Abstract
Expression and localization of members of the aquaporin (AQP) family (AQP1, 2, 3, 4, and 5) in the kidney of the musk shrew (Suncus murinus) was examined by immunohistochemistry. AQP1 was expressed in the proximal tubules and in the thin limb of the loops of Henle. AQP1 was the only water channel expressed in the proximal nephron examined, indicating that AQP1 may be an independent water transporter in the proximal nephron. AQP2 and AQP5 were localized to the apical cytoplasm of the cortical to medullary collecting duct (CD) cells and AQP3 and AQP4 were localized to the basal aspect of the cortical to medullary CD cells. AQP3 expression was weaker in the cortical cells compared with the medullary cells, whereas AQP4 was strongly positive throughout the CD. These indicate that the CD is the main water reabsorption segment of the nephron and is regulated by AQPs. Indeed, apical water transport of CD cells of the musk shrew may be controlled by both AQP2 and AQP5. The characteristic expression pattern of the AQPs in this animal provides a novel animal model for elucidating the regulation of water reabsorption by AQPs in the mammalian kidney.
A
The musk shrew (Suncus murinus) is a small animal that exhibits several primitive mammalian characteristics (Shigehara 1985; Kimura and Tohya 1989; Endo et al. 1992) and has been used as a laboratory animal model for the study of basic mammalian physiology and pathology (Kimura et al. 1996; Takata et al. 1999). In the musk shrew kidney, histological characteristics are shown in a thin cortex, relatively small renal corpuscles, and simply convoluted glomerular capillary (Kitoh et al. 1985). The study of the body fluid regulation mechanism in this animal will lead to knowing the basic system in many mammals. In this study we examined the expression and localization of several AQP subtypes in the nephron of the musk shrew kidney including AQP1, AQP2, AQP3, AQP4, and AQP5. There are some key differences in the expression and localization of the AQP subtypes between rodents and human, as previously reported.
Materials and Methods
Animals
Twenty adult male musk shrews, 16 to 24 weeks of age and weighing 55–60 g, were used in this study according to procedures approved by The Animal Care and Use Committee at Hyogo College of Medicine. Animals were kept in separate cages and maintained under a 12-hr light/dark cycle at a constant temperature of 25C. Food and water were given ad libitum until sacrifice. Animals were anesthetized with diethyl ether and sodium pentobarbital (40 mg/kg, IP). For reverse transcription (RT)-PCR and Western blotting examination, animals were killed by decapitation, and kidneys were removed. Kidneys were immediately frozen in liquid nitrogen and stored at −80C until use. For histochemical experiments, an overdose of pentobarbital was injected into the animals and intracardially perfused with PBS followed by Bouin's fixative.
RT-PCR
Total RNA of the kidney was isolated with TRizol reagent (Invitrogen; Carlsbad, CA) according to the manufacturer's protocol. Single-stranded cDNA was synthesized with reverse transcriptase and oligo (dT) primer (Invitrogen). cDNA was used for PCR with Taq DNA polymerase (Hot Master; Perkin-Elmer, Watertown, MA) with primers designed using rodent, human, and musk shrew cDNA data in DDBJ (Table 1). PCR amplification was performed on 1 μl of cDNA in a thermal cycler for 35 cycles. Each cycle consisted of a heat denaturation step at 94C for 30 sec, annealing of primers at 55–60C for 30 sec, and polymerization at 72C for 30 sec. RNA without RT was used for the negative controls of RT-PCR with no positive amplification being observed.
Antibodies
Anti-AQP antibodies in the present study were as follows: polyclonal rabbit anti-AQP1 antibody (PAb) (Chemicon; Temecula, CA), rabbit anti-AQP2 PAb (Almone Labs; Jerusalem, Israel), goat anti-AQP4 PAb (Santa Cruz Biotechnology; Santa Cruz, CA), and goat anti-human AQP5 PAb (Santa Cruz Biotechnology). A polyclonal rabbit anti-AQP3 PAb was raised against the C-terminal 18 amino acids estimated from the AQP3 cDNA sequence of the musk shrew (DDBJ accession no. AB275385). Secondary antibodies used for brightfield microscopic examination included biotin-conjugated donkey anti-rabbit IgG PAb (Jackson ImmunoResearch; West Grove, PA) or donkey anti-goat IgG PAb (Jackson ImmunoResearch). Cy3 or FITC-conjugated donkey anti-rabbit IgG (Jackson ImmunoResearch) and anti-goat IgG PAb (Jackson ImmunoResearch) were used for fluorescent studies. Horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG PAb and rabbit anti-goat IgG PAb (Zymed; South San Francisco, CA) were used for Western blotting.
Oligonucleotides used for RT-PCR a

aChoice of oligonucleotides was carried out according to the sequences available in DDBJ (see text).
Western Blotting
Musk shrew kidneys were homogenized in 10 mM Tris–HCl (pH 7.4) containing 1% Triton X-100, 2 mM EDTA, and protease inhibitor cocktail (Complete; Roche Diagnostics, Basel, Switzerland). The homogenate was centrifuged at 12,000 × g for 20 min, and the supernatant was collected. SDS-PAGE was performed with the buffer and urea gel according to the condition in Neely et al. (1999). The supernatant was denatured in 62.5 mM Tris–HCl (pH 7.4) containing 10% glycerol, 2% SDS, 50 mM dithiothreitol, and 0.01% bromophenol blue. SDS-PAGE was performed with the 4.0% acrylamide stacking gel containing 2 M urea and 7.5% acrylamide separating gel containing 4 M urea. Separated proteins were transferred to the polyvinylidene difluoride membrane, and immunostaining was carried out. Primary antibodies were diluted as follows: AQP1, 1:4000; AQP2, 1:20,000; AQP3, 1:4000; AQP4, 1:2000; and AQP5, 1:2000; and membranes were incubated with primary antibodies overnight at 4C. Control examination was performed using the antibodies preincubated with their specific peptide provided by the companies. Secondary antibodies were diluted as follows: HRP-conjugated anti-rabbit IgG, 1:200,000 and HRP-conjugated anti-goat IgG, 1:50,000, and membranes were incubated for 2 hr at room temperature. HRP-chemiluminescence kit (ECL; GE Healthcare, Buckinghamshire, UK) was used for signal detection, and signals were taken with lumino image analyzer LAS-1000 (Fujifilm; Tokyo, Japan).
Immunohistochemistry
Immunohistochemical analysis was performed on 6-μm kidney sections derived from Bouin's fluid-fixed paraffinembedded tissue blocks. After deparaffinization and dehydration, antigen retrieval was carried out by autoclaving the sections for 10 min at 120C in 20 mM citrate buffer (pH 6.0). Slides were then treated with 1% H2O2 in methanol for 20 min to block endogenous peroxidase activity. After blocking with Block Ace (Dainippon Sumitomo Pharm; Osaka, Japan) for 1 hr at room temperature, tissue sections were incubated with primary antibodies overnight at 4C. Dilution ratios of each primary antibody were as follows: AQP1, 1:300; AQP2, 1:1000; AQP3, 1:500; AQP4, 1:500; and AQP5, 1:500. Tissue sections were washed in PBS followed by incubation with the relevant biotin-conjugated secondary antibodies diluted 1:200 for 1 hr at room temperature. For brightfield examination, tissues were colorized with the ABC system (Vector Laboratories; Burlingame, CA) and diaminobenzidine (Zymed) with sections being observed under light microscopy (BX-50; Olympus, Tokyo, Japan). Control examination was performed using preincubated primary antibodies with their specific peptide, and positive reactions disappeared (data not shown). Serial sections were stained with hematoxylin and eosin for histological examination. Dual-immunofluorescent histochemistry was performed using mixed primary antibodies. Specimens were examined by confocal laser scanning microscopy (LSM-510; Carl Zeiss, Oberkochen, Germany). Specificity of the secondary antibodies was examined by immunohistochemistry, and no cross-reactivity was observed.
Results
RT-PCR amplification was observed for AQP1, 2, 3, 4, and 5 (Figure 1A), indicating that these AQPs are expressed in the musk shrew kidney. Expression of these AQPs was also detected with Western blotting in the kidney homogenate, and typical major bands were observed in each blot (Figure 1B). AQP3 was reactive to <100-kDa band in addition to the <37-kDa band. This may be the oligomeric AQP3, which was not dissociated in the sample buffer (Roudier et al. 2002). AQP5 has not been previously detected in the mammalian kidney, and this is the first report of the expression of AQP5 in the mammalian kidney.
AQPs examined in this study were localized to the nephron in the kidney tissue (Figure 2). AQP1 was localized to the proximal tubules and followed the loop of Henle (Figures 2A–2D). Although strong reactivity was observed in the straight part of the proximal tubules and the following thin limbs (Figure 2C), weak expression was also noted in the apical part of the convoluted tubules (Figure 2A). AQP1 was localized to the apical and basolateral region of the straight tubules and thin limb (Figure 2C, inset). AQP1 was the only water channel detected in the proximal nephron in this study. The other AQP subtypes (AQP2, AQP3, AQP4, and AQP5) were distributed in the cortical and medullary CD cells (Figures 2E–2L). AQP2 reactivity was localized to the apical cytoplasm of cortical CD cells with reactivity being closer to the apical membrane in the medullary CD cells (Figures 2E and 2F). Unlike AQP2 reactivity, AQP3 was distributed to the basolateral region of the CD cells (Figures 2G and 2H). This reactivity was a little weaker in cortical cells compared with medullary cells. This raises the possibility that the water transport activity by AQP3 may be higher in the distal CD than in the proximal CD. AQP4 was also localized to the basal region of CD cells but, unlike AQP3, there was no difference in the intensity of the immunoreactivity observed between the cortex and medulla (Figures 2I and 2J). AQP5 immunoreactivity was observed in the apical cytoplasm of the cortical CD and was closer to the apical membrane in the medullary CD (Figures 2K and 2L). Cellular distribution of these AQPs in CD cells was examined by confocal laser scanning microscopy following dual-fluorescent staining (Figure 3). AQP2 and AQP4 were located in the apical cytoplasm and basolateral aspect of the cells, respectively (Figures 3A–3C). Both AQP3 and AQP4 colocalized to the basal membrane of the cells (Figures 3D–3F), whereas AQP2 and AQP5 were both distributed over the apical cytoplasm (Figures 3G–3I). Thus, AQPs exhibit either an apical (AQP2 and AQP5) or basal (AQP3 and AQP4) location on CD cells. This indicates that they may play a role in water transport into and out of the cell. Interestingly, a characteristic feature of the musk shrew kidney is the distribution of four different kinds of AQPs within a single cell.
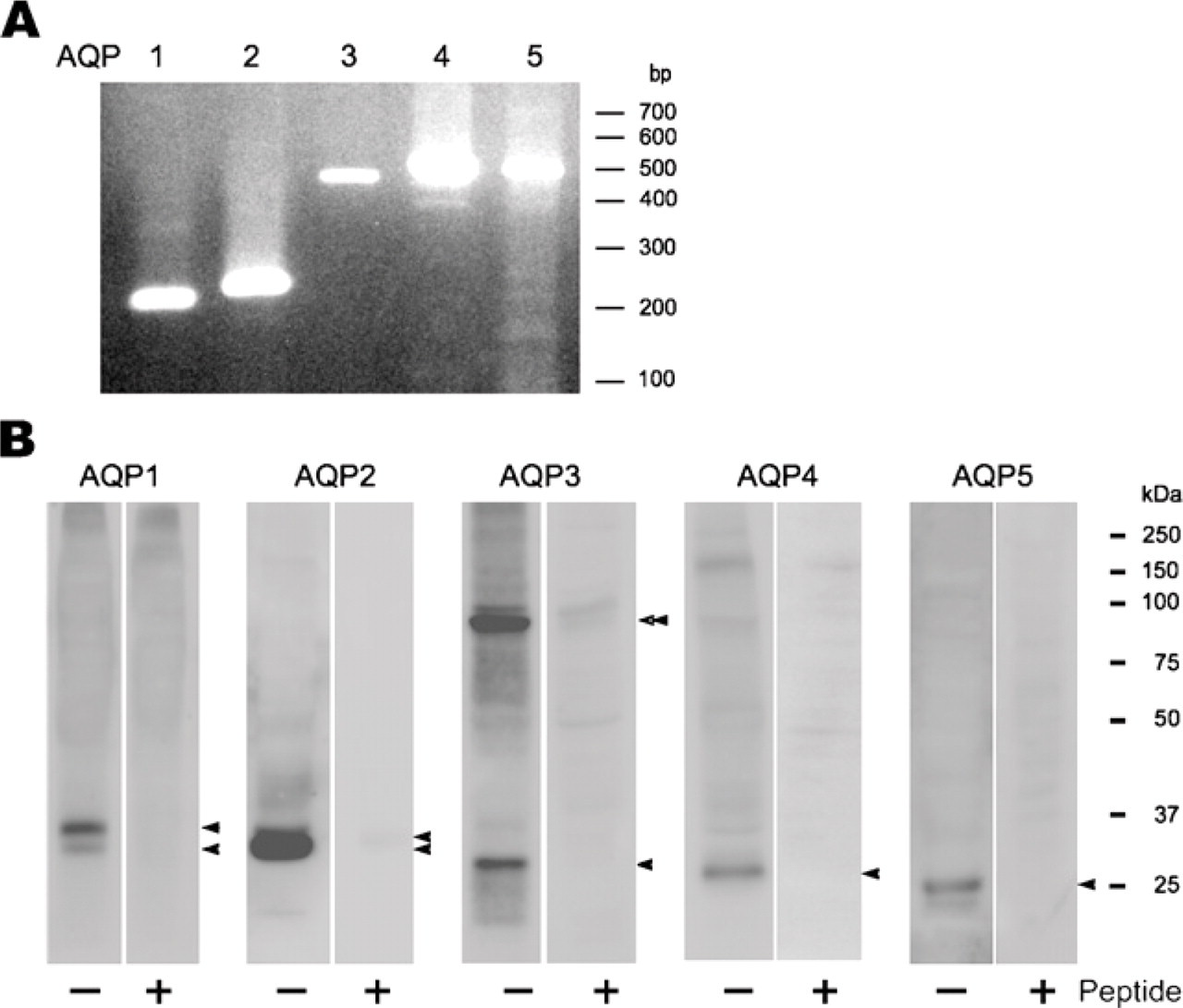
Expression of AQPs by RT-PCR and Western blotting analysis in the musk shrew kidney. (
Discussion
In this study we examined the expression and localization of AQP1, AQP2, AQP3, AQP4, and AQP5 in the kidney of the insectivore, musk shrew (Suncus murinus). AQP1 was expressed in the proximal straight tubules and in the thin limb of the loop of Henle with weak expression evident in the convoluted tubules. AQP1 was the only water channel detected in the proximal part of the nephron in this study. Similar results have been obtained in previous studies of rodent and human kidney (Denker et al. 1988; Sabolic et al. 1992; Nielsen et al. 1993b, c, 1995; Hasegawa et al. 1994; Maunsbach et al. 1997). A large amount of the water is reabsorbed in these segments of the nephron according to the osmotic gradient between the primitive urine and the renal interstitium. AQP1 is localized in both apical and basal plasma membrane in proximal tubules and descending thin limb, consistent with a role in the transcellular water movement across both surfaces of the cells (Nielsen et al. 1993c). Schafer et al. proposed that volume reabsorption in the proximal tubules could be explained by luminal dilution produced by active NaCl in combination with an effective luminal hypotonicity produced by the preferential reabsorption of solutes such as glucose, amino acids, and bicarbonate, which have higher reflection coefficients than the NaCl that remains in the lumen (Andreoli et al. 1978; Schafer et al. 1978; Andreoli and Schafer 1979). Thus, AQP1 may facilitate the water effectively in the proximal tubule. Axial heterogeneity was observed in the convoluted tubules and the straight tubules in the proximal tubules. There is heterogeneity of the expression of AQP1 in the rat and human proximal tubules (Nielsen et al. 1993c; Maeda et al. 1995; Maunsbach et al. 1997). It is not clear how the high water permeability is maintained in the convoluted tubules. There is the possibility that the other types of channel homologous or water transport systems are present in the convoluted tubules. As a result, AQP1 is functionally conserved in the mammalian nephron. AQP1 may act as the main channel for water reabsorption in the proximal tubules of mammals.
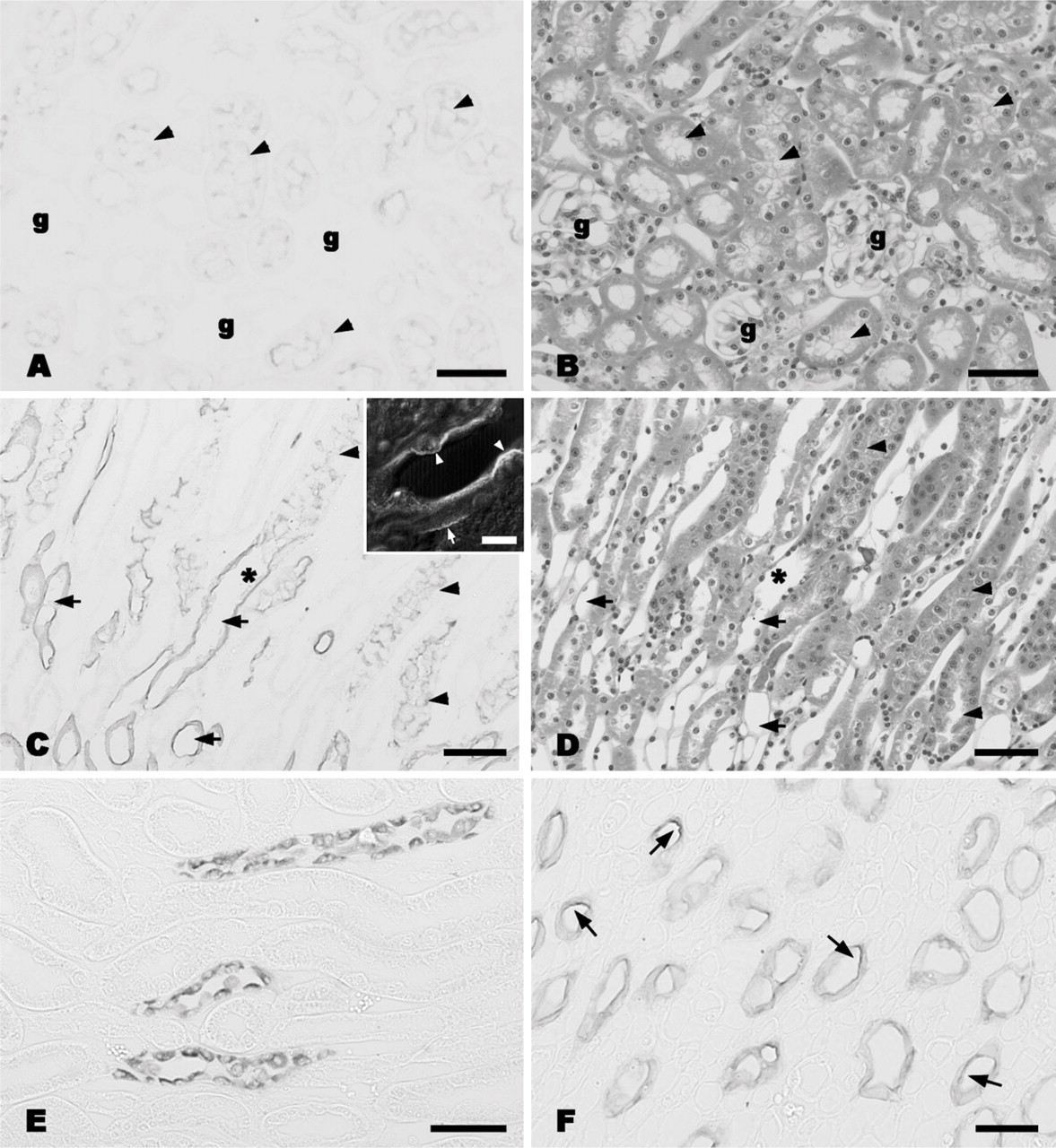
Immunohistochemical localization of AQPs in the musk shrew kidney. (A) AQP1 in the cortex. Weak but positive reaction is observed in the apical site of the proximal convoluted tubules (arrowheads). Glomeruli are indicated with g. (
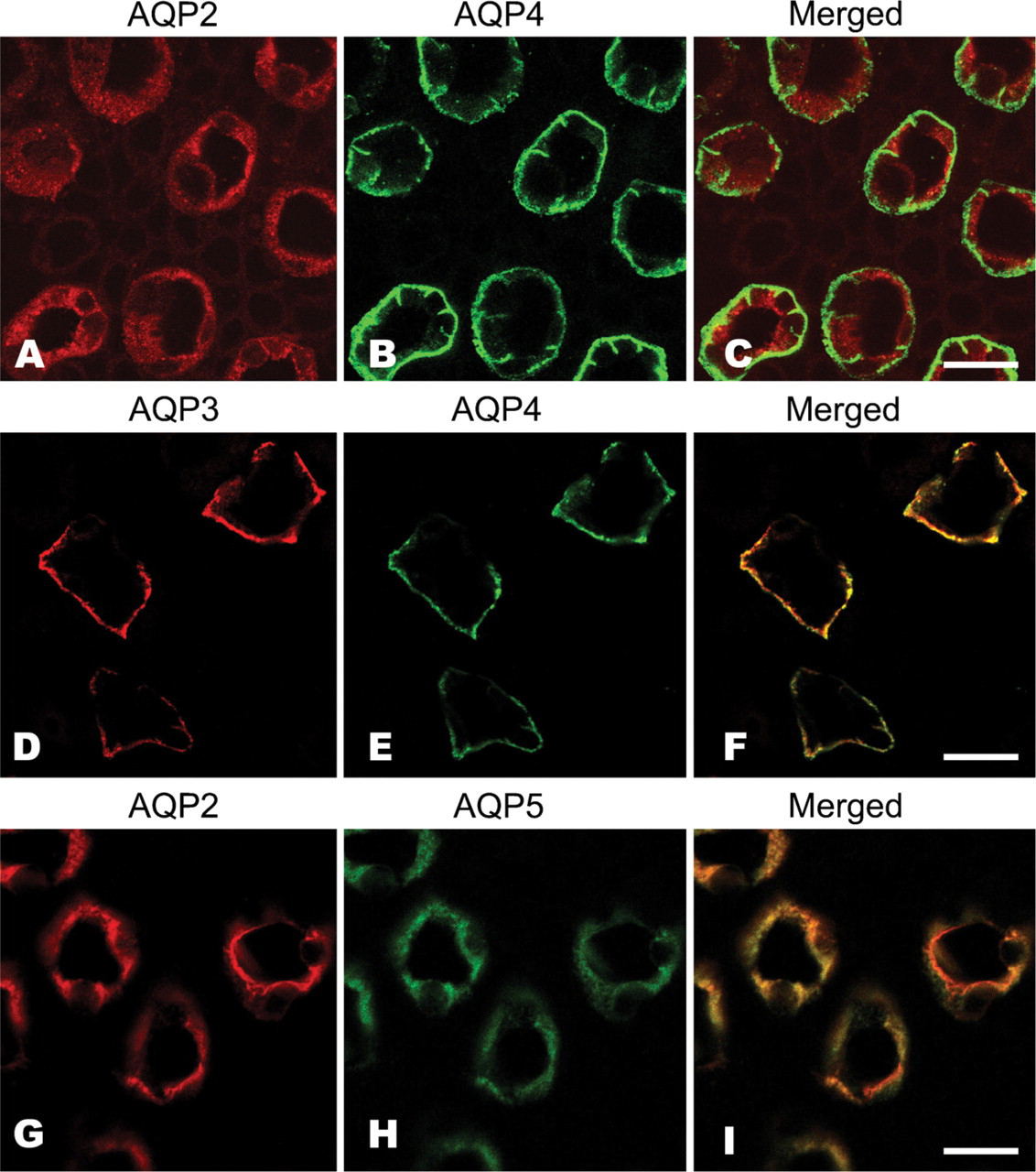
Dual immunofluorescent histochemistry of AQPs in musk shrew nephrons. AQP2 (
Four subtypes of AQPs (AQP2, AQP3, AQP4, and AQP5) were all localized to the CD cells. Whereas AQP2, AQP3, and AQP4 have been reported to be detected in CD cells in rodents and human (see reviews, Takata et al. 2004; Verkman 2005), expression of AQP5 has not been detected in mammalian kidney. Interestingly, AQP2 and AQP5 colocalized at the apical cytoplasm of the cells, whereas AQP3 and AQP4 were at the basolateral membrane. It is known that AQP2 translocates from the intracellular compartment to the plasma membrane of the principal cell of the CD following stimulation by vasopressin (Fushimi et al. 1993; Nielsen et al. 1993a, 1995; DiGiovanni et al. 1994; Marples et al. 1995; Yamamoto et al. 1995). The similar localization of AQP2 observed in this study suggests that the same regulatory mechanism may be present in the musk shrew kidney. On the other hand, AQP5 is known to be commonly expressed in secretory epithelial cells, such as those within the salivary gland, lachrymal gland, eye, and airway of mammals (Raina et al. 1995; Matsuzaki et al. 1999, 2003; Gresz et al. 2001; Wang et al. 2003; Maeda et al. 2005). Whereas AQP5 was not detected in the rat kidney (Raina et al. 1995), AQP5 expression in the CD cells of the musk shrew kidney suggests that regulation of AQP5 expression may differ among mammalian species. In a similar fashion to AQP2, AQP5 is translocated from the intracellular compartment to the plasma membrane as the response to muscarinic or adrenergic receptor stimulation in rat parotid acinar cells (Ishikawa et al. 1998, 1999) and via cAMP in mouse lung epithelial cells (Yang et al. 2003). Although it is not known whether regulation of AQP5 translocation in the musk shrew CD cells of the kidney is similar to that operating in the epithelial cells of the rodent parotid and lung, there seem to be at least two different systems for regulating water transport by AQP2 and AQP5 in CD cells in the musk shrew kidney. If this primitive mammal shows a basic AQP expression pattern, it is necessary to inspect the AQP5 expression in the kidney in other mammalian species.
AQP3 and AQP4 were localized to the basal region of the CD in the musk shrew. They were exclusively fixed to the basolateral aspect of the cells. Colocalization of these AQPs is well known in rodents (Echevarria et al. 1994; Ishibashi et al. 1994; Ma et al. 1994; Ecelbarger et al. 1995; Frigeri et al. 1995; Terris et al. 1995; van Hoek et al. 2000). It suggests that water transport at the basal side of CD cells may involve AQP3 and AQP4, and this may be a conserved pattern in mammals. Differing levels of AQP3 immunoreactivity in the musk shrew were demonstrated between the cortical and medullary section of the CD, thereby suggesting that different water transport activity may be present in these parts of the CD. In the rat, AQP3 is abundant in the cortical as well as the medullary CD (Ecelbarger et al. 1995; Terris et al. 1995), whereas AQP4 is mainly abundant in the inner medulla (Terris et al. 1995). It is interesting that the expression pattern of these AQPs suggests that these AQPs could replace each other in the musk shrew. AQP3 knockout mice exhibit a marked urinary concentrating defect with severe polyuria (Ma et al. 2000), whereas mice lacking AQP4 exhibit a mild urinary concentrating defect (Ma et al. 1997; Chou et al. 1998). Consideration of the different expression levels of these two AQPs in this study and the phenotype of AQP3 or AQP4 knockout mice suggests that the renal reabsorption activity of AQP4 might be superior to AQP3 in the musk shrew. The exact importance of the effect of this quantitative difference in expression level of these AQPs upon water transport is unclear, but there are some differences in water permeability between the CD of the cortex and that in the medulla.
Physiological characteristics of the musk shrew are revealed by blood biochemistry analysis (Nagai et al. 1984; Hasegawa and Sugawara 1985; Ozawa et al. 1985; Tsukahara et al. 1990). The musk shrew exhibits a higher concentration of blood urea nitrogen and a lower concentration of serum lipids such as total cholesterol, triacylglycerol, phospholipid, and nonesterified fatty acid compared with rodents (Hasegawa and Sugawara 1985). Moreover, serum concentrations of thyroid hormones differ between the rat and musk shrew (Ozawa et al. 1985; Tsukahara et al. 1990). To date, no differences have been reported in urine production between the animals, and it is therefore not known whether these serum biochemistry differences are directly or indirectly related to differences in the urinary production system of the nephron. If the expression of AQPs in the nephron varies with the genetic and/or living environment of animals, then it is likely that the functional importance of the AQPs may be clarified by studying the expression and distribution of AQPs in various mammalian species.
Footnotes
Acknowledgements
This study was supported in part by a Grant-in-Aid for Researchers, Hyogo College of Medicine, Hyogo, Japan. We thank Ms. Megumi Hatta and Mr. Katsumi Gion (Department of Anatomy, Hyogo College of Medicine) for secretarial assistance.
