspension system and the aqueous system were measured at 600 nm using a UV-Vis spectrophotometer (PerkinElmer) at different concentrations (0.00, 0.04, 0.08, 0.12, 0.16, 0.20, 0.24, 0.28, and 0.32 g/L). The transmittance as a function of concentration, used as the standard working curves for TF449B both in D5 and water media, were plotted respectively.
For each cotton fabric, one part of the collected raffinate was analyzed by UV-Visible (UV-Vis) spectrometry. Conse-quently, the adsorption ratio of TF449B, namely
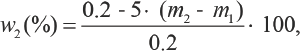
The error of the standard working curves,
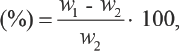
Pseudo-First-Order Kinetic Model
The pseudo-first-order kinetic model can be expressed by Lagergren equation (Eq. 3).23–27 If the change curve of
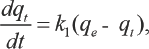
When the Lagergren equation is used to analyze kinetic data, it is generally linearized as in Eq. 4. It describes the linear relationship between ln(
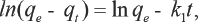
Pseudo-Second-Order Kinetic Model
The pseudo-second-order kinetic model is be represented in Eq. 5.21,22,28,29 Its linear form is shown in Eq. 6.
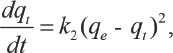
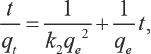
The value of R 2 determines whether the adsorption behavior of TF449B accords with the pseudo-second-order kinetic model or not.
Results and Discussion
Standard Working Curves
To quantify the adsorption ratio of TF449B, the standard working curves of TF449B in D5 medium and water were evaluated. Both the softener/D5 suspension system and the traditional aqueous system liquid mixtures were opaque. The transmittance of the liquid mixtures was significantly influenced by TF449B concentration (Fig. 3). The transmittance decreased with increased TF449B concentration.
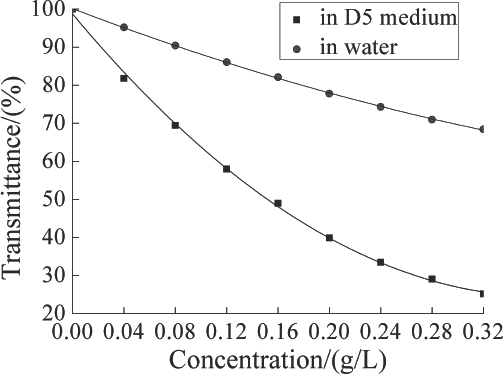
Transmittance vs. TF449B concentration in D5 and water media.
For both systems, Fig. 3 indicates that the relationship between the transmittance and the concentration of TF449B were quadratic. The two equations can be described by Eq. 7, and the R 2 is presented in Table I.
TF449B Adsorption Standard Working Curves in Different Media

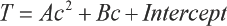
The plots of Eq. 7 for both D5 and water media had R 2 values of 0.999, which indicates the equations accurately described the relationship between the transmittance of the finishing liquid and the TF449B concentration (Table I).
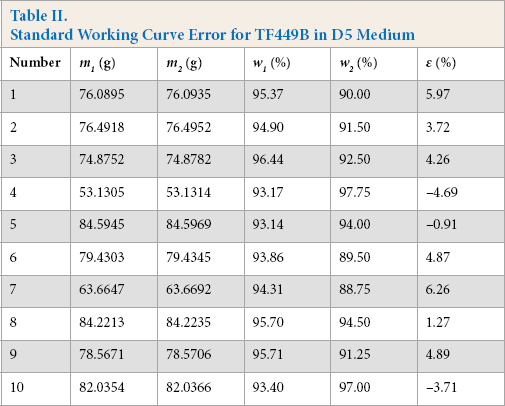
Standard Working Curve Error
To investigate the accuracy of the standard D5 medium working curve, the error was calculated (Table II). The maximum error observed was within ± 6.26%, which was acceptable. Therefore, it can be concluded that the adsorption ratio of TF449B in D5 medium was accurately expressed by the standard working curve. In addition, the applied method was simple and rapid.
Standard Working Curve Error for TF449B in D5 Medium
SEM Images
Because there were a large number of cotton loops on the surface of the cotton towel, the cotton towel showed a heterogeneous and rough surface. For this reason, it was difficult to measure the surface of the cotton towel by SEM. However, when cotton yarns withdrawn from the cotton towel were straightened and then placed together side-by-side, almost all of the cotton yarns were on the same plane. Hence, the quality of SEM images was significantly improved, with the distribution of TF449B on the fiber surface easily observed (Figs. 4a and b).
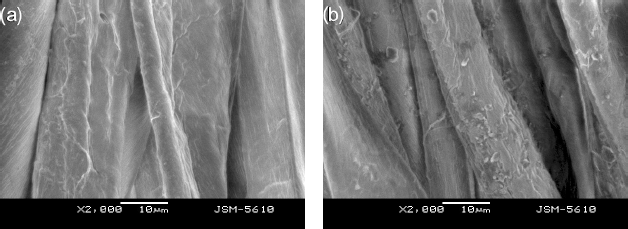
SEM images at 2000× of (a) original cotton samples and (b) cotton yarns treated with the TF449B -D5 suspension system at 1% concentration.
From Fig. 4a, the surface of cotton fibers without TF449B were clean and smooth. Compared to the original cotton, the cotton treated by the softener/D5 suspension system at a 1% concentration was adsorbed as many granular solids on the fiber surface (Fig. 4b).
Only D5 and TF449B were present in the softener/D5 suspension system. Additionally, D5 can be easily volatilized at high temperature, whereas TF449B is very stable at high temperature. For these reasons, during the drying process of the cotton yarns treated with the softener/D5 suspension system, D5 should gradually volatilize into the surrounding atmosphere, and TF449B should stay on the cotton fibers. Therefore, it is suggested that the granular solids on the surface of cotton fibers were composed of TF449B.
D5 is a cyclic molecule constituted by silicon-oxygen bonds and TF449B is an amide softener that possesses an aliphatic chain. Therefore, the affinity between TF449B and D5 is extremely low. However, there are a large number of hydroxyl groups on the surface of cotton fibers. Hence, the affinity of TF449B for cotton fibers is very good. TF449B can easily adhere to cotton fibers by Van der Waals forces. Consequently, TF449B should easily transfer from the D5 medium to the cotton fiber surface.
Adsorption Kinetic Curves
To study the adsorption of TF449B on cotton fabrics in the D5 medium and in water, the finishing temperatures of the softener/D5 suspension system were set at 25 °C and 40 °C, and the finishing temperatures of the aqueous system at 40 °C and 50 °C. The experimental results are presented in Fig. 5.
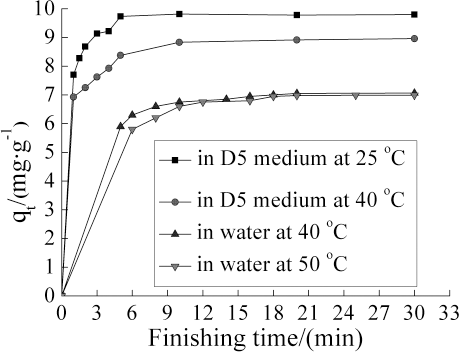
TF449B adsorbed vs. finishing time in different media at different temperatures.
For the softener/D5 suspension system, finishing temperature had a huge influence on the adsorption rate of TF449B and the adsorption maximum. When the finishing temperature was 25 °C, the
For the traditional aqueous system, the curves showed the same general increase as that in D5 medium, but the influence of finishing temperature was not so prominent to the adsorption rate of TF449B and the adsorption maximum. Furthermore, it took at least 20 min to achieve adsorption equilibrium, which indicated a slow rate of adsorption in water. A possible reason for the fast adsorption of TF449B (Fig. 5) onto cotton from D5 was the difference in affinity of the softener for D5 and cotton. Once TF449B in D5 contacted cotton fibers, it preferred to stay on the fibers. Because of TF449B's high affinity in D5 for cotton fibers, the amount of TF449B adsorbed on cotton fibers at equilibrium in the D5 medium was extremely high (nearly 100%), compared to that in the water medium (Fig. 5).
TF449B can be easily dissolved in water at about 60 °C, indicating that TF449B has a good affinity with water, as well as with cotton fibers when the TF449B in the water medium was used. The adsorption of TF449B on cotton fibers in the aqueous system depended on the distribution of TF449B between water and cotton fibers. Although TF449B in the water medium had a good affinity with cotton fibers, it was quite difficult for TF449B to achieve a high adsorption on cotton fibers in a short time. As a consequence, the adsorption rate of TF449B in the water medium was slower than that in the D5 medium. TF449B in the water medium took a longer time to reach an equilibrium state of adsorption than that in D5 medium. At the equilibrium state, the adsorption maximum of TF449B depends on its distribution between water and cotton fibers.
Pseudo-First-Order Kinetic Model Fitting
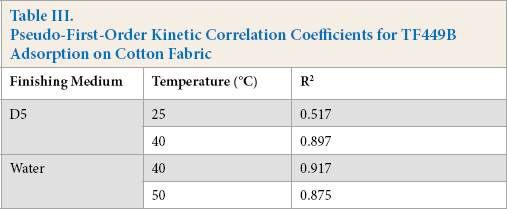
The adsorption of TF449B at different temperatures on cotton fabrics was investigated to explore whether it accorded with the pseudo-first-order kinetic model or not. The fitting line plots of the equations in the D5 and water media are shown in Figs. 6a and b, respectively. The distribution of all the data was equivalently scattered. Table III shows the values of R
2
at different temperatures in the two systems. Both systems show very low correlation coefficients (R
2
< 0.92), showing that the linear equation was not suitable to describe the adsorption process of TF449B on cotton fabrics in both D5 and water media. This implies that the change curve of
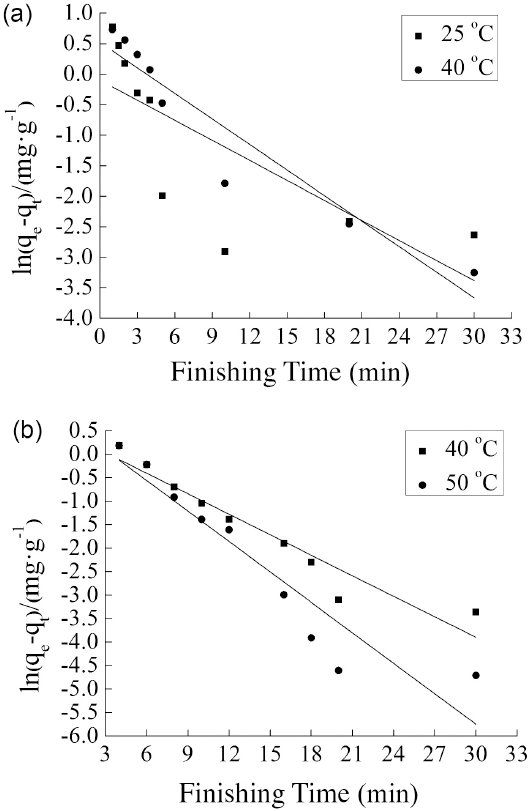
Pseudo-first-order-kinetics plots for TF449B adsorption from (a) D5 medium and (b) water medium.
Pseudo-First-Order Kinetic Correlation Coefficients for TF449B Adsorption on Cotton Fabric
Pseudo-Second-Order Kinetic Model Fitting
Because the pseudo-first-order kinetic model did not describe the adsorption of TF449B in the softener/D5 suspension system and the traditional aqueous system, the pseudo-second-order kinetic model was investigated. The fitting line plots of
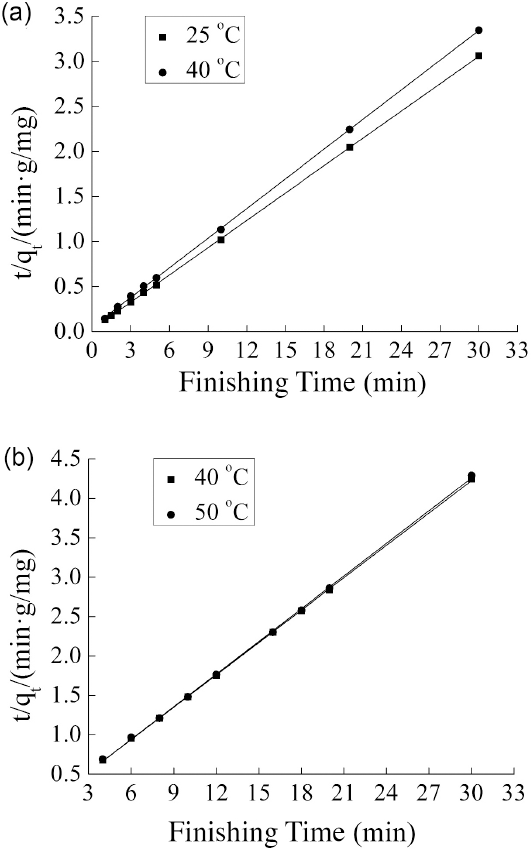
Pseudo-second-order-kinetics plots for TF449B adsorption from (a) D5 medium and (b) water medium.
Pseudo-Second-Order Kinetic Correlation Coefficients for TF449B Adsorption on Cotton Fabric

The slopes of the fitting lines for D5 media at 25 °C and 40 °C were different (Fig. 7a), which was caused by the different adsorption ratios of TF449B at these two temperatures. However, using the aqueous system, because the adsorption ratio of TF449B was basically the same at 40 °C and 50 °C, the two fitting line plots of
Furthermore, for the softener/D5 suspension system, the
From the above, it can be concluded that the pseudo-sec-ond-order kinetic model can well describe the adsorption behavior of TF449B on cotton fabrics in both D5 and water media. In addition, TF449B was observed to absorb on the surface of cotton fibers by SEM.
Conclusions
The adsorption kinetics of amide softeners on cotton fibers were investigated in D5 and water media. It was shown by SEM that TF449B only absorbed on the surface of cotton fibers. The adsorption ratio of amide softeners was quantitatively and accurately characterized by the standard working curves of transmittance as a function of concentration. In addition, a high correlation coefficient (0.999) was obtained when the adsorption data were modeled by pseudo-second-order kinetics. The pseudo-second-order kinetic model agreed well with the dynamic behavior of amide softener adsorption on the cotton fabrics surface in D5 and water media under various initial softener concentrations and temperatures. The adsorption rate of TF449B in the D5 medium was significantly faster than that in water, and the equilibrium adsorption in the D5 medium (nearly 100%) was also higher than that in water. In conclusion, the softener/D5 suspension system is a promising candidate for water-free softener finishing.
