Abstract
Work-related musculoskeletal disorders (WMSDs), also known as repetitive strain injuries of the upper extremity, frequently cause disability and impairment of the upper extremities. Histopathological changes including excess collagen deposition around myofibers, cell necrosis, inflammatory cell infiltration, and increased cytokine expression result from eccentric exercise, forced lengthening, exertion-induced injury, and repetitive strain-induced injury of muscles. Repetitive tasks have also been shown to result in tendon and neural injuries, with subsequent chronic inflammatory responses, followed by residual fibrosis. To identify mechanisms that regulate tissue repair in WMSDs, we investigated the induction of periostin-like factor (PLF) and periostin, proteins induced in other pathologies but not expressed in normal adult tissue. In this study, we examined the level of PLF and periostin in muscle, tendon, and nerve using immunohistochemistry and Western blot analysis. PLF increased with continued task performance, whereas periostin was constitutively expressed. PLF was located in satellite cells and/or myoblasts, which increased in number with continued task performance, supporting our hypothesis that PLF plays a role in muscle repair or regeneration. Periostin, on the other hand, was not present in satellite cells and/or myoblasts.
Keywords
W
Histopathological changes including excess collagen deposition around myofibers, cell necrosis, inflammatory cell infiltration, and increased cytokine expression result from eccentric exercise, forced lengthening, exertion-induced injury, and repetitive strain-induced injury of muscles (McCully and Faulkner 1986; Fritz and Stauber 1988; Stauber et al. 1988, 1996; Johansson and Sojka 1991; Stauber and Smith 1998; Barbe et al. 2003, 2008; Elliott et al. 2008, 2009). Muscle exposure to repeated high velocity stretch also resulted in sustained fibrotic myopathy [i.e., an increase in non-contractile tissues (Stauber et al. 1994, 2000)]. Unfortunately, recovery from fibrotic changes is negligible, even with complete cessation of strain/activity for up to 12 months (Stauber et al. 2000), highlighting the importance of understanding the development and subsequent management of WMSDs.
Repetitive tasks have also been shown to result in tendon and neural injuries, with subsequent chronic inflammatory responses (Järvinen et al. 1997; Barbe et al. 2003, 2008; Clark et al. 2003, 2004; Elliott et al. 2008, 2009), followed by residual fibrosis (Clark et al. 2003, 2004). Direct damage to tendons results in collagen dysplasia and fibroblast proliferation (Allampallam et al. 1996; Carpenter et al. 1998; Archambault et al. 2001). With overuse, there is increased tendon cross-sectional area, increased collagen fiber disorganization, increased interfibrillar glycosaminoglycan, hypercellularity, and a change in cell morphology from elongated spindle-shaped cells to more-rounded, plump cells (Soslowsky et al. 2000). Overuse also increases the expression of cartilage-specific genes and reduces the expression of tendon-specific genes in the supraspinatus tendon in a rat model of tendon overuse (Archambault et al. 2007). These abnormal characteristics are suggestive of tendon pathology, resulting in pain and inflammation, ultimately leading to impaired use of the affected limb.
In spite of a widespread occurrence of these disorders, the underlying pathomechanisms are poorly understood, thereby impeding recommendations for effective interventions. To identify mechanisms that regulate tissue remodeling in WMSDs, we propose the study of molecules such as periostin-like factor (PLF), a protein also induced in other pathologies but not expressed in normal adult tissue (Litvin et al. 2005, 2007; Rani et al. 2009). PLF is expressed in adults under pathological overloading conditions, such as failing human hearts, injured blood vessels, volume-overloaded rat hearts (Litvin et al. 2006, 2007), fractured bone (Zhu et al. 2009), and bones exposed to repeated overuse (Rani et al. 2009). A related protein, periostin, is also expressed in multiple cancers and is implicated in cell motility and metastases (Sasaki et al. 2001; Matei et al. 2002; Yoshioka et al. 2002; Sasaki et al. 2003; Bao et al. 2004; Shao et al. 2004; Kudo et al. 2006; Erkan et al. 2007; Tilman et al. 2007; Choi et al. 2008; Fukushima et al. 2008; Puglisi et al. 2008; Ruan et al. 2009).
Here, the level of PLF in muscle, tendon, and nerve pathologies resulting from overuse is studied in a rat model of WMSDs developed by Barr and Barbe and described in detail in Barbe et al. (2003) and Clark et al. (2004). In this model, animals perform a voluntary, repetitive reaching and handle-pulling or grasping task at low to high reach rates and force levels as described in Table 1 in Elliott et al. (2009). Performance of a high-repetition, negligible force (HRNF) (four reaches/min, <5% of maximum grip strength) task for 8–12 weeks induced declines in motor performance and coordination, myofiber damage, fibrotic compression of the median nerve, a modest yet significant 9% decline in nerve conduction velocity, and local and systemic inflammation in forearm nerves and musculoskeletal tissues (Barbe et al. 2003, 2008; Clark et al. 2003; Barr and Barbe 2004; Al-Shatti et al. 2005). When high force [60% maximum pulling force (MPF)] is added to moderate [moderate-repetition, high-force (MRHF)] or high repetition [high-repetition, high-force (HRHF)], increased declines in median nerve conduction velocity (15% and 17%, respectively), sensorimotor deficits, and widespread inflammatory changes in both nerve and musculoskeletal tissues are induced (Clark et al. 2004; Elliott et al. 2009). In a recent study, PLF content in forelimb bones from rats performing the HRHF task showed an increase that was linked temporally to adaptive bone remodeling (Rani et al. 2009), but a decrease when bone resorption reached significant levels. In contrast, periostin was constitutively upregulated in response to task performance.
In this study, we examined the level of PLF and periostin in muscle, tendon, and nerve using immunohisto-chemistry and Western blot analysis. We hypothesized that PLF and periostin will be induced upon performance of HRHF tasks and in response to myofiber, tendon, and nerve damage. We suggest that each protein has a role in repair and regeneration of muscle, tendon, and nerve, as suggested by our findings in bone in the same animal model (Rani et al. 2009). It is well documented that populations of previously inactive adult stem cells known as satellite cells are recruited in the repair and regeneration process in response to skeletal muscle damage (Bischoff 1994). Therefore, we also examined PLF and periostin immunoreactivity in satellite cells and/or myoblasts. Periostin and PLF both were induced in tendons, skeletal muscles, and nerves in response to task performance. PLF demonstrated a clear progressive increase with continued task performance, whereas periostin was constitutively expressed. We found that PLF was located in satellite cells and/or myoblasts, which increased in number with continued task performance, supporting our hypothesis that PLF plays a role in muscle repair or regeneration. Periostin, on the other hand, was not expressed by satellite cells and/or myoblasts.
Materials and Methods
Subjects
The Temple University Institutional Animal Care and Use Committee approved experiments in compliance with National Institutes of Health guidelines for the humane care and use of laboratory animals. Young adult, female Sprague Dawley rats (14–16 weeks of age at onset of task) were obtained from ACE (Boyertown, PA) and housed in the central animal facility in separate cages in a 12-hr light/dark cycle with free access to water.
Behavioral Apparatus
The force apparatus was custom-designed (by Dr. Ann Barr and Custom Medical Research Equipment; Glendora, NJ) and integrated into an existing commercially available operant training system (Med Associates; Georgia, VT). Detailed specifications of the testing chamber and force apparatus have been previously described (Clark et al. 2004; Rani et al. 2009).
Performance of the Repetitive Motion Task
Thirty-six rats were randomized into a HRHF group (n=24), a normal control group (n=6), and a trained control (TC) group (n=6). Training and task performance details have been described in detail previously (Clark et al. 2004; Rani et al. 2009). Briefly, the HRHF rats and TC rats learned to reach and pull a handle at a specified rate and force. Rats were cued to reach at a target rate of 8 reaches/min, although they overreached and had an average reach rate of 12 reaches/min. They were also trained to pull a force handle between minimum [(55% of MPF), as determined in control rats], and maximum (65% MPF) force criteria for at least 50 msec. If these force and time criteria were met, a reward light was turned on and a food pellet (45 mg purified formula pellet; Bioserve, Frenchtown, NJ) was dispensed into a trough, which the rat could only reach by releasing the handle. During an initial 4–5-week training period, access to food was restricted to motivate rats to learn the task. Some animals may have undergone a short period (no more than 7 days) of weight reduction to 80% of weights of the agematched control animals that did not undergo food restriction. Once the animals learned the task, they rapidly gained weight and were maintained at ±5% of age-matched control rat weights. Rats were weighed twice weekly, and food adjusted accordingly. A cohort of six rats used as normal controls (n=6) did not undergo task shaping or food restriction. The TC group (n=6) underwent the training and the food restriction but did not proceed to the HRHF task regimen and were later euthanized at the 12-week endpoint of the HRHF experiment.
Once the HRHF animals were able to perform the task consistently, they performed the task regimen at a mean rate of 12 reaches/min at 60 ± 5% MPF, for 2 hr/day, 3 days/week (Monday, Wednesday, Friday) for 3, 6, 8, or 12 weeks (n=6 for each time point). The daily task was divided into four 0.5-hr training sessions separated by 1.5 hr.
Tissue Preparation for Immunohistochemistry
Normal control (n=3), TC (n=3), and HRHF rats per week (n=3 per weekly endpoint) were analyzed for immunohistochemistry. Following euthanasia by lethal overdose (Nembutol, 120 mg/kg body weight), animals were perfused transcardially with 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4). The entire upper extremity was postfixed “en bloc” by immersion overnight. Following fixation, muscle, tendon, and nerve tissues were separated as flexor and extensor masses from the bones, cryoprotected in 30% sucrose in phosphate-buffered saline (PBS), and then frozen-sectioned on a cryostat into 15-μm longitudinal sections. Two millimeter segments of the proximal forelimb flexors were also collected and sectioned into 15-μm cross-sections. The sections were mounted on charged-coated slides (Ultrastick; Corning, Lowell, MA) and stored at −20C for future use.
Immunohistochemistry and Quantification
Sections, on slides, were heated on a slide tray at 37C for 20 min before immunostaining to ensure adhesion to the charged slide. For peroxidase staining, sections were treated with 3% H2O2 in methanol for 30 min to inhibit endogenous peroxidase activity, washed in PBS, and then blocked with 4% goat serum in PBS-T (PBS with 0.1% Triton X-100) for 30 min at room temperature. Sections were incubated overnight at 4C with primary antibody [anti-PLF at 1:1000 (Litvin et al. 2006), anti-periostin at 1:750 (Zhu et al. 2008), and anti-PAX7 at 1:10 (anti-mouse IgG; Developmental Studies Hybridoma Bank at the University of Iowa)]. The next day, sections were washed and incubated with biotinylated anti-rabbit IgG (Vector Laboratories, Inc.; Burlingame CA) at a concentration of 1:500 in 5% goat serum/PBS for 30 min at room temperature. Sections were washed in PBS and treated for 30 min as described in the ABC signal amplification kit (Vector Laboratories, Inc.), and signal was visualized using DAB. DAB-stained sections were counterstained with hematoxylin, and examined using bright-field microscopy. In negative controls, the primary antibody was omitted during processing (Zhu et al. 2008).
Satellite cells and/or myoblasts were quantified on DAB-stained sections. Images were captured to a computer by a camera interfaced with an upright light microscope. PAX7-positive satellite cells and/or myoblasts were manually counted in a 0.003-mm2 area at 1000× magnification. Thirty-six random regions per slide were evaluated in three slides per animal and three animals per group, to obtain the total number of satellite cells and/or myoblasts per group. The number of satellite cells and/or myoblasts per mm2 was plotted against the weeks of task performance to determine the effect of task performance on the number of satellite cells and/or myoblasts.
Fluorescent staining was used to examine colocalization of PLF or periostin with PAX7 with secondary antibodies tagged with different fluorescent tags. Peroxidase staining was used for quantification purposes because the chemical DAB signal is reliably retained over time, providing the opportunity for quantification of staining. For fluorescent staining, sections on slides were baked at 37C for 20 min to ensure adhesion onto charged slides. Slides were washed in PBS and blocked with 4% goat serum/PBS-T for 2 hr at room temperature. Sections were incubated overnight at 4C with primary antibody diluted in 2% bovine serum albumin (BSA)/PBS (anti-PLF at 1:1000, anti-periostin at 1:750, and anti-PAX7 at 1:10). Slides were washed three times in PBS before incubation with appropriate Cy2- or Cy3-tagged secondary antibodies (Jackson Immuno-Research Laboratories, Inc.; West Grove, PA) at a concentration of 1:200 in 2% BSA/PBS for 1 hr at room temperature in the dark. Excess secondary antibody was removed by washing sections in PBS before incubation with 4',6-diamidino-2-phenylindole (DAPI) at a concentration of 1:1000 in 2% BSA/PBS for 20 min in the dark. Slides were washed three times in PBS, mounted with anti-fade reagent (Slowfade Light Antifade Kit; Molecular Probes, Carlsbad, CA), and examined under appropriate wavelengths using fluorescent microscopy. In negative controls, the primary antibody was omitted during processing.
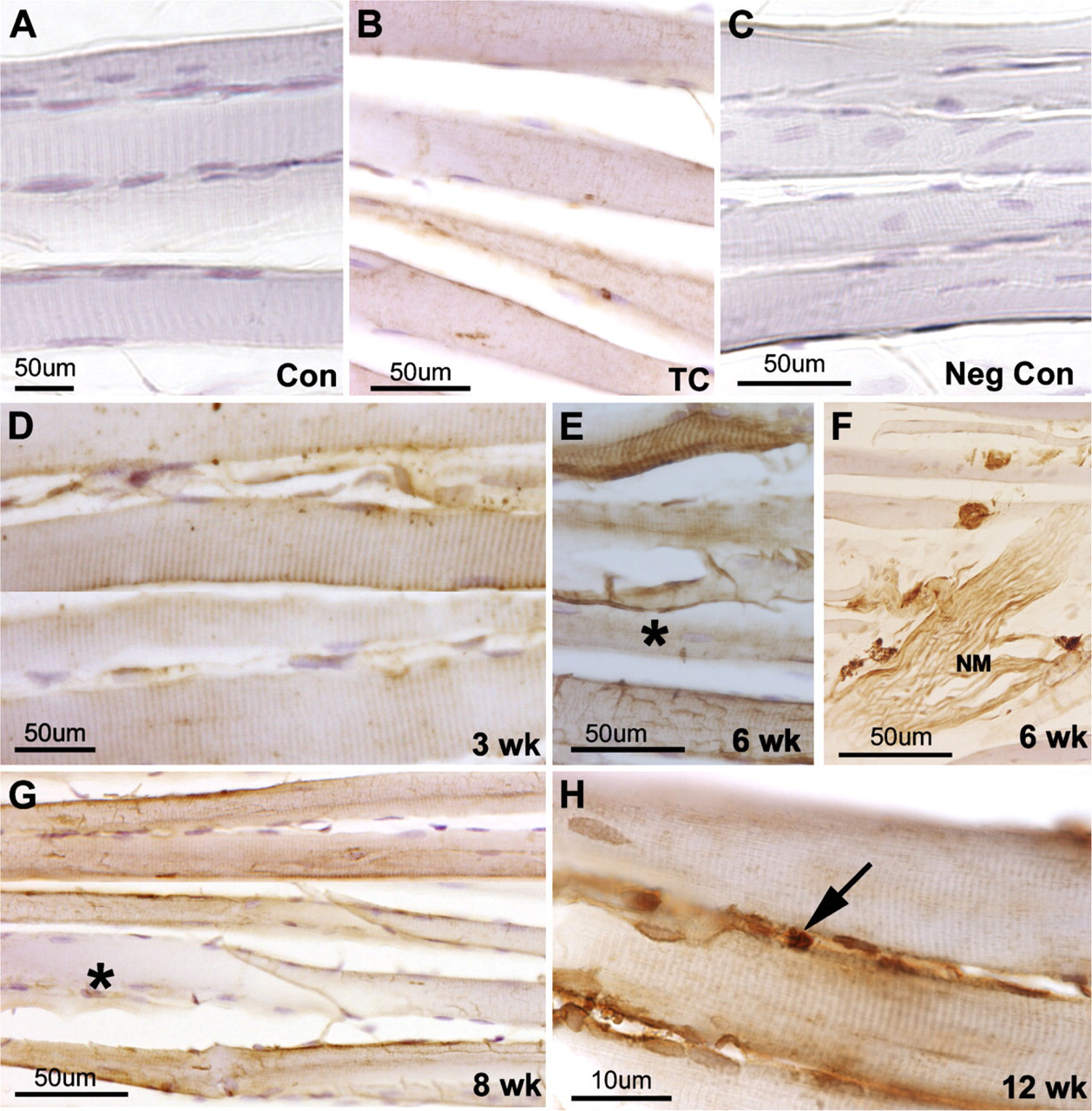
Forelimb muscles isolated from control, trained control (TC), or experimental rats [3-, 6-, 8-, or 12-week high-repetition, high-force (HRHF)] were cryosectioned and immunoreacted to periostin-like factor (PLF) antibody. (A) Normal control (Con) animals did not show PLF localization. (B) PLF was increased in TC animals. (C) Section from a 6-week HRHF time point on which primary antibody was omitted and which was used as a negative control (Neg Con). (D-G) HRHF animals (3-week, 6-week, and 8-week) showed increased localization of PLF in the muscle fibers, and muscle fibers appeared moth-eaten, with some fibers showing higher localization of PLF than others (asterisk). (H) Higher magnification of 12-week HRHF muscle showing the immunolocalization of PLF in muscle fibers as well as satellitelike cells (arrow).
Western Blot Analysis
Muscles and tendons obtained from normal control (n=3), TC (n=3), and HRHF rats (n=3) were homogenized in radioimmunoprecipitation assay buffer [25 mM Tris HCl, pH 7.6, 150 mM NaCl, 1% NP-40, 1% sodium deoxycholate, 0.1% SDS, protease inhibitor cocktail (Sigma; St. Louis, MO)] and incubated overnight at 4C, supernatant collected, and stored at −80C. For Western blot analysis, 25 μg of the protein sample was mixed with 5× Laemilli sample buffer (10% SDS, 50% glycerol, 25% β-mercaptoethanol, 300 mM Tris HCl, pH 6.8, and 0.04% bromophenol blue), boiled for 5 min, and resolved by 10% SDS-PAGE. Protein samples were transferred to nitrocellulose membrane for 1 hr at 4C, and membranes were blocked in 5% non-fat dry milk in PBS with 0.1% Tween-20 (PBST) for 1 hr and incubated in 0.3 μg/ml PLF or periostin-specific primary antibody overnight at 4C. Membranes were washed in PBST, incubated with 0.2 μg/ml of horseradish peroxidase (HRP)-conjugated goat antirabbit IgG secondary antibody (Pierce; Rockford, IL) for 1 hr at room temperature, and chemiluminescent signal was detected using the enhanced chemiluminescence (ECL) kit (Pierce). Blots were stripped, washed, and reprobed with 0.2 μg/ml glyceraldehyde-3-phosphate dehydrogenase (GAPDH)-specific primary antibodies overnight at 4C. The next day, membranes were washed with PBST and incubated with 0.2 μg/ml of HRP-conjugated goat anti-mouse IgG secondary antibody (Pierce) for 1 hr at room temperature, and the chemiluminescent signal was detected on X-ray film using the ECL kit (Pierce). Densitometric analysis (α imager) was performed on X-ray films to normalize the data against GAPDH.
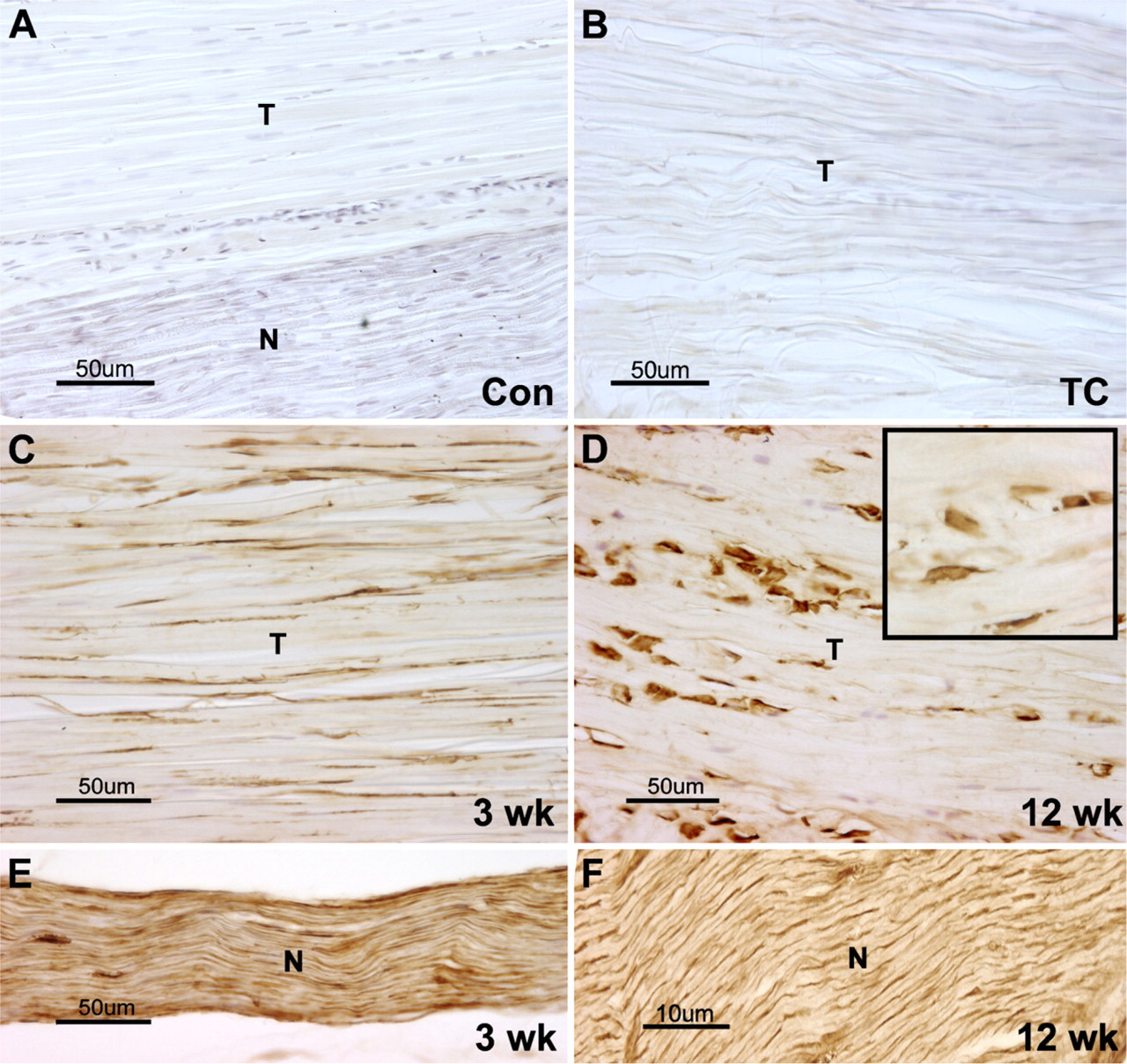
Forelimb tendon isolated from control or experimental rats (3-, 6-, 8-, or 12-week HRHF) were cryosectioned and immunoreacted with PLF antibody. (A,B) No PLF was detected in the normal control or TC animal tendon (T) or nerve (N). (C,D) PLF was detected at high levels in the tendons of 3- and 12-week HRHF animals, and the shape of the fibroblast nuclei changed from elongated (C) to more-rounded and plump (D) and inset at higher magnification showing rounded nuclei). (E,F) High amounts of PLF were also seen in nerves from 3- and 12-week HRHF animals.
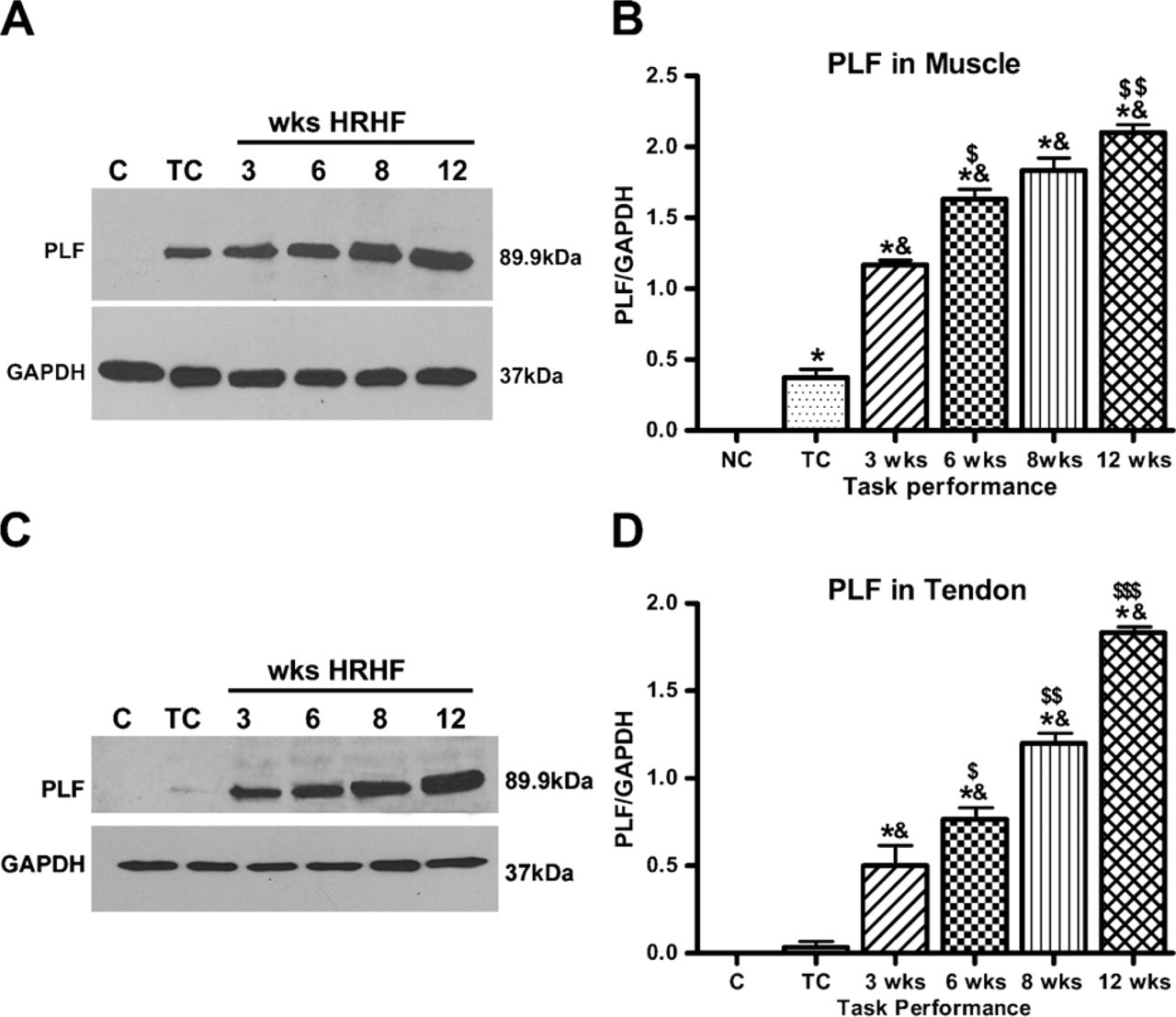
(A) Western blot analysis using protein samples from muscles for controls (C), TC, and 3-week-12-week HRHF animals showed an increase in PLF levels. (B) PLF increased significantly in task-performing animals compared with normal controls (NC) (∗ p<0.05) and compared with TCs (&p<0.05). PLF levels also increased significantly at week 6 compared with week 3 ($ p<0.05) and at week 12 compared with week 8 ($$ p<0.05). (C) Western blot analysis using protein samples from tendons for C, TC, and 3-12-week HRHF animals showed an increase in PLF levels. (D) PLF increased significantly in task-performing animals compared with normal controls (∗ p<0.05) and compared with TCs (& p<0.05). Increase in PLF levels was also observed after each week of task performance and is denoted by $ symbols, where $ denotes increase in week 6 from week 3, $$ denotes increase in week 8 from week 6, and $$$ denotes increase in week 12 from week 8. Western blot data are the mean of three independent experiments. GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Statistical Analyses
Univariate ANOVAs (Prism; Graphpad Software, La Jolla, CA) were used to determine the effect of task performance by week on the upregulation of PLF or periostin protein and the number of satellite cells and/or myoblasts. Posthoc analyses were carried out by the Bonferroni method for multiple comparisons, with results compared with baseline (week 1) or control levels, and adjusted p values reported. A p value of <0.05 was considered significant for all analyses.
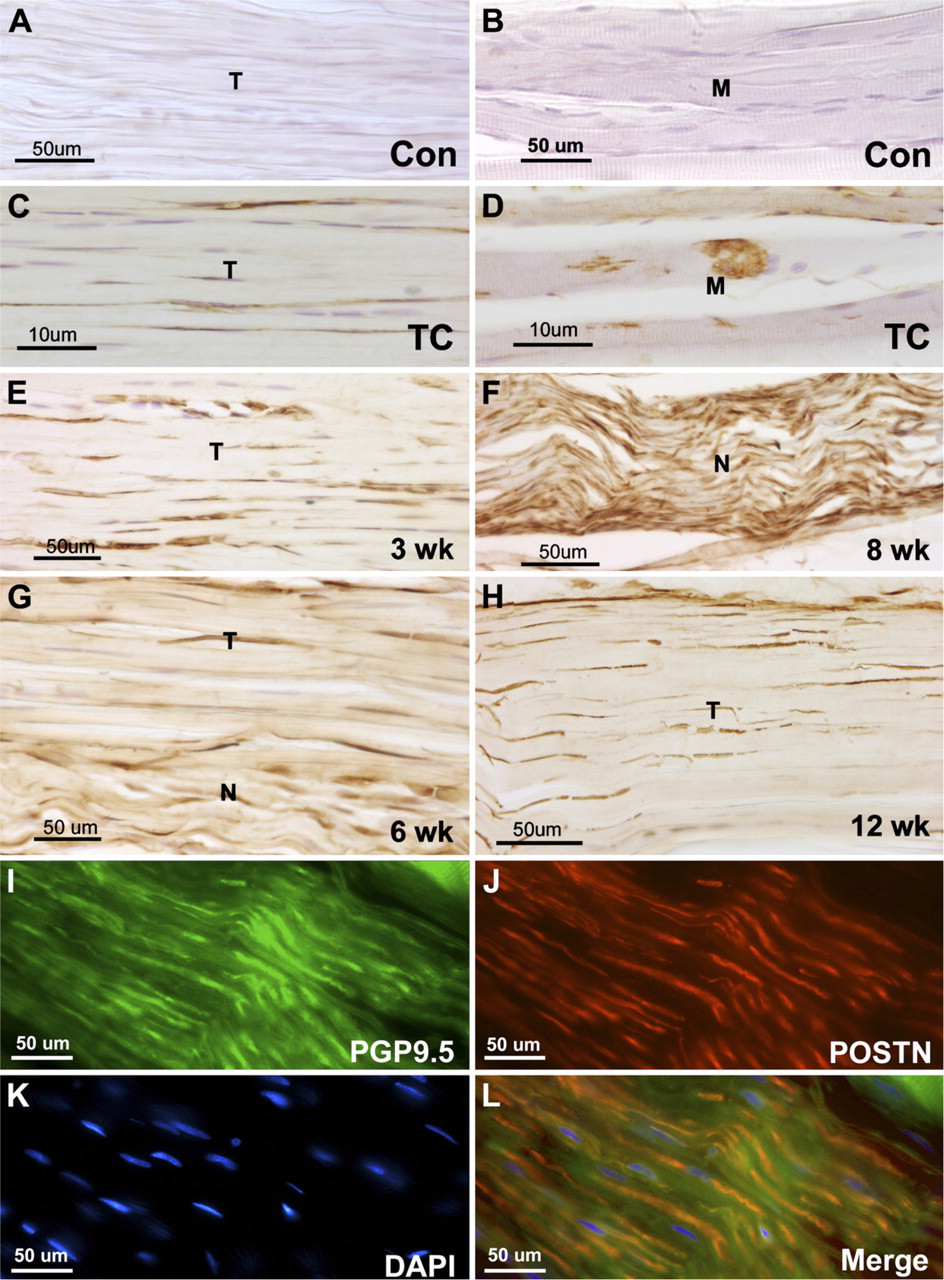
Forelimb muscles isolated from control (Con), TC, or experimental (3-, 6-, 8-, or 12-week HRHF) rats were cryosectioned and immunoreacted with periostin or PGP9.5 antibodies. (A,B) Periostin was not detected in tendon (T) or muscle (M) of control animals. (C,D) Periostin was present in tendons and muscles of TC animals. (E-H) Periostin was observed in tendons and nerves (N) of experimental animals. (I-L) The neuronal marker PGP9.5 (green) was used on 6-week HRHF sections to confirm that periostin (POSTN, red) was localized to axons in peripheral nerves in the forelimb. 4',6-Diamidino-2-phenylindole (DAPI) was used as a nuclear stain (K).
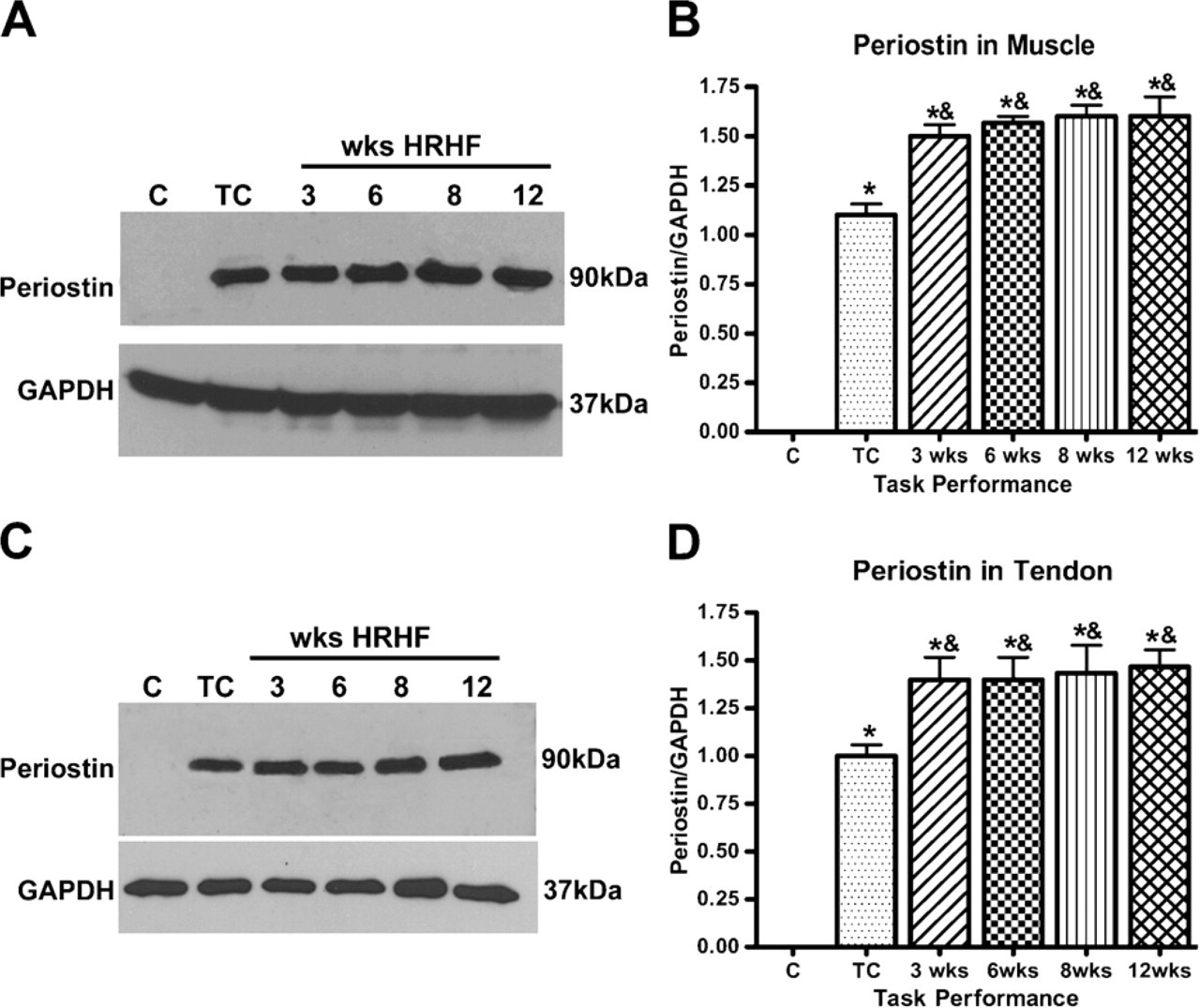
Western blot analysis using protein samples from C, TC, and 3-12-week HRHF animals for muscle (A,B) and tendon (C,D) showed an increase in periostin in experimental animals compared with C and TC animals. Periostin increased significantly in both muscle and tendons of experimental animals compared with normal controls (∗ p<0.05) and TCs (& p<0.05). Western blot data are the mean of three independent experiments.
Results
PLF and Periostin Induction in Forelimb Muscle, Tendon, and Nerve
PLF was not present in the flexor forelimb muscles of the normal control animals (Figure 1A); however, PLF was present at low levels in TC muscles (Figure 1B). A section on which the primary antibody was omitted was used as negative control (Figure 1C). PLF was seen in muscle fibers that appeared moth-eaten in 3-week HRHF rats (Figure 1D); PLF continued to be present in moth-eaten-appearing muscle fibers as well as in normal-appearing muscle fibers in HRHF animals that continued to perform the task for 6 to 12 weeks, although the relative amount of staining differed in individual fibers (less PLF in fibers marked by asterisk as indicated in Figures 1E and 1G) (Figures 1E–1H). PLF was also present in neuromuscular junction structures (Figure 1F), and in small cells outside the muscle fibers (Figure 1H). These latter cells were satellite-like cells in appearance, based on their size and anatomical location (marked by arrow in Figure 1H).
We also observed increased PLF in forelimb flexor tendons and nerves with HRHF task performance. In normal control and TC animals, PLF was not present in either tendons or nerves (Figures 2A and 2B), the latter in contrast to muscle of TC animals, in which there was a small increase in PLF levels and immunostaining. When animals performed the HRHF task for 3 weeks, PLF was present in tendons (Figure 2C) and nerves (Figure 2E), compared with normal controls and TCs (Figures 2A and 2B vs Figures 2C and 2E). PLF was also present in HRHF 12-week tendons (Figure 2D) and nerves (Figure 2F). By week 12, fibroblasts were rounded in appearance (insert at high magnification) as opposed to their normally elongated, spindle-shaped appearance as seen at week 3 (cf. Figures 2C and 2D).
Western blot analysis performed on tissues of a second cohort of animals confirmed that the amount of PLF increased in forelimb flexor muscles and tendons with weeks of task performance, compared with normal controls (Figures 3A and 3B, and 3C and 3D, respectively). Similar results were seen in three Western blot replicates using muscle protein samples from different rats for each blot, and the data depicted graphically in Figure 3B showed that PLF increased significantly in task-performing animals compared with normal controls (p<0.05) and compared with TCs (p<0.05). PLF levels were also increased significantly at week 6 compared with week 3 (p<0.05) and at week 12 compared with week 8 (p<0.05). Similar results were seen in three Western blot replicates using tendon protein samples from different rats for each blot. The data are depicted graphically in Figure 3D. PLF increased significantly in task-performing animals compared with normal controls (p<0.05) and compared with TCs (p<0.05). An increase in PLF levels was also observed after each week of task performance.
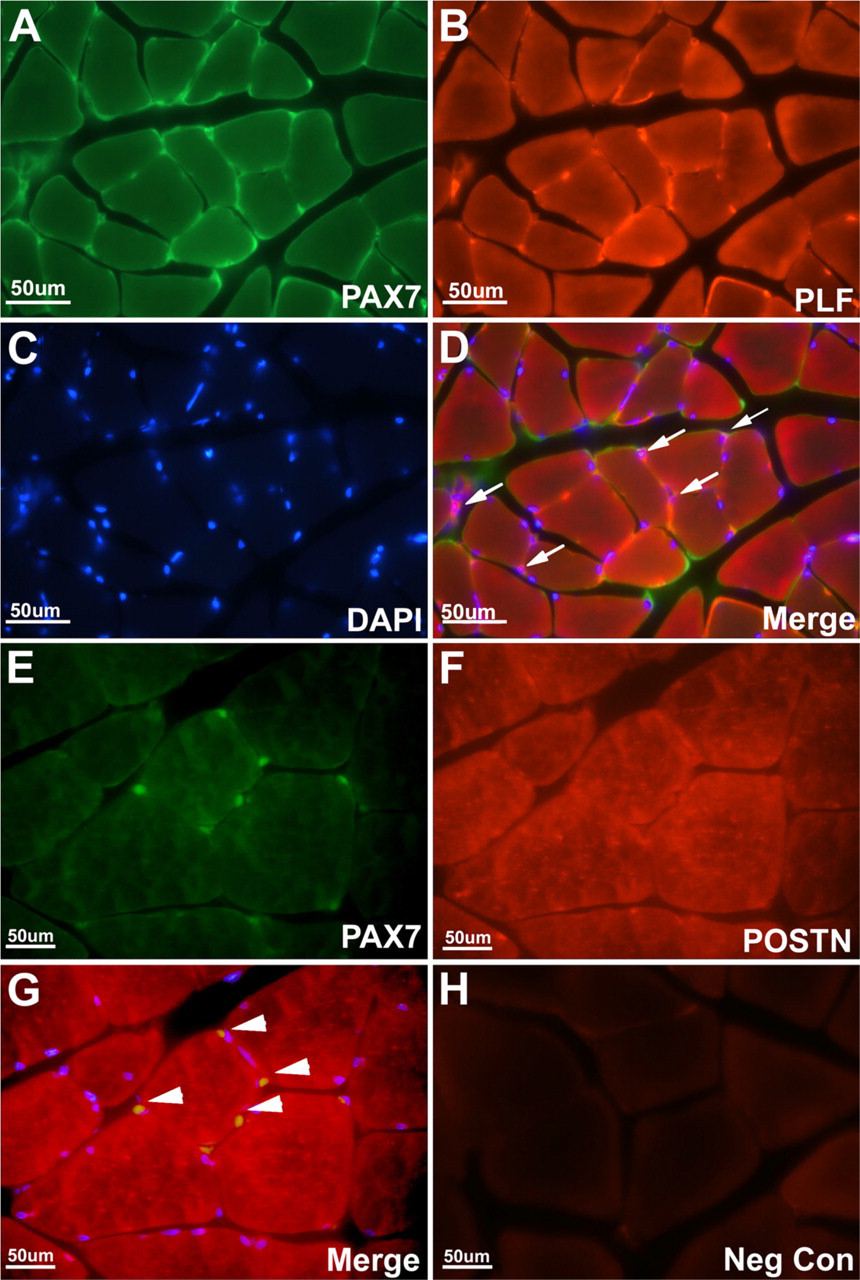
Forelimb muscles isolated from control or experimental rats (12-week HRHF) were cryosectioned and immunoreacted with PLF, periostin (POSTN), or PAX7 (satellite cell-specific marker) antibody. (A) PAX7 (green) was detected in satellite cells and/or myoblasts; (B) PLF (red) was present in the muscle fibers and satellite cells and/or myoblasts in experimental animals. (C) DAPI was used as a nuclear stain. (D) Overlay of PLF and PAX7 cells showing co-localization of these two proteins in satellite cells and/or myoblasts, as indicated by white arrows. (E) PAX7 (green) was detected in satellite cells and/or myoblasts; (F) periostin (red) was present in the muscle fibers in experimental animals. (G) Overlay showed no colocalization of periostin and PAX7 in satellite cells and/or myoblasts, as indicated by white arrowheads. (H) A tissue section on which primary antibody was omitted was used as a negative control.
Immunohistochemical analysis of periostin in normal control, TC, and HRHF rats revealed a different pattern of localization compared with PLF. Periostin localization was absent in normal control tendon and muscle (Figures 4A and 4B, respectively). In contrast to PLF, periostin was present in tendon fibroblasts (Figure 4C) and in nerves (data not shown), although periostin was present in muscles of TC rats (Figure 4D), which was similar to PLF immunolocalization in TC rats (Figure 1B). Periostin continued to be present in tendon fibroblasts and in nerves of weeks 3–12 HRHF animals (Figures 4E–4H). The neuronal marker PGP9.5 (Figure 4I) was used on 6-week HRHF sections to confirm that PLF (data not shown) and periostin (Figures 4J and 4L) were localized to axons in peripheral nerves in the forelimb. DAPI was used as a nuclear stain (Figure 4K).
The results obtained by immunohistochemistry for periostin were confirmed by Western blot analysis (Figures 5A and 5C). Periostin content increased in response to the training (in TCs) compared with normal controls, and levels increased significantly after animals moved from the training regimen to the task regimen; however, there was no significant change in response to increased task duration in muscles (Figure 5A) or tendons (Figure 5C). Similar results were seen in three Western blot replicates using samples from different rats for each blot. The data are depicted graphically in Figures 5B and 5D. Periostin increased significantly in task-performing animals compared with normal controls (p<0.05) and compared with TCs (p<0.05).
Satellite Cells and/or Myoblasts and PLF Expression
Sectioned muscle tissue from experimental animals (12 HRHF) reacted with PAX7 antibody, a marker for satellite cells and proliferating myoblasts (Figure 6A), and PLF antibody (Figure 6B) demonstrated colocalization of the two proteins (Figure 6D), suggesting that PLF is expressed by satellite cells and/or myoblasts (colocalization depicted by arrows in Figure 6D). DAPI was used as a nuclear stain (Figure 6C). In contrast, PAX7-positive cells (Figure 6E) and anti-periostinstained fibers (Figure 6F) did not show any colocalization, as marked by white arrowheads in the merge image (Figure 6G). Negative control for immuno-staining was performed without the use of primary antibody, on muscle tissue from a 12-week HRHF experimental animal (Figure 6H). The increase in PLF in muscle following weeks of task performance probably resulted from an increase in the number of satellite cells. Therefore, the number of satellite cells in DAB-stained sections was determined (Figure 7). PAX7-positive cells increased significantly with task performance over time (Figure 8).
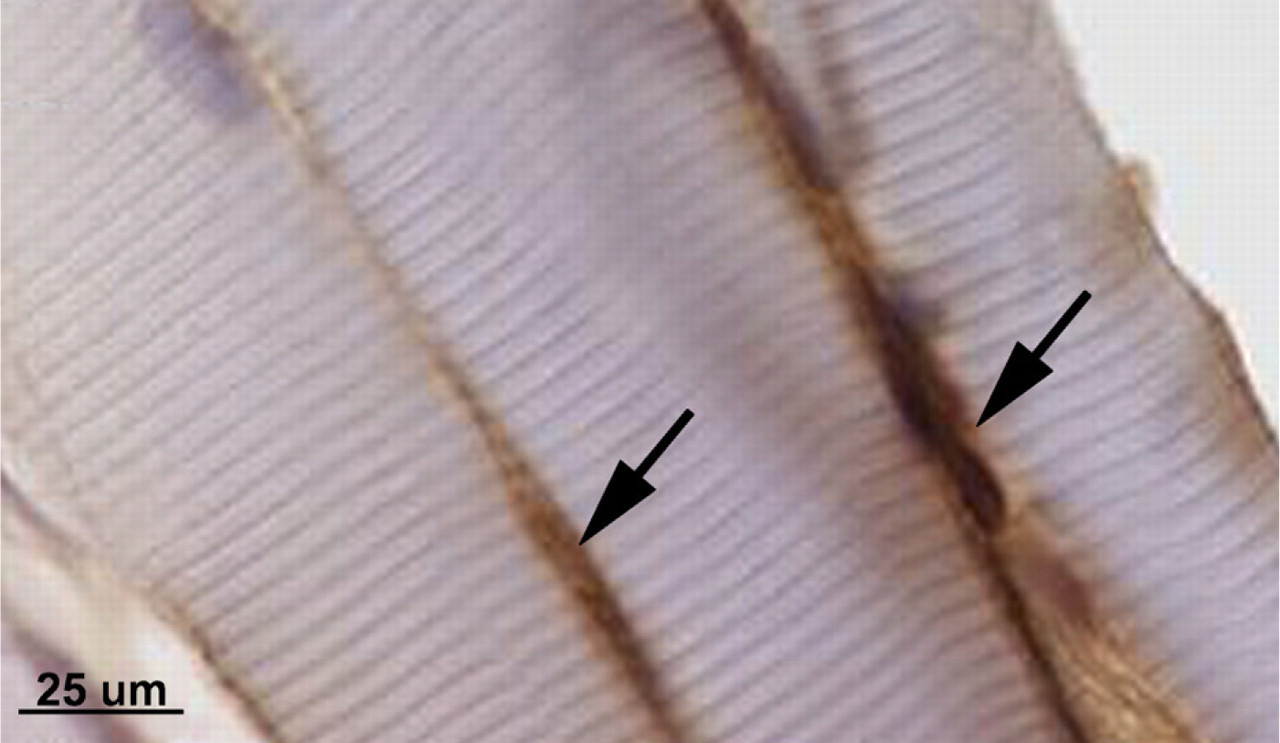
Muscle sections immuno-reacted with PAX7 and counterstained with hematoxylin show PAX7-positive satellite cells and/or myoblasts as indicated by black arrows.
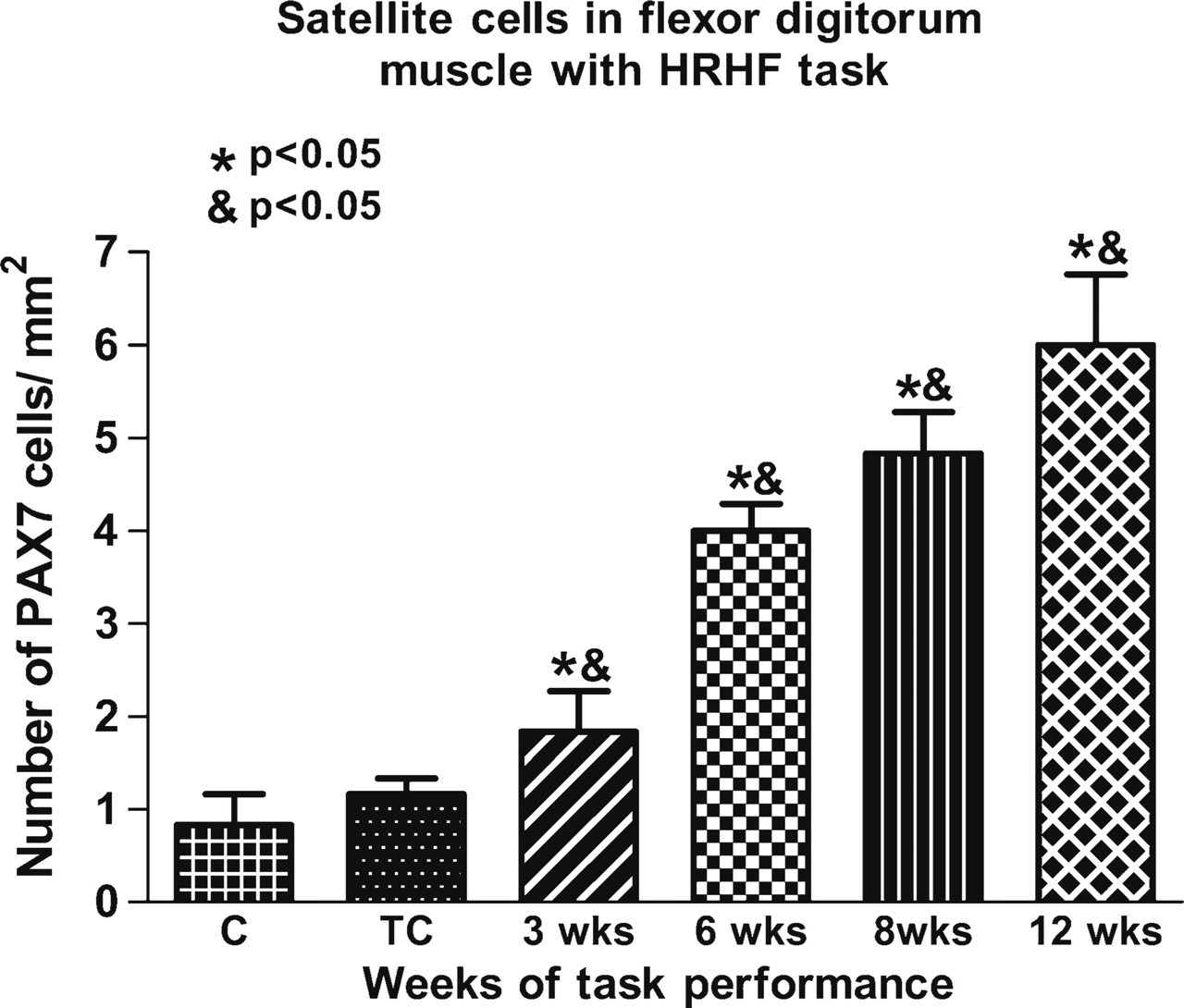
Thirty-six fields per section and three slides per group were counted to obtain number of satellite cells and/or myoblasts. Graph shows number of satellite cells and/or myoblasts per mm2 against treatment groups. All experimental animals showed significant increase in number of satellite cells and/or myoblasts over weeks of task performance, compared with normal controls (∗ p<0.05) and TCs (& p<0.05).
Discussion
In our model of upper-extremity WMSDs, disruption of myofibers at musculotendinous junctions in forearm flexors in the reach limbs of HRNF animals was reported, as well as increased degraded myelin protein in the median nerve and decreased nerve function, each indicative of injury (Barbe et al. 2003). Task exposure associated with increases in pro-inflammatory cytokines and ED1 macrophages in musculoskeletal and nerve tissues, suggestive of injury-induced inflammation, increased with higher demand tasks (Barbe et al. 2003, 2008; Elliott et al. 2008, 2009). Furthermore, increased PLF and periostin content in forelimb bones of animals after performance of the HRHF task was reported by Rani et al. (2009). Therefore, we hypothesized that forelimb musculotendinous and nerve tissues of HRHF animals may also have increased PLF and periostin expression. In tendons, we observed a significant increase in PLF in the HRHF group, in contrast to both normal and TC groups, in which PLF was not present. Periostin was also not present in tendons of normal control animals. However, periostin was induced in tendons in TC animals (an increase not observed for PLF), and its levels did not increase further in tendons with duration of task performance. Future investigation is required to further the understanding of the specific role of these two proteins in injured tendons. However, a difference in the temporal patterns of PLF and periostin suggests that these alternatively spliced products of a single gene may have different roles. The common feature between periostin and PLF is their shared homologies, whereas their main differences are located at the COOH terminus (exon 17 in PLF and exon 21 in periostin) (Litvin et al. 2004).
Our data also demonstrated that PLF is induced in forelimb skeletal muscle by HRHF task performance and that PLF levels and localization increased with task duration. With skeletal muscle damage, quiescent muscle adult precursor cells called satellite cells become activated. Discovered by Katz (1961) and Mauro (1961), satellite cells, located under the basal lamina of muscle fibers, constitute a reserve population of cells able to proliferate in response to injury, and give rise to muscle fibers as well as more satellite cells (Blaveri et al. 1999). These cells contribute myonuclei to growing muscle fibers and are the main, if not the only, cell type that contributes to muscle regeneration (Bischoff 1990a,b, 1994). There is little or no turnover of satellite cells in non-damaged, non-myopathic adult skeletal muscle (Decary et al. 1997). Satellite cells migrate to the site of injury from close proximity, and may enter from elsewhere within the muscle or, in extreme circumstances, from contiguous muscles (Seale and Rudnicki 2000; Hawke and Garry 2001). Therefore, using PAX7 antibody, we determined the presence of satellite cells and early proliferating myoblasts, both of which express this protein (Halevy et al. 2004; Zammit et al. 2004). In addition, we determined whether changes in their numbers were associated with an increase in PLF and/or periostin. Our data indicated that the satellite cell and myoblast population not only increased with task duration, but the marker for these cells (PAX7) also colocalized with PLF, suggesting that PLF-immunoreactive satellite cells and/or myoblasts may play a role in the repair or regeneration process in muscles following injury. Although periostin was up-regulated in skeletal muscle fibers after task performance, it was not expressed in satellite cells and/or myoblasts. This suggests that periostin is not involved in recruitment of satellite cells and/or myoblasts and hence plays a different role following muscle injury. These findings correlate well with those of Goetsch et al. (2003), in which they show that periostin is upregulated in response to injury as an extracellular matrix protein. In contrast, our data suggest that PLF is involved in satellite cell-mediated repair and regeneration following muscle injury. Likewise, in an in vivo model of bone formation, adenoviral overexpression of PLF in the bone marrow cavity resulted in increased bone formation, most likely by recruiting osteopro-genitor cells from the periosteum (Zhu et al. 2009). Therefore, we conclude that both of these isoforms have specific roles in repair and regeneration of tissues after loading or injury, but mediated by different mechanisms. Further investigation will provide insights into the underlying downstream pathways implicating PLF and periostin in the pathogenesis of WMSDs and other repetitive overload injuries to the musculoskeletal system.
