Abstract
The author has recently reported the distribution of the cytoskeleton-associated protein caldesmon in spleen and lymph nodes detected with different antibodies against caldesmon (J Histochem Cytochem 58:183–193, 2010). Here the author reports the distribution of caldesmon in the CNS and ganglia of the mouse using the same antibodies. Western blot analysis of mouse brain and spinal cord showed the preponderance of l-caldesmon and suggested at least two l-caldesmon isoforms in the brain. Immunostaining revealed the predominant reactivity of smooth muscle cells and cells resembling pericytes of many large and small blood vessels, ependymocytes, and secretory cells of the pineal gland and pituitary gland. Neuronal perikarya and neuropil in general displayed no or weak immunoreactivity, but there was stronger labeling of neuronal perikarya in dorsal root and trigeminal ganglia. In the brain, staining of the neuropil was stronger in the molecular layers of the dentate gyrus and cerebellum. Results show that caldesmon is expressed in many different cell types in the CNS and ganglia, consistent with the notion that l-caldesmon is ubiquitously expressed, but it appears most concentrated in smooth muscle cells, pericytes, epithelial cells, secretory cells, and neuronal perikarya in dorsal root and trigeminal ganglia.
Caldesmon is an actin-, myosin-, tropomyosin-, and calmodulin-binding protein existing as different isoforms because of alternative splicing of a single gene (for review, see Sobue and Sellers 1991; Huber 1997; Dabrowska et al. 2004; Wang 2008). The low molecular weight isoform (l-caldesmon) is thought to be ubiquitously distributed in non-muscle tissues (Sobue and Fukumoto 2010), but the high molecular weight isoform (h-caldesmon) is almost exclusively expressed in differentiated smooth muscle cells (smcs). l- and h-caldesmon differ by the insertion of an additional central region in h-caldesmon. h-caldesmon modulates the contraction of smooth muscle by inhibiting actomyosin ATPase activity, which can be reversed by binding to Ca2+/calmodulin or by phosphorylation of caldesmon (Ngai and Walsh 1984; Horiuchi et al. 1986; for review, see Arner and Pfitzer 1999; Kim et al. 2008). l-caldesmon in non-muscle cells influences organization and stabilization of the microfilament network (Kordowska et al. 2006; Morita et al. 2007). Raised serum levels of l-caldesmon were reported to be a potential marker for glioma (Zheng et al. 2005).
Immunostained migrating neurons and densely stained blood vessels were observed in the developing rat brain (Fukumoto et al. 2009). However, which cells in the adult brain express caldesmon appears controversial. Some studies show caldesmon only in blood vessels, others exclusively in neurons. In the normal human brain and in gliomas, caldesmon is expressed in endothelial cells, smcs, and pericytes of blood vessels; in the dura, it is expressed in fibroblasts (Zheng et al. 2003; Zheng et al. 2004). In the rat cortex and hippocampus, smcs of blood vessels and endothelial cells were reported to display caldesmon immunoreactivity (Kreipke et al. 2006). On the contrary, Represa et al. (1995) reported preferential staining of cell bodies and proximal dendrites of rat cortical neurons, cerebellar Purkinje and granule cells, neurons in the dorsolateral nucleus of the thalamus, and a few interneurons in the hippocampus. In an ultrastructural study of the rat hippocampus exclusively, neurons were immunoreactive (Agassandian C et al. 2000). Label was located in dendrites but was absent from axons. In the amygdala, neuronal perikarya and nuclei of a subpopulation of neurons as well as some areas of the neuropil displayed caldesmon immunoreactivity at light microscopy; ultrastructural examination revealed the same intraneuronal distribution as in the hippocampus in addition to labeled nuclei and cytoplasm (Agassandian K and Cassell 2008). Cell culture experiments, however, demonstrated caldesmon in neurites and growth cones of cultured rat and chick neurons (Kira et al. 1995; Alexanian et al. 2001). Similarly, cultured astrocytes displayed caldesmon immunoreactivity (Abd-el-Basset et al. 1991), but glial cells in tissue were not stained (Agassandian C et al. 2000; Zheng et al. 2004; Agassandian K and Cassell 2008).
The role of caldesmon in the brain is not clear. There is evidence that it may have a major role in the developing brain’s vasculature (Zheng et al. 2009). A possible role in neurons could be influencing synaptic plasticity by transferring signals from receptors to the actin cytoskeleton, as proposed by Represa et al. (1995) and K. Agassandian and Cassell (2008). To investigate the expression of caldesmon in different cell types in the CNS and ganglia of the mouse, we have performed an analysis using three of the antibodies recently used to detect caldesmon in the spleen and lymph nodes (Köhler 2010); we also included the antibody used by K. Agassandian and Cassell (2008). We have compared our immunohistochemical results to human tissues stained for caldesmon, as shown in the Human Protein Atlas (Uhlén et al. 2005), and to results from in situ hybridization shown in the Allen Mouse Brain Atlas (Lein et al. 2007).
Materials and Methods
Animals
For immunohistochemistry, 11 C57BL/6 mice of both sexes, ages 3 to 12 months, were obtained from the Institute of Physiology, University of Cologne and Harlan, Horst (The Netherlands). For Western blot analysis, four C57BL/6 mice of both sexes, ages two to four months, were obtained from Harlan, Horst (The Netherlands). The animals were handled according to the guidelines of the animal care committee of the University of Cologne.
Antibodies
Four different antibodies against caldesmon were used: a rabbit polyclonal antibody produced in the Institute of Physiology, University of Cologne, by immunization of rabbits with recombinant mouse h-caldesmon. Full-length h-caldesmon was expressed as Strep-Tag fusion-protein in HEK-EBNA cells and injected into rabbits. The caldesmon-specific antibody was purified from rabbit serum by affinity chromatography. We used it at a concentration of 0.23 µg/ml for immunohistochemistry or 0.03 µg/ml for Western blot. A human l-caldesmon mouse monoclonal antibody (clone 8/L-Caldesmon, catalog #610660) was obtained from BD Transduction Laboratories (Heidelberg, Germany) and used diluted 1:50 or 1:100 for immunohistochemistry or 1:6000 for Western blot. This antibody was raised against a fragment of human l-caldesmon corresponding to amino acids (aa) 251–395, a sequence 93% identical between human and mouse (Basic Local Alignment Search Tool [BLAST] analysis; Altschul et al. 1997) but both present in l- and h-caldesmon. This antibody was used by Zheng et al. (2004) and Bogatcheva et al. (2006). K. Agassandian and Cassell (2008) used a goat anti-caldesmon (N-19) antibody raised against a peptide mapping near the N-terminus of human caldesmon (sc-7575; Santa Cruz Biotechnology, Heidelberg, Germany). We tested this antibody for immunohistochemistry at various dilutions ranging from 1:50 to 1:1000; for Western blot, we diluted it 1:500. A mouse monoclonal antibody to human uterus smooth muscle extract (clone h-CD, catalog #C4562) and, according to the manufacturer, specific for human h-caldesmon was obtained from Sigma-Aldrich (Taufkirchen, Germany) and diluted 1:250 for immunohistochemistry or 1:500 for Western blot. A polyclonal rabbit antibody against aa 36–42 of Aβ42 (catalog #44-344; Biosource, Nivelles, Belgium) that was not expected to stain normal brain and spinal cord was used as control to exclude unspecific staining of polyclonal rabbit IgG. Other antibodies used were a mouse monoclonal to glial fibrillary acidic protein (GFAP) from porcine spinal cord (Debus et al. 1983), clone G-A-5 (Roche, Mannheim, Germany) diluted 1:50 for immunohistochemistry, and a mouse monoclonal to a synthetic peptide from the C-terminus of human GLUT-1 (Acris, Herford, Germany) diluted 1:500 for immunohistochemistry.
Gel Electrophoresis and Immunoblotting
Methods of gel electrophoresis and immunoblotting have previously been published in detail (Köhler 2010). In short, mouse brain, spinal cord, lung, and uterus were washed in cold Tris-base sodium chloride buffer (pH 7.6), frozen in liquid nitrogen, and stored at −80C until use. Tissues were homogenized in lysis buffer and centrifuged at 3000 × g for 20 min at 4C. The supernatant was used. The amount of proteins in the supernatant was determined using a BCA protein assay kit (Perbio, Bonn, Germany). Proteins were separated by 6% SDS-PAGE and electroblotted onto a nitrocellulose membrane, followed by incubation with primary antibodies at 4C overnight. After washing, the blot was incubated with horseradish peroxidase–coupled secondary antibodies to rabbit (Dako, Hamburg, Germany) or mouse (Sigma-Aldrich) or goat IgG (Linaris, Wertheim-Bettingen, Germany). Finally, the blot was washed again and developed with chemiluminescent substrate (Roche).
Tissue Processing
Animals were deeply anaesthetized (0.055 g tribromethanol/100 g body weight intraperitoneally) and perfused via the left ventricle with 0.1 M phosphate-buffered saline (pH 7.4) for 3 min, followed by 4% paraformaldehyde (PFA) or 2% PFA/0.5% glutaraldehyde for 15 min. Brains and spinal cords were removed, postfixed in the same fixative at 4C overnight, dehydrated, embedded in paraffin or frozen in isopentane in liquid N2, and cryoprotected in graded sucrose. Vertebral columns, including spinal cords, were demineralized for 5 days (Korkmaz et al. 2004) before embedding in paraffin. Then, 5-µm-thick brain sections were cut with a microtome in the frontal or sagittal plane; 5-µm-thick sections of spinal cord were cut in the transverse or sagittal plane and mounted on silane-coated slides.
Immunohistochemistry
Immunohistochemistry was performed using the avidin-biotin peroxidase method and nickel-intensified 3,3′-diaminobenzidine (DAB) as chromogen as previously published (Köhler 2010).
Double Staining
For light microscopy double staining, sections were consecutively stained, first with the caldesmon polyclonal antibody and then for GFAP. The bound caldesmon antibody was visualized using DAB with nickel enhancement, which resulted in a deep blue-gray reaction product. Before staining for GFAP, the peroxidase from the previous stain was blocked by incubating the sections in 0.3% H2O2 in TBS for 30 min at room temperature. DAB without nickel enhancement results in a brown reaction product indicating GFAP-antibody binding.
Immunofluorescence double staining was performed with primary antibodies consecutively applied overnight at 4C. Incubation with the first primary antibody (GLUT-1 or clone 8/L-Caldesmon) was followed by incubation with biotinylated anti-mouse IgG (1:250; Dianova, Hamburg, Germany) for 30 min at room temperature and then by Alexa Fluor 488–conjugated streptavidin (1:200; MoBiTec, Göttingen, Germany) for another 30 min at room temperature. The second primary antibody (caldesmon rabbit polyclonal) was detected with Alexa Fluor 546–conjugated anti-rabbit IgG (1:400; MoBiTec).
Digital Imaging
Sections stained for light microscopy were examined with a BX40-microscope (Olympus, Hamburg, Germany) equipped with phase contrast and a ColorView II digital camera (Olympus Soft Imaging Solutions, Münster, Germany). Confocal laser scanning microscopy was performed with a LSM510 confocal microscope (Zeiss, Jena, Germany). Digital images were processed for brightness and contrast using Picture Publisher 8 (Micrografx, Richardson, TX) or Adobe Photoshop 5.0 (Adobe, San Jose, California).
Results
Testing of the Antibodies and Immunoblot Analysis of the Expression of Caldesmon Isoforms
The rabbit polyclonal caldesmon antibody recognized one band at 130 kDa and clearly two bands at ~71/72 kDa in lysates of mouse brain (Fig. 1A) corresponding to reported molecular weights of h- and l-caldesmon (Huber 1997). The same double band at ~71/72 kDa was stained by clone 8/L-Caldesmon, which was used by Zheng et al. (2004) and Bogatcheva et al. (2006), but the higher molecular weight band at 130 kDa was not detected (Fig. 1B), even at five-fold increased protein load (not shown). The goat anti-caldesmon (N-19) antibody, which was used by K. Agassandian and Cassell (2008), recognized bands at 130 kDa and ~71/72 kDa (not shown). When spinal cord was probed, the rabbit polyclonal antibody and clone 8/L-Caldesmon labeled a band at ~71/72 kDa. As in brain, the rabbit polyclonal detected the high molecular weight isoform in addition (Fig. 1A,B).
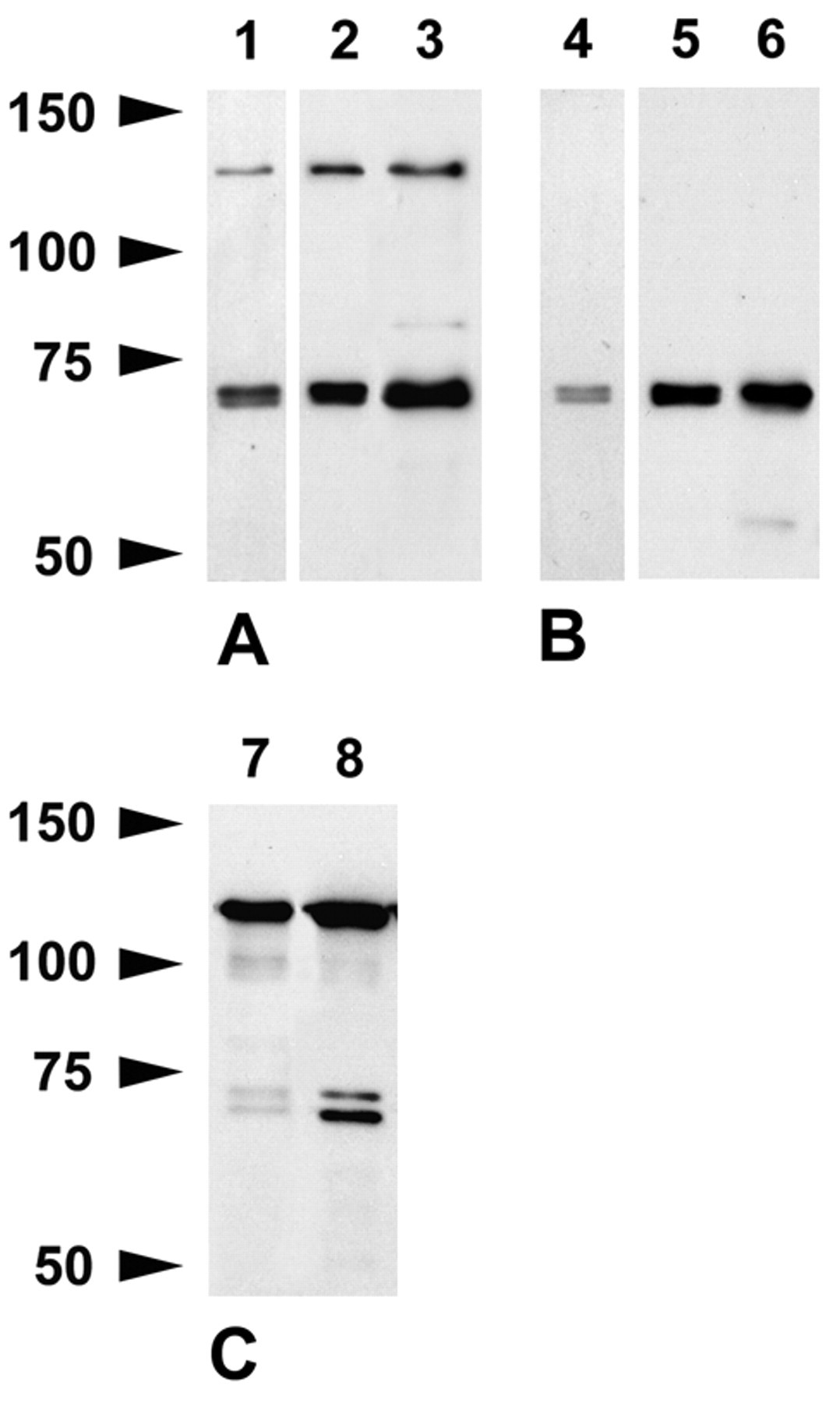
Western blots of brain (lanes 1, 2, 4, 5) and spinal cord (lanes 3, 6) after 6% SDS-PAGE stained with the caldesmon rabbit polyclonal antibody (A) and clone 8/L-Caldesmon (B). Lanes 1 and 4, 10 µg per lane loaded; lanes 2, 3, 5, 6, 20 µg per lane loaded. The rabbit polyclonal and clone 8/L-Caldesmon recognize two closely spaced bands at ~71/72 kDa clearly visible after reduced protein load. The rabbit polyclonal antibody stains a band at 130 kDa corresponding to the molecular weight of h-caldesmon in addition. (C) Western blots of uterus (2.5 µg per lane loaded) show that the rabbit polyclonal antibody (lane 7) and clone 8/L-Caldesmon (lane 8) recognize h-caldesmon as well as two bands within the molecular weight range of l-caldesmon.
In uterus where h-caldesmon is abundant, the rabbit polyclonal antibody and clone 8/L-Caldesmon labeled a band at 130 kDa (Fig. 1C). We did not obtain a band at 130 kDa with the monoclonal antibody against human h-caldesmon, neither in brain nor in lung, which has a comparably high and about equal amount of h- and l-caldesmon (not shown). Taken together, these results demonstrate that the rabbit polyclonal, goat polyclonal (N-19), and clone 8/L-Caldesmon specifically recognize h- and l-caldesmon isoforms in Western blots and that l-caldesmon isoforms predominate in the mouse CNS.
Caldesmon Immunoreactivity in the Mouse Brain
Good results were obtained when PFA-fixed paraffin sections with microwave pretreatment in citrate buffer (pH 6.0) were used. Microwave pretreatment in EDTA buffer (pH 8.0) increased staining of small blood vessels with clone 8/L-Caldesmon but also increased reactivity of the secondary antibody with ependymal cells of the choroid plexus and parts of the neuropil; it caused high background when clone h-CD was used. Immunoreactive blood vessels were more numerous in cryosections; optimal immunoreactivity with clone h-CD required cryosections. Fixation with 2% PFA/0.5% glutaraldehyde precluded staining with the rabbit polyclonal antibody and clone 8/L-Caldesmon; clone h-CD was not tested. Except for sections pretreated in EDTA buffer, staining of sections processed in parallel was absent when the primary antibodies were omitted (not shown). Also, there was no staining when the caldesmon polyclonal antibody was replaced by polyclonal rabbit IgG of irrelevant specificity (anti-Aβ42) at a similar concentration (Fig. 2B).
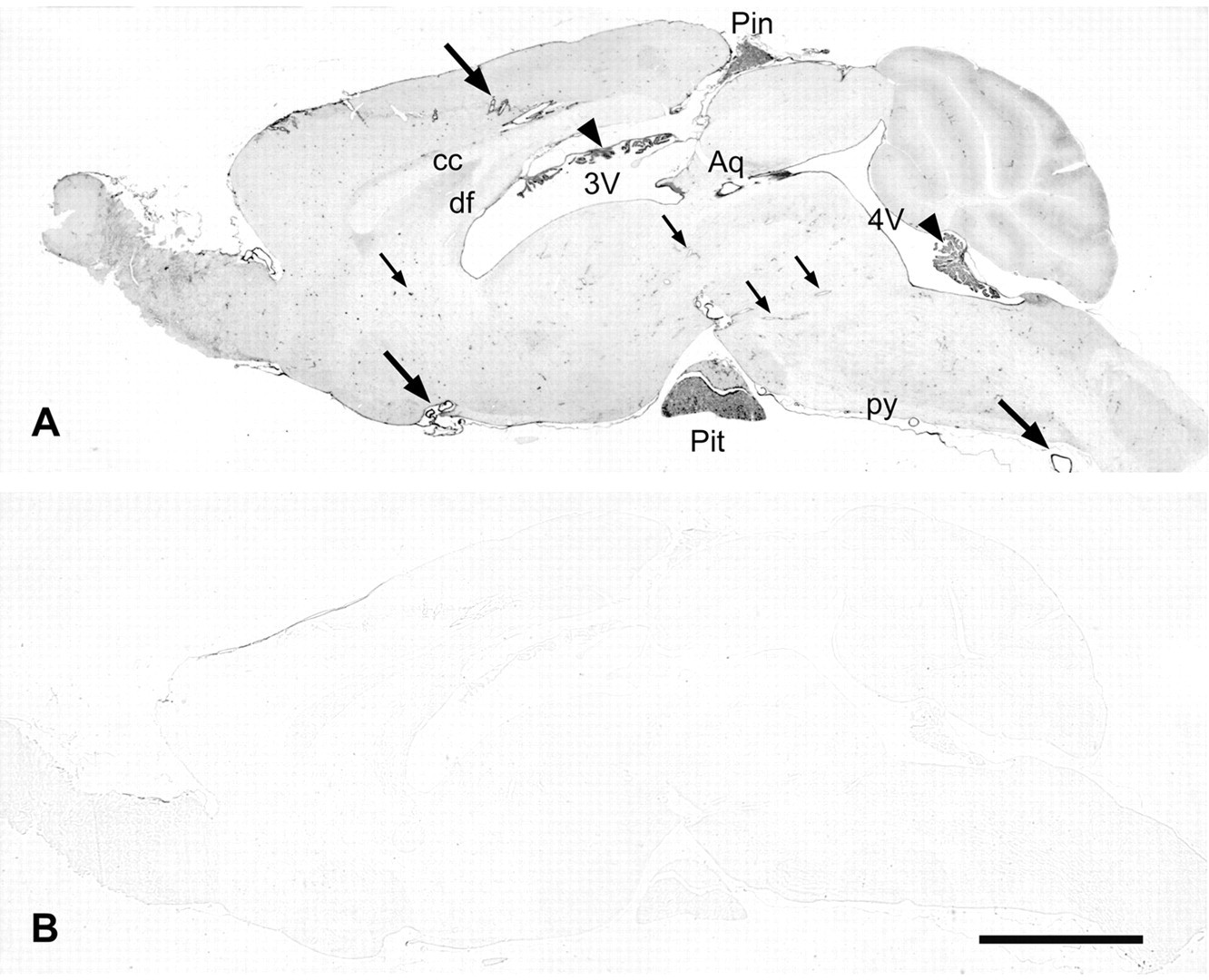
Overview of caldesmon immunoreactivity of the mouse brain. (A) Four percent paraformaldehyde (PFA)–fixed, sagittal paraffin section of a mouse brain stained with the caldesmon rabbit polyclonal antibody after microwave pretreatment in citrate buffer. Larger blood vessels (large arrows), ependymal cells lining the ventricles and aqueduct (3V, third ventricle; 4V, fourth ventricle; Aq, aqueduct), ependymal cells of the choroid plexus (arrowheads), and the pineal (Pin) and pituitary (Pit) gland display strong immunostaining clearly recognizable at low magnification, but medium-sized (small arrows point to examples) and small blood vessels are also stained. If compared to unstained white matter (cc, corpus callosum; df, dorsal fornix; py, pyramidal tract), there is weak immunoreactivity of the neuropil, which is stronger in the molecular layer of the cerebellum. (B) Immunoreactivity is absent after replacement of the caldesmon rabbit polyclonal antibody by an irrelevant rabbit polyclonal antibody against Aβ42. Color development time was the same for both sections. Scale bar = 2 mm.
Expression of h-caldesmon in smcs has been demonstrated in many studies (for review, see Wang 2008), including histopathological studies that used clone h-CD (e.g., Miettinen et al. 1999; Ceballos et al. 2000; Visée et al. 2005; Perez-Montiel et al. 2006; Qiu et al. 2008). Therefore, an antibody against caldesmon should stain smc if it recognizes h-caldesmon. Clone h-CD and the rabbit polyclonal stained smcs in muscle layers and/or blood vessels of intestine, bladder, uterus, spleen, and lymph nodes. Staining of mouse smcs in muscle layers was comparably weak when clone 8/L-Caldesmon was used.
In the mouse brain, immunoreactivity with clone h-CD was confined to smcs in the wall of large blood vessels and to ependymal cells (not shown). The caldesmon rabbit polyclonal antibody and clone 8/L-Caldesmon, which recognized h- and l-caldesmon isoforms in Western blot, produced a very similar pattern. Both stained smcs of large blood vessels and cells resembling pericytes of many smaller blood vessels (Figs. 3A–E, 4A–K). Endothelial cells displayed only little immunoreactivity, as revealed by double staining with the rabbit polyclonal antibody and the endothelium marker GLUT-1 (Fig. 4A–F). Ependymal cells lining the ventricles and aqueduct as well as ependymal cells of the choroid plexus exhibited moderate to strong immunoreactivity with the rabbit polyclonal antibody and clone 8/L-Caldesmon (Figs. 2A, 5D,E). These antibodies also labeled the meninges. In the pituitary gland and pineal gland, secretory cells were stained (Figs. 2A, 5A,B,G,H). In general, sections stained with the caldesmon rabbit polyclonal antibody and clone 8/L-Caldesmon displayed no or only slight labeling of neuronal perikarya (Figs. 2A, 3A–E, 4L). When higher concentrations were tried, the rabbit antibody had a tendency to stain neuronal nuclei. Because, to the best of our knowledge, except for K. Agassandian and Cassell (2008), none of the studies that demonstrated caldesmon in normal mammalian tissues reported nuclear localization of caldesmon, we considered staining of nuclei to be unspecific. Paraffin sections displayed an overall low-level staining of the neuropil with the rabbit polyclonal antibody, slightly contrasting against unstained major fiber tracts (corpus callosum, anterior commissure, fimbria/fornix, axon fascicles of the striatum, white matter of the cerebellum, pyramidal tract; Fig. 2A). In the hippocampus, immunoreactivity of the neuropil was slightly stronger in the oriens layer and stratum radiatum but most conspicuous in the molecular layer of the dentate gyrus (Fig. 4L). This was at least in part attributable to weak staining of astrocytes, as shown by double staining for caldesmon and GFAP (Fig. 4M,N). Immunoreactivity of the neuropil was also stronger in the molecular layer of the cerebellum (Fig. 2A), but perikarya and the proximal major dendrites of Purkinje cells were unstained (Fig. 3B).
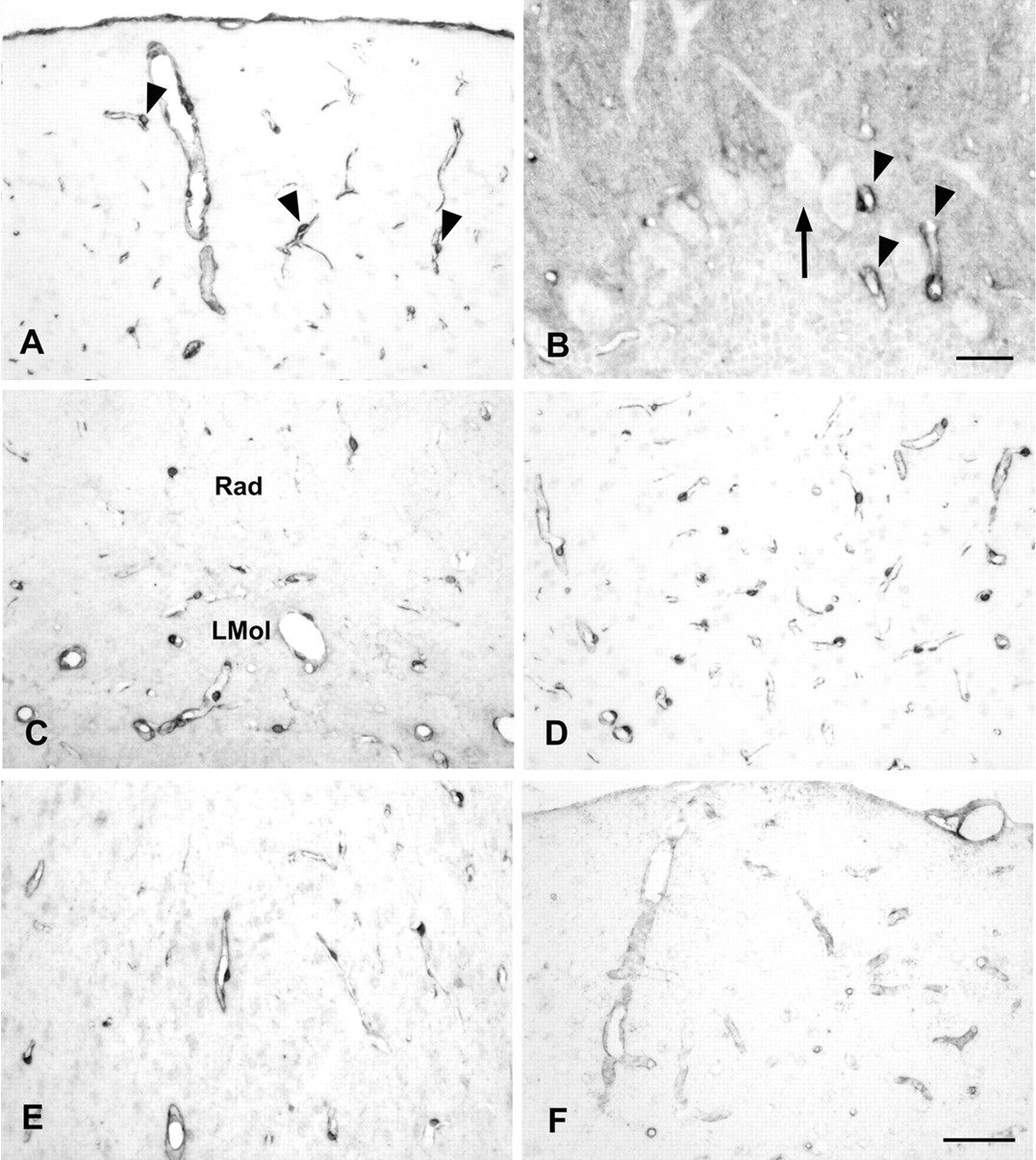
Caldesmon immunoreactivity of blood vessels in different regions of the brain. (A) Four percent paraformaldehyde (PFA)–fixed paraffin section of the cortex stained with clone 8/L-Caldesmon after microwave pretreatment in EDTA buffer. The meninges and blood vessels are immunoreactive. Cell bodies located at the outer surface of the vessel wall, reminiscent of pericytes (arrowheads point to examples), and fine processes are stained. (B) Four percent PFA-fixed paraffin section of the cerebellum stained with the rabbit polyclonal antibody after microwave pretreatment in citrate buffer. Blood vessels are immunoreactive (arrowheads). Perikarya and major dendrites of Purkinje cells (arrow) are unstained. There is also moderate staining of the molecular layer. (C) Four percent PFA-fixed paraffin section of the hippocampus stained with the rabbit polyclonal antibody after microwave pretreatment in citrate buffer showing immunoreactive blood vessels in the stratum radiatum and lacunosum moleculare layer. (D) Four percent PFA-fixed paraffin section of the superior colliculus stained with clone 8/L-Caldesmon after microwave pretreatment in EDTA buffer showing a dense net of immunoreactive small blood vessels. (E) Four percent PFA-fixed paraffin section of the amygdala stained with the rabbit polyclonal antibody after microwave pretreatment in citrate buffer showing immunoreactive blood vessels. (F) Four percent PFA-fixed cryosection of the cortex stained with the goat (N-19) antibody showing immunoreactive blood vessels. LMol, lacunosum moleculare layer; Rad, stratum radiatum. Scale bar (A, C, D, E, F) = 50 µm. Scale bar (B) = 20 µm.
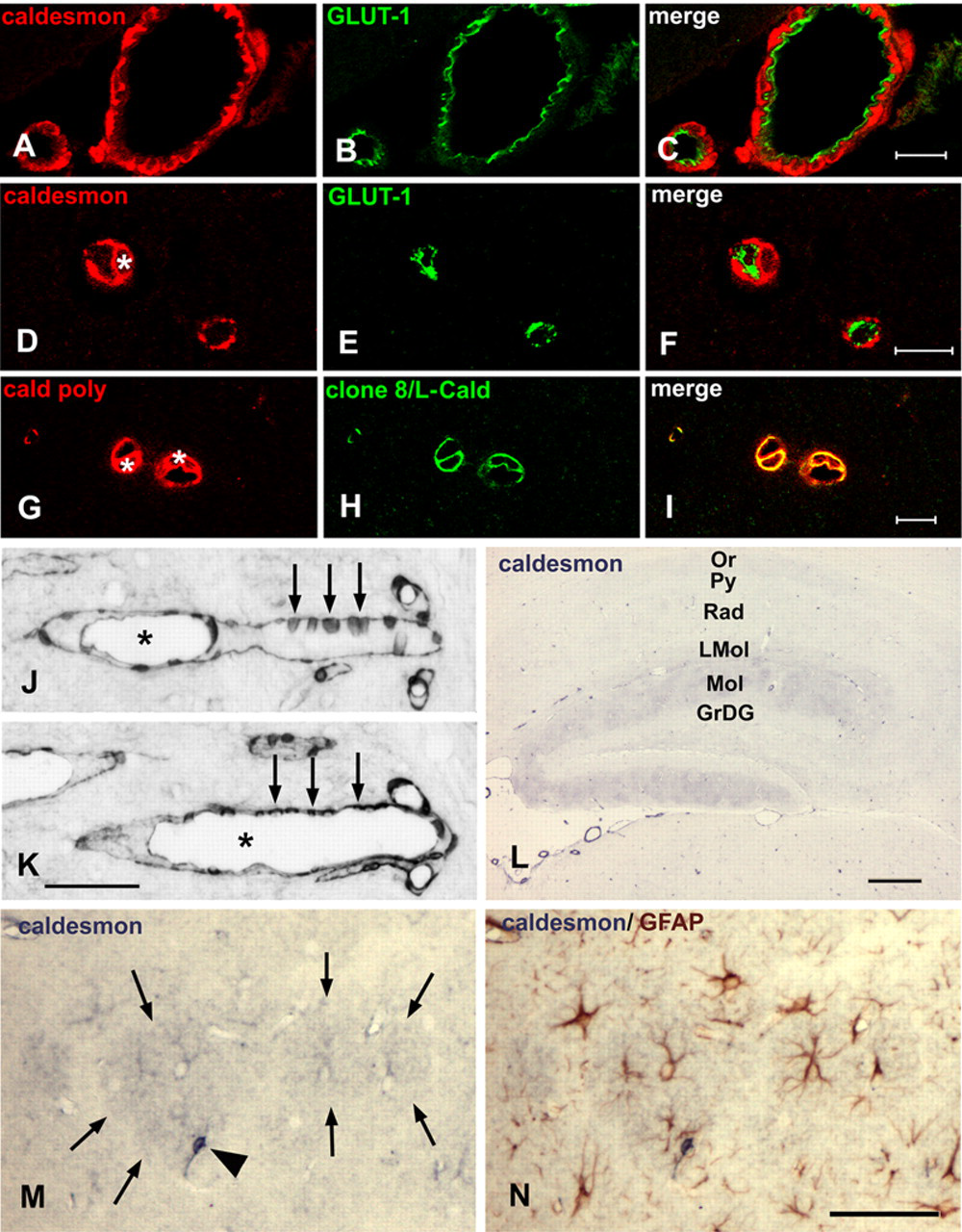
Caldesmon immunoreactivity of smooth muscle cells, pericytes, endothelial cells, and astrocytes. (A–C) Confocal microscopy analysis at 0.5 µm optical thickness of meningeal blood vessels, 4% paraformaldehyde (PFA)–fixed paraffin section. Caldesmon is located in smooth muscle cells. There is only little co-location of caldesmon and the endothelium marker GLUT-1. (D–F) Confocal microscopy analysis at 0.5 µm optical thickness of small cortical blood vessels, 4% PFA-fixed paraffin section. Caldesmon is located in a cell resembling a pericyte (the nucleus is marked by an asterisk in D). There is no co-location of caldesmon and the endothelium marker GLUT-1. (G–I) Confocal microscopy analysis at 0.5 µm optical thickness of small cortical blood vessels, 4% PFA-fixed paraffin section. The rabbit polyclonal antibody and clone 8/L-Caldesmon label the same cells (nuclei are marked by asterisks in G). (J, K) Consecutive 4% PFA-fixed paraffin sections of a medium-sized blood vessel in the brainstem. Cells discontinuously located on the outer surface of the vessel wall (arrows point to examples), reminiscent of pericytes, are stained with the rabbit polyclonal antibody (J) and clone 8/L-Caldesmon (K); the asterisks mark the lumen of the blood vessel. (L) Four percent PFA-fixed paraffin section of the hippocampus. At low magnification, the molecular layer of the dentate gyrus displays moderate and patchy caldesmon immunoreactivity. Note weak immunoreactivity of the oriens layer and stratum radiatum, the staining of blood vessels, but the absence of significant staining of neuronal perikarya, most obviously in the pyramidal and granule cell layers. (M) A section temporarily cover-slipped after staining with the rabbit polyclonal antibody showing the molecular layer of the dentate gyrus at higher magnification. Unstained nuclei and stained coarse processes are visible in the center of the stronger immunoreactive regions (delineated by arrows) resembling astrocytes stained including their fine processes. The arrowhead points to a stronger immunoreactive cell resembling a pericyte. (N) Consecutive staining of the same section for glial fibrillary acidic protein (GFAP) confirms that the moderate caldesmon-immunoreactive cells are astrocytes. The cell resembling a pericyte is not double stained. GrDG, granular layer of dentate gyrus; LMol, lacunosum moleculare layer; Mol, molecular layer of dentate gyrus; Or, oriens layer; Py, pyramidal cell layer; Rad, stratum radiatum. Scale bar: A–C = 20 µm; D–F = 10 µm; G–I = 10 µm; J, K = 50 µm; L = 200 µm; M, N = 50 µm.
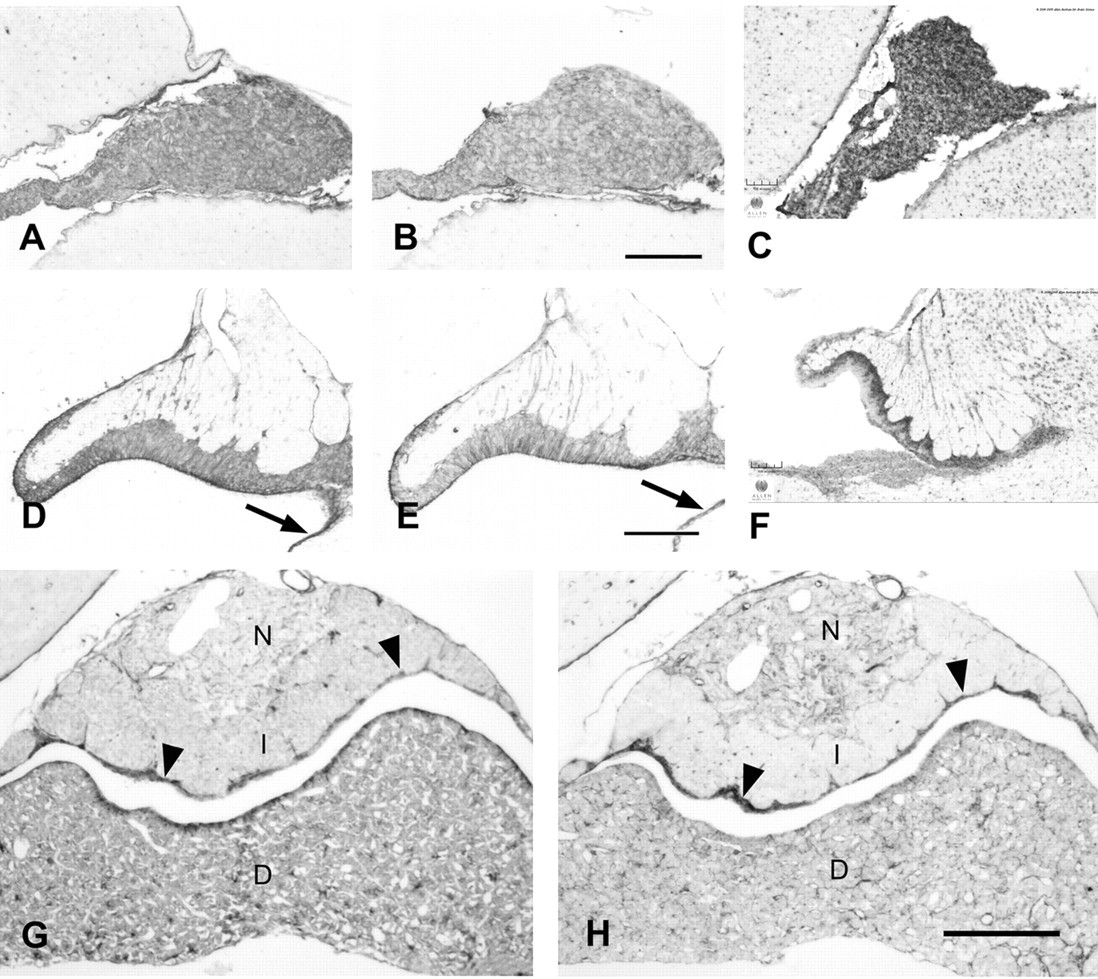
Caldesmon immunoreactivity of ependymal cells and secretory cells. Consecutive 4% paraformaldehyde (PFA)–fixed paraffin sections stained with the caldesmon rabbit polyclonal antibody (A, D, G) or clone 8/L-Caldesmon (B, E, H) after microwave pretreatment in citrate buffer and comparison with results from in situ hybridization as shown in the Allen Mouse Brain Atlas (C, F) (Allen Mouse Brain Atlas [Internet]. Seattle (WA): Allen Institute for Brain Science. 2009. Available from: http://mouse.brain-map.org). (A–C) Secretory cells of the pineal gland are immunoreactive with the rabbit polyclonal antibody or clone 8/L-Caldesmon, consistent with expression of caldesmon as shown in the Allen Mouse Brain Atlas. (D–F) Sagittal sections of the subcommissural organ show immunostaining of ependymocytes; caldesmon immunoreactivity obtained with the rabbit polyclonal antibody or clone 8/L-Caldesmon is located at their ventricular pole and lateral aspect. Results from the Allen Mouse Brain Atlas show expression in the same cells. Arrows point to immunoreactive ependymal cells lining the third ventricle. (G, H) Mouse pituitary gland consisting of pars distalis (D), pars intermedia (I), and pars nervosa (N). The rabbit polyclonal antibody and clone 8/L-Caldesmon label elongated cells in the pars nervosa. There is also strong staining of epithelial cells lining the cleft of Rathke’s pouch (arrowheads). The pars distalis of the adenohypophysis displays strong immunoreactivity of stellate cells and slender elongated processes lining the sinus as well as immunoreactivity of secretory cells with the rabbit polyclonal antibody. Clone 8/L-Caldesmon stains the same cells but secretory cells weaker. Scale bar: A, B = 100 µm; C = 105 µm; D, E = 50 µm; F = 105 µm; G, H = 100 µm.
The goat anti-caldesmon (N-19) antibody, when used diluted 1:50 for staining of 2% PFA/0.5% glutaraldehyde-fixed brain sections (the fixative used by Agassandian K and Cassell 2008), produced an overall punctuate to rod-like staining with foci of stronger staining. It also labeled several small blood vessels. In 4% PFA-fixed frozen sections, mainly blood vessels were weakly to moderately stained (Fig. 3F); in 4% PFA-fixed frozen sections with microwave pretreatment, blood vessels were labeled and there was widespread staining of nuclei.
Caldesmon Immunoreactivity in the Spinal Cord and Ganglia
Sections of spinal cord were only pretreated in citrate buffer (pH 6.0). Staining of spinal cord sections processed in parallel was absent when the caldesmon rabbit polyclonal antibody was replaced by the Aβ42 antibody at a similar concentration (not shown) or when primary antibodies were omitted (Fig. 6B). There was a clear contrast between the unstained white matter and the neuropil of the gray matter also displaying several moderately stained neuronal perikarya with the caldesmon rabbit polyclonal antibody. Blood vessels and ependymal cells lining the central canal displayed stronger immunoreactivity (Fig. 6A); as in the brain, surrounding meninges were labeled with the rabbit polyclonal antibody (Fig. 6A) and clone 8/L-Caldesmon. In the dorsal root ganglion (DRG; Fig. 6C,D) and trigeminal ganglion (not shown), neurons and surrounding satellite cells were immunostained.
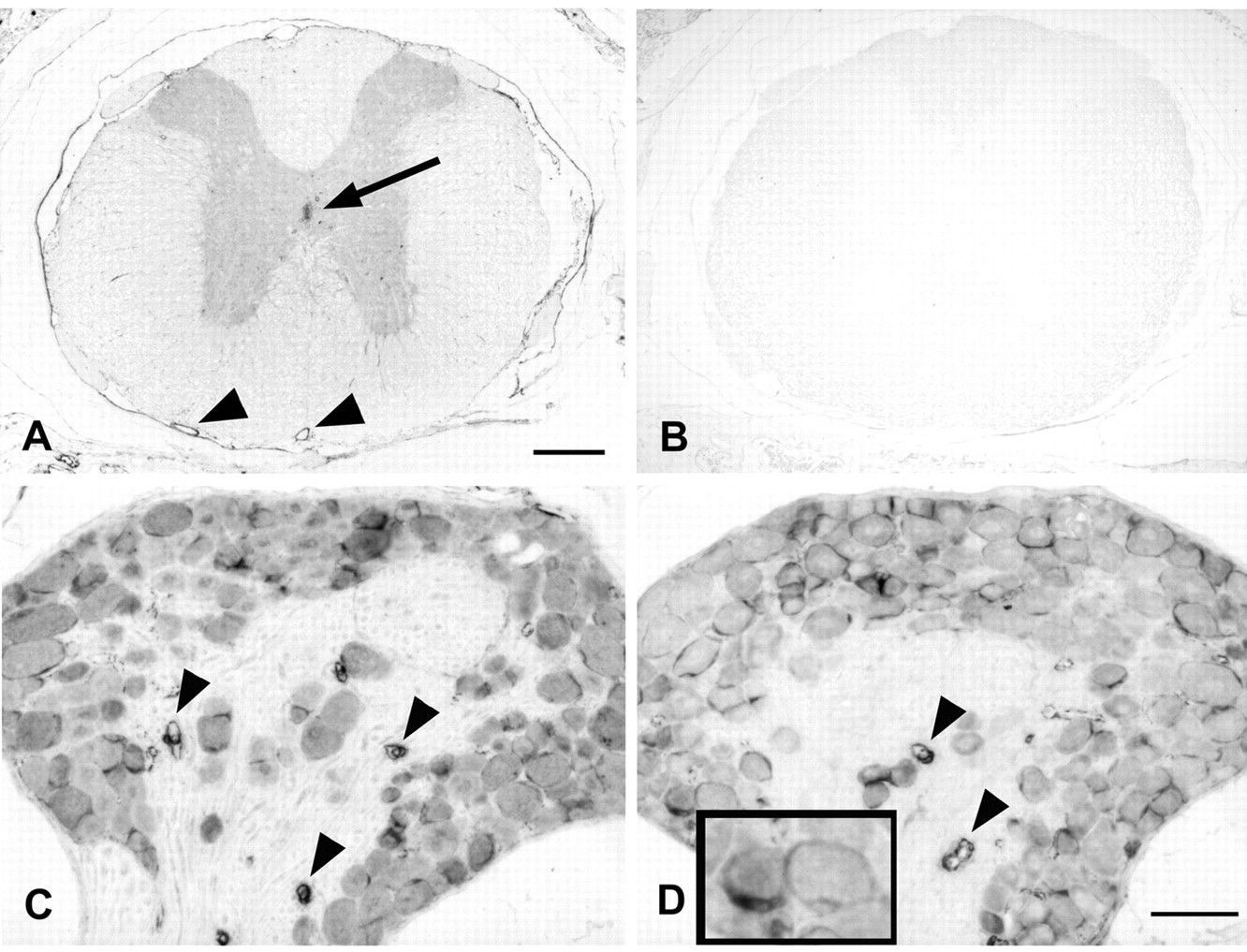
Caldesmon immunoreactivity of mouse spinal cord and dorsal root ganglia (DRGs). Four percent paraformaldehyde (PFA)–fixed paraffin sections of mouse spinal cord and ganglia stained with the caldesmon rabbit polyclonal antibody (A, C) or clone 8/L-Caldesmon (D). (A) Thoracic spinal cord displays moderately stained gray matter, stronger stained ependymal cells lining the central canal (arrow in A), blood vessels (arrowheads), and meninges. Some neuronal perikarya within the gray matter display moderate but distinct immunoreactivity. (B) Staining is absent from a nearby section when the primary antibody was omitted. (C, D) Neurons in the DRG are moderately stained with the rabbit polyclonal antibody and clone 8/L-Caldesmon. Satellite cells (insert in D) and blood vessels (arrowheads) display stronger immunoreactivity. Scale bar: A, B = 200 µm; C, D = 50 µm.
Discussion
Various cell types in the mouse CNS and ganglia displayed caldesmon immunoreactivity consistent with the notion that l-caldesmon is ubiquitously expressed (Sobue and Fukumoto 2010). The pattern of immunoreactivity obtained with the caldesmon rabbit polyclonal antibody and clone 8/L-Caldesmon were very similar; blood vessels were most strongly labeled. These antibodies also detected several cell types which have not previously been reported to express caldesmon: ependymal cells lining the ventricles, the ependymal cell layer of the choroid plexus, secretory cells in the pineal gland, stellate cells in the pituitary gland, astrocytes in the molecular layer of the dentate gyrus, and neurons and satellite cells of dorsal root and trigeminal ganglia.
Western blot analysis supports the specificity of the rabbit polyclonal antibody and clone 8/L-Caldesmon for caldesmon. We could not confirm the specificity of clone h-CD for mouse h-caldesmon by Western blotting. Clone h-CD was used in many histopathological studies to detect human h-caldesmon; the almost exclusive staining of mouse smcs in this study is consistent with what has been reported for human tissues. However, human ependymal cells were not stained with the h-caldesmon antibody (Köhler, unpublished results), and therefore the immunoreactivity of mouse ependymocytes may not be specific. The rabbit polyclonal antibody and clone 8/L-Caldesmon recognized the same double band in the brain with molecular weights corresponding to l-caldesmon, suggesting that l-caldesmon isoforms exist in the mouse brain as has been reported for the rat hippocampus where two-dimensional electrophoresis separated three isoforms of l-caldesmon (Agassandian C et al. 2000).
We have compared our immunohistochemical results to images in the Human Protein Atlas (www.proteinatlas.org) that show human paraffin-embedded tissues stained with three different, well-characterized antibodies against human caldesmon (M3557, mouse monoclonal, Clone h-CD, Dako; HPA008066 and HPA017330, rabbit polyclonals, Atlas antibodies or Sigma Prestige Antibodies). The distribution of caldesmon in human cortex, hippocampus, and cerebellum shown in the atlas (Table 1) and the distribution in spleen and lymph nodes are very similar to the distribution reported here and elsewhere (Köhler 2010) for the mouse. Taken together, results from five different antibodies, obtained in two independent laboratories, show a very similar distribution of caldesmon in human and mouse brain, spleen, and lymph nodes, suggesting that these antibodies recognize caldesmon in tissue sections.
Distribution of Caldesmon in Human Cortex, Hippocampus, and Cerebellum Obtained with Three Different Antibodies against Caldesmon as Shown in the Human Protein Atlas (www.proteinatlas.org, visited October 2010)
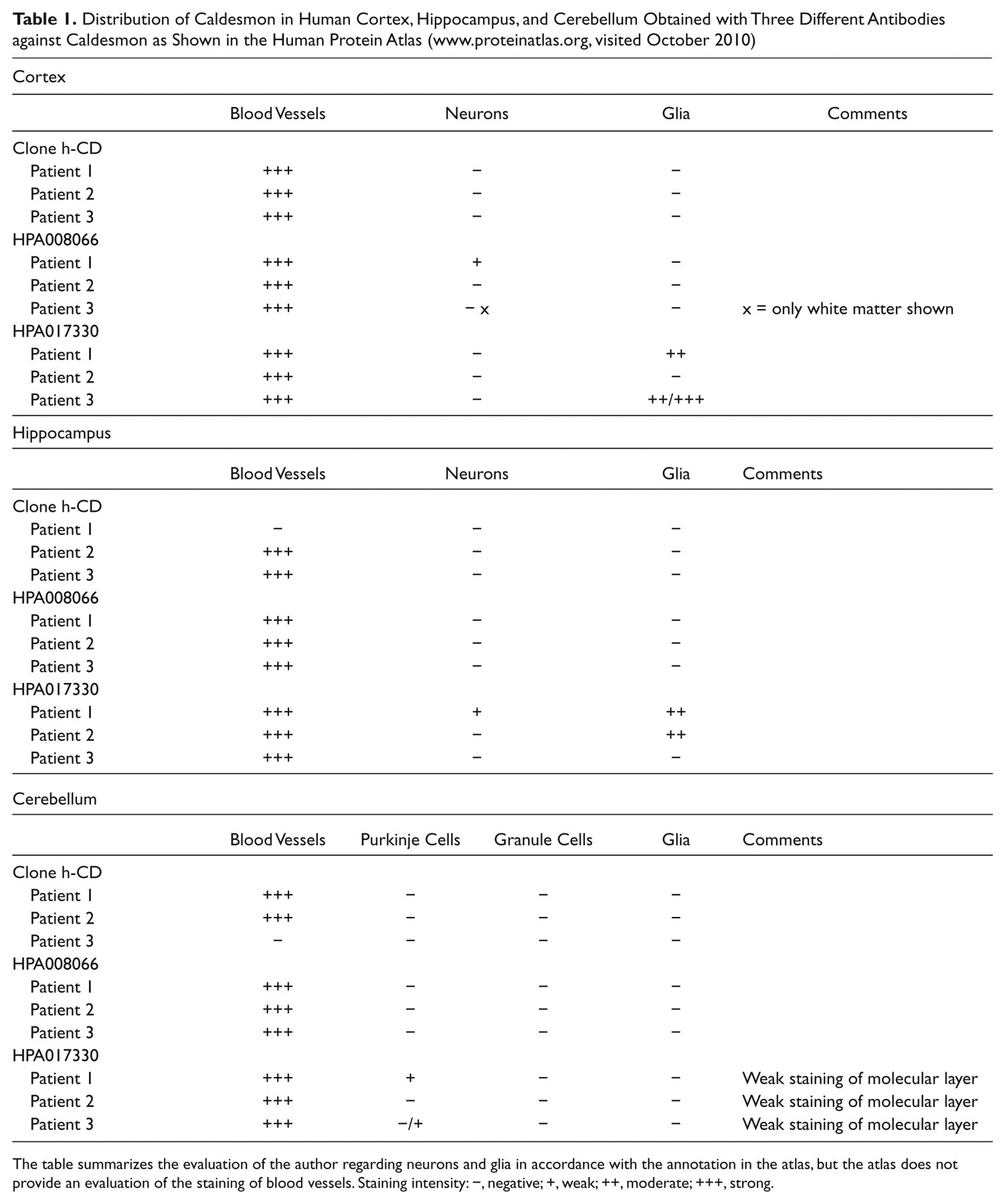
The table summarizes the evaluation of the author regarding neurons and glia in accordance with the annotation in the atlas, but the atlas does not provide an evaluation of the staining of blood vessels. Staining intensity: −, negative; +, weak; ++, moderate; +++, strong.
We have also compared our immunohistochemical results to the Allen Mouse Brain Atlas (www.brain-map.org). Two image series in the atlas show in situ hybridization with two different probes, each defining a sequence that is present in both h- and l-caldesmon. Image series 70919250 presents a comparably weak reaction, whereas image series 68795453 shows stronger staining. The analysis of the degree of caldesmon gene expression in the mouse brain that can be seen in the expression-analysis-view of the atlas is in good agreement with our immunohistochemical results, confirming comparably high expression in meningeal and medium-sized blood vessels, high expression in the choroid plexus, expression in ependymal cells, and comparably high expression in the pineal gland (the pituitary gland is not shown in the atlas). Expression in meningeal and medium-sized blood vessels is often higher than in many neurons. However, there is high expression in granule cells in the dentate gyrus without obvious correlation to our findings, but if caldesmon is localized in dendrites, the absence of immunostaining of neuronal perikarya would not indicate that caldesmon is not present in neurons. We have also compared our results from spinal cord and the DRG to the Allen Mouse Spinal Cord Atlas, which shows expression in blood vessels, in neurons in the gray matter of the spinal cord, in DRGs, and in ependymal cells lining the central canal similar to our immunohistochemical results.
Ban et al. (1984) reported that smcs were immunostained for caldesmon in all of the various organs of rats examined, and thus immunoreactivity of meningeal blood vessels in the brain can be expected. Our results are also in line with Fukumoto et al. (2009), who reported numerous densely stained small blood vessels in the embryonic rat cortex in addition to migrating neurons. Migrating neurons may have an increased content of caldesmon concentrated in the perikaryon. In contrast, Represa et al. (1995), C. Agassandian et al. (2000), and K. Agassandian and Cassell (2008) reported preferential or exclusive caldesmon immunoreactivity of neurons. Represa et al. stained 2% PFA-fixed vibratome sections and used an antibody against pig platelet caldesmon. Possibly, these conditions were optimal to detect caldesmon in neurons. C. Agassandian et al. (2000) presumably used the same antibody and an antibody against chicken gizzard caldesmon that recognized l- and h-caldesmon in Western blots of rat hippocampus. They attributed the higher molecular weight band to caldesmon in blood vessels. Agassandian et al. demonstrated caldesmon only in dendrites, not in perikarya, which is in line with our results. Agassandian and Cassell used an antibody that is commercially available. They confirmed the specificity of this antibody by preabsorbtion with the immunizing peptide. We have shown here that this antibody stains blood vessels if applied on PFA-fixed frozen sections.
Expression of caldesmon has been shown in endothelial cells in culture (e.g., Stasek et al. 1992; Mirzapoiazova et al. 2005; Bogatcheva et al. 2006). Zheng et al. (2004) attributed caldesmon immunoreactivity of normal brain blood vessels in tissue sections to endothelial cells, but Zheng et al. (2003) reported expression in all cells (endothelial cells, smcs, and pericytes) of normal and tumor-associated blood vessels. Kreipke et al. (2006) showed only little staining of endothelial cells in the rat hippocampus under normal conditions similar to our results from the mouse. It is possible that caldesmon is more concentrated in human than in rodent endothelial cells.
Stronger immunostaining of caldesmon in the molecular layer of the mouse dentate gyrus shown in this study was due to weak staining of astrocytes. We also observed some co-location of caldesmon and GFAP in the molecular layer of the cerebellum, consistent with weak immunoreactivity of Bergmann glia, but found no obvious caldesmon immunoreactivity of astrocytes in other regions. This first report about the presence of caldesmon in a subpopulation of mouse astrocytes in situ is in line with a previous report about the expression of caldesmon in cultured mouse astrocytes. Caldesmon was demonstrated in immature astrocytes in culture but not in small stellate astrocytes that appeared in older cultures (Abd-el-Basset et al. 1991). Caldesmon could also be present in other components of the molecular layers of the dentate gyrus and cerebellum because results from the Allen Mouse Brain Atlas show its expression by granule cells in the dentate gyrus and cerebellum.
Interestingly, stronger caldesmon immunoreactivity of the neuropil as reported here corresponds to high concentrations of filamentous actin in the molecular layer of the cerebellum and in the hippocampus, mainly located in dendritic spines but nearly absent from neuronal perikarya (Capani, Martone, et al. 2001). Filamentous actin was also demonstrated in pericytes and some astrocytic processes (Capani, Ellisman, et al. 2001). The effects of caldesmon on polymerizing actin have recently been demonstrated in vitro (Huang et al. 2010). The similar histological distribution of caldesmon and high concentrations of filamentous actin in the brain suggest a role of caldesmon in regulating the assembly of actin in diverse cell types, including neurons, likely to result in different cellular functions. There is presently little experimental evidence for a function of caldesmon in neurons beyond regulating migration and neurite outgrowth (Kira et al. 1995; Alexanian et al. 2001; Fukumoto et al. 2009). K. Agassandian and Cassell (2008) referred to Ferhat et al. (2003) for a role of caldesmon in dendritic spine remodeling, but Ferhat et al. did not mention caldesmon in this article.
The distribution of caldesmon in the brain reported here points to a major role of caldesmon in blood vessels. Pericytes of smaller blood vessels appear as the continuation of contractile smcs, but pericytes are thought to have diverse functions (for review, see Krueger and Bechmann 2010). Subpopulations of pericytes express the smooth muscle isoform of actin (for review, see Allt and Lawrenson 2001) as do subpopulations of reticular cells in spleen and lymph nodes. Pericytes and reticular cells are strongly stained for caldesmon in tissue sections with the rabbit polyclonal antibody and clone 8/L-Caldesmon but not with clone h-CD, suggesting a comparably high content of l-caldesmon in non-muscular cells that are capable of expressing the smooth muscle isoform of actin.
Confirming the significance of the immunohistochemical staining of secretory cells in the pituitary and pineal gland for caldesmon, expression of caldesmon in rat pituitary cells was reported earlier based on immunofluorescence of isolated cells, and a role of caldesmon in exocytosis was suggested (Natarajan et al. 1991; for review, see Dabrowska et al. 2004). Similarly, caldesmon was demonstrated in the apical portion of epithelial cells of several epithelia outside the CNS (Ban et al. 1984; Ishimura et al. 1984; Fujita et al. 1984; Bretscher and Lynch 1985). In the brain of the mouse, this intraepithelial localization is clearly visible in the elongated ependymal cells of the subcommissural organ. Filamentous actin has also been demonstrated in the apical portion of ependymocytes (Li et al. 2007; Li et al. 2009).
In conclusion, immunohistochemistry of fixed tissue sections with different antibodies detects caldesmon in the CNS of the mouse predominately in blood vessels but also in several other cell types consistent with a ubiquitous expression of l-caldesmon. Our results suggest marked differences in the concentration of l-caldesmon in different cell types.
Footnotes
Acknowledgements
The excellent technical assistance of Mrs. M. Dinekov is gratefully acknowledged.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and authorship of this article.
