Abstract
Dendritic cells (DCs) are key cells in innate and adaptive immune responses that determine the pathophysiology of Crohn's disease. Intestinal DCs migrate from the mucosa into mesenteric lymph nodes (MLNs). A number of different markers are described to define the DC populations. In this study we have identified the phenotype and localization of intestinal and MLN DCs in patients with Crohn's disease and non-IBD patients based on these markers. We used immunohistochemistry to demonstrate that all markers (S-100, CD83, DC-SIGN, BDCA1-4, and CD1a) showed a different staining pattern varying from localization in T-cell areas of lymph follicles around blood vessels or single cells in the lamina propria and in the MLN in the medullary cords and in the subcapsular sinuses around blood vessels and in the T-cell areas. In conclusion, all different DC markers give variable staining patterns so there is no marker for the DC.
I
For characterization of human DCs, a series of markers have been used. In peripheral blood, five distinct subsets of DCs have been identified (Table 1) (Fithian et al. 1981; Takahashi et al. 1984b; Cochran et al. 1993; Zhou and Tedder 1995; Grouard et al. 1997; Rissoan et al. 1999; Valladeau et al. 1999; Geijtenbeek et al. 2000; Dzionek et al. 2001, 2002; Liu et al. 2001; MacDonald et al. 2002). In addition, myeloid and plasmacytoid DCs can be distinguished (Table 1) (Fithian et al. 1981; Takahashi et al. 1984b; Cochran et al. 1993; Zhou and Tedder 1995; Grouard et al. 1997; Rissoan et al. 1999; Valladeau et al. 1999; Geijtenbeek et al. 2000; Dzionek et al. 2001, 2002; Liu et al. 2001; MacDonald et al. 2002). Baumgart et al. (2005) demonstrated that, in blood of IBD patients during flare-ups of the disease, immature DCs of both myeloid and plasmacytoid origins are reduced, probably because these cells migrate to the gut.
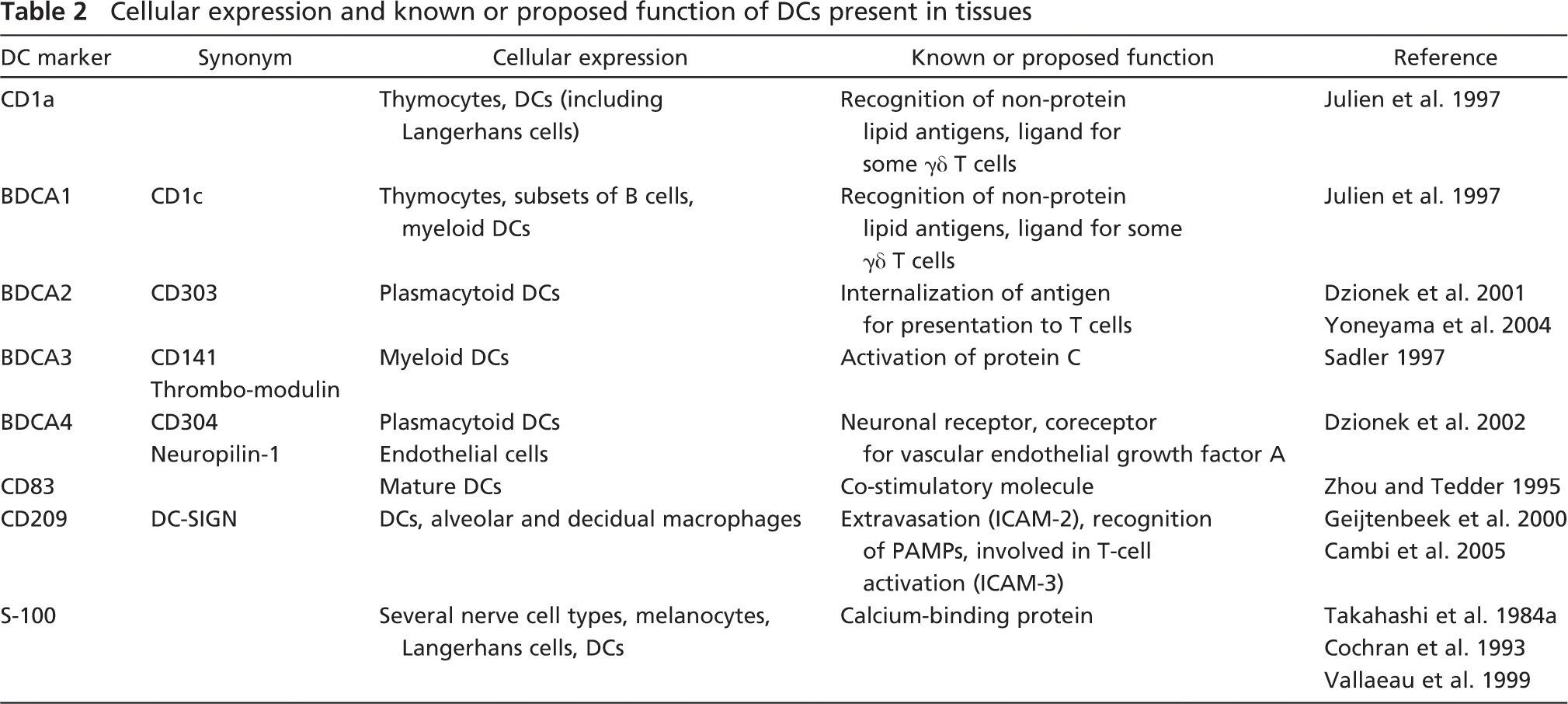
In tissues, three major human DC populations are distinguished, i.e., two myeloid-derived DC populations and one plasmacytoid DC population. Table 2 lists the characteristics of the different DC populations in peripheral tissues (Takahashi et al. 1984b, 2001; Cochran et al. 1993; Zhou and Tedder 1995; Jullien et al. 1997; Sadler 1997; Geijtenbeek et al. 2000; Dzionek et al. 2001, 2002; Yoneyama et al. 2004; Cambi et al. 2005).
In the present study we have determined which DC subpopulations in human colon and MLN can be distinguished when these different markers are used. In addition, we speculate which of these populations may be involved in the pathogenesis of CD. As far as we know, we have performed the first in situ analysis of human intestinal DCs and revealed that in vivo populations in tissues differ from the widely used monocyte-derived DCs generated in vitro (te Velde et al. 2003). Therefore, it is important for a better understanding of the pathophysiology of CD to characterize DC populations in colon and draining lymph nodes in situ. On the basis of the in situ analysis of DC subpopulations, we can determine which populations are of interest for future molecular characterization. These DC populations may be potential targets for future therapy.
Markers used for the characterization of DC populations in blood and tissue
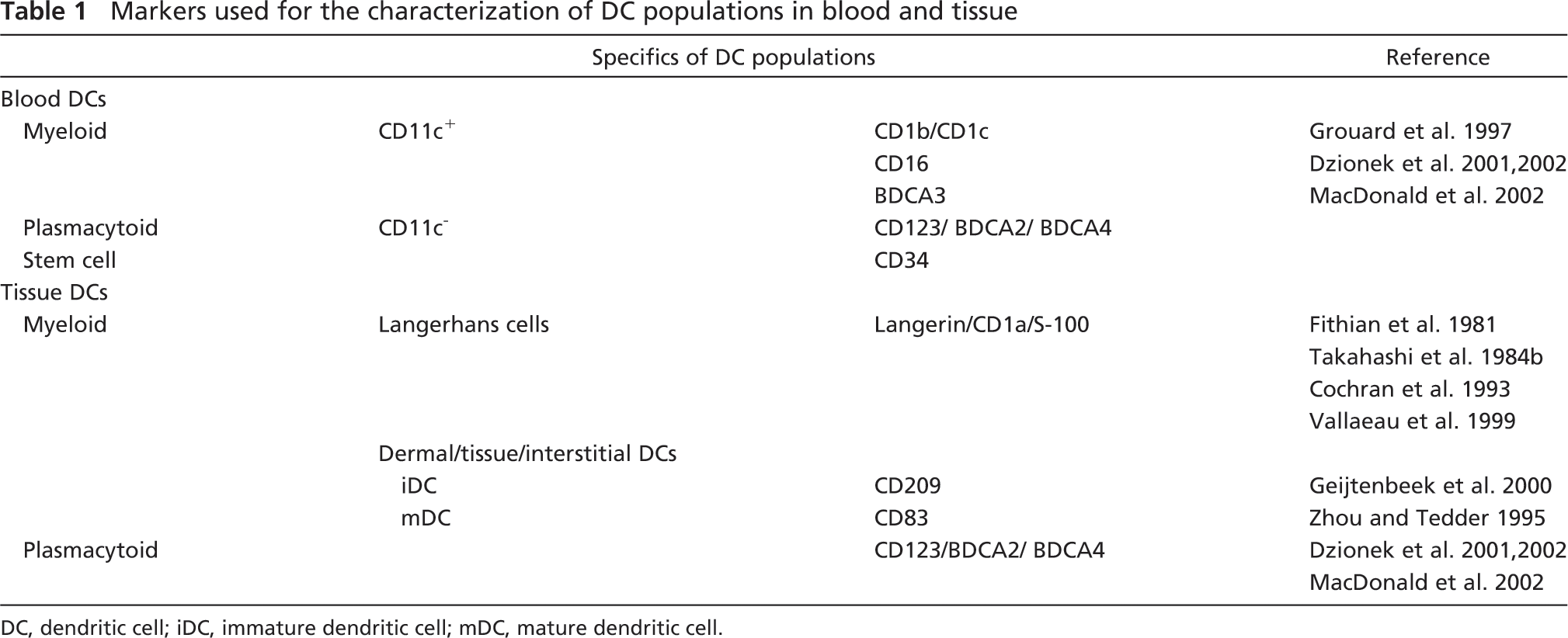
DC, dendritic cell; iDC, immature dendritic cell; mDC, mature dendritic cell.
Cellular expression and known or proposed function of DCs present in tissues
Materials and Methods
Patients and Tissue Samples
Colon and MLNs were obtained with informed consent from patients with CD and non-IBD-related disorders (diverticulitis, polyposis coli, or colon carcinoma) by surgical resection. Non-diseased colon mucosa samples were obtained from patients with colon cancer taken at least 7 cm from the tumor. MLNs that were devoid of cancer metastasis were also obtained from these patients (CD; n=7) and non-IBD-related disorders (n=3). Age range of the CD patients (n=9) was 26–41 years (mean age: 36 years), whereas the age range of patients with non-IBD-related disorders (n=11) was 34–84 years (mean age: 57 years). Prior to the resection procedure, six of the nine CD patients were treated with corticosteroids. After resection, colonic mucosa and MLNs were immediately snap frozen in liquid nitrogen and stored at −80C until cryostat sectioning. Alternatively, samples were fixed in 4% buffered formaldehyde, dehydrated, and embedded in paraffin.
Immunohistochemistry
Frozen Sections
BDCA-1-4 Staining. Serial cryostat sections were cut on a Cryo-Star HM560 (Microm; Walldorf, Germany) and transferred to aminopropyltriethoxysilane (APES)-coated glass slides (StarFrost; Knittel, Klinipath, Duiven, The Netherlands), dried, and stored at −80C until further processing. Tissue sections were defrosted at room temperature, dried, and fixed in ice-cold acetone for 10 min at room temperature. After fixation, tissue sections were rinsed with PBS (pH 7.8), placed in a semi-automatic stainer (Sequenza; Shandon, Breda, The Netherlands), and incubated with normal goat serum (CLB; Amsterdam, The Netherlands) to block nonspecific staining for 10 min. Sections were then incubated with primary antibody for 60 min at room temperature. Mouse anti-human monoclonal antibodies (Miltenyi Biotec; Bergisch Gladbach, Germany) directed against BDCA-1 (AD5-8E7), BDCA-2 (AC144), BDCA-3 (AD5-14H12), and BDCA-4 (AD5-17F6) were used. All primary antibodies were diluted in PBS containing 1% blocking reagent (10,961,760; Roche Diagnostics GmbH, Mannheim, Germany) to block endogenous avidin and biotin activity. Following incubation with primary antibody, sections were rinsed with PBS for 5 min and incubated with biotinylated goat anti-mouse antiserum (Biogenics; Klinipath) for 30 min at room temperature. Next, sections were rinsed with PBS and incubated with streptavidin alkaline phosphatase (ss-AP; Biogenics, Klinipath) for 30 min at room temperature. Sections were then rinsed with PBS containing Tris buffer (0.2 mol/liter, pH 8.5) and incubated with New Fuchsin (Chroma; Kongen, Germany) substrate (containing levamisole to block endogenous AP enzyme activity) for 20 min at room temperature. Sections were counterstained with Gill's hematoxylin, rinsed with distilled water, and mounted in Vecta Mount (Vector Laboratories; Burlingame, CA). Control staining was performed by substituting primary antibody with an isotypic control monoclonal antibody.
Localization in colon and MLN of DC subtypes
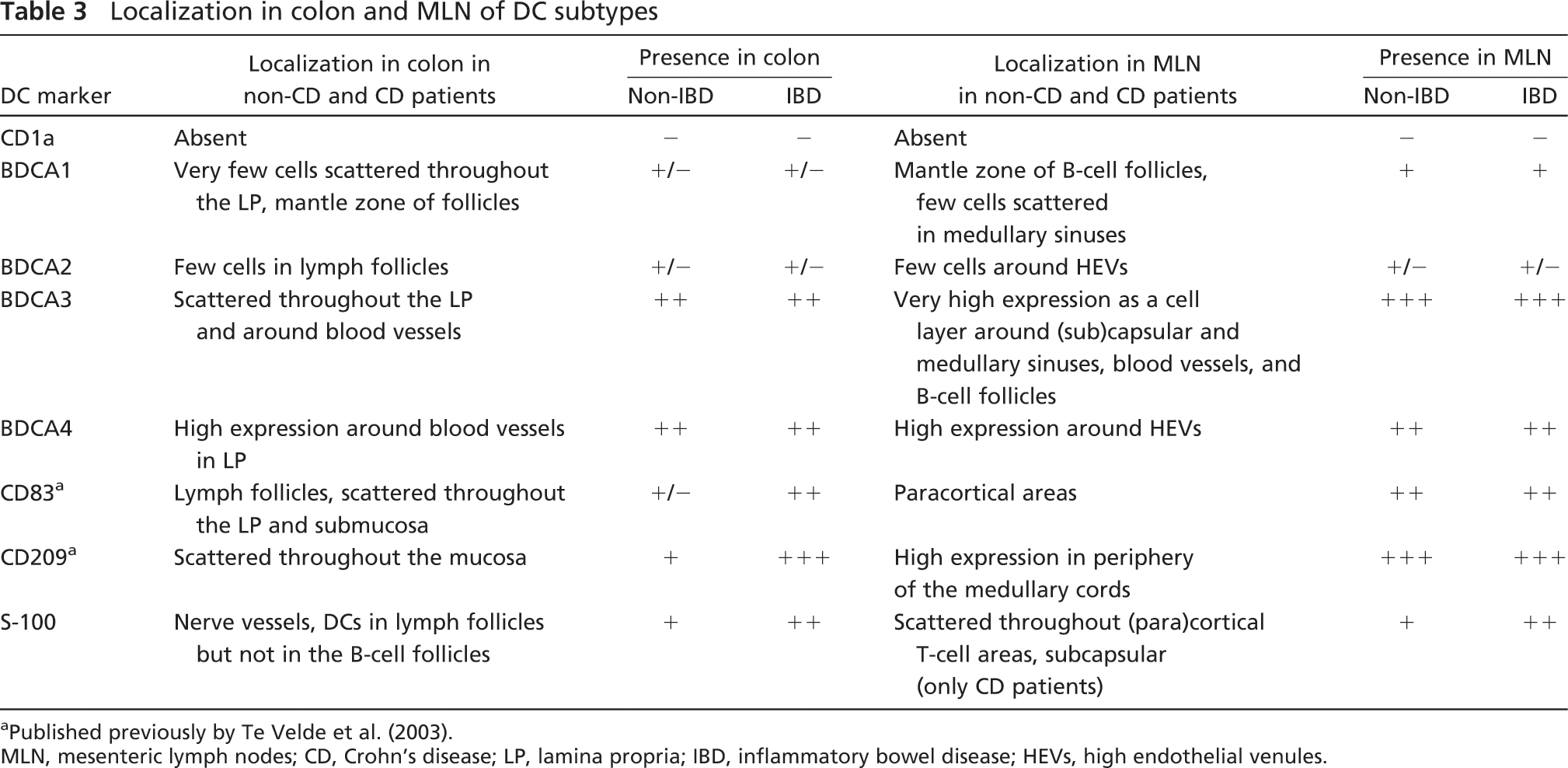
a Published previously by Te Velde et al. (2003).
MLN, mesenteric lymph nodes; CD, Crohn's disease; LP, lamina propria; IBD, inflammatory bowel disease; HEVs, high endothelial venules.
DC-SIGN and CD83 Staining. Frozen sections were stained using a standard alkaline phosphatase protocol as described previously (te Velde et al. 2003). In short, sections were incubated in 5% (v/v) normal goat serum (CLB) to block nonspecific staining for 10 min. Sections were then stained with either anti-CD83 as primary MAb (Beckman Coulter; Mijdrecht, The Netherlands) or with FITC-labeled anti-DC-SIGN (obtained from Dr. Y. van Kooyk; Free University Medical Center, Amsterdam, The Netherlands). Following incubation with primary antibody, sections were extensively rinsed in PBS and incubated with biotinylated goat anti-mouse IgG (Dako; Glostrup, Denmark). Next, FITC-labeled MAb sections were incubated with rabbit anti-FITC antibody (Dako) and goat anti-rabbit antibody (Dako). Subsequently, sections were incubated with streptavidin/biotin-conjugated alkaline phosphatase complex (ABC protocol; Dako). Color was developed using as substrate naphthol AS-MX phosphate (0.3 mg/ml) plus New Fuchsin (0.1 mg/ml; Chroma) in 0.2 M Tris–HCl buffer, pH 8.0 (ABC protocol), and sections were counter-stained with hematoxylin.
Paraffin Sections
Paraffin sections were stretched and dried at 37C overnight, deparaffinized in xylene, and rehydrated in alcohol series. To block endogenous peroxidase activity, sections were treated with 3% H2O2 in methanol for 20 min. Sections were subjected to heat-induced epitope retrieval for 10 min at 95C. To block nonspecific binding sites, sections were incubated with normal goat serum (5% in PBS) for 10 min. Sections were then incubated with primary antibodies CD1a (1:20; Dako) and S-100 (1:4000; Dako) diluted in PBS containing 5% BSA for 1 hr. After incubation with secondary antibodies (Dako), peroxidase activity was demonstrated by using 3, 3-diaminobenzidine tetrachloride (Sigma; St Louis, MO). Finally, sections were counterstained with hematoxylin, dehydrated, and mounted in Pertex (Sigma–Aldrich; Steinheim, Germany). All sections were examined in a double-blind manner by a pathologist.
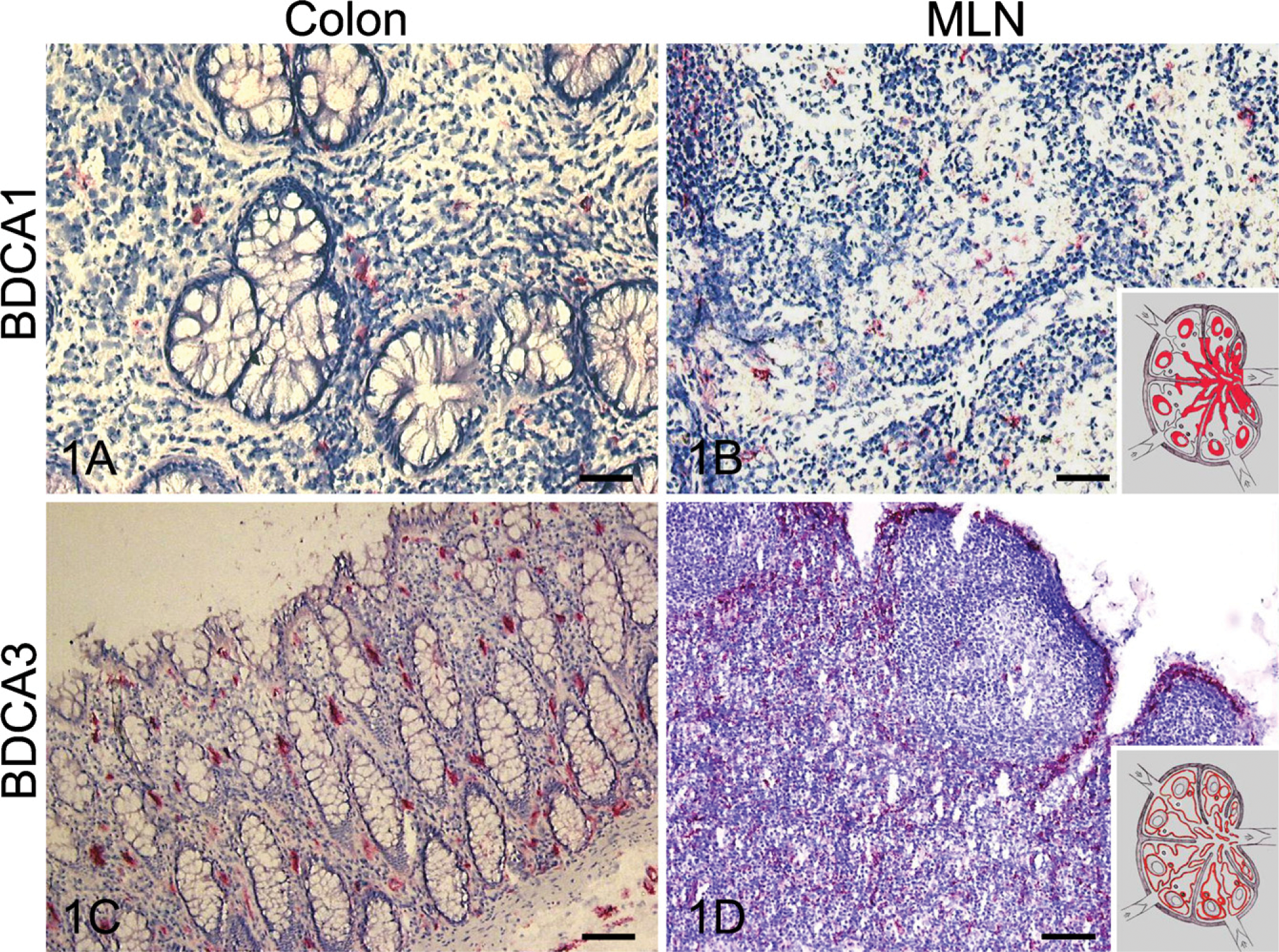
Photographs of the immunohistochemical determination of the expression of the myeloid dendritic cell (DC) markers BDCA1 (
Results
Myeloid DCs
Distribution patterns of myeloid DCs in colonic mucosa and MLNs of non-CD and CD patients using the DC markers BDCA1, BDCA3, S-100, DC-SIGN, CD83, and CD1a are summarized in Table 3.
BDCA1 and BDCA3
In colonic tissue, expression of BDCA1+ cells was mainly observed in association with lymph follicles. Only a few cells were scattered throughout the lamina propria (LP) (Figure 1A). In MLN, expression of BDCA1 was restricted to the mantle zone, and some subsets of cells were scattered throughout medullary sinuses (Figure 1B). BDCA3 expression was found in single cells in the LP of both non-CD and CD patients (Figure 1C). Moreover, BDCA3 was expressed around blood vessels in the LP, submusosa, and muscle layers of the colon. In MLN, BDCA3 was expressed around (sub)capsular and medullary sinuses, blood vessels, and lymph follicles accentuating the frontier between T- and B-cell areas (Figure 1D).
S-100
In MLNs, S-100+ cells were scattered throughout the cortical and paracortical T-cell areas, whereas cells located in B-cell areas did not express S-100 (Figure 2). S-100+ cells were also found in paracortical sinuses in MLNs of CD patients, whereas the paracortical sinuses of non-CD MLNs were completely devoid of S-100+ cells. The number of S-100+ cells was increased in MLNs of CD patients (see Table 3). Lymph follicles in colonic tissue demonstrated a slight increase of S-100+ DCs in CD patients as compared with non-CD patients. S-100+ DCs were also absent in the B-cell areas of the follicles.
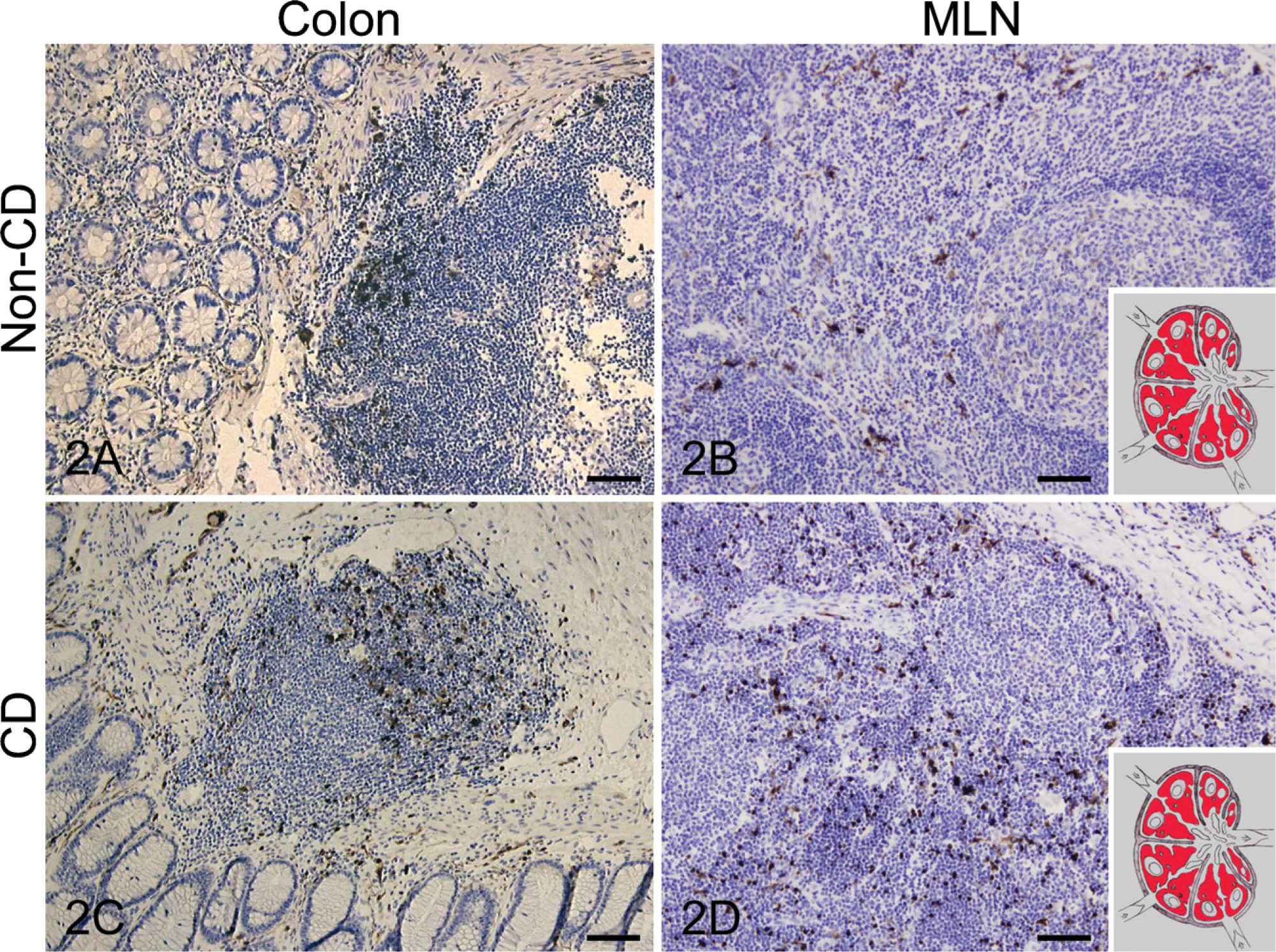
Photographs of the immunohistochemical localization of S-100 expression in colon (
DC-SIGN+ Cells and CD83+
It has been previously demonstrated that DC-SIGN+ cells are scattered throughout the mucosa, and that the CD83+ population is present in aggregated lymphoid nodules and as single cells in the LP (te Velde et al. 2003). In addition, we demonstrate in this present study that, in MLN, expression of DC-SIGN is present in the periphery of medullary cords, which is populated by macrophages and plasma cells. CD83+ cells were found in paracortical zones populated by T cells (Figure 3). There were no differences between CD and non-CD patients.
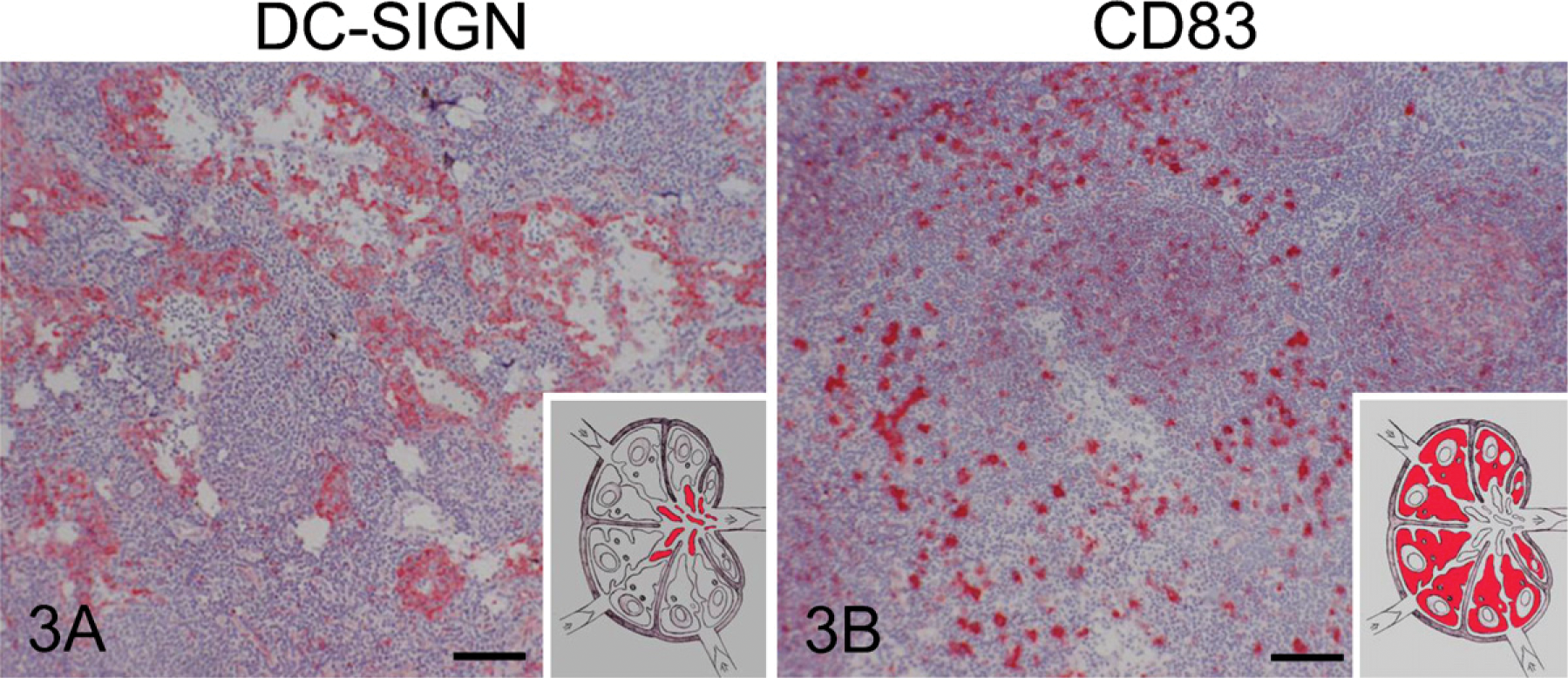
Photographs of the immunohistochemical localization of DC-SIGN (
CD1a Cells
Both colonic tissues and MLNs of non-CD and CD patients were completely negative for CD1a expression (data not shown).
Plasmacytoid DCs
Plasmacytoid DCs are characterized by the expression of BDCA2 and BDCA4. BDCA2 expression of a few cells was found in lymph follicles in colon, whereas BDCA2+ cells were absent in mucosa of both non-CD and CD patients (Figure 4). In contrast, expression of BDCA4 was strong in endothelial cells in LP of both non-CD and CD patients. In MLN, expression of both BDCA2 and BDCA4 was closely associated with high endothelial venules (HEVs).
Discussion
During an immune response, DCs traffic from peripheral tissues into draining LNs through lymphatic vessels. However, it is not yet known which DC populations are present in the colon wall and which DC populations migrate from colon tissue into the MLNs. In humans, these DC populations may play an important role in the pathogenesis of CD. Therefore, we investigated subtypes and their localization of DCs in colon and MLN. We demonstrate here that three different subpopulations of myeloid DCs populate the colon mucosa and MLN of non-CD and CD patients. These populations consist of the following: (1) immature DCs or macrophages that express DC-SIGN, (2) mature DCs that express S-100 or CD83, and (3) mature DCs that express BDCA3. BDCA1 and CD1a expressing DCs were virtually absent in colon as well as MLNs. Immature DCs and macrophages are mainly localized at antigen-capturing sites such as the mucosa and medullary cords, whereas mature DCs are present where antigen is presented, including the T-cell areas in colonic lymph follicles and MLNs.
In general, LNs contain three types of DCs, i.e., interdigitating DCs (IDCs), follicular DCs, and plasmacytoid DCs. IDCs are like Langerhans cells characterized by the expression of S-100, which is a member of the family of calcium-binding proteins (Takahashi et al. 1984a, b; Cochran et al. 1993). Based on the localization of markers, we demonstrated that S-100+ and also CD83+ cells present in the MLN are most likely IDCs, which are mature DCs from myeloid origin and capable of presenting antigens to naive T cells in the paracortical areas. Moreover, in colonic tissue, S-100 as well as CD83 demonstrated a similar expression pattern in lymph aggregates, suggesting that they are involved in similar functional processes. In contrast, S-100 expression in cervical LNs was absent in lymph follicles (Takahashi et al. 2001; Engering et al. 2004). Furthermore, only subcapsular sinuses of CD patients and not of non-CD patients harbor S-100+ DCs, indicating that in CD patients there is an increased influx of mature DCs of MLNs via subcapsular sinuses into the T-cell areas. An increased number of S-100+ DCs was also found in extrafollicular T-cell areas in hyperplastic palatine tonsils of patients with tonsillitis (Papadas et al. 2001), in several autoimmune diseases such as juvenile rheumatoid arthritis (Frosch et al. 2003), and in primary biliary cirrhosis (Demetris et al. 1989), confirming a role for S-100+ cells in inflammatory processes. Moreover, S-100 is expressed by nerve cells and cancer cells and is used as a marker in cases of melanoma, nerve tissue cancer, and several carcinomas (Takahashi et al. 1984a; Daimaru et al. 1985; Monda and Wick 1985; Drier et al. 1987; Wick et al. 1988; Gillett et al. 1990; Cochran et al. 1993).
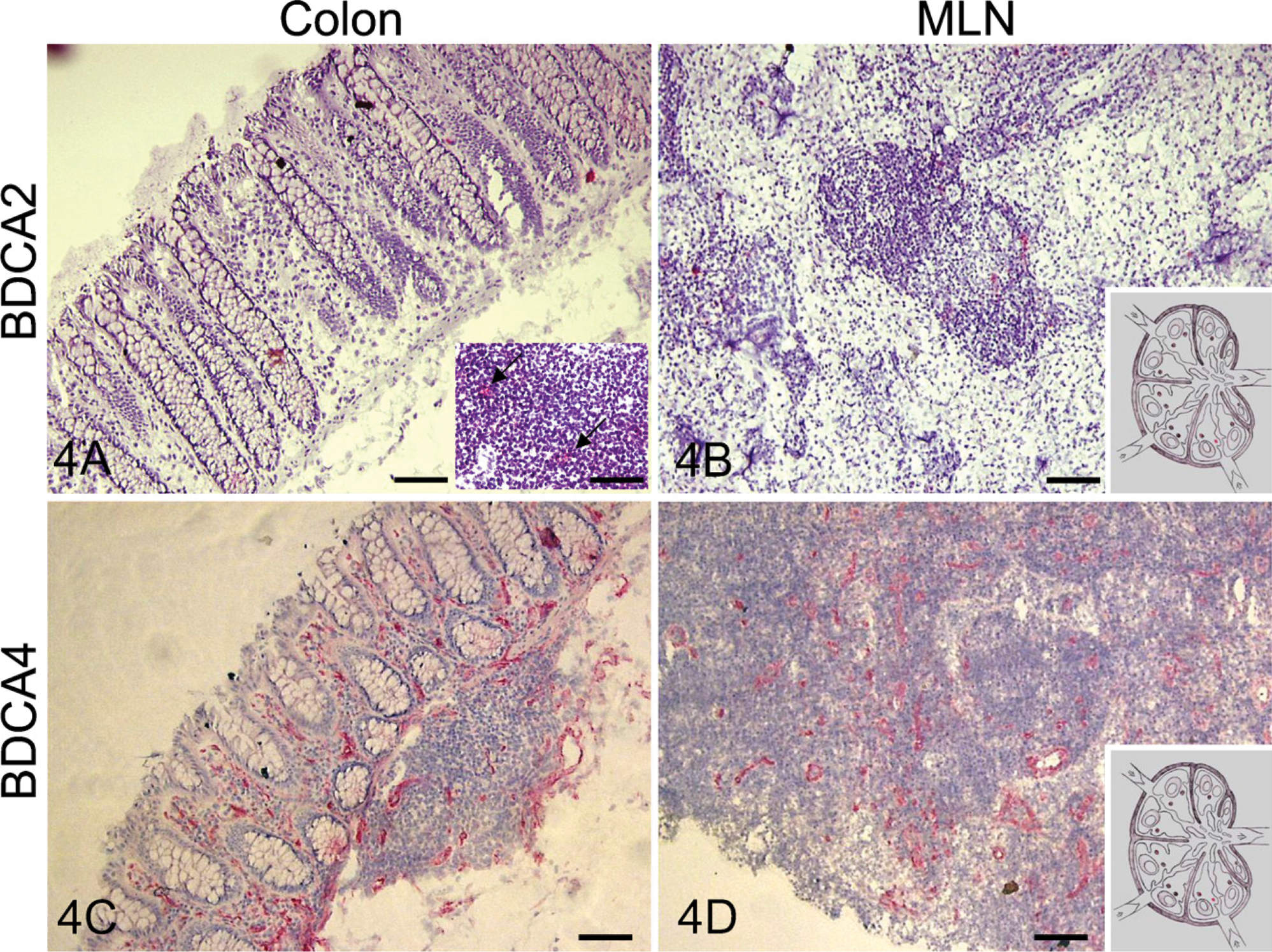
Photographs of the immunohistochemical localization of expression of the plasmacytoid DC markers BDCA2 (
BDCA3+ cells are likely to be IDCs, but because of their specific localization at the border of lymph follicles and sinuses they seem to belong to a myeloid cell type other than S-100+ and CD83+ DCs. These cells may be capable of interacting with B and T cells to induce humeral responses. However, BDCA3+ cells may be more involved in antigen-capturing processes because they are found in LP.
Follicular DCs are characterized by none of the described markers. Expression of BDCA1 in the mantle zone of B-cell follicles may be indicative of follicular DCs; however, expression is not restricted to DCs. B-cell subsets in the germinal center and mantle zone of LNs, marginal zone B cells in the spleen, and a subpopulation of B cells in the peripheral blood also express BDCA1 (Small et al. 1987; Smith et al. 1988; Plebani et al. 1993), which belongs to a family of lipid antigen-presenting molecules (Brigl and Brenner 2004). The observation that BDCA1+ cells were smaller and without the characteristic morphology of DCs suggests that expression of BDCA1 in MLN is due to the presence of B cells rather than DCs.
DC-SIGN is a C-type lectin involved in sampling antigens by recognition of specific carbohydrate structures of autoantigens and foreign antigens and is therefore mainly expressed by immature DCs and macrophages (Geijtenbeek et al. 2000). This is consistent with our earlier findings that DC-SIGN+ cells are scattered throughout colonic mucosa where these cells can encounter antigens. DC-SIGN+ cells were present in medullary cords of MLNs, but in contrast to cervical LNs no expression was detected in the outer zone of the paracortex (Engering et al. 2004).
Although CD1a is mainly used as a marker for Langerhans cells in the skin, expression of this marker is not restricted to this cell type because subsets of interstitial and monocyte-derived DCs also express CD1a (Tazi et al. 1993; Zhou and Tedder 1996; Yoshida et al. 1997). However, CD1a was not detectable in MLNs and colonic tissue of both non-CD and CD patients. Moreover, subsets of DCs in the superficial LNs were CD1a positive, whereas CD1a expression is absent in deeply located LNs including mesenteric, mediastinal, and para-aortic LNs (Takahashi et al. 2001). Additionally, expression of CD1a does not occur in inguinal and liver LNs (Tanis et al. 2004).
Expression of plasmacytoid DC marker BDCA2 has been observed in close association with HEVs, indicating that tissue plasmacytoid DC precursors are blood-derived cells that enter MLN in the HEVs. In agreement with these results, Yoneyama et al. (2004) demonstrated that myeloid DCs migrate via lymph vessels into draining LNs, whereas plasmacytoid DCs enter LNs via the HEVs. BDCA2+ DCs seem to be mainly involved in antigen-capturing processes, and BDCA2 is a C-type lectin transmembrane glycoprotein that internalizes antigens to present to T cells (Dzionek et al. 2001). BDCA2 expression was absent in colonic mucosa of both non-CD and CD patients, which coincides with the results of Middel et al. (2006). BDCA4 is a neuronal receptor of the class 3 semaphorin family and is also expressed by endothelial cells to function as a coreceptor for vascular endothelial growth factor A (Dzionek et al. 2002). This suggests that BDCA4 expression at HEVs and in the LP is related to endothelial cells instead of DCs.
In the current study we have shown that different DC markers give variable staining patterns so that there is no marker for DCs. These different ‘DC markers’ are present in different sites in the colon and MLN in CD patients, indicating that DCs are a heterogeneous group of antigen-presenting cells harboring different functions. Our data indicate that colonic tissue and MLNs harbor at least three different myeloid DC populations. One population consists of immature DCs, which express DC-SIGN and are mainly located at antigen-capturing sites in the mucosa and medullary cords. A second population of DCs expresses BDCA3 and, similar to DC-SIGN+ DCs, also presents as single cells in the mucosa, suggesting that BDCA3+ DCs may be involved in antigen-capturing processes. In the MLN, their location around the lymph follicles suggests a role in the activation of B and T cells. The third DC population consists of mature DCs that express S-100 and CD83. Their location in the T-cell areas in both the colonic lymph follicles as well as the MLN suggests that they have the capacity to present antigen to T cells. DCs from plasmacytoid origin were hardly present in the colon and MLN. Although BDCA4 expression was found around the blood vessels, its expression is probably on endothelial cells instead of DCs.
Future studies will involve the isolation of the described different DC populations for further investigation to utilize favorable results for the development of new therapeutic strategies in the treatment of CD.
Footnotes
Acknowledgements
We thank Esther de Groot and Trees Dellemijn for performing immunohistochemical staining and Kate Cameron for carefully reading the manuscript.
