Abstract
Aquaporin-9 (AQP9) is a water channel membrane protein also permeable to small solutes such as urea, glycerol, and 5-fluorouracil, a chemotherapeutic agent. With the aim of understanding the pathophysiological role of AQP9, we performed an extensive analysis by Western blotting, RT-PCR, and immunolocalization in rat tissues. Western blotting analysis revealed a major band of approximately 32 kD in testis, liver, and brain. Immunofluorescence showed strong expression of AQP9 in the plasma membrane of testis Leydig cells. In liver, AQP9 expression was found to be sex-linked. Male rats had higher levels of AQP9 than female in terms of both protein and mRNA. Moreover, in female livers the expression of AQP9 was mostly confined to perivascular hepatocytes, whereas males showed a more homogeneous hepatocyte staining. No differences in AQP9 expression level related to the age or to protein content of the diet were found, indicating that differences in the liver may be gender-dependent. In the brain, AQP9 expression was found in tanycytes mainly localized in the areas lacking a blood-brain barrier (BBB), such as the circumventricular organs (CVOs) of the third ventricles, the subfornical organ, the hypothalamic regions, and the glial processes of the pineal gland. AQP9 expression in the osmosensitive region of the brain suggests a role in the mechanism of central osmoreception. All these findings show a unique tissue distribution of AQP9 compared to the other known aquaporins.
The most recently cloned water channel, AQP9, has recently been identified in rat liver (Tsukaguchi et al. 1998) and human leukocytes (Ishibashi et al. 1998). Functional studies performed in Xenopus oocytes revealed a broad selective neutral solute channel. AQP9-expressing oocytes manifested a pf similar to that for AQP1 inhibited by HgCl2 and phloretin. Unlike other aquaporins, AQP9 allows permeation of a variety of structurally unrelated solutes, such as carbamides, polyols, purine, and pyrimidines (Tsukaguchi et al. 1998, 1999). It has been speculated that this broad selectivity can have some clinical implications. In fact, AQP9 is also permeable to 5-fluorouracil, a chemotherapeutic agent used in cancer therapy (Tsukaguchi et al. 1998). Tissue localization studies performed by Northern analysis and in situ hybridization in rat have demonstrated the expression of mRNA for AQP9 in liver and testis (Tsukaguchi et al. 1998).
Compared to rat distribution, the human AQP9 mRNA is differently expressed. High AQP9 expression was found in leukocytes and much less in liver. Furthermore, no AQP9 mRNA was found in human testis (Ko et al. 1999; Tsukaguchi et al. 1999). Even though immunolocalization studies are needed to confirm the different tissue distribution of AQP9 protein, Northern blotting data suggest that AQP9 may have different functions in humans and rats.
In this study, as a first step toward understanding the functional role of AQP9, the tissue distribution and the membrane localization of AQP9 in rat tissues were determined by immunohistochemistry and the molecular identity of the AQP9 protein was established. Affinity-purified polyclonal antibodies revealed a unique cellular distribution of AQP9, providing insight into the role of this aquaporin in testis and liver. Moreover, the expression of AQP9 in the osmosensitive region of the rat brain suggests a role for this aquaporin in the mechanism of central osmoreception. While this article was in preparation, an immunolocalization study of AQP9 was reported by another group (Elkjaer et al. 2000). Although our immunolocalization results are in agreement with that study, some substantial differences and contrasting results were obtained, such as the molecular weight of AQP9 protein identified by Western blotting and the lobular expression of AQP9 in rat liver. Moreover, we performed a more detailed morphological study on AQP9 in brain.
Materials and Methods
Antibodies and Animals
Two different commercial affinity-purified rabbit polyclonal antibodies were used for this study. Antibody A was from Alpha Diagnostic International (San Antonio, TX) and antibody C was from Chemicon International (Temecula, CA). Both were produced against an 18-amino-acid synthetic pep-tide within the C-terminal domain of rat AQP9 predicted to be cytoplasmic.
Male and female rats 1 month and 6 months old fed with a standard diet containing 19% protein (w/w) were used for this study. To analyze the effect of the protein diet on liver expression of AQP9, 6-month-old rats were fed with a 22% (w/w) protein diet for 2 weeks before the experiment. Three to five animals for each experimental condition were used.
Immunocytochemistry
Immunofluorescence. Rat liver, testis, uterus, and ovaries were removed, washed in PBS, sliced, and fixed with PBS containing 4% paraformaldehyde (Nicchia et al. 2000). After an overnight incubation with PBS containing 30% sucrose, the tissues were embedded in OCT compound and frozen in liquid nitrogen. Cryostat 4–7-μm sections were incubated with affinity-purified rabbit AQP9 antibodies (5 μg/ml antibody A or 2 μg/ml antibody C) for 1 hr at room temperature (RT). After washings (three times for 5 min) with PBS, sections were incubated for 1 hr with CY3-coupled goat anti-rabbit antibody. Sections were observed using a Leica DMRXA photomicroscope (Leica; Soms, Germany) equipped for epifluorescence, and digital images were obtained with a cooled CCD camera (Princeton Instruments; Princeton, NJ).
Immunoperoxidase. Immunoperoxidase staining was performed as previously described (Nicchia et al. 2000; Frigeri et al. 2001a, b). Briefly, 5-μm sagittal sections of paraffin-embedded brains were collected on poly-lysine-coated slides and deparaffinized. After blocking, the sections were sequentially incubated with (a) primary rabbit affinity-purified anti-AQP9 antibody (5 μg/ml) diluted in TBS overnight at 4C, and (b) secondary antibody, goat anti-rabbit (Dakopatts; Hamburg, Germany) diluted 1:50 in TBS for 30 min at RT followed by the PAP complex, using commercial reagents (Dakopatts). Finally, the sections were treated with 0.06% 3,3′ diaminobenzidine and counterstained with Mayer's hematoxylin for 1 min. Control experiments performed using immunodepleted antibodies showed no staining.
Preparation of Liver, Brain, and Testis Membrane Vesicles and Western Blotting
Rat brain, liver, and testis were removed and homogenized in 0.25 M sucrose, 10 mM Tris-HCl, pH 7.5, containing 1 μg/ml leupeptin, 1 μg/ml pepstatin A, and 1 mM PMSF. After homogenization in a Potter apparatus and centrifugation at 1000 × g for 10 min, a low-speed pellet was prepared by centrifugation at 17,000 × g for 45 min at 4C. The pellet was resuspended at a final concentration of 10–12 μg/μl and overloaded on a discontinuous sucrose gradient consisting of layers of 50% sucrose (7 ml), 23% sucrose (10 ml), 17% sucrose (10 ml), and the low-speed pellet sample (10 ml). The gradient was centrifuged at 55,000 × g overnight using a SW28 rotor and three separated fractions were collected from the bottom and analyzed for Western blotting. F1 (pellet) contained cell debris; F2 collected at the 23–50% sucrose interface contained microsomal and ER vesicles and residual plasma membranes; F3 collected at the 17–23% sucrose interface contained an enriched plasma membrane fraction (Seiler and Fleischer 1982).
SDS-PAGE was performed as previously described (Frigeri et al. 1998) using 13% polyacrylamide gels and affinity-purified antibodies (2 μg/ml diluted antibody A or C) or whole rabbit AQP9 antiserum (1:5000 diluted). AQP9 protein was detected by NBT/BCIP substrates using an alkaline phosphatase-coupled secondary antibody. Revealed membranes were scanned using a UMAX SPEED IIC scanner and Adobe Photoshop software (San Jose, CA) and densitometric analysis was performed using Scion Image software as previously reported (Frigeri et al. 2001a, b). The statistical analysis was conducted by the Student's t-test for unpaired data.
Endoglycosidase Treatment
The fraction 3 of liver and the low-speed pellet of kidney were used for AQP9 and AQP1 deglycosylation experiments, respectively. One hundred μg of each sample was incubated for 10 min at 60C in 50 mM sodium phosphate buffer, pH 7, containing 0.2% SDS and 50 mM β-mercaptoethanol. The denatured samples were then incubated with 4 U of N-glycosidase F (Boehringer Mannheim; Mannheim, Germany) for 4 hr at 37C and finally analyzed by Western blotting.
RT-PCR Experiments
Total RNA was prepared from rat testis, liver, and brain using the TRIzol reagent (Gibco Life Technologies; Beerse, Belgium) and cDNAs were prepared using random primers as previously described (Nicchia et al. 2000). The cDNAs were used to amplify the full-length sequence (888 bp) using specific primers (sense 5′-ATGCCTTCTGAGAAGGACGG-3′; antisense 5′-CTACATGATGACACTGAGCT-3′) or a 599-bp internal fragment (sense 5′-CGAGTTCCTGGGCACCTTTA-3′ and antisense 5′-GCAGTGAAGAGCCTGGGACT-3′) of the AQP9 sequence (Tsukaguchi et al. 1998). To determine differences in the relative amount of AQP9 liver transcript, semiquantitative RT-PCR was performed using the procedure previously described (Frigeri et al. 2001a, b). Relative amounts of AQP9 mRNA were estimated by direct comparison between multiple samples after standardization with co-amplification of 18S rRNA (QuantumRNA kit; Ambion, Austin, TX).
Plasmid Construction and Transfection
The cDNA encoding for human AQP9 was amplified from human blood by RT-PCR experiments and directly ligated into a green fluorescence protein containing expression vector (pcDNA3.1/NT -GFP TOPO Cloning kits; Invitrogen, Carlsbad, CA) with the cytomegalovirus promoter and the gene for resistance to geneticin. Transfection was performed in CHO-K1 cells by use of lipofectin (Ma et al. 1993). Cells were grown in Ham-F12 nutrient mix supplemented with 10% fetal calf serum at 37C in 5% CO2. Twelve hours before transfection the cells were plated in 100-mm dishes. Twenty μg of lipofectin was diluted in 1 ml of serum-free medium and combined with 1 ml of medium containing 5 μg of the recombinant plasmid. The mixture was added to cells and incubated for 7 hr at 37C. The cells were grown for 2 days and then trypsinized and transferred to 10-mm dishes. A selection of cells containing transfected DNA was obtained with a medium containing geneticin (500 μg/ml; Life Technologies, Birmingham, AL) for 10–15 days. Resistant clones were isolated and transferred for expansion and analysis of the presence of the fusion protein. Immunofluorescence of transfected cells was performed as previously described (Nicchia et al. 2000).
Results
Western Blotting and RT-PCR
The first set of experiments was devoted to analysis of the presence of AQP9 protein in several rat tissues. To this purpose, membranes from rat liver were prepared as described in Materials and Methods by differential centrifugation and sucrose discontinuous gradient. Figure 1A shows a typical Western blot obtained using affinity-purified antibodies (antibody A). AQP9 was detected as a protein of approximately 32 kD. In some experiments, a second band of faint intensity appeared at 35 kD. A weak signal was obtained in a total membrane fraction (17,000 × g), whereas AQP9 was strongly enriched in fraction 2, and even more so in fraction 3, after sucrose gradient centrifugation. The specificity of the result was confirmed by using another anti-AQP9 antibody (C) or by using immunodepleted antibodies obtained using antibodies previously reacted with the immunizing peptide (Figure 1B). Confirmation of the antibody specificity was also obtained using transfected cells. CHO cells were stably transfected with an expression vector containing both GFP and the human AQP9 cDNA coding sequences. As shown in Figure 1C, all the cells uniformly expressed AQP9-GFP on the plasma membrane, confirming the correct targeting of the fusion protein. Moreover, when the two antibodies were used to stain the cells, only antibody C crossreacted with the human AQP9 protein (Figure 1D). These data indicate that the two antibodies recognize different epitopes within the rat AQP9 C-terminus sequence.
Expression of AQP9 in brain and testis was analyzed using the same membrane fractionation method (Figure 2A). The same pattern was detected in fraction 3 of the sucrose gradient, indicating that AQP9 is expressed in these two tissues.
RT-PCR experiments were then performed to determine the presence of the specific mRNA for AQP9 in those tissues in which AQP9 protein was found. Using specific rat primers, a fragment of the expected size (~900 bp) containing the full-length AQP9 cDNA was obtained in liver, brain, and testis (Figure 2B). The specificity of the PCR amplification product was verified by sequencing analysis (not shown).
AQP9 has a potential N-linked glycosylation site at Asn142. Therefore, experiments were performed to analyze whether AQP9 is glycosylated. AQP9-enriched membrane fraction F3 from liver was incubated with endoglycosidase, which specifically digests the glucose residues. AQP1 containing kidney membranes was used as a positive control. Figure 2C shows that the relative content and the electrophoretic profile of the two AQP9 immunopositive bands did not change after endoglycosidase treatment, indicating that the protein is not glycosylated.
When livers from male and female rats were separately analyzed, the expression of AQP9 was found to be quite different. Western blotting experiments revealed that the intensity of the AQP9 protein band was significantly stronger in males than in females (Figure 3B), indicating that males have higher liver AQP9 expression than females. To determine whether or not this different protein expression is related to a different gene activity, we analyzed the expression of AQP9 mRNA by competitive RT-PCR experiments. Semiquantitative analysis revealed reliably different levels of AQP9 transcript in male and female livers (Figure 3C).
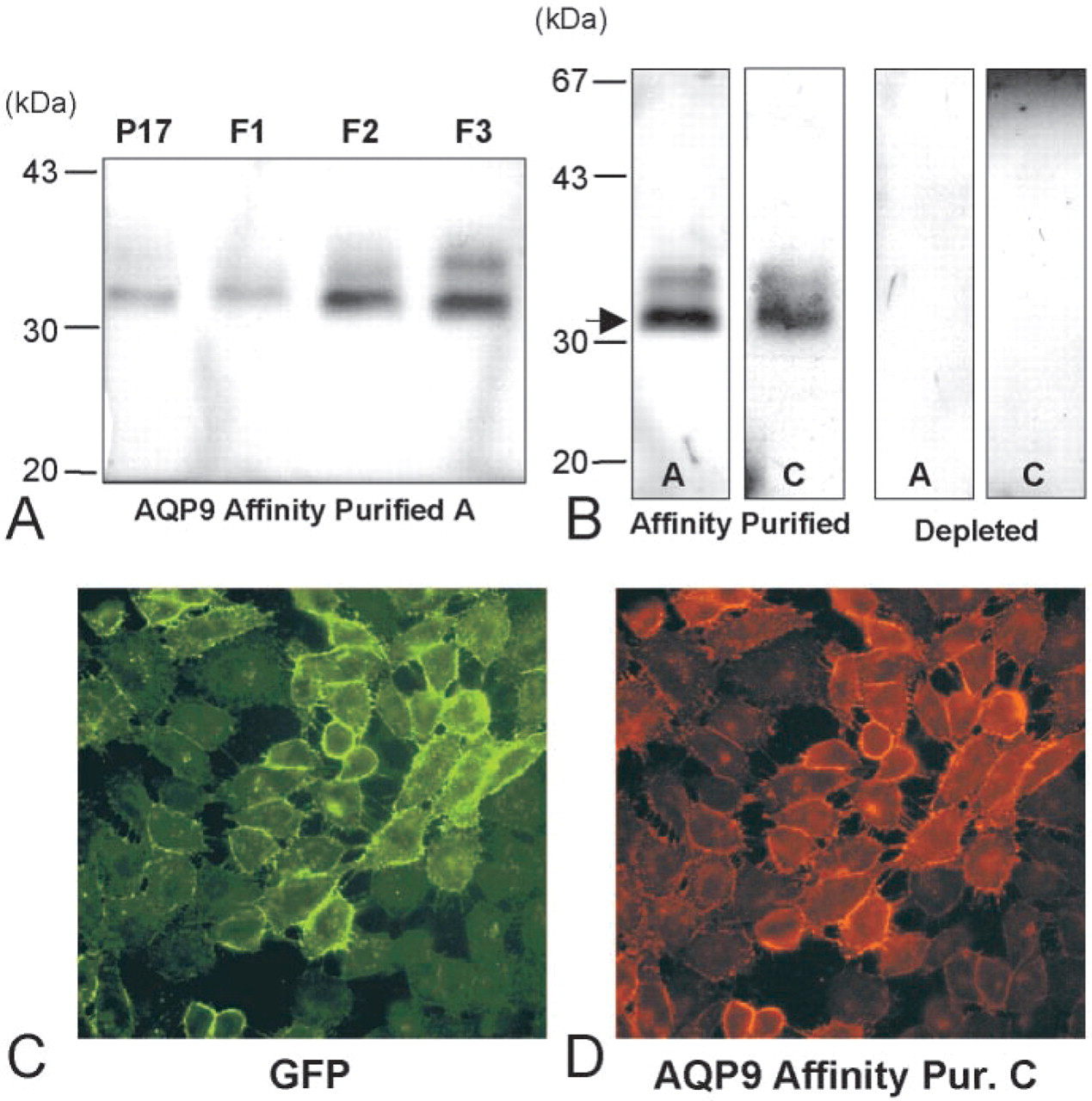
AQP9 antibody characterization by Western blotting and immunofluorescence on transfected CHO cells. (
To analyze whether AQP9 expression in liver was age-dependent or linked to the protein content of the diet, AQP9 protein expression was analyzed by Western blotting experiments. Densitometric analysis results (Figure 3D) showed that no differences were found with age or diet, whereas a significant difference (~20%) in AQP9 expression was found only between males and females.
Immunolocalization Studies
Indirect immunofluorescence staining was used to localize AQP9 in liver, testis, and brain. The two antibodies (A and C) gave identical results.
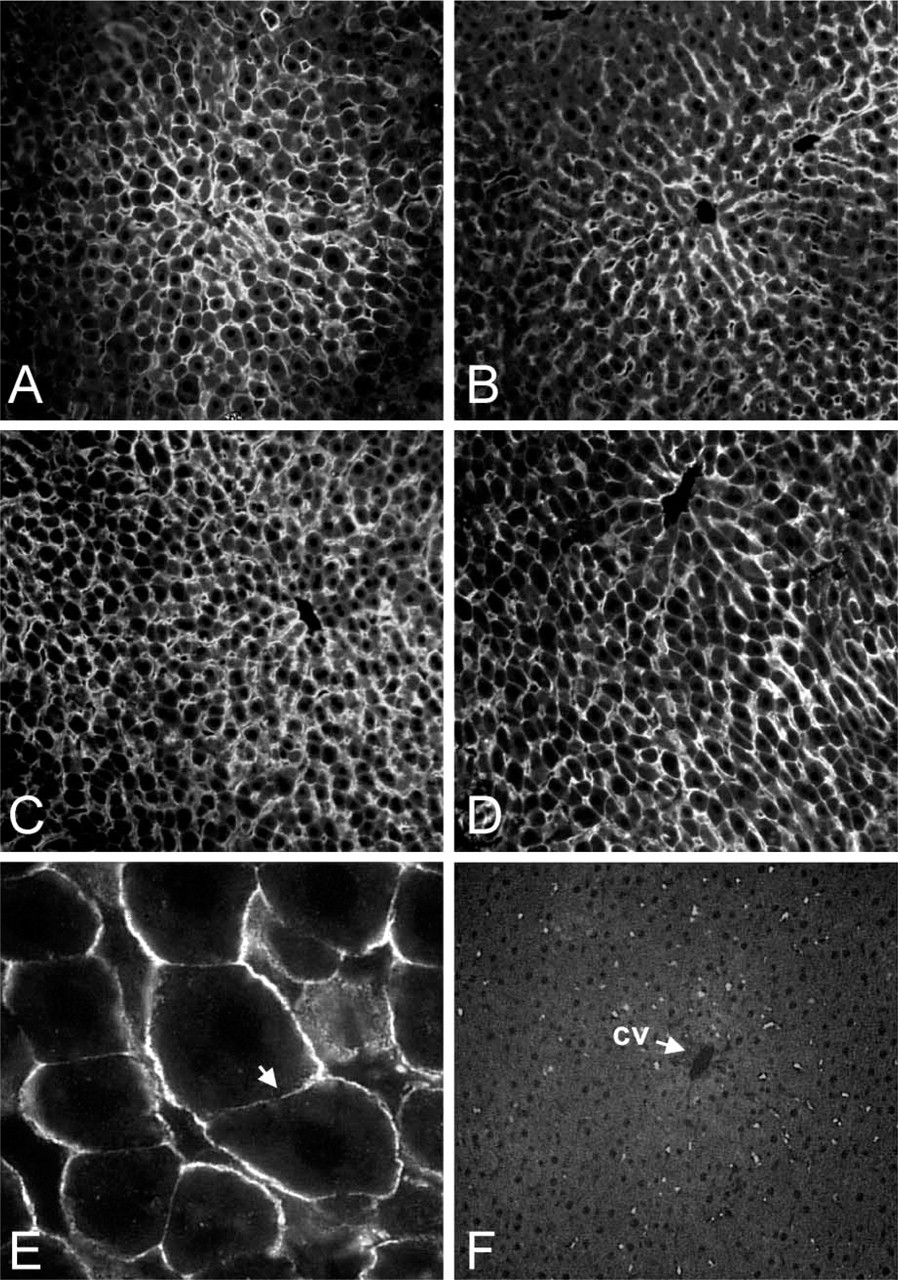
Liver AQP9 expression was analyzed to determine cell distribution and membrane localization and whether AQP9 expression/distribution was dependent on the age and/or the diet of the rats. Affinity-purified antibodies were used to stain cryostat rat liver sections (Figure 4). AQP9 was found only expressed in the hepatocytes. In 1-month (Figure 4A) and 6-month-old (Figure 4B) female rats the expression was strongest in those cells closest to the central vein. In male rats the expression of AQP9 was found to be more homogeneous and involved all the hepatocytes in both 1-month (Figure 4C) and 6-month (Figure 4D) rats. No differences were found in male and female livers of animals fed with a hyperprotein diet (not shown). At higher magnification, the staining was localized in the baso-lateral membrane of the cells, whereas the apical bile canalicular membrane was not labeled (Figure 4E). No other structures were specifically recognized. Control experiments performed after preabsorption of antibodies with the immunizing peptide gave no staining, confirming the specificity of the result (Figure 4F).
Immunofluorescence of rat testis is shown in Figure 5. At lower magnification, AQP9 antibodies recognized interstitial cells of seminiferous tubules (Figure 5A). These cells were histochemically identified as Leydig cells, both isolated or in groups close to blood and lymphatic vessels that surround the seminiferous tubules. No staining was found in the lumen of the seminiferous tubules or in the immature spermatocytes or Sertoli cells. At higher magnification (Figure 5B) the staining was confined to the plasma membrane of the cells. The specificity of the staining was demonstrated by a preabsorbed control (Figure 5B, inset). Immunofluorescence performed in female organs, such as uterus and ovaries, gave no specific staining for AQP9 expression (not shown).
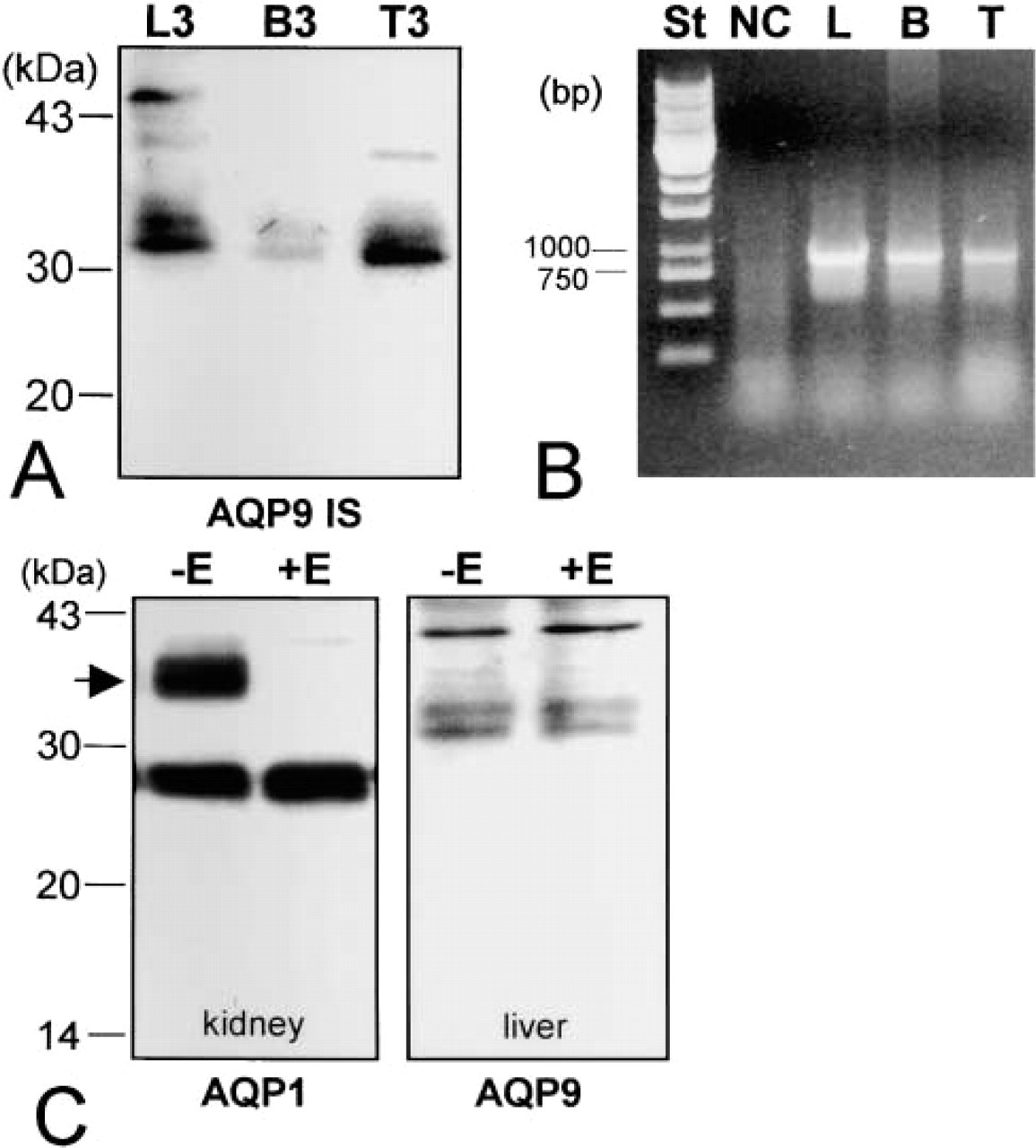
Analysis of AQP9 expression by Western blotting and RT-PCR. (
In brain, the localization of AQP9 was performed using immunoperoxidase staining. The immnocytochemical localization of AQP9 in the brain was performed in the forebrain, including the cerebrum, the diencephalon, and the circumventricular organs (CVOs), and in the pineal gland. Of all brain areas analyzed, a positive signal was found in the CVOs, including the subfornical organ and the hypothalamic regions (Figure 6). Staining was found in a subpopulation of GFAP-positive ependymal cells lacking cilia, and recognized as specialized glial cells (tanycytes) lining the floor of the third ventricle (Figure 6A). Expression of AQP9 was found in the tanycyte bodies and tail process that extended from the ventricular wall into the neuropil and formed end-feet at the vessel surfaces (Figure 6B) in the deep layer. AQP9 immunolabeling was also found in the tanycyte processes ending in the vicinity of the neuronal cells of hypothalmic nuclei (Figure 6C). Strong AQP9 expression was found in the glial limiting membrane of the pineal gland and around the ingrowing vessels (Figure 6D). Fine staining was detectable in the neuroglial processes usually scattered in a random pattern in the pineal stroma or terminating in vessel walls (Figure 6E) or in bulb-like endings on pinealocytes (Figure 6F). No AQP9 labeling was found in the pinealocytes (Figures 5E and 5F) or in the hypothalamic neuronal cells. The specificity of the staining was demonstrated by a preabsorbed control (Figure 6G).
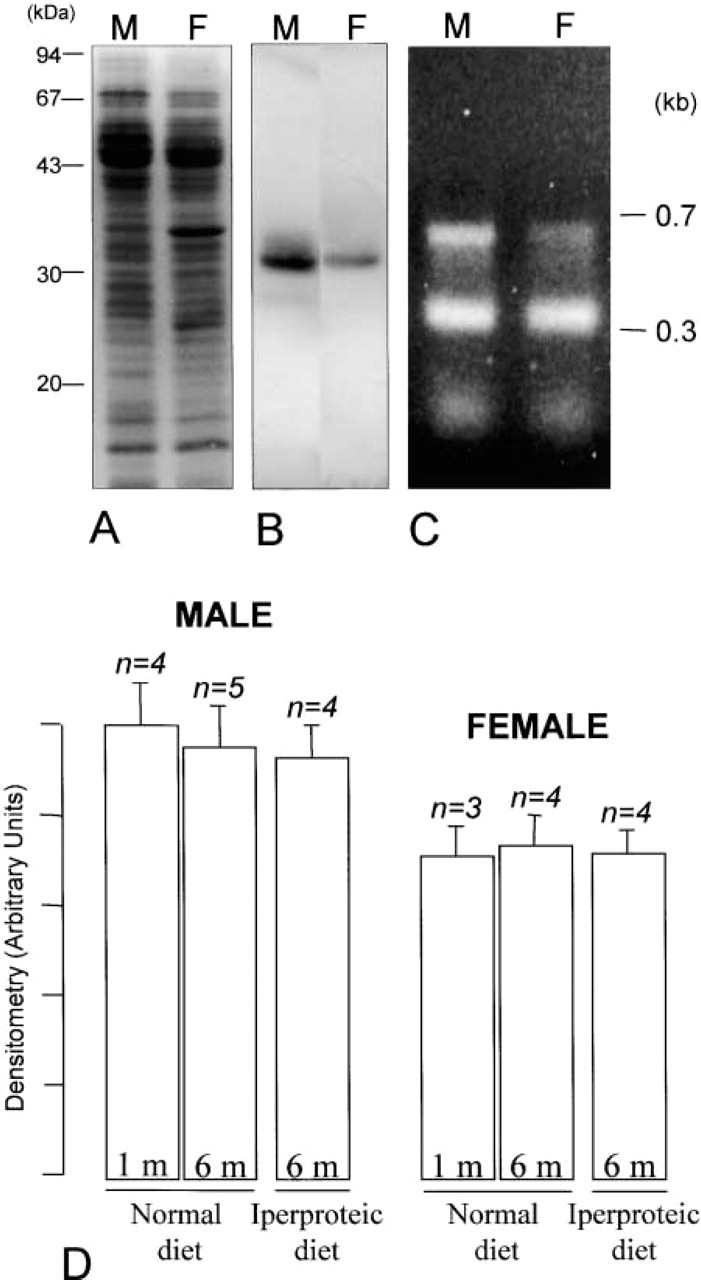
Analysis of AQP9 expression in male and female liver. (
No differences were found between male and female brains (not shown).
Discussion
In this study we examined the tissue distribution and the cellular and subcellular localization of the AQP9 water channel in rat using affinity-purified polyclonal antibodies raised in rabbit against a C-terminal synthetic peptide (see Table 1). Western blotting results show that a major non-glycosylated band was observed, with electrophoretic mobility of approximately 32 kD. The specificity of our results was confirmed by using two different antibodies that, as demonstrated using transfected cells, recognize different AQP9 epitopes, by immunodepletion, and also by immunoprecipitation experiments (not shown). Contrasting with our finding is the molecular weight reported for AQP9 by the group that recently published a similar immunolocalization study (Elkjaer et al. 2000). In that study, a 28-kD predominant band was found in all the analyzed tissues. We have no explanation for this difference.
AQP9 was first cloned from rat and in situ hybridization studies indicated strong expression in hepatocytes (Tsukaguchi et al. 1998). Our results demonstrate that AQP9 expression is confined to the rat hepatocyte basolateral membrane, the plasma membrane facing the sinusoids. The small apical canalicular membrane appeared not to be labeled, even though only electron microscopy experiments would confirm this deduction. A new and interesting finding of the present study is the different expression observed between male and female rats. In liver from female rats the immunofluorescence signal was progressively more intense in hepatocytes close to the central vein (peri-venous hepatocytes), whereas in the liver from male rats expression of AQP9 was found in both peri-venous and periportal hepatocytes. Moreover, our results demonstrate that AQP9 protein content was significantly greater in males and was pretranslationally controlled. It is well established that many hepatic functions are expressed in a sexually dimorphic fashion. A number of hepatic enzymes (Simon et al. 1996), plasma membrane receptors (Mode 1993), and transcription factors (Lahuna et al. 1997) have been shown to be sexually dimorphic. To check for possible factors that would influence AQP9 expression in liver, we analyzed the effect of age and the dietary protein content. One-month- and 6-month-old rats were chosen because they represent two different periods of sexual maturity. The results indicate that neither the sexual maturity nor the dietary protein content appeared to influence AQP9 expression or distribution. Our results suggest that AQP9 is expressed differentially in hepatocytes within the liver plate and that this expression could be gender-dependent. Regarding the possible function of AQP9 in liver, a direct role of this channel in making the water component of bile is not likely because AQP9 appears not to be expressed in the apical membrane of hepatocytes (Yano et al. 1996; Garcia et al. 2001). However, the expression of AQP9 in the basolateral membrane could represent the counterpart expression of an unknown apical water channel necessary for bile formation. Future studies are necessary to clarify whether either or both of these two possibilities are correct.
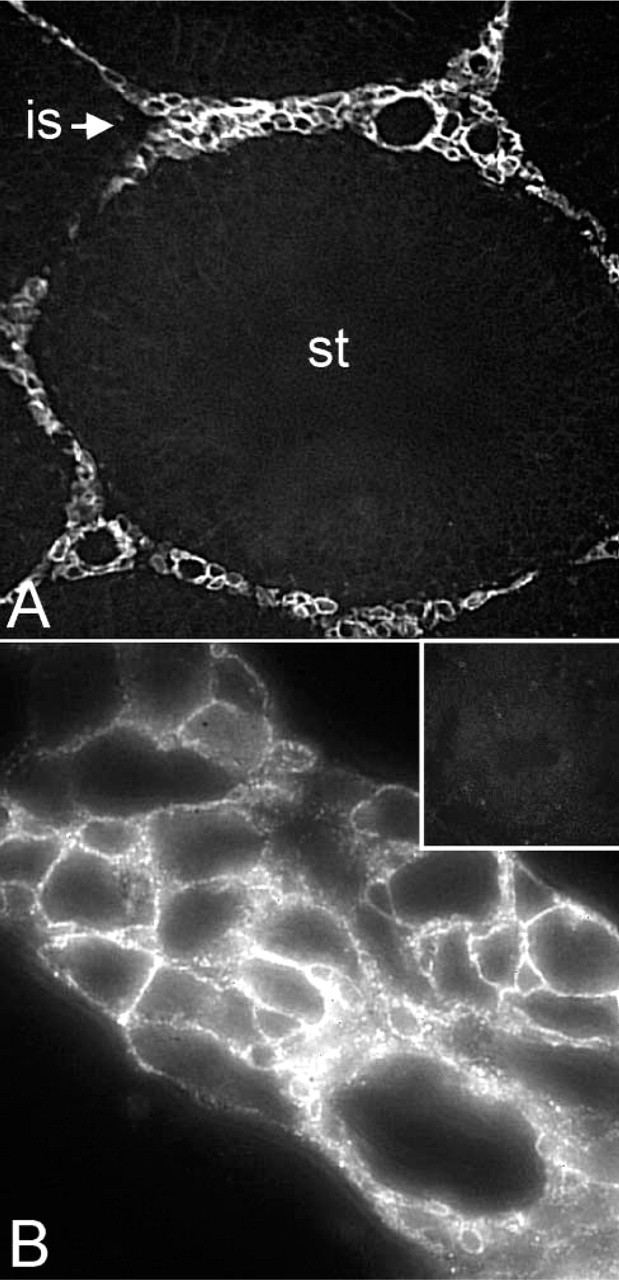
Immunofluorescence detection of AQP9 in testis. (
Immunofluorescence localization of AQP9 in rat liver using purified polyclonal antibodies. (
Expression of AQP9 in rat tissue a
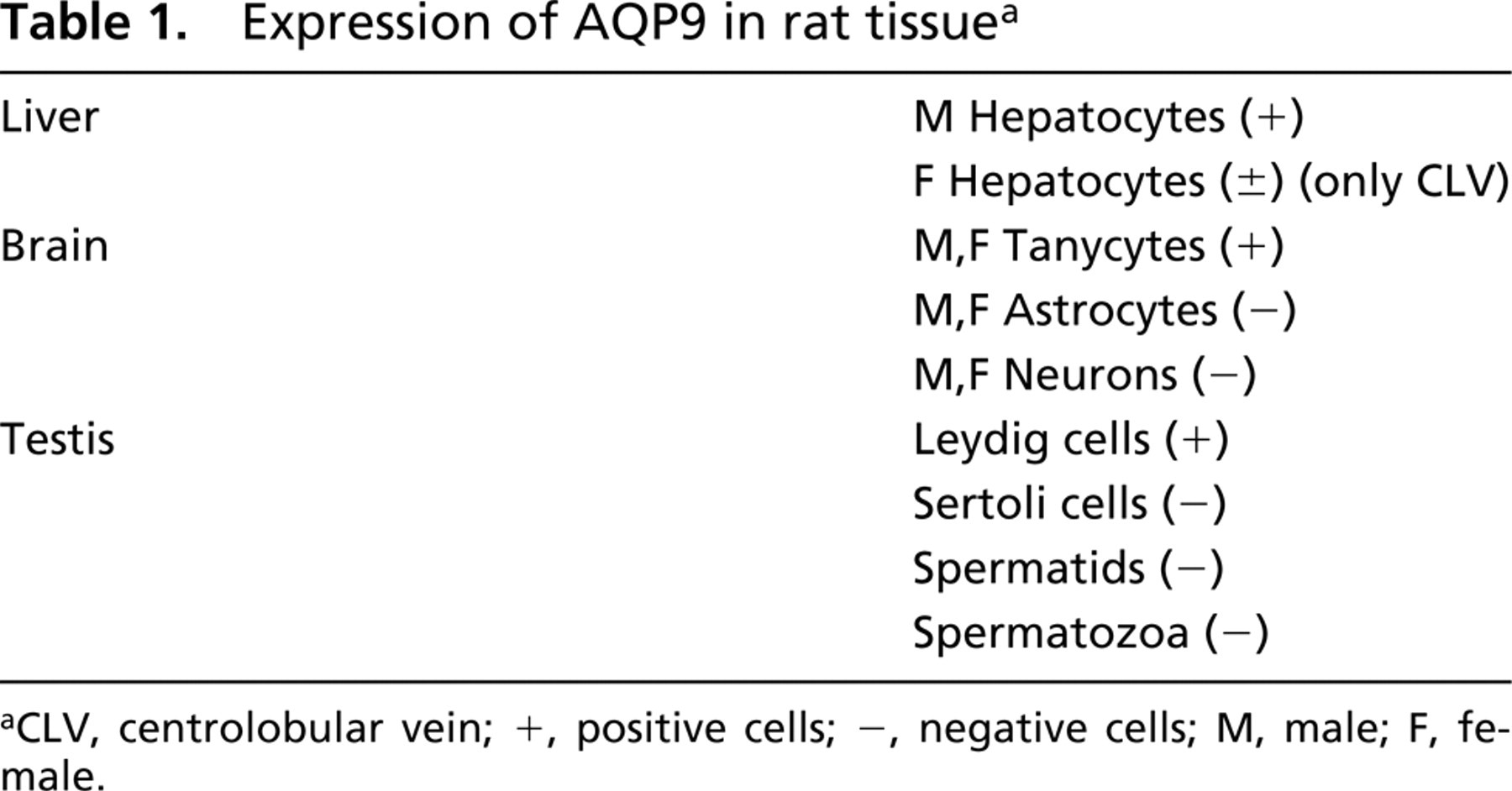
CLV, centrolobular vein; +, positive cells; -, negative cells; M, male; F, female.
AQP9 is also highly permeable to urea in addition to having water transport properties. The liver is a major site of production and elimination of urea suggesting that AQP9 in liver may function as a urea channel (Saez 1994; Tsukaguchi et al. 1998,1999). If this is the case, the membrane localization of AQP9 suggests a role in hepatocyte urea elimination as well as an exit route for other solutes, such as purines and pyrimidines. It is possible that the hepatocyte expression of AQP9 depends on the metabolic state of the liver and thus on the activity of the cells (Tsukaguchi et al. 1998). To clarify the role of AQP9 liver expression related to protein methabolism, we analyzed the effect of an hyperproteic diet. Although the diet effect needs to be more extensively analyzed, we found no evident effect of dietary protein increase.
In testis, AQP9 was found to be expressed in the plasma membrane of interstitial Leydig cells. These endocrine cells secrete the male sex hormone testosterone under the control of endocrine, paracrine, and auto-crine factors (Saez 1994; Dufau 1988). Moreover, Leydig cells participate in the production/regulation of testicular interstitial fluid. The presence of AQP9 channels in these cells may be related to the production of testicular fluid. Leydig cells have the property of changing their shape and volume under hormonal stimulation by LH, which has a positive effect on steroid secretion (Saez 1994). This suggests that AQP9 is involved in the volume changes that occur during hormonal stimulation and therefore in testosterone secretion. Further studies are required to elucidate this aspect.
In the brain, we found an AQP9 expression mainly localized in the areas lacking a blood-brain barrier, such as the CVOs of the third ventricles, the subfornical organ, and the hypothalamic regions. It is important to note that CVOs share common structural features, such as a weakened BB barrier and specialized ependymal cells, which are important for CNS homeostasis and for regulating osmoreceptor functions. Therefore, the presence of AQP9 staining in the ventricle ependymal layer and in the tanycytic glial cells that form a foot-like expansion close to the blood vessels of the subfornical organs suggests that this water channel may be involved in the production/reabsorption of cerebrospinal fluid and in the control of the specific neuronal activity of the CNS. A close relation between the absence of BB barrier and AQP9 expression is further demonstrated by the presence of this water channel in the perivascular glial processes of the pineal gland, whose topographical relationship to the cavity of the third ventricle has been demonstrated to play an important role in the CSF (Hewing 1980). Moreover, we found AQP9 expression in neuroglial processes close to neurons of the hypothalamus and to pinealocytes. This AQP9 expression close to neuroendocrine cells, not previously reported (Elkjaer et al. 2000), is also interesting. Staining of the neuroglial processes in these permeable and secretory areas of the brain suggests a close relation between this water channel and the release of hypothalamic and pineal hormones. Our results therefore lead to an interesting hypothesis that the AQP9 water channel, present on the glial processes close to highly permeable vessels and neurosecretory cells, detects changes in plasma osmolality and interacts as a transducer with neuronal and secretory cells.
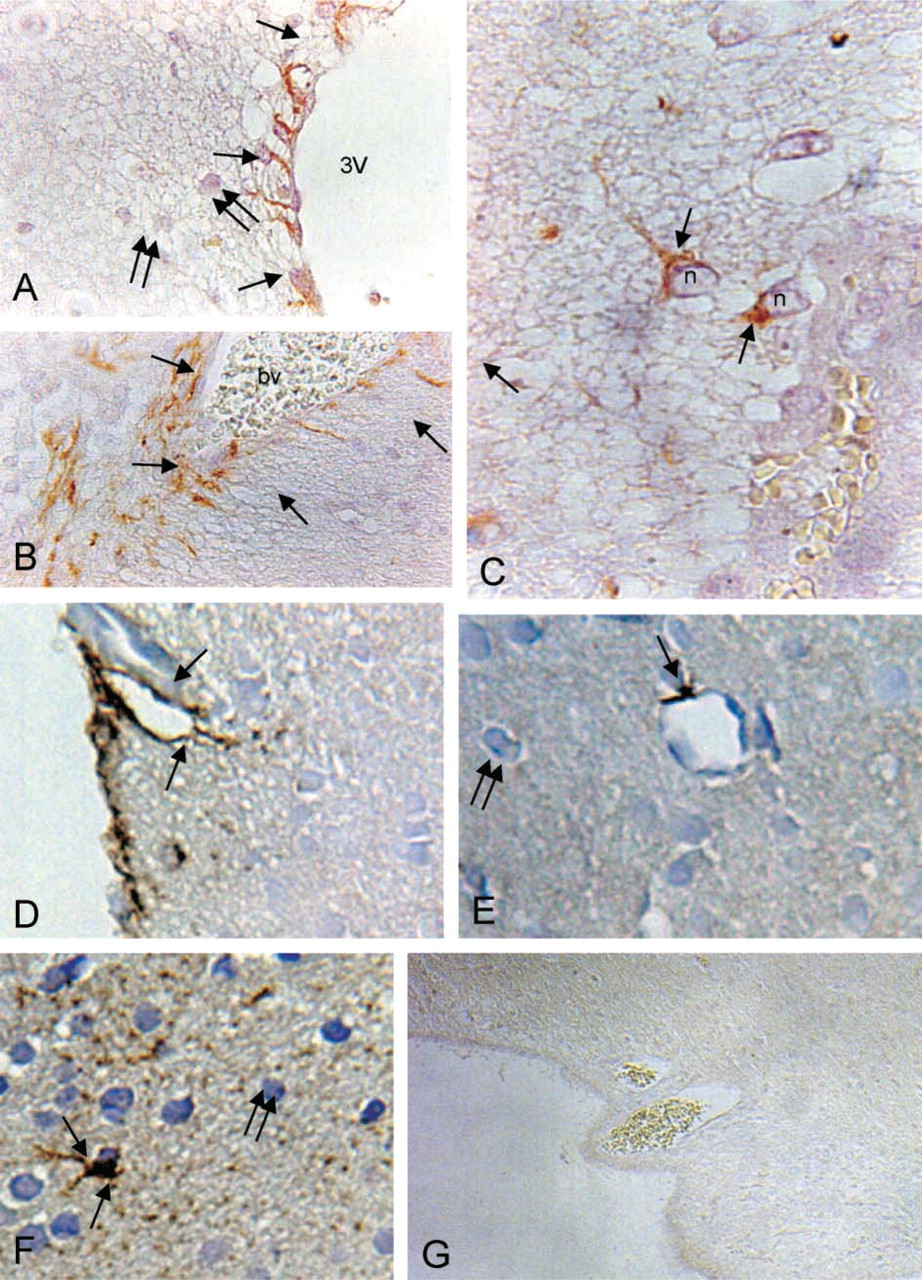
Immunoperoxidase localization of AQP9 in rat brain. (
Footnotes
Acknowledgments
Support by Telethon-Italy (grant no. 983).
We thank Raffaello Peragine, Alessandra Colella, and Dr Luca Liuzzi for excellent technical assistance.
