Abstract
Cytoglobin (Cygb) is a novel tissue hemoprotein relatively similar to myoglobin (Mb). Because Cygb shares several structural features with Mb, we hypothesized that Cygb functions in the modulation of oxygen and nitric oxide metabolism or in scavenging free radicals within a cell. In the present study we examined the spatial and temporal expression pattern of Cygb during murine embryogenesis. Using in situ hybridization, RT-PCR, and Northern blot analyses, limited Cygb expression was observed during embryogenesis compared with Mb expression. Cygb expression was primarily restricted to the central nervous system and neural crest derivatives during the latter stages of development. In the adult mouse, Cygb is expressed in distinct regions of the brain as compared with neuroglobin (Ngb), another globin protein, and these regions are responsive to oxidative stress (i.e., hippo-campus, thalamus, and hypothalamus). In contrast to Ngb, Cygb expression in the brain is induced in response to chronic hypoxia (10% oxygen). These results support the hypothesis that Cygb is an oxygen-responsive tissue hemoglobin expressed in distinct regions of the normoxic and hypoxic brain and may play a key role in the response of the brain to a hypoxic insult.
T
Hemoglobin and myoglobin (Mb) are abundant hemoproteins that have been extensively examined (Wittenberg and Wittenberg 1989,1990; McMahon et al. 2002; Garry et al. 2003). Hemoglobin is a heterotetrameric hemoprotein located within erythrocytes and functions to transport ligands such as oxygen and nitric oxide within the mammalian circulatory system (Finch and Lenfant 1972; Salhany et al. 1973; McMahon and Stamler 1999; stamler 2003). Mb is a monomeric hemoprotein restricted to cardiomyocytes and oxidative myofibers in skeletal muscle (Wittenberg and Wittenberg 1989,2003). Recent studies using pharmacological, transgenic, and gene-disruption strategies support a functional role for Mb in the storage and transport of oxygen, as an inactivator of nitric oxide and as a scavenger of reactive oxygen species within striated muscle (Wittenberg 1965; Wittenberg et al. 1985; Wittenberg and Wittenberg 1987; Doeller and Wittenberg 1991; Garry et al. 1998; Godecke et al. 1999; Flogel et al. 2001,2004; Grange et al. 2001; Meeson et al. 2001; Mammen et al. 2003).
Recently, two new tissue hemoglobins, neuroglobin (Ngb) and cytoglobin (Cygb), have been identified in mammals. Ngb is tissue restricted to neuronal tissue (Burmester et al. 2000; Mammen et al. 2002; Zhang et al. 2002), whereas Cygb is more broadly expressed in adult tissues (Kawada et al. 2001; Burmester et al. 2002; Trent and Hargrove 2002). Cygb is a 190-amino acid hemoprotein with a calculated molecular mass of 21 kDa and has a sequence that is highly conserved in the human, rat, and mouse. Unlike Mb and Ngb, the Cygb protein is encoded by four rather than three exons, and its open reading frame is composed of 570 nucleotide base pairs (Burmester et al. 2002; Trent and Hargrove 2002). However, like the other tissue hemoglobins, the heme-binding domain is encoded by exon 2 (Trent and Hargrove 2002).
Studies exploring the functional role(s) for Cygb in mammalian tissues are limited. Several studies have identified the potential ligands and their binding kinetics to Cygb (Trent and Hargrove 2002; de Sanctis et al. 2004a, b; Fago et al. 2004; Sugimoto et al. 2004; Weiland et al. 2004; Sawai et al. 2005), but limited data are available regarding the expression pattern of this novel hemoprotein (Burmester et al. 2002; Trent and Hargrove 2002; Schmidt et al. 2004), and there are no studies assessing expression of Cygb during embryogenesis or in response to chronic hypoxia. Therefore, in the present study we utilized in situ hybridization (ISH) to examine the spatial and temporal expression pattern of Cygb during murine embryogenesis. Due to the relative similarity in the protein structure between Cygb and Mb (Burmester et al. 2002; Trent and Hargrove 2002), we compared the expression pattern of this novel hemo-protein during development with Mb. In addition, we compared the spatial expression of Cygb with Ngb, the only other known hexacoordinated hemoglobin in vertebrates. Finally, we further examined the response of Cygb to chronic hypoxia in adult mice. These studies further characterize a novel tissue hemoglobin that is expressed and induced in tissues sensitive to hypoxic stress.
Materials and Methods
Animals and Hypoxia Chamber
Adult ICR and C57BL/6 mice (2–4 months of age) were handled and used in accordance with National Institutes of Health (NIH) and University of Texas Southwestern Medical Center's Institutional Guidelines. A plexiglass hypoxic chamber (87 × 42 × 45 cm) was engineered to maintain a constant hypoxic environment of 10% oxygen, and it was monitored continuously for oxygen and carbon dioxide concentrations, temperature, and humidity. A detailed description of the hypoxic chamber has been previously published (Mammen et al. 2002,2003).
Embryo Tissue Processing
ICR strain mouse embryos from timed pregnancies were harvested at gestational days E9.5, E12.0, E13.5, and E16.5 and staged according to somite numbers and age-dependent anatomic features. Embryos were fixed in 4% paraformaldehyde (PFA)/ diethylpyrocarbonate (DEPC)-PBS overnight at 4C, dehydrated, and embedded in paraffin for routine rotary microtomy (Shelton et al. 2000).
Adult Tissue Processing
In accordance with NIH and Institutional Guidelines for animal use, tissues were harvested from normoxic and hypoxic C57BL/6 adult male mice. Each mouse was anesthetized with Avertin (Sigma-Aldrich; St Louis, MO), and a thoracotomy was performed to expose the heart. Following transcardiac perfusion with ~10 ml of ice-cold DEPC-PBS, mice were subsequently perfused with 10 ml of 4% PFA/DEPC-PBS (Garry et al. 1996; Shelton et al. 2000; Mammen et al. 2002). Brains were harvested, postfixed for 14 to 16 hr at 4C, and then stored in DEPC-PBS. Brains were then cryoprotected with 10% sucrose DEPC-PBS and frozen for sled cryotomy (Garry et al. 1996; Shelton et al. 2000; Mammen et al. 2002). Survey of the brain was conducted with 40-μm sled cryotome sections cut at 120-μm intervals. The survey included the entire rostral-to-caudal extremes of the coronal mouse brain.
Vector Constructs
Fragments of the Mb, Ngb, and Cygb genes were amplified from murine heart and brain cDNA, respectively, using primer pairs that spanned the intron between exon 2 and exon 3. Mb primer set produced a 503-bp amplicon and was prepared as previously published (Parsons et al. 1993). Ngb primer set produced a 157-bp amplicon as previously described (Mammen et al. 2002). Cygb primer set produced a 396-bp amplicon. The oligonucleotide primer set for the Cygb amplicon was purchased from Sigma-Genosys (The Woodlands, TX), and the sequences utilized for amplification were as follows: Cygb forward primer: 5′-CCCGGCTGTATGCCAACT-3′; Cygb reverse primer: 5′-GGTCACGTGGCTGTAGATGA-3′.
Mb, Ngb, and Cygb amplicons were gel extracted using the Qiaquick Gel Extraction Kit (Qiagen; Valencia, CA) ligated into either the pGEM-3Z (Mb insert; Promega, Madison, WI) or the pCR II (Ngb and Cygb inserts; Invitrogen, Carlsbad, CA) plasmids and transformed utilizing the TA Cloning Kit-Dual Promoter (Invitrogen) (Parsons et al. 1993; Garry et al. 1996; Shelton et al. 2000; Mammen et al. 2002). Purified plasmids were isolated and sequenced to determine the orientation of the subcloned Mb and Cygb products.
RNA Isolation and Semiquantitative RT-PCR
Total RNA was isolated from embryonic (E17.5), neonatal (postnatal day 2; P2) and adult (2–4 months of age) murine brains utilizing the TriPure Isolation Kit (Roche Diagnostics Corporation; Indianapolis, IN). Reverse transcription (RT) was performed using Superscript II RNase H-Reverse Transcriptase (Invitrogen) to obtain cDNA. Briefly, 6 μg of total RNA was used in each reverse transcription reaction (60 μl). Two μl of diluted cDNA was used as a template for the PCR reaction in a 12-μl reaction volume. Semiquantitative RT-PCR analysis utilizing RNA isolated from embryonic, neonatal, and adult brains was performed as previously described (Garry et al. 1996; Shelton et al. 2000; Mammen et al. 2002) under conditions in which the abundance of each amplified cDNA varied linearly with input RNA. The primer set used to assess Cygb expression in these experiments was as follows: Cygb forward primer: 5′-TGCATGACCCAGACAAGGTA-3′; Cygb reverse primer: 5′-GGTCACGTGGCTGTAGATGA-3′
To ensure equal loading among samples, RT-PCR of the various cDNA samples was performed using the forward and reverse primers for 18s-ribosome (18s-RIB): 18s-RIB forward primer: 5′-CTCAACACGGGAAACCTCAC-3′; 18s-RIB reverse primer: 5′-TGCCAGAGTCTCGTTCGTTAT-3′.
Northern Blot Analysis
The Strip-EZ DNA Probe Synthesis Kit (Ambion; Austin, TX) was utilized to construct the Cygb radioactive probe. Twenty five ng of linearized Cygb cDNA and 5 μl of [α32P]dATP (1000 Ci/mmol; Amersham Biosciences, Piscataway, NJ) were used to produce the radioactive Cygb cDNA probe. A commercially purchased Northern blot membrane (MTN blot; BD Sciences, Palo Alto, CA) containing full-length transcripts from whole embryos at distinct stages of embryogenesis was incubated with the Cygb radioactive probe in an Ultrahyb solution (Ambion) at 42C overnight. Hybridized membranes were exposed to radiographic film at −80C for various lengths of time (6–48 hr). β-actin cDNA was provided with the Northern blot membrane and used as a control probe to ensure equal loading of the lanes with RNA.
Riboprobe Synthesis for ISH
Templates for antisense and sense Mb, Ngb, and Cygb riboprobes were linearized from the Mb-pGEM-3Z plasmid, Ngb-pCR II plasmid, and Cygb-pCR II plasmid, respectively. Radiolabeled Mb, Ngb, and Cygb RNA probes were synthesized from 500 ng of template by in vitro transcription (Maxiscript kit; Ambion) in the presence of 60 μCi of [35S]dUTP (Amersham Biosciences; Piscataway, NJ) as previously described (Garry et al. 1996; Mammen et al. 2002; Shelton et al. 2000). SP6 and T7 RNA polymerases were used to generate antisense and sense probes, respectively. Unincorporated ribonucleotides were removed by passing the transcription products over MicroSpin G-50 columns (Amersham Biosciences). Riboprobes were stored for 1–2 days at −80C prior to performing ISH.
ISH
ISH analyses were performed according to procedures previously described (Garry et al. 1996; Shelton et al. 2000; Mammen et al. 2002). Briefly, 5-μm paraffin sections and 40-μm sled cryotome sections mounted on silane-treated microscope slides (Vectabond Reagent; Vector Laboratories, Burlingame, CA) were dewaxed, permeabilized, and acetylated prior to hybridization at 70C. For hybridization, riboprobes were diluted in a mixture containing 50% formamide, 0.75 M NaCl, 20 mM Tris-HCl, pH 8.0, 5 mM EDTA, pH 8.0, 10 mM NaPo4, pH 8.0, 10% dextran sulfate, 1X Denhardt's, and 0.5 mg/ml tRNA. Following hybridization, sections were rinsed with increasing stringency washes, subjected to RNase A (2 μg/ml, 30 min at 37C), and dehydrated prior to dipping in K5 nuclear emulsion gel (Ilford; Cheshire, UK). Autoradiographic exposure ranged from 21 to 35 days. Following development, slides were counterstained with hematoxylin.
Western Blot Analysis
Protein from whole brain was isolated from normoxic mice and mice exposed to 1 week of chronic hypoxia (10% O2). Western blot analysis was performed according to a previously published protocol (Garry et al. 1998; Mammen et al. 2003). A polyclonal rabbit anti-Cygb antibody was designed and engineered by the Antibody Core Facility at the University of Texas Southwestern Medical Center. This polyclonal rabbit anti-Cygb serum (1:100 dilution) was used as the primary antisera, which was detected using a horseradish peroxidase-conjugated secondary antiserum. This antibody was specific for Cygb as determined by Western blot analyses using extracts of C2C12 myoblasts that expressed Cygb-green fluorescent fusion protein (data not shown). The mouse monoclonal α-tubulin antibody (1:3,000; Sigma) was used as a standard to ensure equal protein loading of the lanes. Band intensity was quantitated using a computerized digital analysis program (Image J 1.24; Scion Corporation, Frederick, MD).
Microscopy and Photomicrography
Cygb expression was visualized using a Laborlux-S microscope stand (Leitz; Wetzler, Germany) equipped with Plan-Apochromatic optics, a standard brightfield condenser, and a Mears low-magnification darkfield condenser (MearsCorp; Waltham, MA). Photomicrographs were obtained with an Optronics VI-470 CCD camera (Optronics; Goleta, CA) and a Power Macintosh G4 equipped with a Scion CG-7 frame grabber and Image J software (Scion Corporation).
Statistical Analysis
Data analysis of Cygb gene expression during brain development and between normoxic and hypoxic wild-type brains was performed using a two-tailed paired Student's t-test. Data values were reported as the mean ± SEM.
Results
Spatial and Temporal Expression Pattern of Cygb During Embryogenesis
A goal of this study was to define the developmental expression pattern of Cygb during murine embryogenesis and to provide a comprehensive analysis of the spatial expression pattern of Cygb in the normoxic and hypoxic adult murine brain. Northern blot analysis revealed that Cygb was predominantly expressed during the later stages of murine embryogenesis (Figure 1). There was limited Cygb expression during early and mid-gestational ages, but there was a significant increase in Cygb expression by E17.0 days post coitus (dpc) (Figure 1).
To validate our results from the Northern blot analysis and to further examine the spatial expression pattern of Cygb during embryogenesis, we performed ISH on sections from sequentially staged embryos (E9.5, E12.0, E13.5, and E16.5). Cygb expression during embryogenesis was limited and was restricted to focal regions within the developing embryo (Figure 2 and Figure 3). Specifically, there were distinct regions of expression within the head (i.e., head mesenchyme, subventricular zone of the dorsal anterior thalamus, the anterolateral region of the cortex, pituitary gland, submucosa of the oral-nasal cavity, lingual swelling, and ventral portion of the neural tube) (Figures 2D-2F and Figure 3B). In addition, at E16.5 dpc there was focal expression in the dermal epithelium (data not shown).
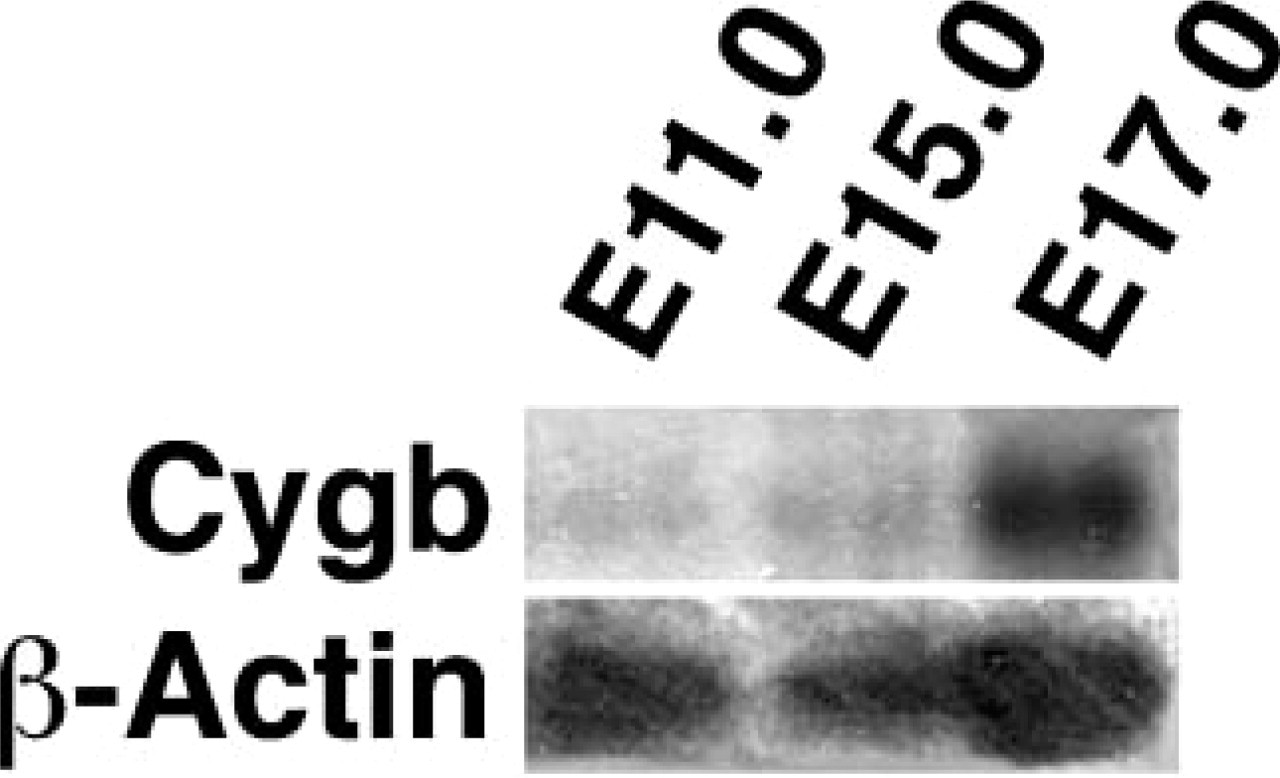
Increased cytoglobin (Cygb) expression during the later stages of embryogenesis. Northern blot analysis of total RNA isolated from specific stages of murine development reveals limited Cygb expression at midgestational ages but more prominent expression at E17.0. β-actin is used as an RNA loading control.
To demonstrate that our embryos were well preserved, we examined the expression pattern of Mb, which was restricted to the developing heart and striated skeletal muscle (Figure 3A). Mb was expressed as early as E9.5 dpc and was restricted to the developing cardiac ventricles (data not shown). By E12.0 dpc there was intense signal representing Mb transcript levels in the compact layer of the cardiac ventricle and the somites (Figure 3A). This robust expression is in contrast to the pattern of Cygb at E12.0 dpc, where expression is of lower intensity and is restricted to the head mesenchyme, lingual swelling, and ventral portion of the neural tube (Figure 2 and Figure 3).
Increased Cygb Expression in the Developing Brain
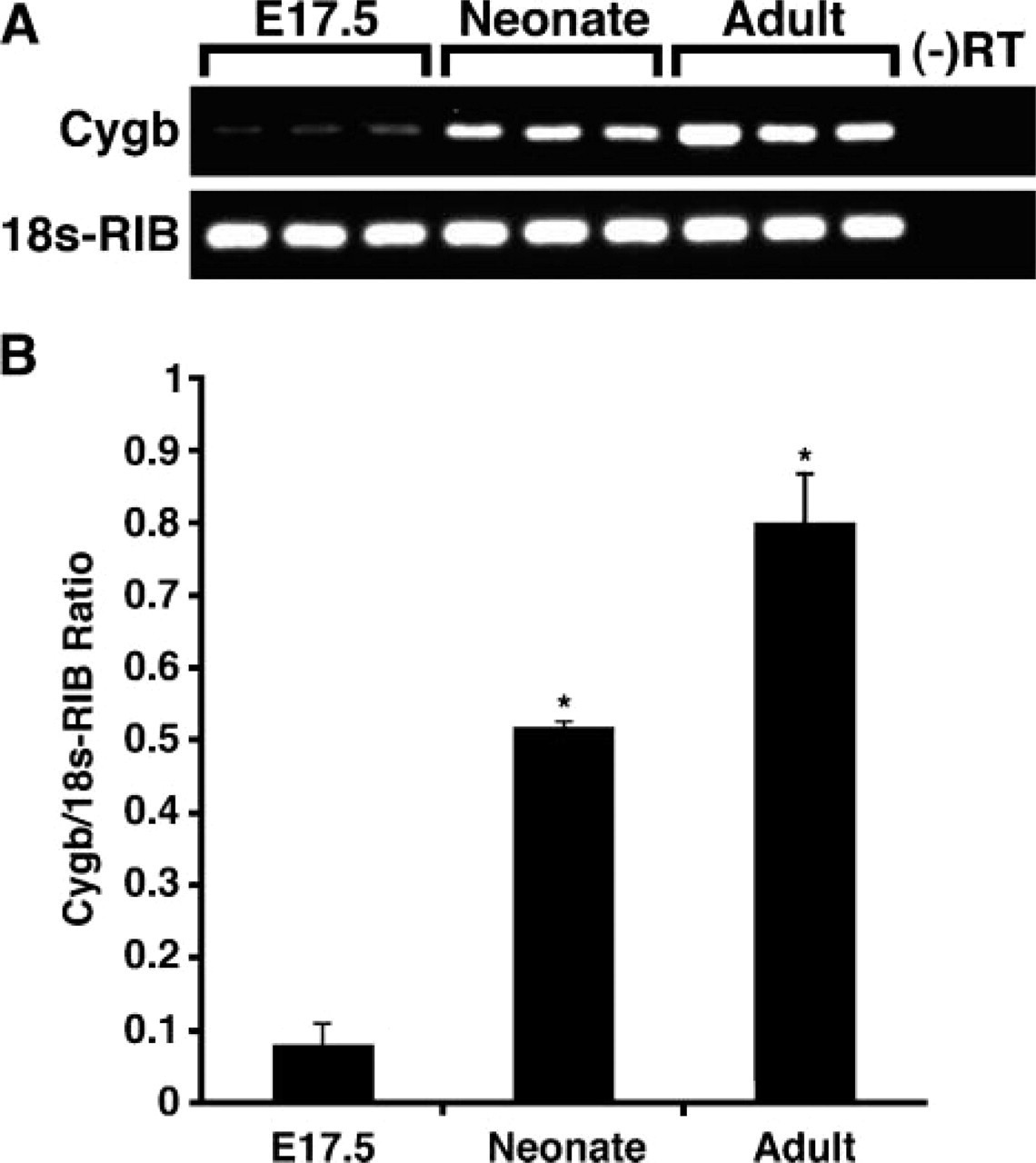
Having demonstrated increased Cygb transcript levels during the latter stages of embryogenesis, we further investigated Cygb mRNA expression levels in the neonatal and adult brains. RT-PCR techniques indicated a significant increase in Cygb transcript levels in the neonatal (7–fold increase) and the adult (10-fold increase) brains compared with the brains harvested from E17.5 embryos (Figures 4A and 4B; E17.5, 0.08 ± 0.03; neonate, 0.52 ± 0.01; adult, 0.8 ± 0.07; ∗ p<0.0005, neonate or adult vs E17.5 brains; n=3 in each group).
Cygb Is Expressed in the Adult Normoxic Brain
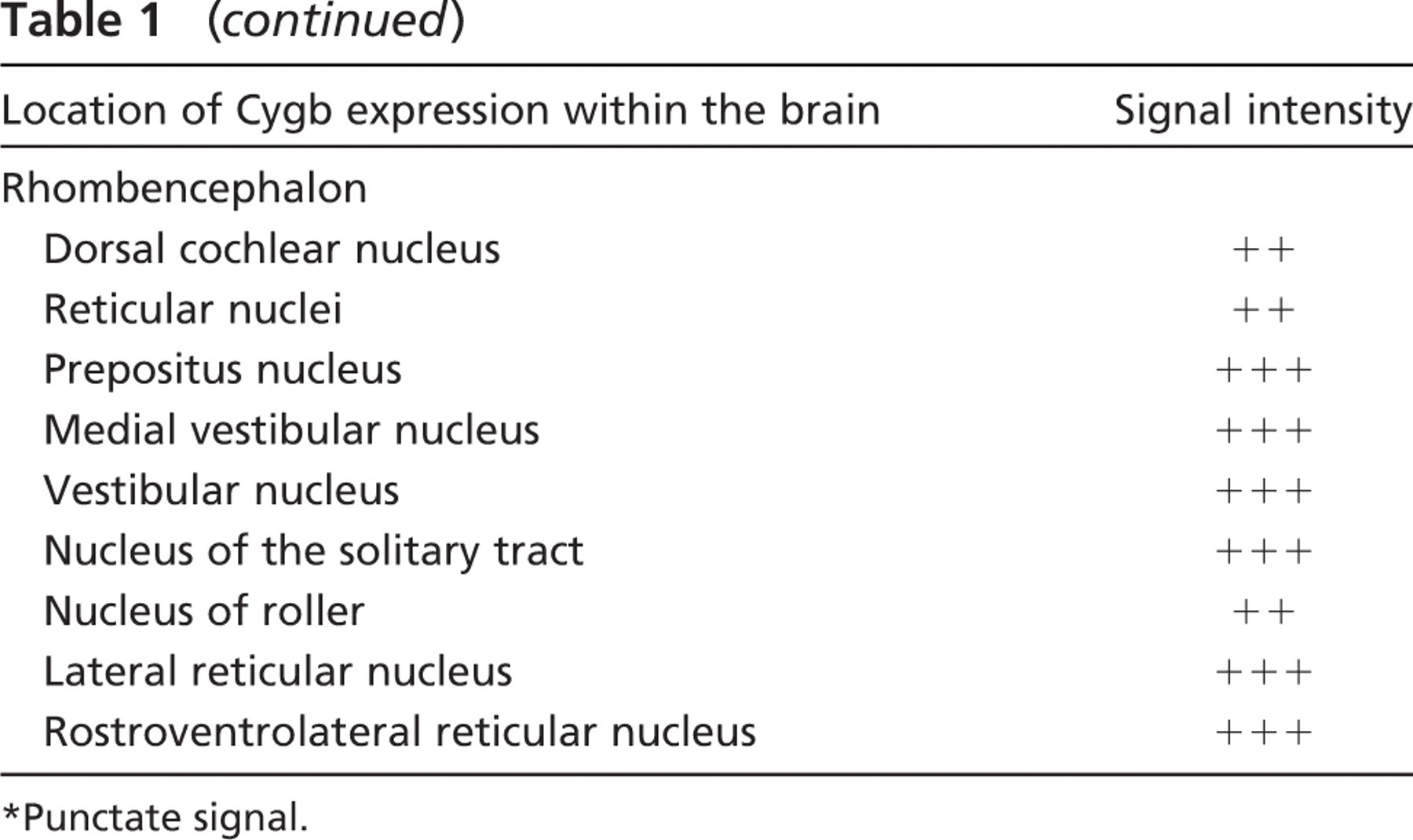
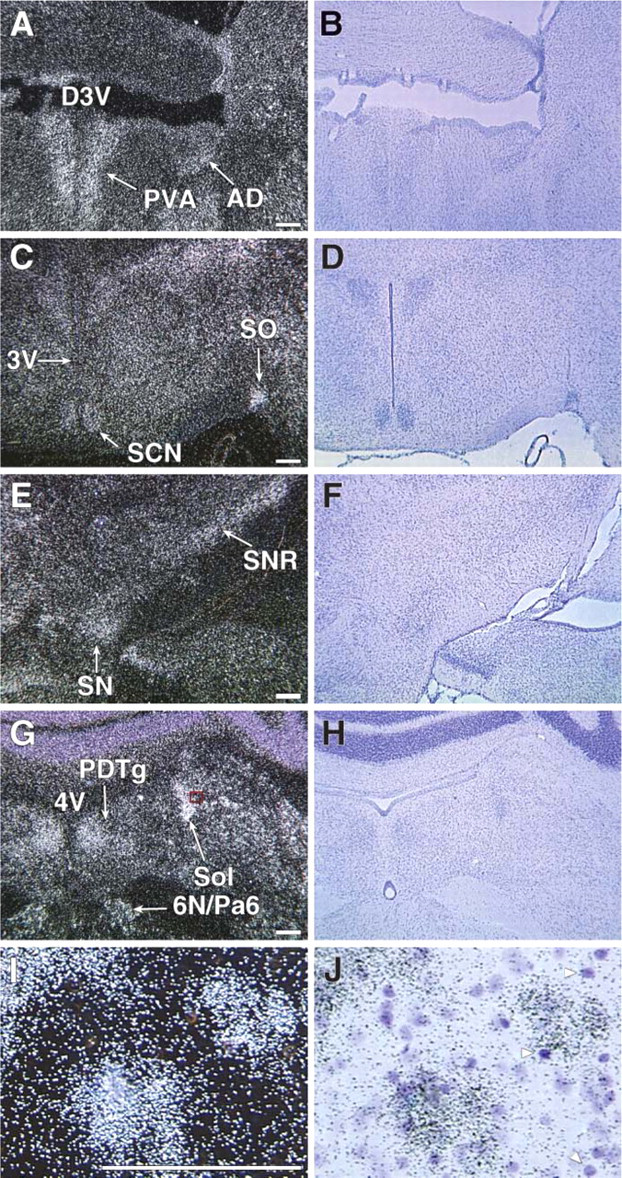
Having observed a marked induction of Cygb associated with the growth of the brain, we undertook a detailed analysis of Cygb expression in the unperturbed, normoxic adult brain. Using ISH techniques on frozen sections of the adult brain, we demonstrated intense signal representing Cygb expression localized to distinct regions of the adult brain. A comprehensive examination of the entire adult brain revealed varying intensity in Cygb transcript levels from robust expression to completely absent signal (Table 1; Figure 5 and Figure 6). Table 1 provides a detailed list of the location and signal intensity of Cygb expression throughout the brain. In the rostral aspect of the brain, the Cygb signal was robust in the piriform cortex, the medial septal nucleus, and in the nucleus of the vertical and horizontal limbs of the diagonal band (Table 1). Moderate punctate Cygb expression was observed in regions of the adult brain that are involved in circadian rhythm and cardiovascular control (i.e., paraventricular thalamic nucleus, suprachiasmatic nucleus, substantia nigra, and the nucleus of the solitary tract) (Figures 5A, 5C, 5E, and 5G). Punctate signal was observed in the olfactory nucleus (especially in the lateral and posterior areas) and in the ventral pallidum (Figure 6A). Less intense signal was observed in the lateral accumbens shell and in the dorsal endopiriform nucleus (Table 1). A diffuse signal was evident throughout the cingulate, motor, and insular cortex (Figures 6A, 6C, 6E, and 6G).
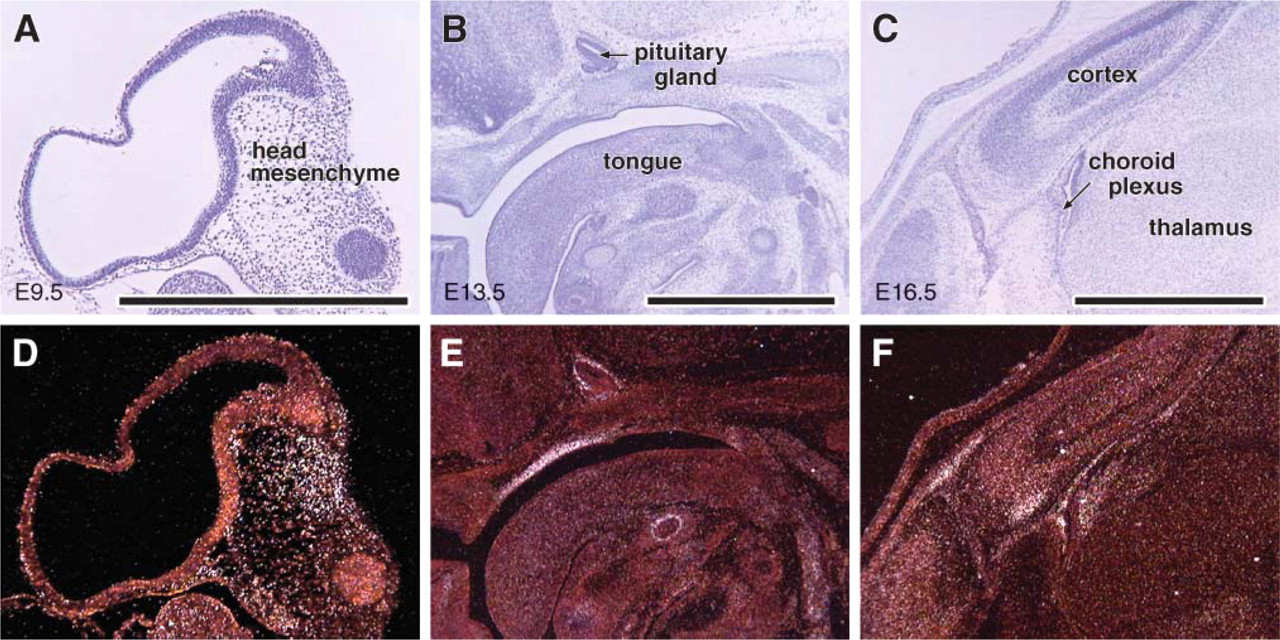
Restricted Cygb expression during embryogenesis. In situ hybridization (ISH) was utilized to hybridize parasagittal sections of embryos at various stages of development [i.e., E9.5, E13.5, and E16.5 days post coitus (dpc)] with an antisense Cygb 35S riboprobe. High magnification brightfield (
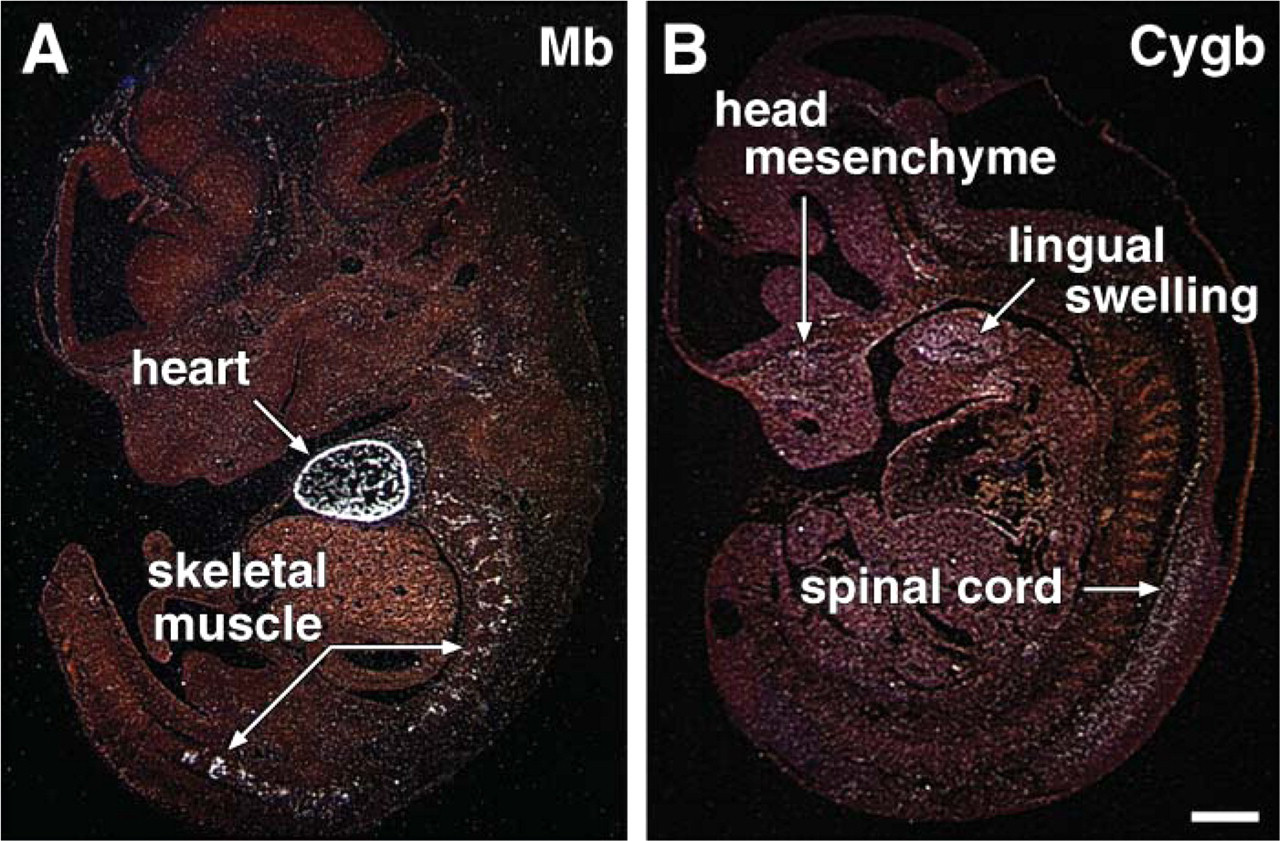
Absent Cygb expression but marked myoglobin (Mb) expression in the somites and developing heart. (
In the hippocampus, expression was strongest in the granular cell layer of the dentate gyrus (Figures 6E and 6G). Weaker, punctate signal was evident throughout the pyramidal cell layer of CA 1, 2, and 3 (Figures 6E and 6G). In the thalamus, the medial habenular nucleus was strongly positive as well as the reticular thalamic nucleus (Figure 6E). Weaker punctate staining was evident throughout the amygdala (Table 1).
Cygb expression was prominent in the mammillary body, especially in the premammillary, supramammillary, and lateral mammillary nuclei (Table 1). Expression was also evident in the interstitial nucleus of Cajal and the intrapeduncular nucleus (Figures 6G and 6I). In the pontine nuclei, Cygb expression was also prominent. In the tegmentum, there was dramatic punctate expression in the reticulotegmental nucleus of the pons, as well as the pedunculopontine tegmental nucleus and the laterodorsal tegmental nucleus (Figure 6I). Within the medulla there was signal in the locus ceruleus, prepositus nucleus, medial vestibular nucleus, medial cerebellar nucleus, vestibular cerebellar nucleus, and nucleus of the solitary tract as well as the lateral and rostroventrolateral reticular nuclei (Figures 5G and 6K). Additional regions throughout the brain expressed Cygb at a lower level, and these results are outlined in Table 1. Sections hybridized with the sense Cygb RNA probe were negative (data not shown).
Enhanced Cygb expression in the adult murine brain. (
Cytoglobin (Cygb) expression in the normoxic brain

Punctate signal.
Using ISH techniques, we further examined the cellular population that expressed Cygb in the adult brain. Our studies revealed that Cygb appears to be localized to the neuron and is not present in either the supporting glial cells or the vasculature. Examination of the signal at high magnification using both darkfield and bright field microscopy confirmed that Cygb was present in the gray matter but absent in the vasculature (Figures 5I and 5J).
Increased Cygb Expression in the Adult Hypoxic Brain
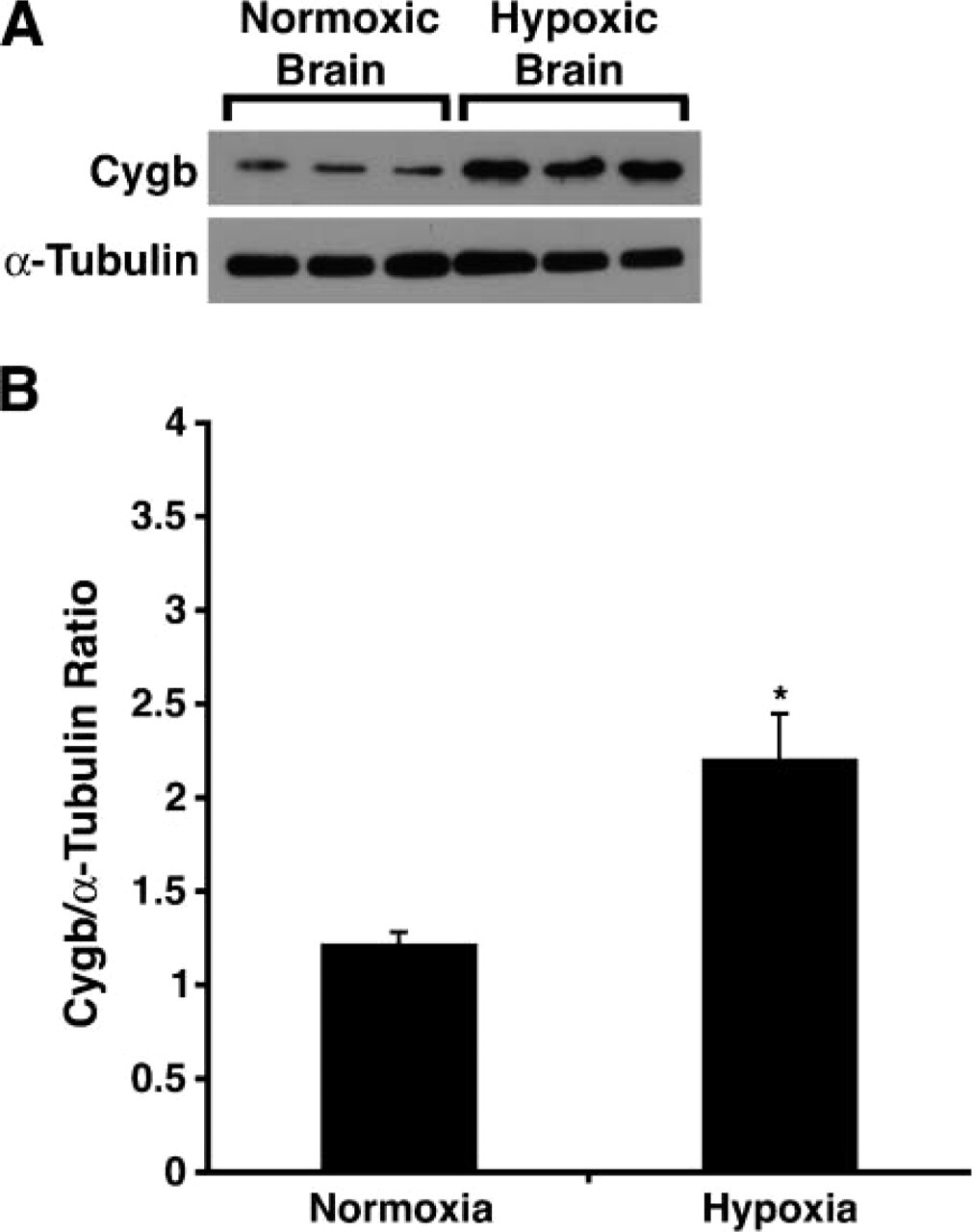
Having previously demonstrated that Mb expression (in the adult murine heart) and not Ngb expression (in the adult murine brain) was induced by chronic exposure to low oxygen tension (Mammen et al. 2002,2003) and due to the relative structural similarity between Mb and Cygb (Burmester et al. 2002; Trent and Hargrove 2002), we undertook experiments to determine whether chronic hypoxia resulted in altered expression of Cygb (i.e., new regions of Cygb expression vs enhanced Cygb signal). Adult mice were exposed to chronic 10% oxygen for a 1-week period. Although there were no new regions of Cygb expression in response to hypoxia, there was an increase in the intensity of the Cygb signal indicating an increase in transcript levels within the hypoxic brain (Figure 6). The increased intensity of the Cygb signal was observed predominantly in regions of the brain that have previously been shown to be responsive to oxidative stress (i.e., hippocampus, thalamus, and hypothalamus) (Figures 6F and 6H) (Won et al. 2002; Erikson et al. 2004; Adam-Vizi 2005; Aksenova et al. 2005; Chong et al. 2005; Dringen 2005; Mariani et al. 2005). Western blot analysis confirmed an increase in Cygb protein expression, and quantification of these results revealed a 2-fold increase of this hemoprotein in the hypoxic brain compared with the normoxic brain (Figures 7A and 7B; 2.20 ± 0.25 vs 1.22 ± 1.07, respectively, ∗ p<0.01; n=3 in each group).
Prominent Cygb expression within neurons of the adult normoxic brain. (
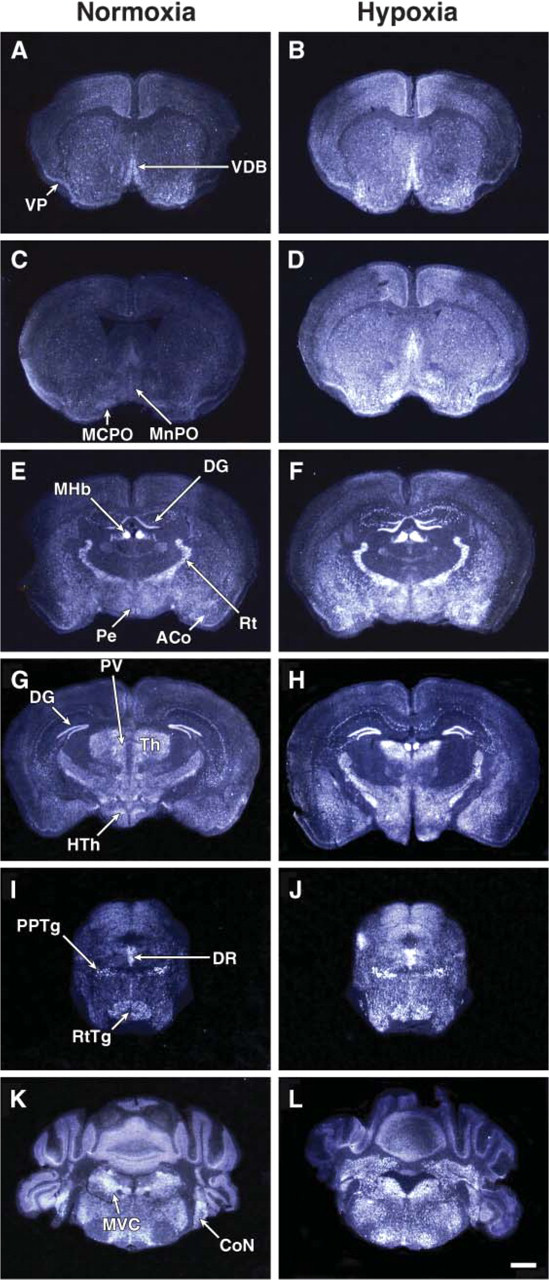
ISH of Cygb expression in the adult normoxic and hypoxic brains. Darkfield illuminations of coronal frozen sections of brains from mice exposed to either normoxic (21% O2;
Differential Expression of Cygb and Ngb in the Adult Brain
We have previously reported that Ngb is also expressed in focal regions of the adult brain. Furthermore, in contrast to Cygb, no significant changes in Ngb expression were observed in the adult mouse brain following chronic exposure to 10% oxygen (Mammen et al. 2002). Using ISH techniques, we compared the neural expression of Cygb and Ngb in the normoxic adult mouse brain. Throughout the amygdala, Cygb was differentially expressed, compared with the expression of Ngb as demonstrated in Figures 8A and 8B and as previously reported by our laboratory (Mammen et al. 2002). To demonstrate authenticity or fidelity of the Ngb riboprobe, we illustrated strong Ngb expression in the pedunculopontine nucleus (Figure 8D). Moreover, Cygb and Ngb are coexpressed in the peduculopontine nucleus (Figures 8C and 8D). These expression studies support the hypothesis that Cygb and Ngb may have distinct functional roles in the adult brain.
Enhanced Cygb expression in the adult hypoxic brain. (
Discussion
The present study provides three key observations regarding the biology of Cygb. Our study is the first comparative analysis of the expression patterns of Cygb and Mb during murine embryogenesis. In comparison to Mb, there was limited Cygb expression within the developing embryo; however, there was a marked induction of Cygb expression in the neonatal and adult brains. Second, we have demonstrated that Cygb, unlike Ngb, is induced in response to chronic hypoxia, and we provide the first detailed analysis of Cygb expression within the brain under normoxic and chronic hypoxic conditions. Robust Cygb expression was observed in regions involved in neurogenesis and the control of the cardiovascular system in the adult normoxic brain. Signal intensity was qualitatively increased in these regions with chronic hypoxic exposure. Finally, we demonstrated that Cygb transcript is expressed within the gray matter and not the vasculature.
Cygb Expression in Regions of the Brain Responsive to Oxidative Stress and/or Hypoxia
In contrast to the embryonic expression pattern for Mb, Cygb has limited expression during embryogenesis. Our data further demonstrate a 10-fold increase in Cygb expression in the adult brain compared with the embryonic brain at E17.5 dpc. The majority of this increase occurred between E17.5 dpc and the neonatal period. Postnatal development of the brain is characterized by a marked growth of neuronal tissue. Neurogenesis occurs predominately in the subventricular zone adjacent to the lateral ventricles and the dentate gyrus of the hippocampus (Eriksson et al. 1998; Nilsson et al. 1999; Eriksson and Wallin 2004). Neurogenesis also occurs to a limited degree in the neocortex of the forebrain and the substantia nigra. Cygb is expressed in these regions and within the dentate gyrus where the expression of this hemoprotein is robust and characterized by punctate signal in the substantia nigra and the embryonic forebrain (Figure 2F, Figure 5G, Figure 6E, and Figure 7A). Induction of Cygb during neuronal growth may reflect a role in cytoprotection or oxygen homoeostasis in metabolically active regions of the brain. Future studies will be necessary to mechanistically decipher the functional role of Cygb during postnatal growth of the brain.
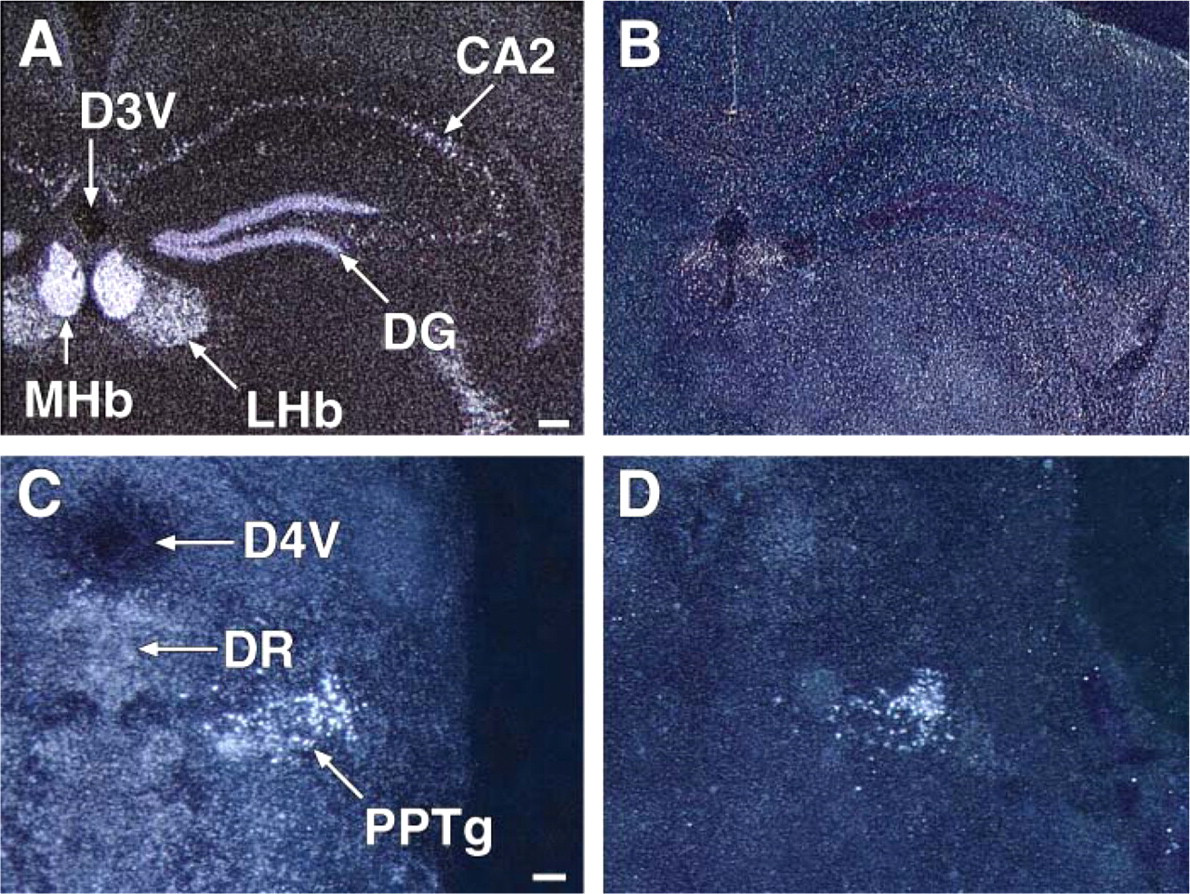
Hippocampus and dentate gyrus of the adult normoxic brain reveals robust Cygb but minimal neuroglobin (Ngb) expression. Darkfield illuminations of coronal frozen sections at the level of the hippocampus were hybridized with either an antisense Cygb 35S riboprobe (
In addition, there are a variety of metabolic and cardiovascular changes that occur during the postnatal period. The hypothalamus, where there is moderate to strong Cygb expression, forms the major portion of the ventral region of the diencephalons and functions to regulate distinct metabolic processes and autonomic functions. Specifically, the paraventricular nucleus of the hypothalamus is associated with the maintenance of metabolic homeostasis. It has been demonstrated to be associated with the regulation of food intake, neural response to oxidative stress, and regulation of the metabolic rate and autonomic nervous system (Schaefer et al. 1993; Duan et al. 1997; Schlenker et al. 2001; Felder et al. 2003; Shih et al. 2003; Schlenker 2005).
The distinct neural expression pattern of Cygb is in contrast to Ngb expression within the brain. Ngb is the only other known tissue hemoglobin expressed within the mammalian brain, and our laboratory has previously demonstrated that Ngb is also localized to distinct regions of the brain (Mammen et al. 2002). However, there is minimal overlap in the neural expression patterns of these two tissue hemoglobins. In particular, regions of the brain that have increased Cygb expression such as the hippocampus and the dentate gyrus lack Ngb. Although there is moderate to intense transcript expression of Ngb in certain regions of the brain that respond to stress (i.e., stria terminalis, nucleus of the solitary tact, periaqueductal gray, amygdala, and subfornical organ), these regions are distinct from the cardiovascular control centers of the brain with intense Cygb expression (i.e., paraventricular thalamic nucleus, suprachiasmatic nucleus, substantia nigra, and the nucleus of the solitary tract) (Mammen et al. 2002).
Marked Upregulation of Cygb Within the Hypoxic Brain
To evaluate the role of Cygb and oxygen homeostasis, adult mice were exposed to chronic hypoxia (10% oxygen) for a 1-week period. Although there are two previous studies that have demonstrated an alteration of Cygb expression in HN33 cells and rat tissue exposed to acute hypoxia (<48 hr) (Fordel et al. 2004; Schmidt et al. 2004), the present study is the first to undertake a comprehensive analysis of Cygb expression in response to chronic hypoxia in the adult mouse brain. Unlike the previous studies, which only utilized real-time PCR to assess Cygb expression, our study utilized different techniques to demonstrate that Cygb is upregulated at both the transcriptional and translational levels in response to chronic hypoxia.
Chronic hypoxia initiates and promotes a multitude of cellular responses as well as physiological responses in the whole animal (Hoppeler et al. 1990; Howald et al. 1990; Hoppeler and Desplanches 1992; Desplanches et al. 1996; Caro 2001; Hoppeler and Vogt 2001). In the brain, it has been demonstrated that hypoxia promotes neurogenesis within the dentate gyrus (Bossenmeyer-Pourie et al. 1999a,b,2000,2002; Daval and Vert 2004). There is a disruption of metabolic homeostasis and a switch in the metabolic substrate that is utilized to fuel ATP production (Xu et al. 2005). Finally, there are several cardiovascular adaptations that occur with chronic hypoxic exposure including the development of tachypnea and tachycardia, presumably to promote oxygen delivery to hypoxic tissues (Yu et al. 1999; Mammen et al. 2003; Campen et al. 2004,2005a,b). Neural control of many of these cardiovascular adaptations occurs within the thalamus and hypothalamus. Thus, our data suggest that the transcription and translation of Cygb may be linked to the process of cytoprotection and metabolic/cardiovascular homeostasis.
In contrast to the 2-fold induction of Cygb protein expression within the hypoxic brain, we previously observed no significant increase in Ngb expression within the hypoxic brain (Mammen et al. 2002). Although both of these tissue hemoglobins are expressed in focal regions of the brain and are both hexacoordinated hemoproteins, the differential response of Cygb and Ngb to chronic hypoxia highlights the different cellular roles these globins may play within a stressed cell. Future studies utilizing genetic technologies will be necessary to further examine the functional role(s) of Cygb and Ngb in the normoxic and hypoxic brain.
Cellular Localization of Cygb Within the Brain
An additional finding outlined in the present study is the observation that Cygb mRNA expression is not expressed in the vasculature but rather is localized to the gray matter, presumably in the neuron. This is an important observation as there is controversy regarding the localization of Cygb within specific organs (organ-specific or parenchymal cells vs fibroblasts). Kawada et al. (2001) and Tateaki et al. (2004) reported that, in the liver, Cygb is expressed only in stellate cells, which are important in the development of hepatic fibrosis, but is absent in hepatocytes. In contrast, Geuens et al. (2003) demonstrated that Cygb is expressed in organ-specific cells (i.e., neurons, hepatocytes, and renal glomerular epithelial cells) rather than in the supporting stroma. Therefore, our data in the adult brain support the findings from Geuens et al. that Cygb is not expressed in non-neuronal cell populations (i.e., vasculature, fibroblasts, etc.) but rather within the gray matter.
In summary, our data demonstrate that the spatial and temporal expression patterns of Cygb are distinct compared with the expression of Mb during murine embryogenesis. Although Cygb is expressed in adult murine heart and skeletal muscle, the embryonic expression pattern indicates that Cygb is likely not essential for cardiac or skeletal muscle development but rather may play an important role in neural development (Burmester et al. 2002; Trent and Hargrove 2002; Schmidt et al. 2004). In addition, unlike Ngb, the selective expression pattern of Cygb within the adult brain and its marked upregulation in response to hypoxia suggests Cygb is a stress-responsive gene that may function in cytoprotection and the maintenance of metabolic or cardiovascular homeostasis. The detailed expression pattern of Cygb within the brain and the precise location of enhanced expression with hypoxia provide insight regarding its potential functional role within the neuron. Future gene disruption and transgenic strategies that modulate the levels of Cygb will be important to fully elucidate the role of this hemoprotein.
Footnotes
Acknowledgements
This study was supported by grants from the American Heart Association-Texas Affiliate (to PPAM), Donald W. Reynolds Clinical Cardiovascular Research Center (to DJG and PPAM), GlaxoSmithKline Research Foundation (to PPAM), and the National Institutes of Health, Grant HL-076440 (to PPAM) and Grant HL-63788 (to DJG).
