Abstract
Crohn’s disease (CD) involves immune system interactions with intestinal tissue, driven by pro-inflammatory cytokines like Tumor Necrosis Factor (TNF-α). Adalimumab, targeting TNF-α, regulates associated inflammatory responses. Despite being humanized, it may induce immunogenic processes, affecting treatment effectiveness. Thus, monitoring serum adalimumab and anti-drug antibody (ADA) levels can optimize therapy. Understanding genetic factors influencing adalimumab response can enhance personalized treatment and improve patient quality of life. We aimed to quantify adalimumab serum levels, assess test interchangeability, detect ADA, examine immune complex formation, and investigate genetic phenotypes related to immunogenicity in CD patients. Seventy CD patients in the maintenance phase with adalimumab were classified into active (CDA) and remission (CDR) groups. Adalimumab concentration was determined via enzyme-linked immunosorbent assay (ELISA—Promonitor) and lateral flow assay (Quantum Blue), with assay interchangeability assessed statistically. ADA and immune complex formation were quantified using ELISA assays. DNA was genotyped for the genes ATG16L1, CD96, and CD155. No significant differences in adalimumab serum concentrations were observed between groups, regardless of the assay. However, a statistical difference between the tests indicated measurement disparity (P = 0.003), with moderate agreement (Lin’s correlation of 0.247). ADA was detected in 4 of 27 of the patients with infratherapeutic levels, 3 in the CDA group and 1 in the CDR group. Analysis of immune complexes revealed significantly higher concentrations in the CDA group (P = 0.0125). The genotypic evaluation revealed significant associations for the CD96 CC (wild-type) genotype with higher CRP levels, colonic involvement, and infratherapeutic levels of adalimumab. ATG16L1 CC genotype was associated with higher CDEIS and fecal calprotectin values, while the variant (TT) genotype had lower platelet counts. The effectiveness of treatment with adalimumab was not directly related to higher medication levels in this cohort. The disparity between tests indicates the need to use only one test in patient follow-up to ensure accuracy in therapeutic monitoring. Genotypic differences highlight the correlation between the wild genotype for CD96 and ATG16L1 with unfavorable laboratory and endoscopic response to adalimumab. Finally, the more significant levels of immune complexes in the CDA group indicate an association with a worse response to adalimumab.
Introduction
Crohn’s disease (CD), categorized as an inflammatory bowel disease (IBD), is a chronic condition that impacts the gastrointestinal tract, resulting in inflammation and structural damage to intestinal tissue. It exhibits a recurring pattern throughout its duration.1,2 Due to its multifaceted nature and the lack of a definitive cure, achieving an effective treatment remains challenging. The therapeutic objective requires identifying medications that target the underlying inflammatory mechanisms to achieve both clinical and endoscopic remission, thereby improving the quality of life for patients. This approach aims to mitigate complications, reduce the frequency of hospitalizations, and diminish the necessity for surgical interventions.3–5
So forth, monoclonal antibodies have been incorporated into the therapeutic approach for CD for more than two decades.5,6 One of them is named adalimumab, a humanized monoclonal antibody that demonstrates the ability to block the effects of tumor necrosis factor-alpha (TNF-α). Demonstrating efficacy, the drug has succeeded in inducing and maintaining mucosal healing among CD patients, including those with severe disease.7–9 Despite the evident advantages, a substantial cohort of patients manifests undesired immune responses to this therapy, attributed to the immunostimulatory nature of these medications.10–13 The immunogenicity process involves the compound endocytosis, the subsequent activation of T and B cells, and the release of anti-drug antibodies (ADA), compromising the medication’s efficacy.13,14 Genetic factors, treatment management, and dosing strategies are potential elements correlated with therapeutic failure, contributing to enhanced clinical outcomes. Implementing a therapeutic approach that involves monitoring serum adalimumab and ADA levels offers benefits in attenuating adverse effects. Such an approach facilitates therapeutic adjustments, which leads to better clinical management.
The significant correlation observed between the detection of ADA and the reduction in serum medication levels emphasizes the potential of this monitoring strategy.12,13,15 This relationship arises from stimulating lymphocyte proliferation, which generates complex antibodies with anti-TNF-α, forming immune complexes. These complexes favor the clearance of the drug through the reticuloendothelial system, promoting a decrease in serum levels of the medication.16–19 The formation of immune complexes between a therapeutic agent and its target may trigger the loss of immunological tolerance and promote the generation of an antibody response directed to the drug. 20 Therefore, serum monitoring of anti-TNF-α medications has become a standard practice. 21 Several studies have demonstrated a correlation between drug levels and clinical and endoscopic improvements, including mucosal healing. Additionally, this approach has been associated with reduced recurrence, higher remission rates, and decreased need for hospitalizations and surgical interventions.3,4 The 2017 AGA guideline emphasizes that empirical dose adjustments may be inappropriate, particularly for patients experiencing immune-mediated or mechanistic failures. 22
Several commercial assays are available to measure serum levels of anti-TNF-α, facilitating the management of IBD. The performance, interchangeability, and correlation between an ELISA and a lateral flow assay for infliximab and adalimumab were recently performed. It was found that there was good agreement for adalimumab levels but a weak correlation for infliximab. 23 Similarly, another study investigated the interchangeability between a lateral flow assay and ELISA for infliximab, identifying significant differences and moderate agreement. 24 Although rarely addressed in the literature, the comparison between tests has proven beneficial in identifying methodologies that offer more accurate results, ensuring the precise measurement of serum drug levels during the patient’s follow-up.
Moreover, evaluating specific polymorphisms associated with drug response may choose patients for a more favorable treatment. 25 Mutations in the ATG16L1 gene (Autophagy-related gene 16-like 1), directly associated with CD, have been identified, and polymorphisms of this gene showed increased substantial benefits from adalimumab therapy, leading to a more effective therapeutic response. 26 Similarly, genetic polymorphisms of the CD96 were associated with response to adalimumab therapy. Their findings suggested that genetic variations within the CD96-CD155 pathway may heighten the likelihood of producing ADA. 27 Since immunogenicity has several determinants, researchers have sought to elucidate additional factors that may contribute to identifying non-responders to anti-TNF-α therapies. The impact of genetic variability on such immunogenic processes remains a topic of substantial uncertainty. Consequently, investigations have focused on exploring the relationship between genetic variations and the occurrence of therapeutic failure.26,27
Thus, identifying a correlation between a patient’s genetic profile, clinical characteristics, and therapeutic responses is pivotal to advancing personalized approaches within medical practice. Hence, we aimed to: (1) assess the serum levels of CD patients treated with adalimumab at a Brazilian tertiary center, (2) compare two methods for measuring serum adalimumab concentrations: the lateral flow assay and the enzyme-linked immunosorbent assay, (3) investigate the occurrence of immunogenic processes associated with the medication’s use, and (4) identify the genotypic profile of these patients for three specific genes. Additionally, we conducted a comparative analysis of two established commercial assays to explore potential interchangeability between them.
Method
Patients and ethical considerations
In this cross-sectional observational study, from 2020 to 2023, 70 patients with CD were included. The inclusion criteria for this study encompassed patients with a confirmed clinical and endoscopic diagnosis of CD who were undergoing regular maintenance therapy with adalimumab, with a minimum of 2 months in the maintenance phase. Additionally, participants were required to attend scheduled medical appointments consistently. According to objective criteria, patients were allocated as belonging to the activity (CDA) or remission (CDR) groups. Disease activity was assessed by colonoscopy (active disease defined as CDEIS—Crohn’s Disease Endoscopic Index of Severity—score ≥5 or presence of deep ulcers in at least one intestinal segment) or nuclear magnetic resonance enterography (MRE) (active disease defined as the presence of deep ulcers in at least an intestinal segment). Exclusion criteria included patients who had not undergone an objective diagnostic assessment, such as ileocolonoscopy or magnetic resonance enterography, within 1 month before the peripheral blood collection or who did not have such assessments scheduled within 1 month following the blood collection.
All the patients enrolled signed an Informed Consent Form. The study was approved by the Research Ethics Committee (CAAE no. 92894418.4.0000.5404). The patients provided written informed consent to participate in the study. This study follows Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 28
Serum levels of adalimumab, anti-drug antibody levels, and formation of immunocomplexes
Adalimumab concentration measurement was performed using both classical enzyme-linked immunosorbent assay (ELISA) (Promonitor®—Progenika Biopharma, S.A., Spain) and lateral flow assay (Bühlmann® Quantum rapid test—Blue—Bühlmann Laboratories AG Schönenbuch, Switzerland), adhering to manufacturer’s instructions. The ELISA test exhibited lower and upper limits of quantification for serum adalimumab of ⩽0.024 and ⩾12 μg/mL, respectively, while the lateral flow assay they were ⩽1.3 and ⩾35 µg/mL. For patients whose measurements exceeded the upper limit of quantification (ULOQ), we assigned a value of ULOQ + 1 (13 for the ELISA method and 36 for the lateral flow assay). Conversely, for those whose measurements fell below the lower limit of quantification (LLOQ), we recorded the value as 0 in both assays. 29 Therapeutic levels were defined according to both assays (lateral flow assay and ELISA) as 5–12 µg/mL.
ADA detection was performed using ELISA assay (Promonitor®—Progenika Biopharma, S.A., Spain), and values surpassing 10 AU were considered positive, according to the kit datasheet. The quantification of circulating immunocomplexes was conducted using ELISA assay (Bühlmann® CIC-CIQ EIA—Bühlmann Laboratories AG Schönenbuch, Switzerland) following the manufacturer’s instructions. Both tests were drug-sensitive assays used to evaluate adalimumab and ADA serum levels using an ELISA assay. For all ELISA assays, the outcomes were compared against a standard curve employing a sample of known concentration provided in the kits.
Genotyping for polymorphisms
Genotyping was conducted on 66 out of the 70 patients included in the study, as four patients declined to provide samples. Genomic DNA was isolated from peripheral blood leukocyte samples using lithium chloride and proteinase K, followed by electrophoresis in a 1% agarose gel stained with ethidium bromide to confirm its integrity. DNA concentration and purity were assessed using a NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies®, USA).
The genotypes of polymorphisms for ATG16L1 (rs10210302), CD96 (rs9828223), and CD155 (rs203710) were determined through real-time PCR employing the TaqMan® genotyping assay. PCR reactions were carried out using patient DNA, SNP Genotyping Assay Mix 1X (comprising two probes and fluorochromes FAM® and VIC®—Life Technologies®, USA), TaqMan® Universal PCR Master Mix 1X (containing Taq DNA polymerase, dNTPs, and optimized buffer solution—Life Technologies®, USA), and sterile water. Amplification conditions involved initial activation of Taq DNA polymerase at 95°C for 10 min, followed by 45 cycles of incubation at 92°C for 15 s and 60°C for 1 min. Genotyping results were analyzed utilizing the TaqMan® Genotyper program (Applied Biosystems®, USA).
Statistical analysis
A descriptive analysis was expressed as the median ± interquartile range (Q1–Q3). Normality was evaluated using the Kolmogorov-Smirnov test with a threshold of P > 0.1. Serum adalimumab levels were examined about disease activity, the use of concomitant immunomodulatory drugs, and antibody anti-adalimumab presence using the non-parametric Mann-Whitney test, while categorical variables were assessed with the Chi-Square test (χ2). A significance level of 5% was adopted for all statistical tests. Statistical analysis and graphs were generated using GraphPad Prism software, version 8 (California, USA).
The following analyses were performed to assess the interchangeability and comparability among the two methods (ELISA and lateral flow assay) for serum levels of adalimumab. The Wilcoxon matched-pairs signed-ranks test was employed to evaluate the differences between ELISA and Quantum Blue results within the measurement range. Non-parametric methods were utilized due to the data’s deviation from a normal distribution. The agreement between results obtained by ELISA and lateral flow assay was evaluated using Passing-Bablok regression analysis and the Bland-Altman method. Passing-Bablok regression analysis 30 investigates the linear relationship between variables X and Y (ELISA and rapid test), providing confidence intervals for the slope (B) and intercept (A). These parameters are used to determine if there is a significant difference between B and 1, and between A and 0. If the 95% confidence interval (CI) for the slope includes the value one and the 95% CI for the intercept contains the value 0, this indicates the absence of proportional and constant differences between the methods, respectively. The analysis included only patients whose adalimumab values were within the detection range of both tests (24 patients). The CUSUM linearity test 31 was used to verify if the residuals were randomly distributed around the regression line, to assess linearity in the relationship between the methods and the applicability of the regression model. All statistical analyses were performed using Stata Statistical Software, version 14 (College Station, TX, USA).
For genotyping, the Shapiro-Wilk normality test was used to assess the distribution of continuous variables. Frequency comparisons were conducted using the Chi-square test to analyze categorical variables. Statistical analysis was performed by GraphPad Prism software, version 8 (California, USA).
Results
Clinical and demographic data
Among the patients included in the study, 43 were classified as CDA and 27 as CDR. The Crohn’s Disease Activity Index (CDAI) median was substantially higher in the CDA group (178.2) compared to the CDR group (113), indicating more active disease in the CDA cohort. Similarly, the Crohn’s Disease Endoscopic Index of Severity (CDEIS) median was elevated in the CDA group, with a mean score of 6.9, reflecting more severe and persistent inflammation. Magnetic resonance imaging also revealed ongoing signs of inflammation in CDA patients, further corroborating the presence of active disease. Laboratory results demonstrated that CDA patients had significantly lower albumin median levels (3.9 g/dL) than CDR patients (4.25 g/dL). Additionally, CRP median levels were elevated in the CDA group (5.32 mg/L) relative to the CDR group (2 mg/L), and fecal calprotectin median levels were markedly higher in CDA patients (1000 µg/g) compared to CDR patients (30 µg/g) (Table 1).
Demographic and clinical characteristics of the participants.
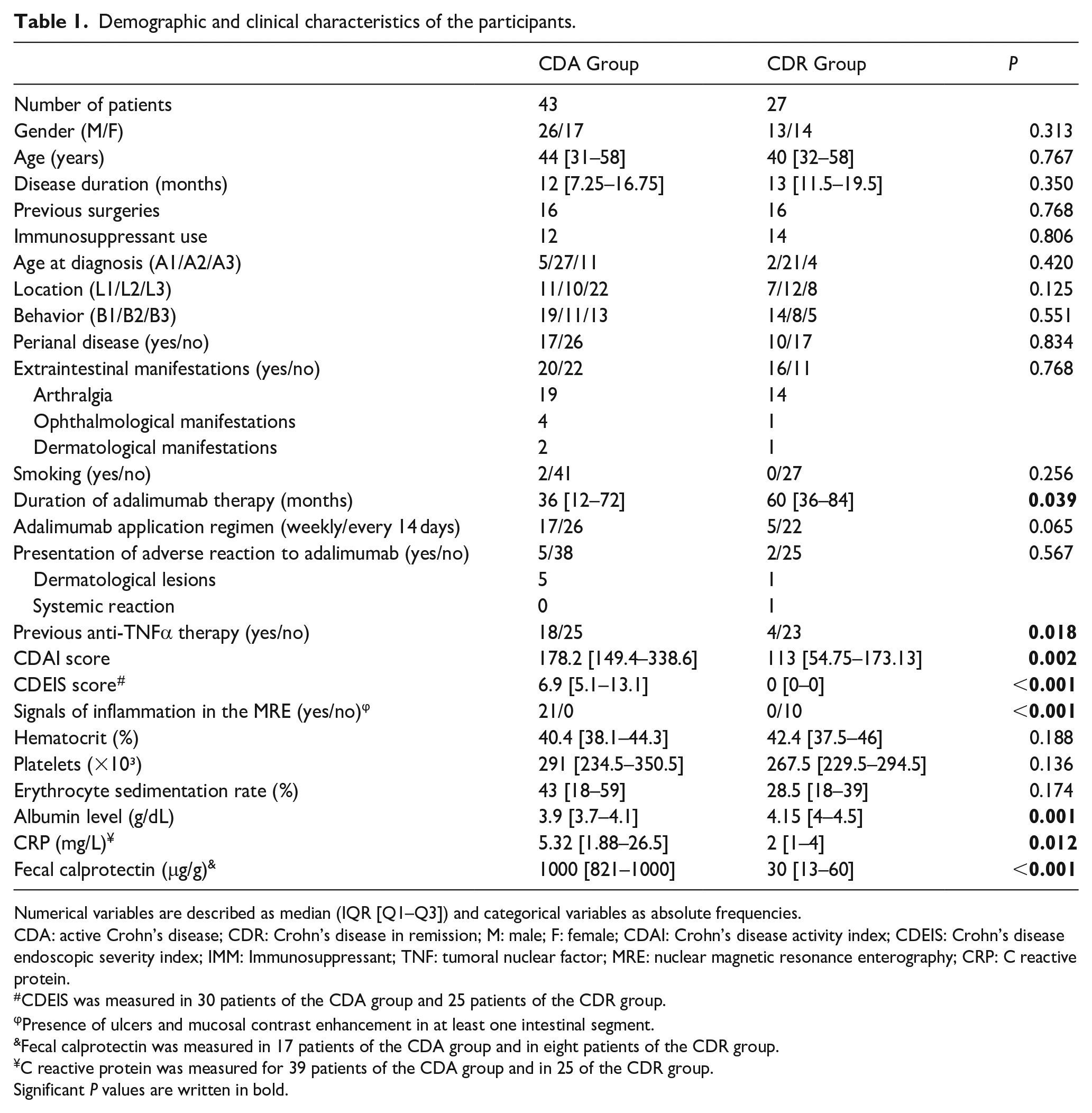
Numerical variables are described as median (IQR [Q1–Q3]) and categorical variables as absolute frequencies.
CDA: active Crohn’s disease; CDR: Crohn’s disease in remission; M: male; F: female; CDAI: Crohn’s disease activity index; CDEIS: Crohn’s disease endoscopic severity index; IMM: Immunosuppressant; TNF: tumoral nuclear factor; MRE: nuclear magnetic resonance enterography; CRP: C reactive protein.
CDEIS was measured in 30 patients of the CDA group and 25 patients of the CDR group.
Presence of ulcers and mucosal contrast enhancement in at least one intestinal segment.
Fecal calprotectin was measured in 17 patients of the CDA group and in eight patients of the CDR group.
C reactive protein was measured for 39 patients of the CDA group and in 25 of the CDR group.
Significant P values are written in bold.
Adalimumab serum levels in Crohn’s disease patients with activity and remission
Adalimumab serum concentrations were assessed using Quantum-Blue assay (Figure 1a) and Promonitor (Figure 1b). No statistically significant differences concerning medication levels were detected between the two groups. Stratifying CDA patients based on therapeutic levels (Table 2), the Quantum-Blue assay showed that nine patients had infratherapeutic levels (20.94%), 11 had ideal levels (25.58%), and 23 had supratherapeutic levels (53.48%). In contrast, employing the Promonitor assay, 12 patients had infratherapeutic levels (27.90%), 10 had ideal concentrations (23.25%), and 20 had supratherapeutic levels (48.85%). Among CD patients in remission (Table 3), the Quantum-Blue assay revealed that three had infratherapeutic levels (11.11%), seven had therapeutic levels (25.92%), and 17 had supratherapeutic levels (62.97%). Similarly, for the Promonitor assay, three patients had infratherapeutic levels (11.11%), eight had therapeutic levels (18.60%), and 16 had supratherapeutic levels (70.29%).

Adalimumab monitoring in patients with active or remission Crohn’s disease. Serum levels were measured using the Quantum-Blue (a) and Promonitor (b) assays. CDA: n = 43, CDR: n = 27. (c) Serum levels of adalimumab in CD patients with and without immunomodulators, measured by the Quantum-Blue assay, and (d) Promonitor assay. For patients whose measurements exceeded the upper limit of quantification (ULOQ), we assigned a value of ULOQ + 1 (13 for the ELISA method and 36 for the lateral flow assay). For those whose measurements fell below the lower limit of quantification (LLOQ), we recorded the value as 0 in both assays. For patients without immunomodulators (IMM−): n = 35; for those with immunomodulators (IMM+): n = 34.
Number of patients in each serum level category—group of patients with active Crohn’s disease.

Number of patients in each serum level category—group of patients with Crohn’s disease in remission.

Furthermore, we examined adalimumab serum concentration in the context of concomitant immunomodulatory medication with TNF-α. No significant differences in adalimumab serum levels were observed in the group of patients using or not an immunomodulator when measured by either the Quantum-Blue (Figure 1c) or Promonitor assay (Figure 1d). However, there was a tendency towards higher serum levels of adalimumab in patients using concomitant immunomodulators when assessed with the Quantum-Blue assay (P = 0.0554).
Comparative analysis of ELISA and lateral flow assay for evaluating adalimumab serum concentrations
To evaluate the interchangeability of the tests, we analyzed whether both methods consistently measured adalimumab serum levels. A statistically significant difference (P = 0.003) was observed in the medians of the two tests, being 5.49 (IQR 2.97–6.08) for Promonitor and 8.2 (IQR 5.3–13.2) for Quantum Blue. This data indicates a disparity between their measurements (Figure 2a), with a considerably higher dispersion in Quantum-Blue results. Our analysis revealed concentration values below the LLOQ and above the ULOQ for both tests. The CUSUM test suggested that linear regression was appropriate for evaluating the relationship between the variables (P > 0.20), leading us to proceed with the Passing-Bablok test (Figure 2b).

Interchangeability between assays. (a) Box plots compare adalimumab concentrations (mg/mL) between Quantum-Blue and Promonitor tests, wherein the horizontal line denotes the median (*P = 0.003), indicative of a statistically significant difference. (b) Linear regression analysis for adalimumab levels using the Passing Bablok method revealed Lin’s correlation coefficient of agreement of 0.247. (c) The Bland-Altman plot was used to represent the comparative analysis between the two measurements, where the difference between the measured concentrations (in mg/L) is plotted on the y-axis. In comparison, the average of the two measurements (also in mg/L) is represented on the x-axis. The Spearman coefficient between the differences and the mean of the measurements corresponds to −0.603, with P = 0.002.
Serum adalimumab concentrations within the detection range were assessed in 23 patients using both assays. The median concentration for Quantum Blue was 8.2, while for Promonitor, it was 5.50, indicating that Quantum Blue results are approximately 8.25% higher than those from the Promonitor. There was no significant difference from zero (3.526; 95% CI: 0.842–4.742) in the Passing-Bablok regression intercept, but the slope was less than one (0.266; 95% CI: 0.085–0.583), indicating a proportional difference between the tests. Furthermore, Lin’s correlation coefficient of agreement was 0.247, demonstrating a moderate agreement between the methods. A Bland-Altman analysis was also performed to evaluate the degree of agreement between the assays, showing moderate agreement (Spearman correlation of −0.603, P = 0.002) (Figure 2c). We found two values outside the limits of agreement, both below the expected range, suggesting a tendency for the Promonitor assay to yield lower values than the Quantum Blue assay.
Levels of anti-drug antibodies and formation of immunocomplexes in Crohn’s disease patients
To evaluate the occurrence of immunogenic processes, first, we measured the presence of anti-adalimumab antibodies (ADA) in patients who presented infratherapeutic levels (n = 12 detected by Quantum-Blue and n = 15 detected by Promonitor). We employed the Promonitor assay for ADA levels, adhering to the manufacturer’s specifications, delineating positive results as values surpassing 10 AU/mL. Of the 12 patients with infratherapeutic levels of adalimumab, according to the Quantum-Blue test, four (33.3%) were positive for ADA, with three patients in the CDA and one in the CDR group. Among the 15 patients who presented infratherapeutic levels of adalimumab according to the Promonitor test, three (20%) were positive, two belonging to the CDA group and one in the CDR group. Among the cohort of 27 patients who presented infratherapeutic adalimumab levels, nine had this result by both tests, and 14.8% (4/27) presented positive ADA (Figure 3a). We compared serum levels between the activity and remission groups, excluding patients with ADA positivity. However, no significant differences were observed (Supplementary Figure 1).

Immunogenicity in Crohn’s disease patients under adalimumab. (a) The Venn diagram shows positive ADA among patients with infratherapeutic adalimumab levels, determined by two diagnostic assays. The green region represents the patients (n = 3) identified as infratherapeutic exclusively by the Quantum-Blue test. The yellow region denotes the patients (n = 6) identified as infratherapeutic solely by the Promonitor test. The intersection area indicates the patients (n = 9) classified as infratherapeutic by both diagnostic tests. (b) The immune complex formation in patients with Crohn’s disease was measured using the ELISA assay.
We then evaluated the formation of immune complexes in all samples, which are aggregations of antibodies produced in response to biological medication. Significant differences were observed in the levels of immune complexes between the groups, with active patients exhibiting higher concentrations than patients in remission (Figure 3b).
Assessment of polymorphism genotypes for ATG16L1, CD96, and CD155
For the genotypic evaluation, we selected three genes: ATG16L1, CD96, and CD155. We could not identify patients with the AA variant genotype of the Single Nucleotide Variants (SNV) in the CD155 gene, so we did not proceed with the polymorphism assessment for this gene. However, we conducted a frequency analysis among wild-type homozygotes (GG) and heterozygotes (GA) for categorical variables (Supplementary Table 1). Among the evaluated variables, we found no association between the homozygosity/heterozygosity of this gene and specific characteristics, disease severity, or response to adalimumab.
To analyze the actual categorical variables for the genes with SNV variations, we separated them by target genes: ATG16L1 was represented in Table 4, and CD96 in Table 5. For ATG16L1, we found a significant difference in platelet counts, which were lower in carriers of the variant genotype (TT). Regarding CD96, the frequency of the CC genotype was higher in patients with colonic CD than those with the CT or TT genotypes (88.0% versus 12.0%, P = 0.05). Furthermore, we found a significant association between infratherapeutic levels, as measured by the Promonitor test, and the presence of the CD96 wild-type genotype. Three of the four patients positive for ADA had the CD96 wild genotype.
Comparison of frequencies of the ATG16L1 gene polymorphisms concerning the categorical variables.

n = 66 (patients who were included in the genotyping).
CDAI: Crohn’s disease activity index.
One patient did not have CDAI and platelet levels in the medical records. P-values in bold indicate a statistically significant difference, defined as P < 0.05.
Comparison of frequencies of CD96 gene polymorphisms concerning the categorical variables.

n = 66 (patients who were included in the genotyping).
CDAI: Crohn’s disease activity index.
One patient did not have CDAI and platelet levels in the medical records. P-values in bold indicate a statistically significant difference, defined as P < 0.05.
For the analysis of continuous variables, we found that patients with the wild-type CC genotype for the CD96 gene had higher C-reactive protein (CRP) values (Table 6) compared to patients with the variant genotype (TT) or heterozygous form (CT). Additionally, patients with the CC genotype for ATG16L1 had higher CDEIS values than those with the variant genotype, both alone and as heterozygotes (Table 7). Similarly, for fecal calprotectin, patients with the CC genotype for ATG16L1 showed increased levels compared to those with other genotypes, exhibiting twice calprotectin levels (Table 8).
Comparison between wild, variant, and heterozygous genotypes of the ATG16L1, CD96, and CD155 genes concerning the C Reactive Protein (CRP) levels.

For ATG16L1, CD96, CD155 genotypes distribution concerning the CRP levels, n = 61.
CRP: C-reactive protein. NA: not applicable.
CRP level was obtained from 61 patients who were genotyped. P-values in bold indicate a statistically significant difference, defined as P < 0.05.
Comparison between wild, variant, and heterozygous genotypes of the ATG16L1, CD96, and CD155 genes concerning the Crohn’s Disease Endoscopic Index of Severity (CDEIS).

For ATG16L1, CD96, CD155 genotypes distribution concerning the CDEIS, n = 37.
CDEIS: Crohn’s Disease Index of Severity. NA: not applicable.
CDEIS was calculated in 37 patients who were genotyped. P-values in bold indicate a statistically significant difference, defined as P < 0.05.
Comparison between wild, variant, and heterozygous genotypes of the ATG16L1, CD96, and CD155 genes considering fecal calprotectin levels.
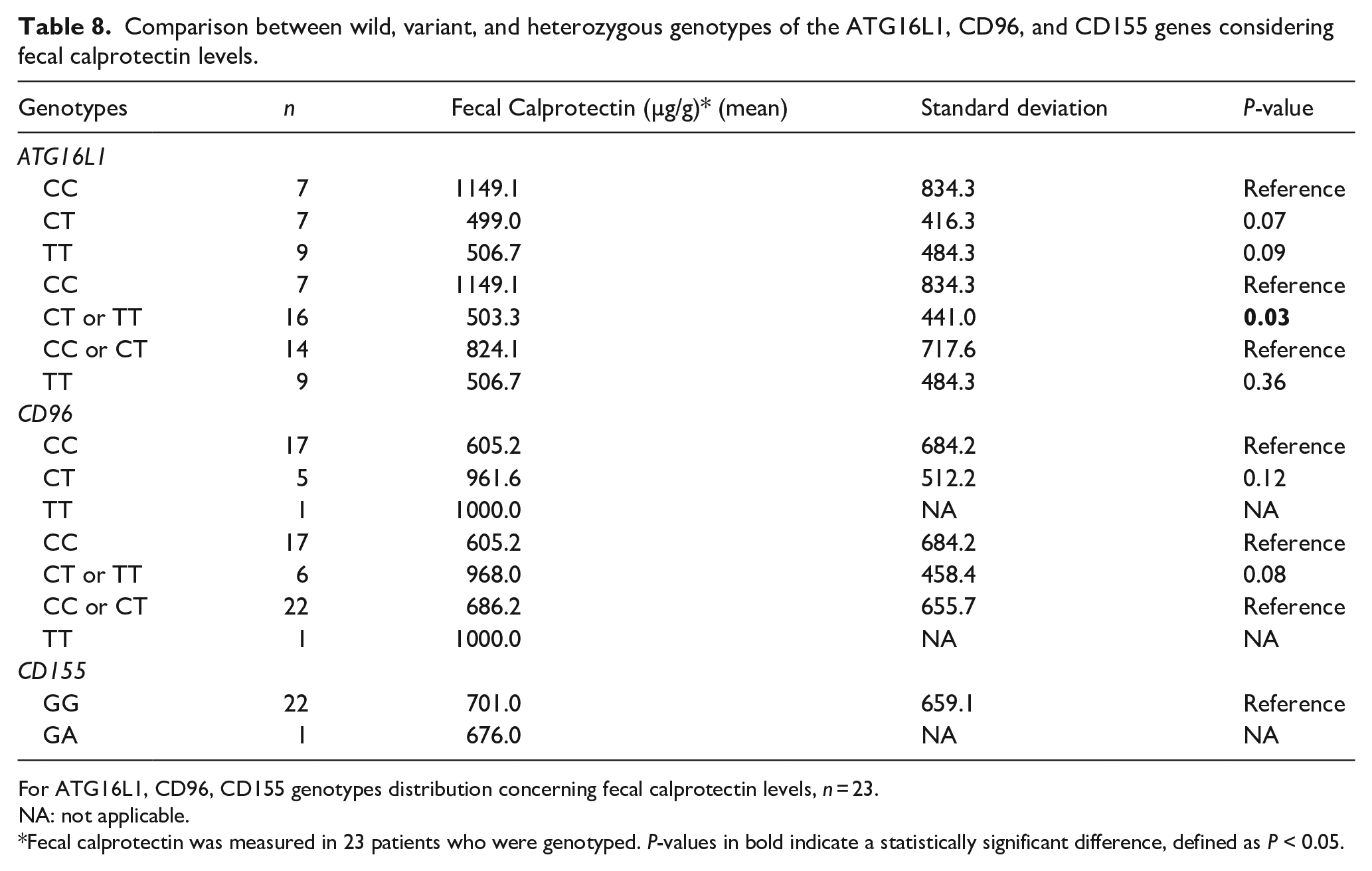
For ATG16L1, CD96, CD155 genotypes distribution concerning fecal calprotectin levels, n = 23.
NA: not applicable.
Fecal calprotectin was measured in 23 patients who were genotyped. P-values in bold indicate a statistically significant difference, defined as P < 0.05.
Our results showed that despite its humanized composition, adalimumab can provoke immunogenic responses that potentially diminish treatment efficacy. Monitoring adalimumab serum levels, detecting ADA, assessing immune complex formation, and evaluating genetic polymorphisms are pivotal for advancing precision medicine. These approaches enable tailored treatment strategies aligning with individual patient profiles, optimizing therapeutic outcomes with available medications. Our study also underscores the necessity of adhering to a consistent testing methodology to ensure reliability and consistency in monitoring data. This leads to a multifaceted approach to understanding and treating CD, integrating drug monitoring, immunological mechanisms, and genetic factors (Figure 4).

Precision medicine in the context of adalimumab therapy for Crohn’s disease entails a comprehensive approach to treatment optimization. A proposal with a personalized approach for CD patients under adalimumab includes meticulous therapeutic monitoring (trough-level drug monitoring and anti-drug antibody detection), immune complexes assessment, and genetic polymorphisms evaluation, which may improve patients’ outcomes and quality of life. These integrated strategies may contribute to optimizing the use of adalimumab, ensuring tailored therapeutic regimens that align with individual patient characteristics and precision medicine.
Discussion
Anti-TNF-α monoclonal antibodies are widely used as a therapeutic strategy for CD, establishing themselves as one of the most prevalent treatment modalities. Despite their impact on IBD management, loss of response remains a challenge, undermining the maintenance of patient remission. 11
Quantifying serum levels is a strategy that facilitates more effective control of therapy, proving highly useful for clinical management, with numerous studies focusing on understanding the correlation between adalimumab levels and their clinical and endoscopic implications. 32 We aimed to evaluate the potential relationship between serum adalimumab levels and CD activity/remission. No significant differences were observed between serum levels and disease activity in our cohort, regardless of the measurement test used. Our findings align with those of Ward et al., 33 who found no correlation between adalimumab levels and disease activity indices.
The literature describes immunomodulators combined with anti-TNF-α therapy as more effective than monotherapy, particularly in infliximab treatment. 34 No differences in serum levels were observed in our study cohort with concurrent use of the immunomodulator and adalimumab. However, a trend was noted when measured using the Quantum-Blue assay. These findings again align with those of Ward et al., 33 who found no influence of adalimumab alone or combined with an immunomodulator, on serum drug levels, and also with Arieira et al., 35 who did not find a correlation between the use of azathioprine and the pharmacokinetics of adalimumab.
Chanchlani et al. 36 observed benefits in utilizing immunomodulators when administered concurrently or before initiating adalimumab therapy, with a significant difference in ADA production. However, introducing immunomodulators following the initiation of anti-TNF-α treatment did not confer any advantage in this aspect. In our cohort, most patients started with adalimumab monotherapy, adding an immunomodulator only when the disease activity recurred, clinically, endoscopically, or both. This sequential approach may have contributed to the observed lack of efficacy in combination therapy.
Serum monitoring in IBD is extensively well-documented, showing its efficacy in personalized therapeutic management. The commercial assays for measuring serum levels must exhibit effectiveness, safety, and reliability in their detection. Several tests are available for this measurement, with ELISA and lateral flow assay being the most commonly utilized methodologies. Both approaches are effective and reliable,37,38 but new studies are still needed to assess the interchangeability between these methods.
Our objective was to assess whether the methodologies exhibited comparable behaviors and efficiency. Our analysis of interchangeability between the Quantum Blue and Promonitor assays revealed a significant difference but a moderate correlation. These findings align with Gomes et al., 24 who similarly identified a significant difference and a moderate correlation when evaluating serum levels of infliximab using the same tests. Cheli et al. 23 assessed the performance, interchangeability, and correlation between an ELISA assay (Chorus Promonitor®) and a lateral flow assay (RIDA QUICK®) for infliximab and adalimumab, showing a good agreement for adalimumab levels but a weak correlation for infliximab.
Our findings indicate that serum levels were higher when measured using the Quantum Blue assay than those assessed by the Promonitor assay, consistent with Cherry et al.’s, 39 who report higher adalimumab levels measured by rapid tests than with ELISA assays. Toja-Camba et al., 40 which employed the same comparison methods as ours, reported no significant differences between the assays for adalimumab, which contrasts with our findings. While the Spearman correlation observed in their study for both drugs indicated a strong correlation between the two assays, our results demonstrated a moderate correlation.
Our study demonstrates that both tests are practical, but switching assays during treatment is not advisable. It is recommended to consistently use the same methodology throughout the entire treatment to avoid potential inappropriate changes in results and, consequently, in therapy.
Lateral flow assays for therapeutic drug monitoring are valuable tools for associating drug levels with disease control and outcomes in CD patients. Verstockt et al. 41 investigated the relationship between adalimumab serum concentrations, ADA development, and long-term clinical outcomes in anti-TNF-naïve CD patients. They found that low serum drug levels at week 4 increased the risk of developing ADAs by week 12, leading to a higher frequency of treatment optimization and demonstrating a reduced likelihood of achieving sustained clinical benefit. This was primarily attributed to primary non-response or secondary loss of response to adalimumab.
Many studies have reported a positive correlation between higher drug levels and improved disease activity in CD. However, our findings differed, as we found no significant association between activity/remission and adalimumab levels. CD, a heterogeneous condition, is affected by factors that can impact treatment outcomes independently of drug levels. These include disease location, strictures and fistulas, and severity. Furthermore, specific therapeutic thresholds may limit additional clinical benefit from higher drug levels. 42
In our study, 41.86% of CDA patients switched to adalimumab after failing previous anti-TNF treatment, compared to 14.81% of patients in the CDR group. Primary loss of response (inefficacy at 14–24 weeks of treatment 43 ), occurred in two patients from the CDA group, while 41 patients from the same group experienced secondary loss of response, often linked to ADA or reduced drug efficacy over time.
Adalimumab has biological advantages over infliximab due to its reduced immunogenic potential and full human composition, unlike infliximab, which has a 25% murine portion. 44 However, response rates between chimeric monoclonal antibodies (infliximab) and humanized ones (adalimumab and certolizumab pegol) show no significant differences. 45 Among our patients with infraterapeutic adalimumab levels, 33.3% and 20% tested positive for ADA, depending on the assay. These rates were slightly higher than West et al., 46 17%, and Sun et al., 47 18.7%.
Studies show that IBD patients face an annual loss of response of 13%–21% after remission induction.11,48–50 Schultheiss et al. 51 demonstrated that with anti-TNF-α treatment, the response risk tripled after the first year but was lower in responders after 2 years. Similarly, in our cohort, patients with active disease had been using adalimumab for a shorter duration than those in remission (P = 0.0390), suggesting that long-term users experience better efficacy in its use.
When circulating ADA binds to the medication, immune complexes form and are quickly cleared, affecting the pharmacokinetics of the monoclonal antibody and consequently contributing to the loss of response.52–55 Studies on infliximab showed that these immune complexes are correlated with short-term adverse events occurring shortly after infusion, such as headaches, nausea, fever, and dyspnea.56–58 In our patients, we found a significant difference in the presence of immune complexes between those with active disease and those in remission, suggesting a link to disease activity likely caused by the presence of ADA and their consequent therapeutic failure.
ADAs are immune system responses directed against a therapeutic drug, recognizing it as a foreign entity and producing antibodies that can hinder treatment efficacy. These antibodies can lower the drug’s therapeutic levels and trigger adverse reactions. 14 Immune complexes form when antibodies bind to antigens, including drugs or foreign proteins. The immune system typically clears these complexes, but their accumulation in tissues can lead to inflammation or tissue damage. 20 In biological therapies, immune complexes may form when ADA binds to the drug or its metabolites, affecting treatment safety and efficacy. In our study, while we did not assess the specific composition of these immune complexes, their presence in patients with negative ADA suggests that their composition is likely not limited to anti-drug antibodies alone.
Our research underscores the need to study the composition of antibodies that constitute the immune complexes. Their presence in CD patients supports additional evidence of the potential autoimmune etiology of the condition; this is consistent with da Silva et al., 59 who identified substantial regulation of pathways associated with B cell functionality. Immune complexes have been well-documented in the literature concerning autoimmune diseases, including circulating immune complexes in autoimmune hepatitis associated with Sicca syndrome and renal disease. 60 Similarly, Yamane et al. 61 found a correlation between immune complexes in patients’ saliva and Sjögren’s syndrome.
In addition to the classic mechanisms of therapeutic failure, such as ADA and immune complex formation, studies have highlighted the influence of genetic variability’s impact on monoclonal antibody responses. 62 Thus, we evaluated the role of genes previously linked to adalimumab response, focusing on ATG16L1 and CD96 polymorphisms. In our cohort, patients carrying the wild-type genotype CC for ATG16L1 exhibited higher CDEIS and fecal calprotectin values, suggesting that the variant genotype is associated with remission. These findings corroborate Nuij et al.’s 26 findings, who observed better adalimumab response in patients with variant genotypes. Furthermore, the authors found no association between the response to the drug and the occurrence of side effects with the polymorphisms, a result consistent with our results.
Our findings also corroborate those of Koder et al., 63 who reported that patients with the CC genotype for ATG16L1 exhibited a long-term loss of response. In contrast, variant genotypes predicted better response to adalimumab. Similarly to our results, at least one variant allele was sufficient for a better response. Barber et al. 64 also observed that a T allele, in homozygosity or heterozygosity, led to a more favorable response to adalimumab.
Among the four positive ADA patients, three had the CD96 wild-type genotype, including two of the CDA group, which might be associated with insufficient serum levels, immunogenicity, and more severe disease. Aterido et al. 27 identified an association between ADA production and the presence of the variant allele for CD96. A limitation of our study, apart from environmental differences exerting epigenetic influence, is the smaller sample size tested for ADA, which did not allow us to perform statistical correlations with the genotypes compared to the study of Aterido et al.
Including only patients within the detection range of the assays to analyze interchangeability may also be considered a potential study limitation because of the decrease in sample size. Another limitation is the absence of a statistical test for sample size calculation, as this study was observational. Other limitations of our study include that it was a cross-sectional and not a longitudinal study, it did not have access to the patient’s outcomes, its methodology with only one test for assessing ADA, and also the restricted number of patients included in the study, resulting from the research conducted during the COVID-19 pandemic when only patients with urgent needs were seen in the clinics. This may have affected the genetic analysis, potentially explaining the absence of SNVs for the CD155 gene, with a larger sample size increasing the chance of finding variants.
Conclusion
In conclusion, our study revealed that most patients with positive ADA during disease activity exhibited low adalimumab levels. The effectiveness of treatment with adalimumab was not directly related to higher medication levels. Additionally, both serum measurement methodologies of adalimumab levels demonstrated significant differences but moderate agreement, highlighting the importance of maintaining the same monitoring test during the anti-TNFα therapy. Genotypic differences underline the correlation between the wild genotype for CD96 and ATG16L1 with unfavorable laboratory and endoscopic response to adalimumab. Finally, CD patients with disease activity were accompanied by significantly higher levels of immune complexes, indicating an association with non-response to adalimumab. Further investigation is needed to depict the nature of these immune complexes, which may better explain the immunogenicity involved in the biological therapy in CD and unveil its etiopathogenesis towards an auto-immune disease.
Supplemental Material
sj-docx-1-iji-10.1177_03946320251319379 – Supplemental material for Therapeutic drug monitoring and immunogenetic factors associated with the use of adalimumab in Crohn’s disease patients
Supplemental material, sj-docx-1-iji-10.1177_03946320251319379 for Therapeutic drug monitoring and immunogenetic factors associated with the use of adalimumab in Crohn’s disease patients by Livia Moreira Genaro, Juliana Carron, Marina Moreira de Castro, Ana Paula Menezes de Freitas Franceschini, Gustavo Jacob Lourenço, Cristiane Kibune Nagasako Vieira da Cruz, Glaucia Fernanda Soares Rupert Reis, Livia Bitencourt Pascoal, Juliana Delgado Campos Mello, Isabela Machado Pereira, Millene Leal Nascimento, Priscilla De Sene Portel Oliveira, Ligiana Pires Corona, Maria de Lourdes Setsuko Ayrizono, Carmen Silvia Passos Lima and Raquel Franco Leal in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-docx-2-iji-10.1177_03946320251319379 – Supplemental material for Therapeutic drug monitoring and immunogenetic factors associated with the use of adalimumab in Crohn’s disease patients
Supplemental material, sj-docx-2-iji-10.1177_03946320251319379 for Therapeutic drug monitoring and immunogenetic factors associated with the use of adalimumab in Crohn’s disease patients by Livia Moreira Genaro, Juliana Carron, Marina Moreira de Castro, Ana Paula Menezes de Freitas Franceschini, Gustavo Jacob Lourenço, Cristiane Kibune Nagasako Vieira da Cruz, Glaucia Fernanda Soares Rupert Reis, Livia Bitencourt Pascoal, Juliana Delgado Campos Mello, Isabela Machado Pereira, Millene Leal Nascimento, Priscilla De Sene Portel Oliveira, Ligiana Pires Corona, Maria de Lourdes Setsuko Ayrizono, Carmen Silvia Passos Lima and Raquel Franco Leal in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-pdf-3-iji-10.1177_03946320251319379 – Supplemental material for Therapeutic drug monitoring and immunogenetic factors associated with the use of adalimumab in Crohn’s disease patients
Supplemental material, sj-pdf-3-iji-10.1177_03946320251319379 for Therapeutic drug monitoring and immunogenetic factors associated with the use of adalimumab in Crohn’s disease patients by Livia Moreira Genaro, Juliana Carron, Marina Moreira de Castro, Ana Paula Menezes de Freitas Franceschini, Gustavo Jacob Lourenço, Cristiane Kibune Nagasako Vieira da Cruz, Glaucia Fernanda Soares Rupert Reis, Livia Bitencourt Pascoal, Juliana Delgado Campos Mello, Isabela Machado Pereira, Millene Leal Nascimento, Priscilla De Sene Portel Oliveira, Ligiana Pires Corona, Maria de Lourdes Setsuko Ayrizono, Carmen Silvia Passos Lima and Raquel Franco Leal in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
We thank Prof. Tristan Torriani for the English editing.
Authors’ contributions
Livia Moreira Genaro: Data curation; Formal analysis; Investigation; Methodology; Project administration; Validation; Writing—original draft. Juliana Carron: Data curation; Formal analysis; Writing—review & editing. Marina Moreira de Castro: Formal analysis; Writing—review & editing. Ana Paula Menezes de Freitas Franceschini: Data curation. Gustavo Jacob Lourenço: Formal analysis, Writing—review & editing. Cristiane Kibune Nagasako Vieira da Cruz: Data curation, Writing—review & editing. Glaucia Fernanda Soares Rupert Reis: Data curation, Writing—review & editing. Livia Bitencourt Pascoal: Formal analysis; Writing—review & editing. Juliana Delgado Campos Mello: Methodology; Writing—review & editing. Isabela Machado Pereira: Writing—review & editing. Millene Leal Nascimento: Writing—review & editing. Priscilla De Sene Portel Oliveira: Writing—review & editing. Ligiana Pires Corona: Formal analysis; Investigation; Methodology; Writing—review & editing. Maria de Lourdes Setsuko Ayrizono: Data curation; Writing—review & editing. Carmen Silvia Passos Lima: Methodology; Writing—review & editing. Raquel Franco Leal: Conceptualization; Data curation; Formal analysis; Funding acquisition; Methodology; Resources; Supervision; Writing—review & editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the São Paulo Research Foundation (FAPESP) [Grant numbers #2020/01924-5 for L.M.G.; #2021/04349-4 for A.P.M.F.F.; #2021/10265-8 for C.S.P.L. and #2023/12810-9 for J.C.] and by the National Council for Scientific and Technological Development (CNPq) [Grant numbers #140462/2020-1 for LMG and #302557/2021-0 for R.F.L.].
Ethics approval and consent to participate
This study was approved by the Ethics Committee of the University of Campinas (CAAE: 92894418.4.0000.5404). All patients signed the informed consent form.
Consent for publication
All patients signed the informed consent form described in the Methods section.
Informed consent
Written informed consent was obtained from all subjects before the study.
ORCID iDs
Availability of data and materials
All data generated or analyzed during this study are included in this article and in the additional files.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
