Abstract
The expression of endothelin-1 (ET-1), which has been proposed to have a potential autocrine/paracrine role, varies during the menstrual cycle, and therefore, ET-1 may be involved in the cyclic change of the human endometrium. However, neither the synthesis nor the degradation of ET-1 in the endometrium has been determined in detail. We investigated endothelin-converting enzyme-1 (ECE-1), which converts big-ET-1 to active ET-1, and neutral endopeptidase (NEP), which cleaves and inactivates ET-1 in human endometrium in vivo and in vitro. Western blot analysis demonstrated that the change in the expression of ECE-1 during the menstrual cycle differed from that of NEP in the endometrium. ECE-1 was expressed by endometrial epithelial cells, whereas NEP was predominantly expressed by stromal cells in vivo and in vitro. In conclusion, our results suggest that spacio-temporal expression of two endopeptidases, ECE-1 and NEP, involved in the synthesis and degradation of ET-1, might regulate ET-1 action in human endometrium.
Keywords
T
Endothelin-1 (ET-1), identified as a long-lasting vasoconstrictor, was originally reported to possess a potential paracrine role in the regulation of uterine blood flow (Economos et al. 1992; Salamonsen et al. 1992; Marsh et al. 1996). We have recently reported that ET-1 has a paracrine effect of Akt phosphorylation and cell proliferation on endometrial stromal cells (ESC) (Iwase et al. 2006). However, the metabolism of ET-1 in the human endometrium has not been entirely defined. Although it is known that neutral endopeptidase (NEP; EC 3.4.24.11), a membrane-bound endopeptidase (Kenny 1993; Papandreou et al. 1998; D'Orleans-Juste et al. 2003), degrades ET-1, the synthesis of ET-1 is a more complicated process. Prepro-ET-1, a 212-aminoacid peptide, is the first product of the ET-1 gene. This precursor is transformed to pro-ET-1 by removal of a short sequence by a signal peptidase, then to big-ET-1 via the activity of a maturing enzyme of the subtilisin family, namely furin (Turner and Murphy 1996). The conversion of big-ET-1 to ET-1 occurs via a unique processing event catalyzed by endothelin-converting enzyme (ECE), which has been reported to be a zinc-dependent metalloendopeptidase homologous with NEP (Turner and Murphy 1996; Gomazkov 1998). More recently, we demonstrated that cell surface aminopeptidases play important roles in regulating the local function of bioactive peptides in human endometrium (Ando et al. 2002; Toda et al. 2002). These results led us to hypothesize that two homologous cell surface peptidases, ECE-1 and NEP, which have different functions in the metabolism of ET-1, regulate ET-1 action in human endometrium. In the present study, we evaluated the expression and localization of ECE-1 and NEP in human endometrium during different phases of the menstrual cycle, and in primary cultured endometrial epithelial and stromal cells.
Materials and Methods
Tissue Collection
We retrieved endometrial biopsy specimens from the pathology files at Nagoya University Hospital, Nagoya, Japan as described previously (Ando et al. 2002). The biopsy specimens of 39 patients, aged 34-43 years, were available for examination (14 proliferative phase, 8 early secretory phase, 8 mid-secretory phase, 9 late secretory phase). Endometrial dating was strictly defined histologically in accordance with the endometrial dating criteria (Noyes and Haman 1953; Noyes et al. 1975) by an expert in this field (T. N.). The use of pathology specimens for this research was approved by the institutional review board. These samples, which had been fixed in 10% formalin and embedded in paraffin, were used for immunohistochemistry. For Western analysis, fresh endometrial tissues were collected by curettage from women aged 34-46 years during hysterectomy at Nagoya University Hospital as described previously (Ando et al. 2002). The fresh endometrial tissue samples were allocated to one of six groups: early proliferative phase (n=3); mid-proliferative phase (n=5); late proliferative phase (n=6); early secretory phase (n=7); mid-secretory phase (n=6); and late secretory phase (n=8). Samples were also collected from patients aged 34-44 years who had received an oral administration of estrogen (n=3) [one Premarin tablet daily (0.625 mg conjugated estrogens made from pregnant mare's urine), Wyeth Lederle Japan; Tokyo, Japan] or of an estrogen-progestin combination (n=4) [one Dolton tablet daily (500 μg norgestrel and 50 mg ethinyl estradiol), Nihon Schering; Osaka, Japan] for 21-28 days before hysterectomy. Informed consent was obtained from each patient before the study. Curetted samples were washed with cold PBS, and then stored at −80C until used for protein extraction.
Western Blot Analysis
The frozen tissue samples were homogenized using a motordriven Teflon pestle for 10 min on ice in PBS extraction buffer containing 1% Triton X-100 and protease inhibitors (1 mM phenylmethylsulfonyl fluoride, 1 μg/ml aprotinin, and 10 μg/ml leupeptin). Tissue extracts were obtained as supernatants after centrifugation at 15,000 × g for 30 min at 4C, and stored at −80C. Protein concentrations were determined using a protein assay kit (Bio-Rad Laboratories; Hercules, CA). The protein extract (10 μg) was separated by 10% SDS-PAGE, and transferred to a nitrocellulose membrane. After blocking in PBS containing 5% skim milk for 1 hr, membranes were immunoblotted with mouse anti-NEP Ab (1:100) (NCL, Novocastra Laboratories Ltd.; Newcastle, UK) or rabbit anti-ECE-1 Ab (1:200) (Zymed Laboratories Inc.; San Francisco, CA). Immunoreactivity for ECE-1 was confirmed using another anti-ECE-1 antibody (R and D Systems, Inc.; Minneapolis, MN). Immunoreactive proteins were stained using the enhanced chemiluminescence system (Amersham Biosciences Corp.; Piscataway, NJ). Relative band density normalized by β-actin was determined from light scans of the resulting films using densitometric analysis software.
Immunohistochemistry
Immunohistochemistry was performed as described previously (Ando et al. 2002). Briefly, formalin-fixed, paraffin embedded tissue sections were cut, deparaffinized, and treated at 90C in a microwave oven for heat-induced epitope retrieval. Immunohistochemical staining was carried out based on the labeled streptavidin-biotin method after incubation with 10% normal goat serum in PBS for 10 min. Staining procedures were done automatically using a Ventana BenchMark IHC staining system (Ventana Medical Systems; Tucson, AZ) according to the manufacturer's instructions. The primary antibody against ECE-1 or NEP was diluted 1:100 in PBS. In negative control experiments, the primary antibody was replaced with rabbit IgG. The slides were counterstained with hematoxylin prior to mounting. Staining for ECE-1 and NEP was carried out repeatedly for each sample. Stained sections were observed under an Olympus BH2 microscope (Tokyo, Japan) and photographed using a charge-coupled device color camera (CS600; Olympus).
Primary Culture of Endometrial Epithelial Cells and ESC and in Vitro Decidualization of ESC
Endometrial epithelial cells (EEC) and ESC were separated as described previously (Arnold et al. 2001; Ando et al. 2002; Kobayashi et al. 2004) based on a modification of the work of Satyaswaroop et al. (1979). In brief, endometrial tissue in the late proliferative phase was minced into small pieces (∼1 mm3), and these pieces were filtered through a cell strainer consisting of 100-μm pore size nylon mesh (Becton Dickinson and Co.; Franklin Lakes, NJ) to remove blood cells. Then the minced tissue was incubated with stirring at 37C for 20 min in PBS, 0.5% collagenase (Wako; Osaka, Japan), and 0.1 mg/ml DNase (Sigma; St. Louis, MO). The tissue digest was vigorously pipetted and passed over a cell strainer consisting of 70-μm pore size nylon mesh (Becton Dickinson and Co.). The epithelial cells were retained in nylon mesh sieves, and the stromal cells passed through to the receptacle below. The epithelial cells were rinsed and back-flushed out of the sieves onto a 60-mm sterile dish. The stromal cells collected from the lower receptacle were suspended and plated onto a 60-mm sterile dish. The EEC and ESC were cultured in RPMI 1640 medium (Sigma) containing 10% fetal calf serum (Invitrogen; Carlsbad, CA), 100 IU/ml penicillin, and 100 μg/ml streptomycin. The purity of EEC and ESC was assessed by morphological determination using light microscopy. Each cell population was routinely >98% pure as assessed by phase microscopy. The ESC were cultured with progesterone (10-6 M; Sigma), 17β-estradiol (10-8 M; Sigma), and (Bu)2cAMP (1 mM; Sigma) for 10-12 days at 37C in a humidified atmosphere of 5% CO2 in air to induce in vitro decidualization. In vitro decidualization was assessed by evaluating morphological changes and assaying the prolactin concentration in the medium.
Western Blotting for ECE-1 and NEP of EEC and ESC
Primary cultured EEC and ESC with or without decidualization were lysed in radioimmunoprecipitation buffer (10 mM Tris-HCl, pH 7.4, 150 mM NaCl, 1% Nonidet P-40, 5 mM EDTA, 1% sodium deoxycholate, 0.1% SDS, 1.2% aprotinin, 5 μM leupeptine, 4 μM antipain, 1 mM phenylmethylsulfonylfluoride, and 0.1 mM Na3VO4). Cell lysates were clarified by centrifugation at 13,000 × g at 4C for 15 min, diluted in 2× sample buffer (125 mM Tris-HCl, pH 6.8, 4% SDS, 10% glycerol, 0.2% bromphenolblue, and 4% 2-mercaptoethanol), resolved by 10% SDS-PAGE, and immunoblotted with 1:5,000 anti-NEP Ab, anti-ECE-1 Ab, or anti-β-actin Ab (Santa Cruz Biotechnology, Inc.; Santa Cruz, CA) as described above.
RNA Preparation and RT-PCR
Total RNA was isolated from EEC and ESC in 100-mm dishes using TRIzol reagent (Invitrogen; Carlsbad, CA) following the manufacturer's protocol. An RT reaction with 1 μg total RNA was carried out with a Gene Amp RNA PCR kit (Perkin-Elmer Corp.; Norwalk, CT). Thereafter, 1-ul aliquots of the RT reaction products underwent PCR. The following sets of oligonucleotide primers were used: ECE-1, 5′-ACT TTG GTG GCA TAG GTG TCG-3′ (sense) and 5′-TGC AAA GCC CAG GAA GAA GAG-3′ (antisense), which correspond to 1790-1810 and 2113-2133 of ECE-1 cDNA, respectively (Schmidt et al. 1994; Shimada et al. 1995); NEP, 5′-GTG CCC AGC AGT CCA ACT CAT TGA AC-3′ (sense) and 5′-CCC CAT TTC TGT GGT GTT GGC AAG TC-3′ (antisense), which correspond to 1697-1722 and 2285-2310 of NEP cDNA, respectively (Malfroy et al. 1988; Shipp et al. 1988; Liu 2000); glyceraldehyde-3-phosphate dehydrogenase (GAPDH), 5′-GGG GAG CCA AAA GGG TCA TCA TCT-3′ (sense) and 5′-GAG GGG CCA TCC ACA GTC TTC T-3′ (antisense), which correspond to 399-424 and 614-635 of GAPDH cDNA, respectively (Tokunaga et al. 1987). Amplification was performed using Taq polymerase (Perkin-Elmer) over 35 cycles for ECE-1 and NEP, and 25 cycles for GAPDH. Each cycle consisted of denaturation at 94C for 1 min, annealing at 54C for 1 min, and extension at 72C for 1 min.
Results
ECE-1 and NEP Expression in Human Endometrial Tissue
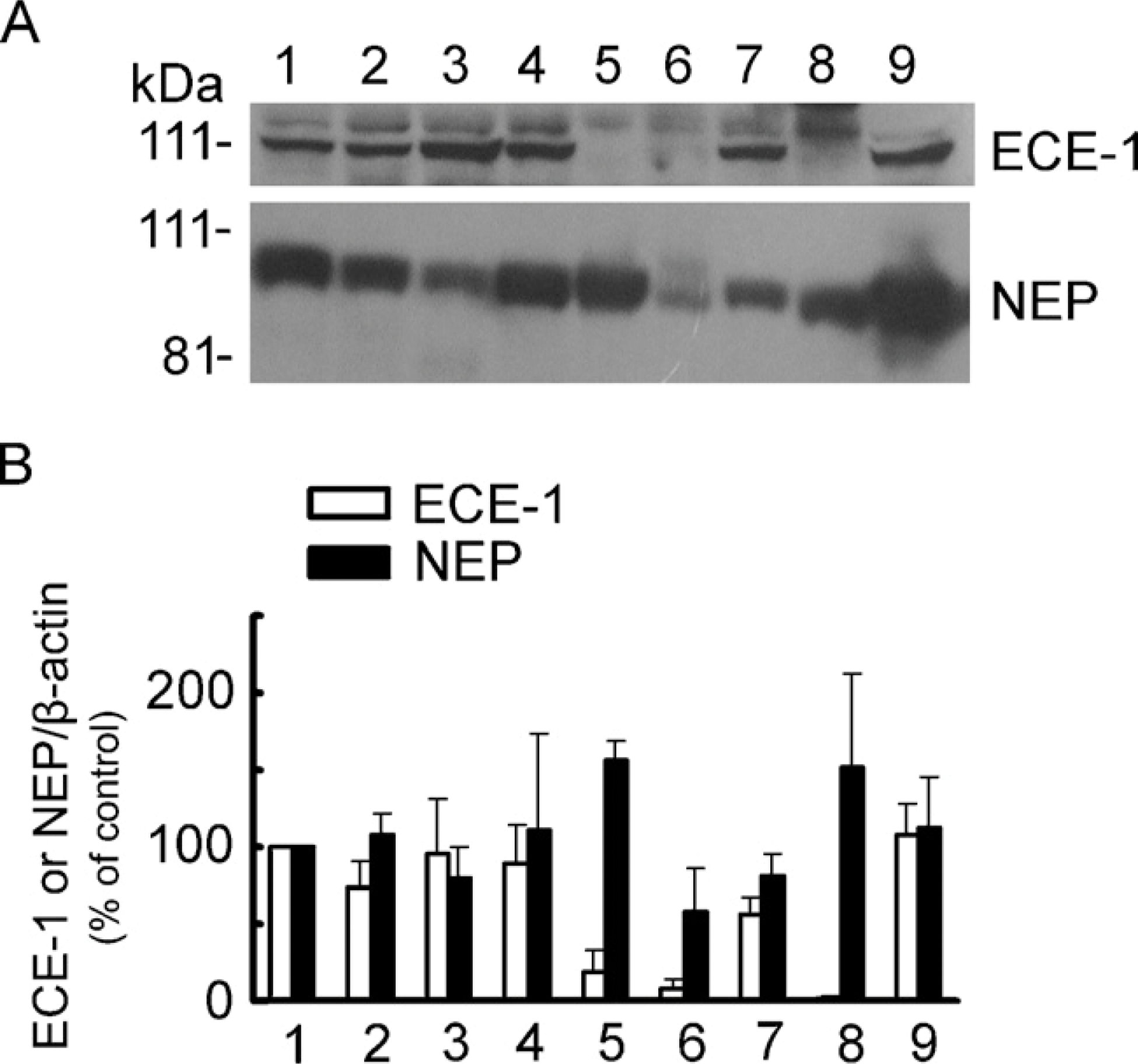
First, to confirm the expression of ECE-1 and NEP in the human endometrium in vivo, Western blot analysis and immunohistochemistry were performed. Both ECE-1 and NEP were detected in the endometrial tissue samples using Western blotting. Immunoreactive bands of ECE-1 were conspicuous in the samples in the mid-and late proliferative phases, and the early and mid-secretory phases, and also in the specimens from patients treated with estrogen tablets. However, ECE-1 was less abundant in the samples from the late secretory phase, progestin-exposed endometrium, and decidua than in the above samples (Figure 1A, upper panel). Lower bands around 111 kDa represented most of ECE-1, which were confirmed by another anti-ECE-1 antibody (data not shown). Upper bands, which showed relatively weak immunoreactivity and slightly different expression profiles compared with lower bands, might correspond to different glycosylation forms. On the other hand, immunoreactive bands of NEP were conspicuous in the samples from the proliferative and secretory phases, especially in the mid- and late secretory phase endometrium. In addition, the level of NEP was higher in the samples of cases treated with estrogen/progestin for 2–3 weeks compared with those treated with estrogen only (Figure 1A, lower panel). Densitometric analysis of relative band intensity revealed that exposure to progesterone tends to suppress ECE-1 expression but upregulate NEP expression (Figure 1B).
(
Immunohistochemistry revealed the contrast in localization of ECE-1 and NEP. ECE-1 was localized in the cytoplasm and the apical membrane in the luminal and glandular epithelium during the proliferative phase. In the secretory phase, ECE-1 was localized mainly to the apical membrane of the glandular epithelium. In contrast, NEP was mainly expressed by stromal cells and was observed in the entire stromal region throughout the menstrual cycle (Figure 2). The immunohistochemistry results shown here are representative of most samples. Both ECE-1 and NEP exhibited distinct distribution and variation in staining intensity, depending on the cell type as well as menstrual cycle phase (Table 1). These results indicate that ECE-1 and NEP, which show different distribution in the human endometrium, are regulated differently by ovarian sex steroid hormones.
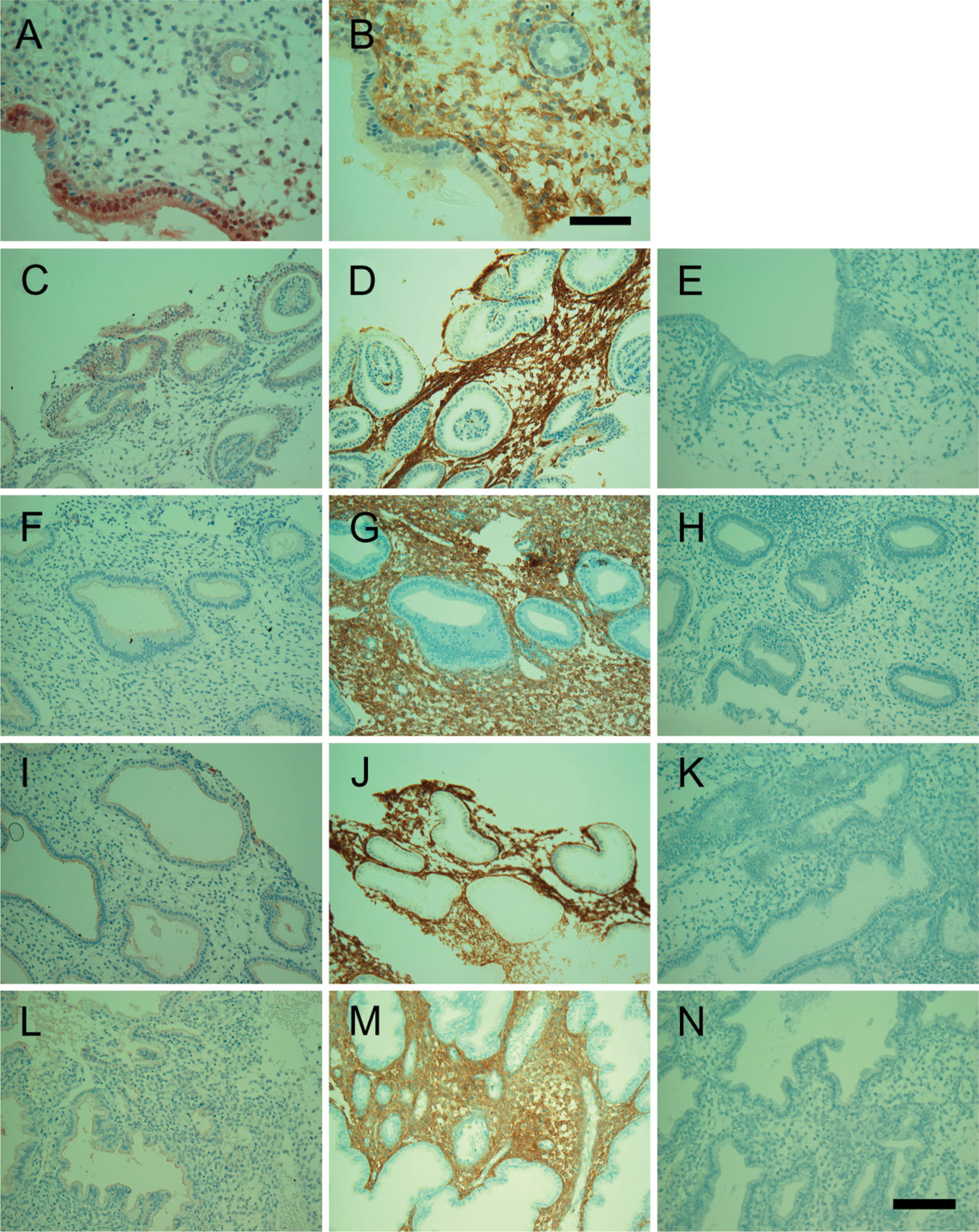
Immunohistochemical localization of ECE-1 and NEP in the human endometrium in the proliferative phase (
Immunostaining of endometrial structures with specific antibodies for ECE-1 and NEP

Staining intensity is expressed as negative (−), occasional or weak (+/−), moderate (+), or strong (++). ECE-1, endothelin-converting enzyme-1; NEP, neutral endopeptidase.
In Vitro Expression of ECE-1 and NEP in Primary Cultured EEC and ESC
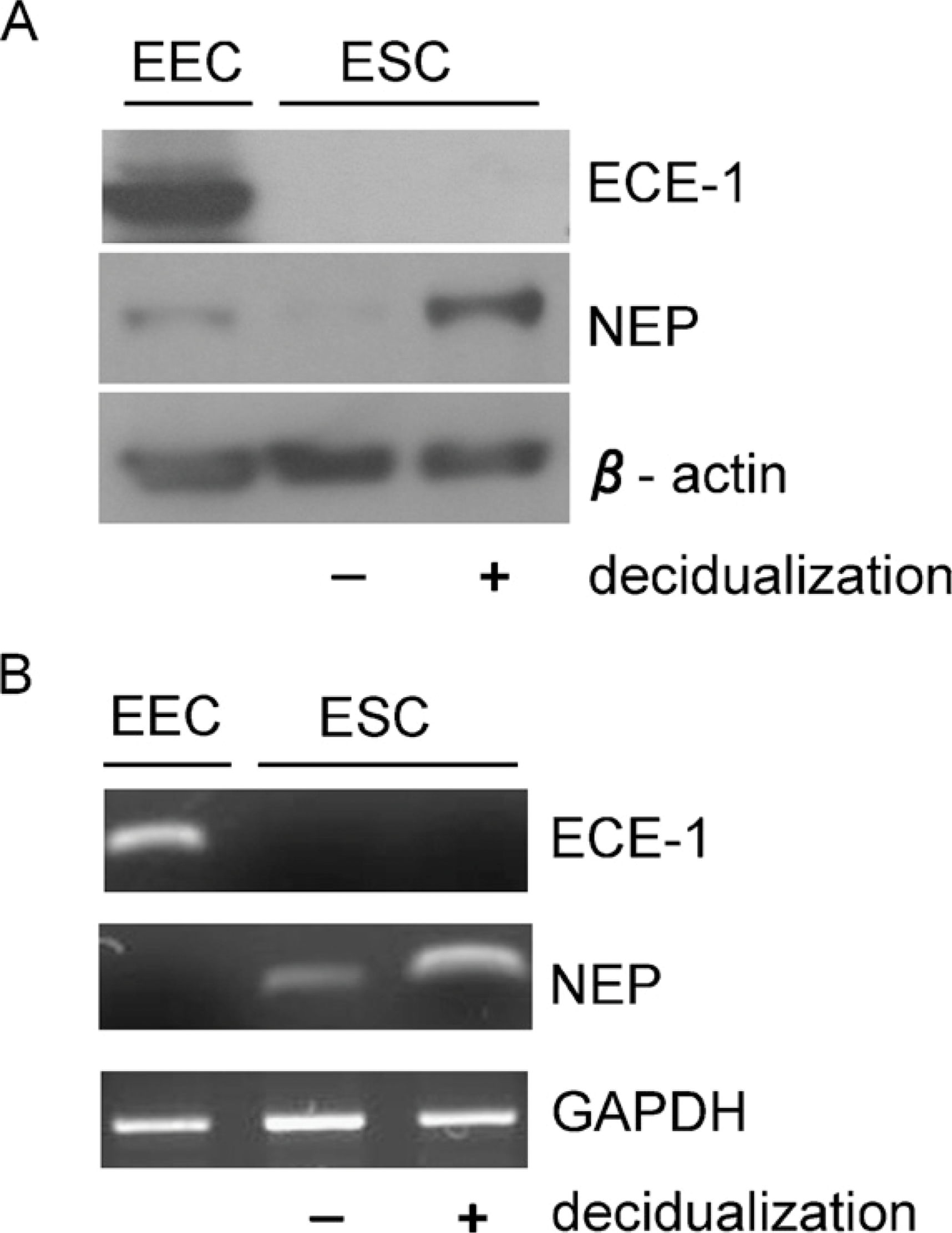
We next performed Western blotting and RT-PCR using primary cultured EEC and ESC to assess the expression of ECE-1 and NEP in vitro. Western blotting showed that EEC, not ESC, with or without decidualization, expressed ECE-1 and that decidualized ESC expressed NEP, whereas EEC and non-decidualized ESC possessed much less NEP (Figure 3A). RT-PCR showed results similar to those of Western blotting (Figure 3B). These results suggest that the difference in localization of ECE-1 and NEP in immunohistochemistry corresponds to the difference of their expression in EEC and ESC.
Discussion
Previous immunolocalization studies showed that ET-1 is present throughout the cycle in both epithelial cells and stromal cells (Cameron et al. 1992; Salamonsen et al. 1992; Ohbuchi et al. 1995). Inhibition of the passage of proteins through the Golgi apparatus with the ionophore monensin resulted in an accumulation of ET in the epithelium, not stroma, suggesting that ET synthesis occurs primarily in the endometrial epithelium (Salamonsen et al. 1992), whereas primary cultured ESC released ET-1 in vitro (Kubota et al. 1995). We have recently demonstrated that primary cultured EEC are the major source of ET-1 (Iwase et al. 2006). Considering that ECE-1 is responsible for converting big-ET-1 to ET-1, our findings support that EEC expressing ECE-1 are the predominant source of ET-1. Our results also suggest a possibility that the cyclic changes in the expression of ET-1 are implicated in ECE-1 expression in the human endometrium. However, ECE have three isoforms, ECE-1, -2, and -3, which have different characteristics (D'Orleans-Juste et al. 2003), and there is a possibility that ESC express other ECE isoforms. Moreover, ECE-1 has four isoforms (ECE-1a, -b, -c, and -d), which show different subcellular localization (Meidan et al. 2005). In the current study, Western blotting showed ECE-1 in human endometrium to be approximately 110-120 kDa, slightly smaller than the originally reported molecular mass (130 kDa). There is a possibility that the different glycosylation status of ECE-1 isoforms in various tissues might result in different molecular masses, although RT-PCR targeting identical ectodomains in each of the isoforms in this study failed to reveal which isoforms of ECE-1 are expressed. Therefore, more studies to investigate the profile of ECE expressed by human endometrium are needed to clarify the production of endometrial ET-1.
(
NEP has been thought to inactivate its peptide substrates, including ET-1, and therefore, to be involved in the cyclic change of the human endometrium. We also demonstrated that the proliferative effect of ET-1 is regulated by NEP expressed by decidualized stromal cells in vitro. Our results with immunohistochemistry and Western blotting for NEP in the current study almost coincide with those of our previous report. The increase of NEP in the mid- and late secretory phase in vivo and in primary cultured decidualized ESC in vitro suggests that progesterone might upregulate the expression of NEP. Zheng et al. (2006) have recently reported that progesterone induced transcription of the NEP gene by the coordinate action of multiple androgen-responsive elements. The discrepancy in our results between in vivo and in vitro expression of NEP in non-decidualized ESC may be explained in part by differences in the exposure to hormones, including sex steroid hormones as well as various cytokines and growth factors supplied through endometrial blood vessels; i.e., primary cultured ESC without decidualization are not exposed to such substances. Further study is required to reveal how the expression of NEP is regulated in ESC.
Our extensive studies on peptidases indicate that various peptidases are expressed in and localized to certain parts of the human endometrium. We previously reported that aminopeptidase A expressed in ESC disappears with decidual change (Ando et al. 2002), and that placental leucine aminopeptidase (P-LAP)/oxytocinase localized to epithelial cells shows a change in subcellular distribution during the menstrual cycle (Toda et al. 2002). We also found that adipocyte-derived leucine aminopeptidase, a homolog of P-LAP, which produces peptides present on the class I major histocompatibility complex, is expressed in the endometrial epithelium (Shibata et al. 2004). It is of interest that various peptidases, which have different substrates and different functions, show different profiles of expression and localization in the human endometrium. In the present study, we found that two related endopeptidases, ECE-1, which is involved in ET-1 synthesis, and NEP, which inactivates ET-1, exhibit different localizations in the endometrium and different expression patterns during menstruation. Our results indicate that the cooperative action of EEC and ESC in the regulation of ET-1 might be involved in the cyclic changes in the human endometrium. In summary, our findings indicate that EEC and ESC regulate the activation and inactivation of ET-1, which may be involved in cell proliferation in ESC, cooperatively with ECE-1 and NEP. Further study will help define the influence of the cooperative action of EEC and ESC on the cyclic developmental change of the human endometrium.
