Abstract
Periostin-like factor (PLF) and Periostin are alternatively spliced mRNAs. Our findings are the first to show similarities and differences between PLF and Periostin location using isoform-specific antibodies. The differences in when and where they are present during mouse embryogenesis suggest that they may have different functions. Using immunostaining techniques, we observed that PLF was highly expressed at 12.5 days postconception (dpc) in the intermediate and outer zones of most brain regions, spinal cord, cranial and spinal nerves, and chondrocytes in developing bone and in the heart wall. By 16.5 dpc, PLF was also present in ameloblasts and odontoblasts in developing teeth, and by 19.5 dpc, PLF was present at low levels only in vagal nerve bundles, discrete white matter bundles in the brain, and chondrocytes of developing ribs. Periostin, on the other hand, was absent at 12.5 dpc from dorsal spinal cord and from cranial and spinal nerves. By 16.5 dpc, Periostin was present in many spinal nerves, but absent thereafter, and at 19.5 dpc, Periostin was present in chondrocytes in developing bone but not in neural tissues. The different spatial and temporal location of PLF and Periostin in cartilage and bone cells suggests different roles for these proteins in endochondral bone formation. The early expression of PLF in brain differentiation zones and in developing axon bundles and nerves suggests that it may facilitate axon growth.
P
Sequence analysis of PLF identified an N-terminal signal sequence, suggesting that the protein is secreted; a putative nuclear localization sequence (NLS), suggesting that it may be translocated to the nucleus; one potential N-linked glycosylation site; and four fasciclin domains each containing 150 amino acids (Litvin et al. 2004). Proteins that contain fasciclin domains are related to Fasciclin, identified in insects (Zinn et al. 1988). In both Drosophila and grasshoppers, fasciclin I is expressed on the surface of a subset of commissural axon pathways in the embryonic central nervous system (CNS) and on sensory axonal pathways in the peripheral nervous system (PNS) (McAllister et al. 1992). Although the details on the molecular mechanisms involved are unclear, fasciclin I mediates interactions between cell surfaces in the nervous system. Therefore, in examining the temporal and spatial location of PLF, we paid particular attention to the developing nervous system.
We rely on data obtained from studies on βIG-H3 and Periostin to provide clues about the role of PLF in the structure and function of cells. This is the first report to examine differences between PLF and Periostin localization during embryogenesis using isoform-specific antibodies. βIG-H3 was first identified in adenocarcinoma cells treated with transforming growth factor-β (Skonier et al. 1992). It has a signal sequence at the N terminus, an Arg-Gly-Asp sequence at the C terminus, and four Fas domains (Skonier et al. 1992). Transcripts of βIG-H3 are detected in connective tissues including cartilage during embryogenesis (Ferguson et al. 2003). βIG-H3 is expressed in preosteoblasts, mediates osteoblast adhesion, and inhibits osteoblast differentiation (Thapa et al. 2005). In addition, it is secreted from various cell types and is detected in nuclei of human bladder smooth muscle cells and fibroblasts (Billings et al. 2000). The major function of βIG-H3 as a secreted protein is to mediate cell spreading, adhesion, proliferation, and migration (reviewed by Litvin et al. 2005).
Periostin was first identified in MC3T3-E1 osteoblast-like cells. The major difference between PLF and Periostin is at the C-terminal region (Litvin et al. 2004). Periostin is expressed in osteoblasts in vitro and in periosteum and periodontal ligament tissues in vivo. It has several isoforms (PLF being one of them). Periostin is secreted and supports MC3T3-E1 cell adhesion and spreading (Horiuchi et al. 1999). Periostin is expressed in the teeth and its surrounding tissues during development but not in CNS tissues (Goetsch et al. 2003; Suzuki et al. 2004). It is also expressed and functions in other cell types and tissues, as reviewed by Litvin et al. (2005). Mice deficient in Periostin display dwarfism, incisor enamel defects, and an early-onset periodontal disease–like phenotype (Rios et al. 2005). Moreover, it has been shown that Periostin is regulated by Twist, a transcription factor, which is essential for intramembranous ossification (el Ghouzzi et al. 1997; Oshima et al. 2002).
In our previous study, we showed using in situ hybridization that Periostin isoform mRNAs are detected in cartilage and developing bone at 16.5 days postconception (dpc) (Litvin et al. 2004). However, we have yet to explore the localization of each isoform, Periostin, and PLF protein in other tissues. The primary objective of this paper was to examine the temporal and spatial expression of PLF and Periostin immunohistochemically using PLF and Periostin-specific antibodies in developing mouse embryonic tissues and to compare their spatial and temporal location.
Materials and Methods
Tissue Collection
Mouse embryos that were 12.5, 13.5, 16.5, and 19.5 dpc were collected from mouse dams after euthanasia using sodium pentobarbital (120 mg/kg body weight). Three to five embryos were collected per age group. The embryos were immersion fixed in 4% paraformaldehyde in phosphate buffer (pH 7.4), processed for paraffin embedding, and sectioned. Sections were 5 μm thick, were cut in a cephalic to caudal direction along the long axis, and were mounted on charged and coated glass slides (Fisher Plus; Fisher Scientific, Fair Lawn, NJ). Animals (dams) were maintained and used according to the principles in the National Institutes of Health Guide for the Care and Use of Laboratory Animals (US Department of Health and Human Services, Publication No. 86-23, 1985) and guidelines established by the Institutional Care and Animal Use Committee of Temple University.
Western Blot Analysis
Tissues were homogenized in radioimmunoprecipitation assay (RIPA) buffer (50 mM HEPES, pH 7.5, 150 mmol NaCl, and 0.1% Tritron X-100) containing protease inhibitors (Sigma; St Louis, MO). Cells in culture were washed in PBS, and proteins were extracted in RIPA buffer containing protease inhibitors. Lysates were incubated on ice for 20 min, passed through an 18-gauge needle twice, and centrifuged at 3000 rpm for 10 min at 4C. Protein concentration was determined using the BCA kit (Pierce; Rockford, IL). Equal protein from each sample (100 μg/lane) was separated by PAGE, and the proteins were transferred to nitrocellulose. The blots were probed with affinity purified polyclonal antibodies specific for either Periostin or PLF. The Periostin-specific antibody was directed against a peptide specific to Periostin (KFIEGGDGHLFEDEEIKR aa 764–781; Invitrogen, Carlsbad, CA), and the PLF-specific antibody was directed against a peptide specific to PLF (EPKIKVIQGSLQPIIKTEG aa 681–699; Litvin et al. 2006). Primary antibody binding was detected with horseradish peroxidase–conjugated goat anti-rabbit secondary antibody and visualized by enhanced chemiluminescence (Amersham; Arlington Heights, IL).
Immunohistochemistry
Paraffin-embedded mouse embryo sections at 12.5, 13.5, 16.5, and 19.5 dpc were deparaffinized and incubated in 1.5% H2O2 for 15 min at room temperature. The slides were blocked with 5% goat serum in PBS for 1 hr and incubated with PLF antibody at a 1:1000 dilution overnight at 4C or in Periostin antibody at a 1:750 dilution overnight at 4C. The PLF-specific antibody is described by Litvin et al. (2006). Subsequent secondary antibody and substrate-chromogen development was carried out using a diaminobenzidine peroxidase kit (Vector Laboratories; Burlingame, CA). Sections were counterstained with nuclear stains, such as hematoxylin, nuclear red, or 4′,6-Diamidino-2-phenylindole, dihydrochloride (the latter as a fluorescent mounting media from Vector Laboratories), or with 1% Alcian blue (a cartilage counterstain). Some sections were also co-immunostained using immunofluorescent techniques after the PLF staining for PGP9.5, a general nerve marker used here to identify nerve fascicles (cat. no. ab8189; Abcam, Cambridge, MA). Sections were first incubated in 10% goat serum with 0.3% triton in PBS for 1 hr and then in the PGP9.5 antibody at a 1:50 dilution in PBS overnight at room temperature. A goat anti-mouse Cy3 (red fluorescent tag; Jackson ImmunoResearch Laboratories, Inc., West Grove, PA) was used for detection. Omission of the primary antibody or replacement of the antibody with normal serum was used as negative controls.
Results
To characterize the temporal and spatial localization of PLF and Periostin in musculoskeletal tissues under conditions of increased work load, inflammation, and injury, we raised PLF-specific and Periostin-specific antibodies against a peptide located in exons 17 and 21, respectively (Litvin et al. 2004). Exon 17 is present in the PLF transcript and not in Periostin, whereas exon 21 is present in Periostin and not in PLF. To confirm the specificity of the PLF antibody, we analyzed total protein isolated from 20.5 dpc embryonic mouse heart, the tissue used to identify PLF (Litvin et al. 2004, 2006). The affinity purified PLF antibody detected a single band corresponding to the full-length PLF protein (molecular mass, 90 kDa; Figure 1Aa). To confirm specificity of the PLF antibody, we competed the PLF antibody with the antigenic peptide (from exon 17) before Western blot analysis and showed that the 90-kDa band was eliminated (Figure 1Ab). In addition, we expressed PLF by adenovirus in COS cells, which are cells that do not express endogenous PLF. Total protein isolated from these cells was analyzed by Western blot, which, when reacted with PLF antibody, identified a single band at 90 kDa. The PLF antibody did not react with recombinant Periostin purchased from GenWay Biotech, Inc. (San Diego, CA) (Figure 1B), confirming the specificity of the PLF antibody. Because exons 17 and 21 are the same size, both Periostin and PLF are estimated to be 90 kDa (Litvin et al. 2004). Next we showed that, in proteins isolated from 17.5 dpc mouse embryonic femurs, the Periostin-specific antibody recognized a band at 90 kDa (Figure 1C). The specificity of the Periostin antibody was also tested by Westernblot against the recombinant protein purchased from GenWay (Figure 1C).
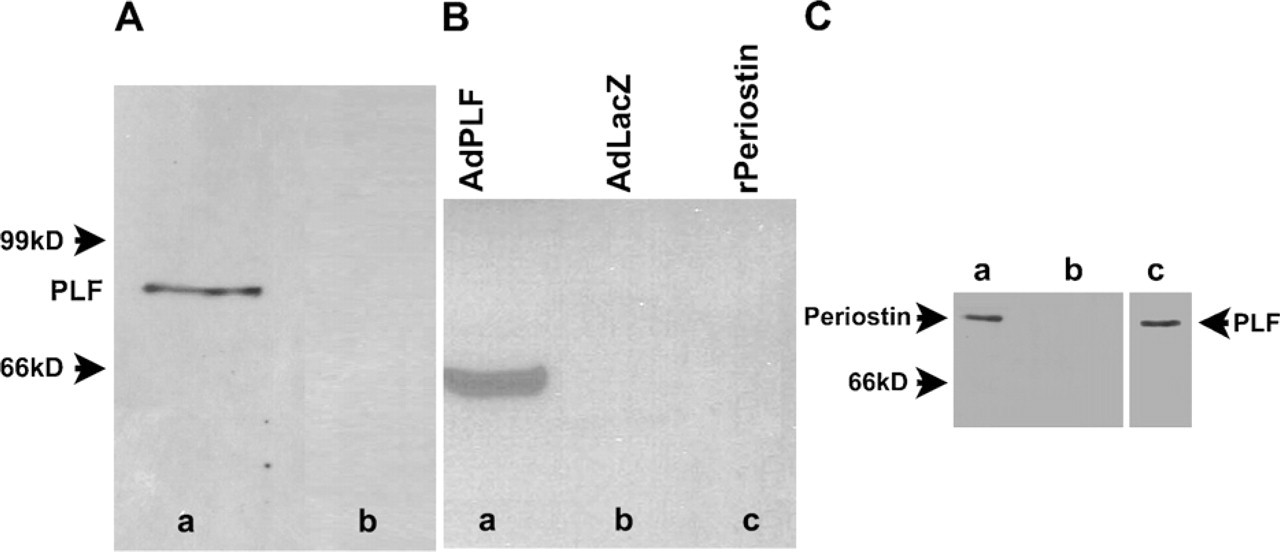
Specificity of Periostin-like factor (PLF) and Periostin-specific antibodies. (
The spatial and temporal localization of both the PLF and Periostin proteins were examined during embryogenesis at 12.5 to 19.5 dpc. These times during embryogenesis where chosen to incorporate stages when organ systems begin to form at 12.5 dpc and mature through 19.5 dpc. Between 12.5 and 19.5 dpc, the various cell types differentiate in the various organ systems. At 12.5 dpc, mesenchymal cells are beginning to condense, and cartilaginous templates for the ribs, vertebrae, limbs, and jaws were observed (Figures 2B and 2H). As development proceeds, at 16.5 dpc, endochondral bone formation is typified by the presence of ossification centers, allowing us to examine the expression of both isoforms in differentiating chondrocytes at various stages of maturation. In addition, we present data on the presence of PLF and Periostin in central and peripheral neural tissues to ascertain whether it has an early and transient expression pattern that may suggest a role in neurogenesis or axonogenesis.
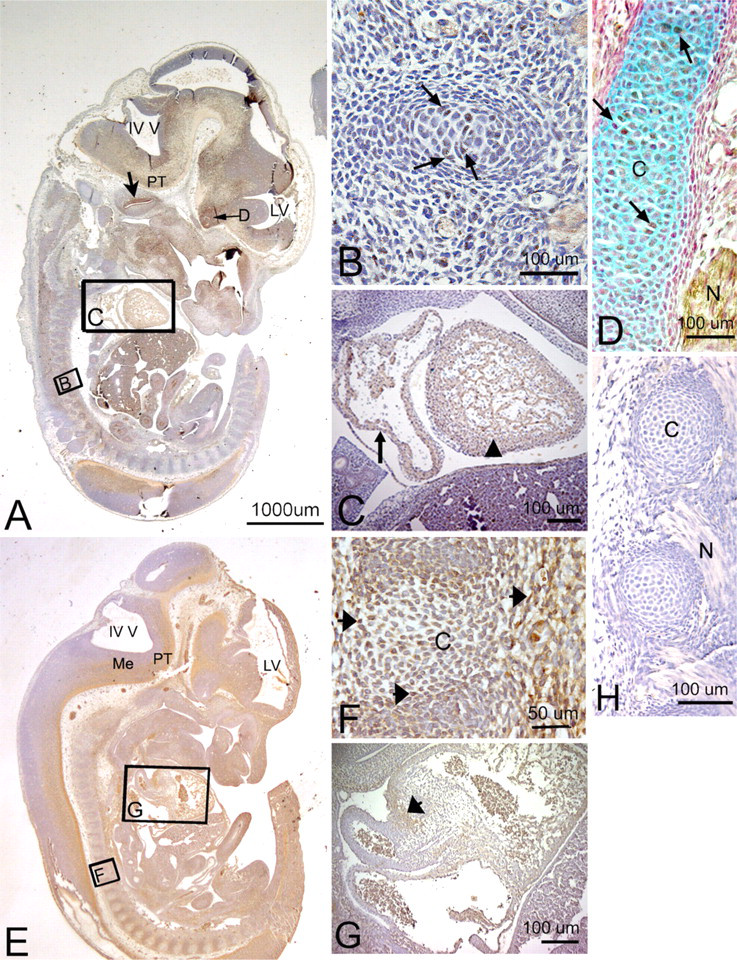
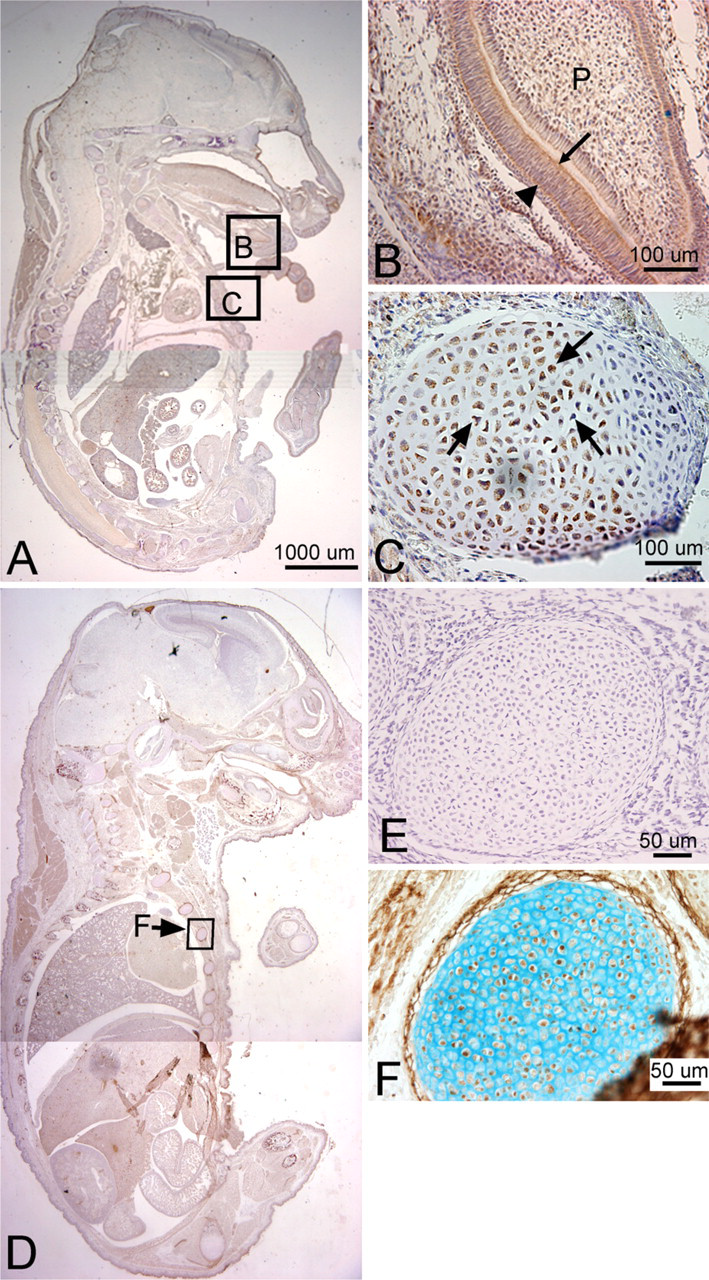
Localization of PLF and Periostin proteins in developing cartilage and myocardium in a mouse embryo at 12.5 days postconception (dpc). Sectioned embryo was probed with PLF antibody (
Localization of PLF in nervous system in embryonic tissues
E, embryonic; V, trigeminal nerve; VII, facial nerve; VIII, vestibulocochlear nerve; IX, glossopharyngeal nerve; X, vagus nerve; +, presence detected immunohistochemically, +++ > ++ > + in staining intensity; −, no staining; NA, not assessed.
At 12.5 dpc
PLF immunoreactivity in mouse embryonic vertebral cartilage at 12.5 dpc was weak (Figures 2B and 2D). Relatively higher staining was detected in the atrium and ventricle of the heart (Figure 2C). In the nervous system, at a stage associated with the onset of neurogenesis and axon outgrowth, intense staining was detected throughout the brain and cord and in processes associated with cranial and spinal nerves (summarized in Table 1). For example, the upper border of the cochlear lumen contained PLF-stained cells and processes, as did the diencephalon region of the optic stalk (Figure 2A). The presence of PLF in the ganglionic eminence (Figures 3A and 3B), medulla, and pons was restricted to the outer differentiating zones (Figure 3C). PLF was absent from the ventricular zone of the ganglionic eminence, presumptive pons, and medulla (Figures 3A–3C), a region that consists of progenitor-undifferentiated cells. PLF was present in the posterior commissure (Table 1) and in longitudinal axon bundles throughout the midbrain and hindbrain, including the pyramidal tract (the cortico-motor descending track; Figures 3B and 3C). Axons bundles from several cranial nerves, the trigeminal, glossopharyngeal, and vagus, as well as spinal nerves associated with the dorsal root ganglion, were intensely positive for PLF (Figures 2D, 3E, and 3F). The PLF-stained vagus nerve could be traced into the thoracic region (Figures 2D and 3E), and a lumbosacral spinal nerve could be traced into the hindlimb (Figure 3B, F). Staining was not detected at this stage in the sensory neuronal cells of the dorsal root ganglion (Figure 3E). Moreover, PLF expression in the basal plate of the spinal cord, which corresponds to the ventral horn of the spinal cord (motor), contained intensely stained axon processes and cells (Figures 3A, 3B, and 3D). This staining pattern contrasts with the more lightly stained alar plate that corresponds to the dorsal horn of the spinal cord (sensory) (Figures 3A and 3D). Table 1 contains a list of other central nervous system structures that stained for PLF.
Localization of PLF (
Periostin immunoreactivity in mouse embryonic vertebral cartilage at 12.5 dpc was widespread throughout the mesenchymal cells (Figure 2F). Staining was also detected in cushion cells in the heart (Figure 2G). In the nervous system, Periostin staining was localized to ventral brain and spinal cord regions at 12.5 dpc (Table 2; Figures 2E and 3G) but absent from cranial and spinal nerves (Figure 3I). For example, Periostin was detectable in the ventral ganglionic eminence, in the outer differentiating zones of the medulla and pons (Figures 2E and 3I), and in the pyramidal tract (Figure 2E). However, in contrast to PLF, no Periostin immunoreactivity was detected in axons of most cranial and spinal nerves (Figure 3I). Although Periostin expression was detected in the basal plate of the spinal cord, none was detected in the alar plate (Figure 3G).
At 13.5 dpc
Relatively low levels of PLF continued to be detected in cartilaginous vertebrae and ribs and in the heart (Figure 4A). Also, low levels of PLF staining could be detected in the ganglionic eminence differentiating field, presumptive medullary and pons differentiating fields, pyramidal tract, and cranial portions of the vagus nerve (Figures 4A and 4B). Strong staining for PLF was detected in the hippocampus ventricular zone and differentiating field, upper edge of the cochlear lumen, and in lumbosacral neuronal processes passing from the spinal cord between developing vertebrae (Figures 4A–4C; Table 1). PLF could also be detected only at very low levels in developing vertebral cells at this stage (Figure 4D).
Higher levels of Periostin than PLF could be detected in cells of cartilaginous vertebrae and ribs and in the heart (compare Figures 4E and 4G to 4A and 4D). Also at 13.5 dpc, Periostin staining could be detected in the ganglionic eminence differentiating field, presumptive medullary and pons differentiating fields, and pyramidal tract (Figures 4E and 4F; Table 2). However, in contrast to PLF, Periostin was absent from the cochlear (compare Figure 4B to 4F). Periostin was clearly detected in developing vertebral chondrocytes (Figure 4G) and in lumbosacral neuronal processes (Figure 4H; Table 2), the latter in contrast to Periostin at 12.5 dpc (which showed no staining of peripheral nerve processes; Figure 3I; Table 2).
At 16.5 dpc
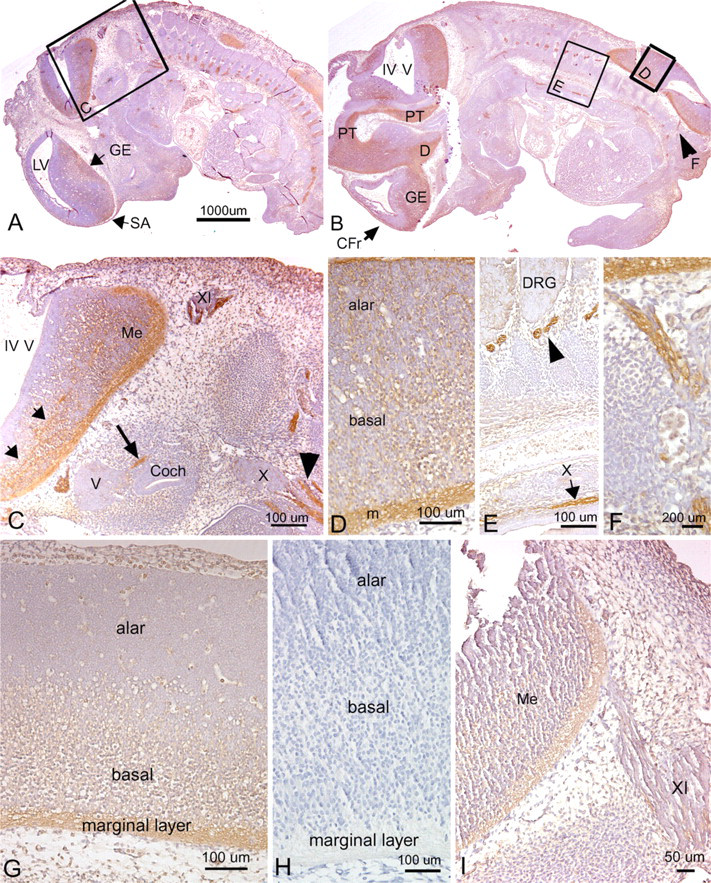
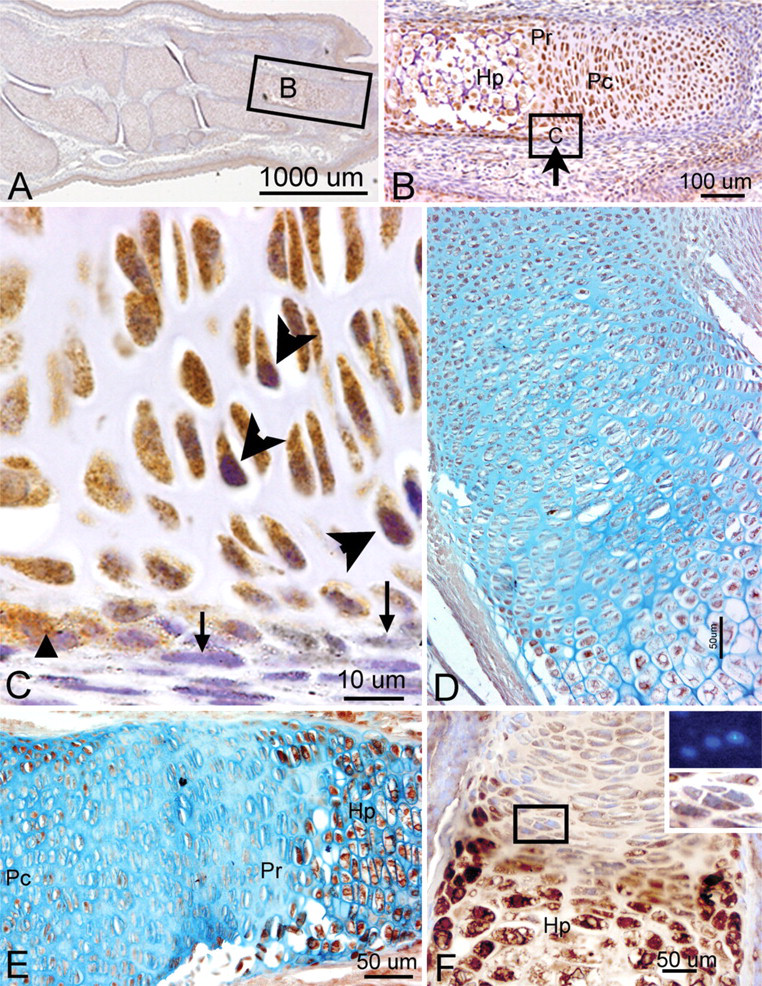
We detected a robust amount of PLF protein in ameloblasts, odontoblasts, cells in the dental pulp of developing teeth at 16.5 dpc (Figure 5B), and chondrocytes in the developing ribs (Figure 5C, arrow) and vertebrae (data not shown). Also, intense PLF staining was detected in developing taste buds, tongue muscles, and adrenal glands (Figures 6A, 6B, and 6D). In neural tissues, PLF protein was detected in the cytoplasm of dorsal root ganglion cells (Figure 6C and inset), staining that was absent at earlier embryonic ages (Figure 3E). By this stage, most PLF staining in brain structures (Figure 6A) had reduced dramatically compared with earlier ages and compared with spinal cord and pituitary staining in these same embryos (Figures 6E–6G; Table 1). In the spinal cord, PLF-positive axon bundles could be followed in longitudinal and dorsoventral directions (Figure 6E) and in the cytoplasm of large neurons located in the basal plate (presumptive ventral horn) of the spinal cord (Figure 6F). In the pituitary gland, discrete areas of intense PLF staining were visible at the outer edges of this gland (Figure 6G). Also at this stage, PLF was detected in proliferating and hypertrophic chondrocytes of the limb (Figures 7A–7D). Interestingly, intense staining was detected in mesenchymal preosteoblasts (bony collar, small arrowhead) and not in cells making up the perichondrium (Figure 7C, small arrow), suggesting that PLF plays a role in osteoblast development. This difference in staining (PLF expression in the bony collar and not in the perichondrium) is also seen at lower magnification in Figure 7B, where the arrow demarcates the boundary between the perichondrium and the bony collar. In addition, PLF showed both cytoplasmic and nuclear localization (Figure 7C, large arrowheads), which supports the predicted amino acid sequence analysis that identified a nuclear localization signal in PLF (Litvin et al. 2004).
Localization of Periostin in nervous system in embryonic tissues
E, embryonic; V, trigeminal nerve; VII, facial nerve; VIII, vestibulocochlear nerve; IX, glossopharyngeal nerve; X, vagus nerve; +, presence detected immunohistochemically, +++ > ++ > + in staining intensity; −, no staining; NA, not assessed.
Periostin immunolocalization was primarily detected in the mesenchymal cells of the perichondrium and to a lesser extent in the chondrocytes of developing bones (Figure 5F). However, Periostin, unlike PLF, was not present in spinal cord or brain regions of embryos that were 16.5 dpc (Figure 5D). In the limb, Periostin staining was strongest in the hypertrophic regions of developing forelimb bones (Figures 7E and 7F) but less visible in the prehypertrophic and proliferating regions (Figures 7E and 7F).
Localization of PLF (
Localization of PLF (
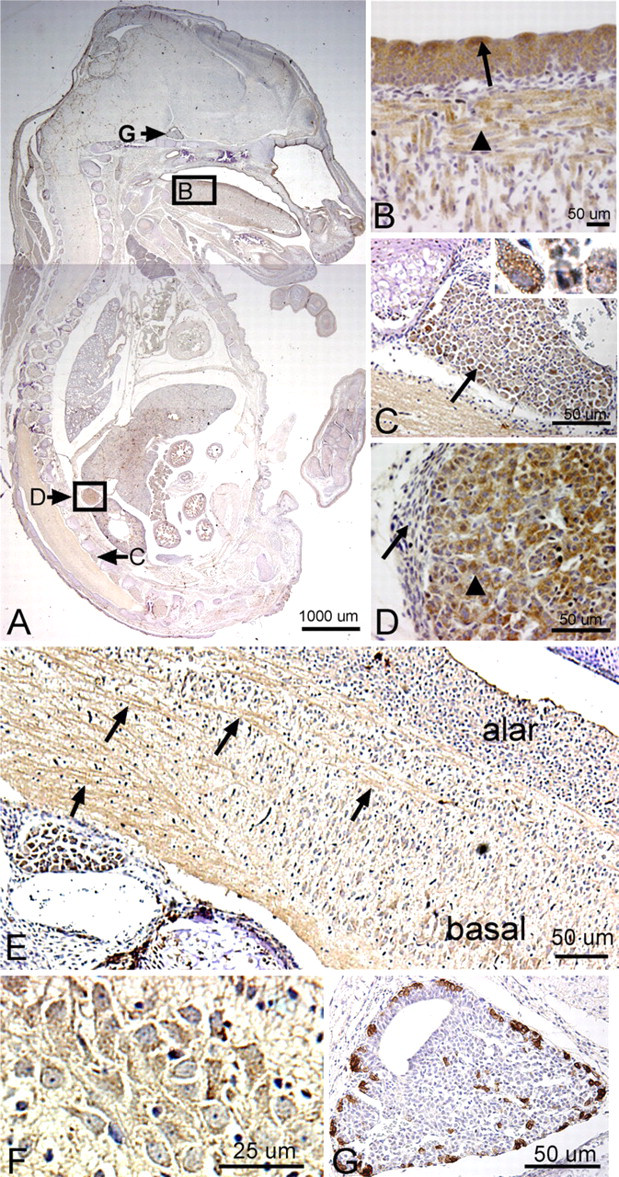
Localization of PLF protein in tongue, DRG, and adrenal gland of a mouse embryo at 16.5 dpc, counter-stained with hematoxylin. (
Localization of PLF (
At 19.5 dpc
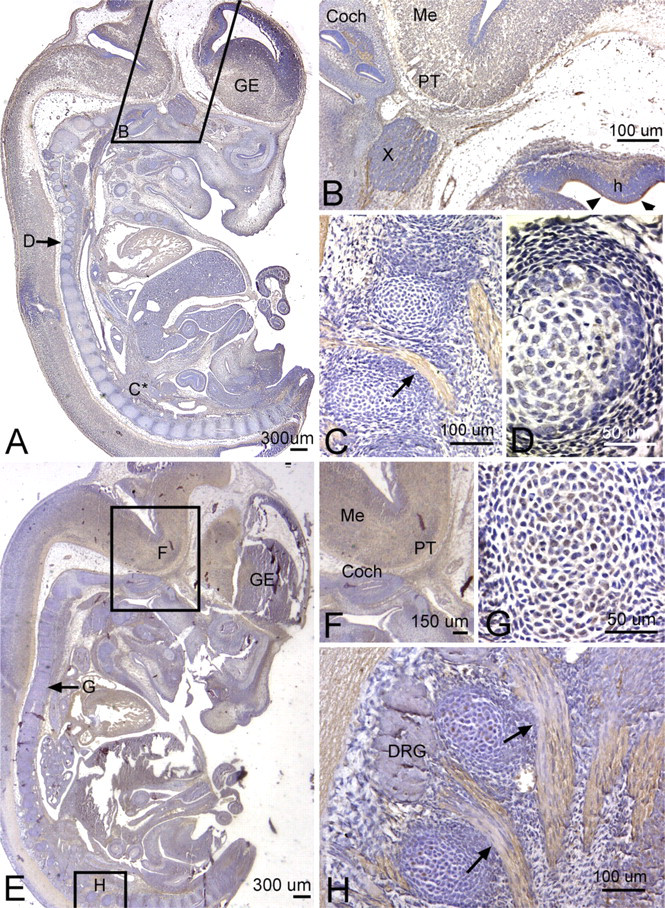
By this embryonic age, PLF staining was greatly diminished in central neural tissues (Table 1). However, it could still be detected as light axonal process staining, particularly in white matter tracts such as the corpus callosum of the neocortical regions and fimbria fornix of the hippocampus (Figures 8A, 8C, and 8D). Most peripheral and cranial nerve staining had diminished considerably by this age. However, the PLF staining could still be detected in the vagus nerve in the thoracic region (Figure 8B). PLF staining colocalized with PGP9.5, a general neuronal marker used to mark axons in this study, in the vagal nerve axons (Figures 8F and 8G). Also, PLF was present in chondrocytes of developing ribs at 19.5 dpc (Figure 8E).
We also examined Periostin staining at 19.5 dpc. We detected little to no staining for Periostin in brain regions and in brain white matter regions at this stage (Figures 9A, 9C, and 9D), although very faint staining was detected in the vagal nerve fascicles in the thoracic region (Figure 9B). Periostin staining was detected in a subset of chondrocytes of developing ribs and rib facet joints (Figures 9E and 9F) and at higher levels in the perichondrium surrounding these structures.
Discussion
The data presented here are descriptive analyses of the temporal and spatial pattern of expression of two protein isoforms, Periostin and PLF, with a focus on bone and nervous tissues. This is the first report on protein localization using isoform-specific antibodies for Periostin and PLF.
Bone development is a complicated biological process, which occurs by two mechanisms: intramembranous and/or endochondral ossification. Intramembranous ossification results in bone formation by osteoblasts, whereas in endochondral ossification, a cartilage template is first formed by chondrocytes and is then replaced by bone. Therefore, chondrocytes have a key role in endochondral ossification, whereas osteoblasts play an essential role in both types of bone formation. We illustrate by immunochemical staining that, during embryogenesis, PLF and Periostin were localized to proliferating and hypertrophied chondrocytes in developing long bones. In addition, mesenchymal cells in the perichondrium adjacent to hypertrophied chondrocytes were intensely stained for PLF, whereas those adjacent to proliferating chondrocytes were unstained. These findings suggest a role for PLF in endochondral bone formation and more specifically in the proliferation and differentiation of chondrocytes. The significance of the presence of PLF in mesenchymal cells in the bony collar suggests a possible role in recruitment of cells to the osteogenic lineage. Periostin was clearly detectable in the perichondrium surrounding developing bones, suggesting a possible role in recruitment of cells to the chondrocyte lineage.
We found PLF immunostaining in subsets of axons/axonal bundles and in several brain and spinal cord regions containing differentiating neurons. Most of this staining was stage specific, with the greatest staining present in neuronal tissues in embryos that were 12.5 and 13.5 dpc. PLF staining was present by 12.5 dpc in many peripheral nerves but was maintained until at least 19.5 dpc in a subset (e.g., the vagus nerve). There was an absence of PLF staining in dorsal root ganglion cells early, but there was strong expression later, by 16.5 dpc. These results are different from the embryonic localization of Periostin in both this study and in a study by Goetsch et al. (2003). Although Periostin was present in peripheral nerves at 11.5 dpc, no staining was observed in dorsal root ganglia or in brain or spinal cord structures in the study by Goetsch et al. (2003) at the ages examined. The very early and transient expression of PLF at 12.5 dpc on axon bundles suggests that PLF helps pioneer the growth of these axons. Periostin appears to stain brain structures or spinal cord structures, as shown in Goetsch et al. (2003).
In contrast, PLF appears to more closely resemble the staining of fasciclin I in commissure axons and other growing axons only as they are establishing their route or during arborization (Zinn et al. 1988; Jay and Keshishian 1990; McAllister et al. 1992; Zhong and Shanley 1995). Fasciclin I is a glycoprotein present in insects and involved in the mediation of homopholic cell adhesion, axonal arborization, and development of presynaptive functions (Elkins et al. 1990; Zhong and Shanley 1995). It is expressed in a stage-specific manner in the developing nervous system of grasshopper embryos on the surfaces of all cell bodies and axons in the PNS, commissural axons, bilateral longitudinal axonal tracts extending the length of the embryo, and several specialized sensory structures of the head (Jay and Keshishian 1990; McAllister et al. 1992). The duration of fasciclin I expression varied, with expression on commissural cell bodies and axons detected only during their formation, but expression was maintained on tracts such as the anterior corpus callosum. The related proteins, fasciclin II and III, are also expressed transiently on subsets of axonal cells, axons, and bundles, and appear on other pathways later in development (Patel et al. 1987; Grenningloh et al. 1991). Because PLF, like fasciclin I, is present in multiple tissues, it is possible that it is involved in morphogenesis in a manner similar to fasciclin proteins. The similarity between PLF and fasciclin staining, both temporally and spatially, suggests that PLF may play a role in neuronal morphogenesis.
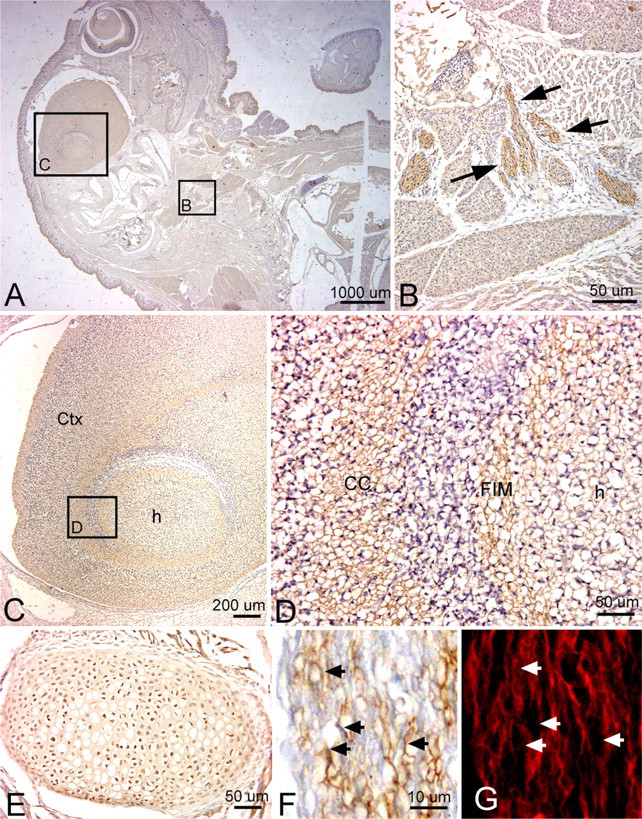
Localization of PLF protein in the brain, peripheral nerves in the thorax, and a rib at 19.5 dpc, counterstained with hematoxylin (
PLF was also detected in the adrenal gland and pituitary. The adrenal gland is made up of two parts: the outer region is called the adrenal cortex and the inner region is called the adrenal medulla. The adrenal cortex is of mesodermal origin and derived from the proliferation of coelomic epithelium, whereas the medulla is of ectodermal origin and derived from the neural crest. The medulla of the adrenal gland consists of sympathetic neural cells. Therefore, a strong signal of PLF in the medulla of the adrenal gland is consistent with localization of PLF in neural tissues. The pituitary is the master gland of the endocrine system, which controls the functions of the other endocrine glands. It is located at the base of the brain and is divided into two lobes: the anterior (adenohypophysis) and the posterior (neurohypophysis). The neurohypophysis consists mainly of axons extending from the hypothalamus. The detection of PLF in the neurohypophysis possibly suggests that PLF acts as a guidance molecule to direct these projecting axons from the hypothalamus to the neurohypophysis. Given that cells in the anterior hypophysis produce hormones necessary for the function of a variety of organs, the biologic function of PLF in this region requires further study.
In summary, the spatial and temporal pattern of localization of Periostin and PLF differ, suggesting different roles for these proteins. PLF may be secreted based on the presence of a signal peptide. It may also be present in the nucleus as suggested by the presence of a nuclear localization signal and as we have shown in embryonic tissues. Therefore, understanding mechanisms by which PLF mediates its effects will require studying its function as a secreted, as well as an intracellular, protein.
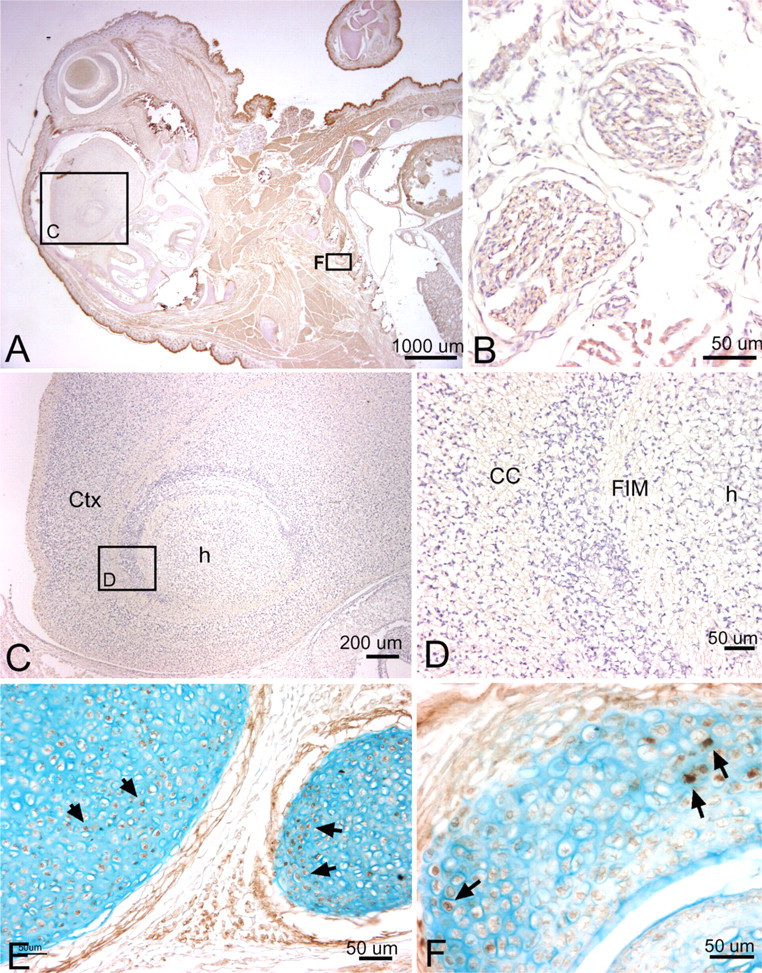
Localization of Periostin protein in the brain, thoracic structures, a rib, and a rib facet joint at 19.5 dpc, counterstained with hematoxylin (
