Abstract
Mucin 1 (MUC1) is a glycoprotein in human endometrium and is abundant at the luminal epithelial surface in the receptive phase. It has a highly glycosylated ecto-domain that contains keratan sulfate chains, that disappears at the time of implantation. In addition, the glycoforms on MUC1 differ in fertile and infertile women. Therefore the aims of this study were investigations on glycosylation of MUC1 with the Thomsen-Friedenreich (TF) epitope on normal human endometrium throughout the menstrual cycle and binding of galectin-1 on the TF epitope in the endometrium and the expression of galectin-1 on the human oocyte. Human endometrial tissue was obtained from 54 premenopausal patients and was immunohistochemically analyzed with monoclonal antibodies against MUC1, TF epitope, galectin-1, and biotinylated galectin-1. In addition, human oocytes were analyzed for TF, galectin-1 expression, and galectin-1 binding. We identified a significant upregulation of MUC1 and TF epitope and, in addition, galectin-1 binding in glandular epithelium and epithelial apical surface tissue from proliferative to secretory phase. With double staining experiments, we identified a coexpression of TF and MUC1 in the early secretory phase and galectin-1 binding to TF during the same period of time. In addition we identified TF epitope and galectin-1 expression plus binding on the human oocyte and irregularly fertilized oocytes. Upregulation of TF epitope on the glandular epithelium and epithelial apical surface tissue in the secretory phase and binding of galectin-1 at the same time show the possibility of galectin-1–mediated trophectoderm binding to the endometrium within the window of implantation.
The Thomsen-Friedenreich (TF) epitope has been known for a long time as a tumor-associated epitope (Springer 1984). The presence of the TF epitope during the early fetal phase, its absence in non-carcinomatous postfetal tissues, and its association with carcinomas suggest that the TF epitope is a stage-specific oncofetal carbohydrate epitope. The TF epitope is a carbohydrate moiety related to blood group epitopes and consists of galactose-β1-3N-acetylgalactosamine (Galβ1-3GalNAc-). In epithelial cells, the TF epitope is carried by MUC1 on the apical surface of these cells. On tumor cells, MUC1 is posttranslationally modified, resulting in incomplete O-glycosylation and exposing the TF epitope (Springer et al. 1990). In addition, in the first trimester of pregnancy, we found strong expression of TF epitope and MUC1 at the apical side of the syncytiotrophoblast directed toward the maternal blood. This expression was consistent in the second trimester of pregnancy, and to a lesser degree in the third trimester (Richter et al. 2000). We also identified positive staining for TF epitope and MUC1 on extravillous trophoblast cells in the decidua during the first and second trimesters of pregnancy. Trophoblast tumor cells of the cell line BeWo, which form a syncytium in vitro, were also positive for TF epitope and MUC1, whereas Jeg3 cells, which are unable to form a syncytium, expressed only MUC1 (Jeschke et al. 2002).
Galectin-1, a prototype galectin, forms non-covalently associated homodimers under physiological conditions. Its two carbohydrate recognition domains preferentially recognize type I and type II N-acetyllactosamine residues present on all complex N-linked and many O-linked glycoproteins (Sparrow et al. 1987; Abbott et al. 1988). Through the recognition of cell surface β-galactosidic residues, the lectin displays a wide range of biological activities involving cell adhesion to the endothelium (Woynarowska et al. 1994,1996). A recent study showed that galectin-1 expression increased in the late secretory phase of the endometrium and in decidua (von Wolff et al. 2005). In a former study, we showed that galectin-1 recognizes appropriate glycoepitopes on the syncytiotrophoblast and on chorionic carcinoma cells (BeWo) (Jeschke et al. 2004). Results demonstrate further that ligation of galectin-1 to Galbeta1–4GlcNAc and Galbeta1–3GalNAc (TF) epitopes on BeWo cells have regulatory effects on human chorionic gonadotropin (hCG) and progesterone production. In addition, we performed binding experiments on irregularly fertilized oocytes using human uterine epithelial Ishikawa cells as a model for TF-expressing glandular epithelial cells of the endometrium. Ishikawa cells represent an optimized model of the human endometrium. It consists of a human uterine epithelial cell line that has maintained polar organization to a high degree (Fukuda and Nozawa 1999; Heneweer et al. 2005).
Therefore the aim of this study was: (a) identification of MUC1 and TF expression in glandular epithelial cells of the endometrium throughout the menstrual cycle; (b) identification of galectin-1 binding to glandular epithelial cells of the endometrium throughout the menstrual cycle; (c) identification of TF expression and galectin-1 binding to the oocyte and irregularly fertilized oocytes; and (d) binding experiments on irregularly fertilized oocytes using human uterine epithelial Ishikawa cells as a model for TF-expressing glandular epithelial cells of the endometrium.
Materials and Methods
Tissue Samples
Samples of human endometrium were obtained from 54 premenopausal, non-pregnant patients undergoing gynecological surgery, either by dilation and curettage or hysterectomy, for benign diseases, mainly uterine leiomyoma. All women had a normal and regular menstrual cycle with no hormonal treatment for 3 months prior to surgery. All pathological and hyperplastical endometrial samples were excluded from this study. Endometrium samples were classified according to anamnestical and histological dating (Dallenbach-Hellweg and Poulsen 1985) into proliferative (n=32), early secretory (n=10), and late secretory phases (n=12) as previously described (Mylonas et al. 2000,2003a,b).
Controlled ovarian stimulation of infertile patients attending the in vitro fertilization (IVF) department of the Ludwig-Maximilians-University Munich was performed using standard protocols, including recombinant follicle-stimulating hormone (FSH) (Gonal-F; Serono, Geneva, Switzerland), and/or human menopausal gonadotrophin (Menogon; Ferring Pharmaceuticals, Kiel, Germany).
Follicular growth was monitored by transvaginal ultrasound and serum levels of estradiol (E-2). hCG (Predalon Organon; AKZO-NOBEL, The Netherlands) was administered 36 hr before oocytes were retrieved by transvaginal ultrasound–guided aspiration. FSH, E-2, and hCG levels were measured using a solid-phase, competitive chemiluminescent immunoassay (DPC Immulite; Diagnostic Products Corp., Los Angeles, CA).
Oocytes of each patient were inseminated by homologous standard IVF or intracytoplasmic sperm injection (ICSI) procedures. Fertilization of the oocytes was assessed by light microscopy 14–18 hr after insemination or ICSI for the presence, number, and morphology of pronuclei. A prerequisite for normal fertilization is the presence of two pronuclei (2 PN), one male, one female, approximately of equal size and of two polar bodies. Such a zygote has a diploid chromosomal status. Abnormal zygotes are either haploid [having only one pronucleus (1 PN)], triploid, or polyploid (having 3 PN or more). 3-PN zygotes may arise through different mechanisms, e.g., by penetration of an oocyte by two spermatozoa, by penetration by a binucleate spermatozoon, or by retention of the first or second polar body chromosomes. A 3-PN zygote may develop further to a cleaved embryo and even reach the blastocyst stage; however, the triploid status is not compatible with normal development, and subsequently its growth is arrested. Therefore, all 3-PN zygotes are always excluded from further culture and are discarded. The normally fertilized (2-PN) zygotes used for the present study were donated by patients who chose not to cryopreserve their excess fertilized oocytes. According to German laws for the protection of embryos, all remaining fertilized oocytes, other than those assigned for embryo transfer, must be either cryopreserved or discarded. Normal unfertilized oocytes were derived from cells that at the time of insemination or ICSI were immature and therefore not suitable for clinical use.
The Ethical Committee of the Ludwig-Maximilians-University of Munich approved the study, and informed consent of the patients was obtained.
Purification and Biotinylation of Galectin-1 From Human Placenta
Galectin-1 was prepared from term placental tissue by lactose extraction with EDTA-MePBS (20 mM sodium phosphate, pH 7.2, 150 mM NaCl, 4 mM 2-mercaptoethanol, 2 mM EDTA; all BioRad, Munich, Germany) and purified by sequential affinity chromatography on asialofetuin Sepharose 4B (Hirabayashi and Kasai 1984) followed by affinity chromatography on lactosyl agarose (GE Healthcare; Freiburg, Germany). Then the protein was purified to homogeneity by anion exchange chromatography on a Resource Q column (GE Healthcare) (Walzel et al. 1999).
The lectin (1 mg/ml) was biotinylated in phosphatebuffered saline (PBS, pH 8.0) by the addition of 40 μl 10 mM biotinyl-N-hydroxysuccinimide in dimethyl sulfoxide (Boorsma et al. 1986). After incubation at room temperature for 1 hr, biotinylated galectin-1 was affinity purified on lactosyl agarose (GE Healthcare). The bound fraction was eluted with 50 mM lactose in EDTA-MePBS. Buffer exchange was performed on a Bio-Gel P6 column (Bio Rad; Munich, Germany) equilibrated with PBS, pH 7.4 (Jeschke et al. 2004).
Cocultivation of Irregularly Fertilized Oocytes and Human Uterine Epithelial Ishikawa Cells
The human endometrial cancer cell line Ishikawa was derived from an endometrial adenocarcinoma (Nishida et al. 1985), derived from the American Type Culture Collection and cultured on superfrost cell culture tissue slides (Langenbrinck; Teningen, Germany) in quadriperm cell culture dishes. The cells were maintained in DMEM (Biochrom; Berlin, Germany) supplemented with 10% fetal calf serum, 5 ml L-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin (Biochrom). Irregularly fertilized oocytes were placed on the free surface of endometrial cell monolayers and cultivated for another 24 hr. After that, cell culture medium was removed and cells were dried and wrapped.
Immunohistochemistry
Immunohistochemistry was performed using a combination of pressure cooker heating and the standard streptavidin-biotin-peroxidase complex with the use of the mouse-IgG-Vectastain Elite ABC kit (Vector Laboratories; Burlingame, CA). Mouse monoclonal antibodies used for these experiments are anti-mucin 1 (PankoMab, 2 μg/ml, mouse IgG; Glycotope, Berlin, Germany) and anti-TF (Nemod TF 2, 2 μg/ml, mouse IgM; Glycotope). In addition, biotinylated galectin-1 was used for immunohistochemical detection of galectin-1 binding diluted to 0.3 μg/ml with PBS and incubated with the slides overnight at 4C.
Briefly, paraffin-fixed tissue sections were dewaxed using xylol for 15 min, rehydrated in ascending series of alcohol (70%, 96%, and 100%), and subjected to epitope retrieval for 10 min in a pressure cooker using sodium citrate buffer (pH 6.0), containing 0.1 M citric acid and 0.1 M sodium citrate in distilled water. After cooling, sections were washed twice in PBS. Endogenous peroxidase activity was quenched by immersion in 3% hydrogen peroxide (Merck; Darmstadt, Germany) in methanol for 20 min. Nonspecific binding of the primary antibodies was blocked by incubating the sections with “diluted normal serum” (10 ml PBS containing 150 μl horse serum; provided by Vector Laboratories) for 20 min at room temperature. Sections were then incubated at room temperature for 60 min with the primary antibodies. After washing with PBS, sections were incubated in “diluted biotinylated serum” (10 ml PBS containing 50 μl horse serum; provided by Vector Laboratories) for 30 min at room temperature. After incubation with the avidin-biotin-peroxidase complex (diluted in 10 ml PBS; Vector Laboratories) for 30 min and repeated washing steps with PBS, visualization was performed with substrate and chromagen 3,3'-diaminobenzidine (DAB; Dako, Glostrup, Denmark) for 8–10 min. Sections were counterstained with Mayer's acidic hematoxylin and dehydrated in an ascending series of alcohol (50–98%). After xylol treatment, sections were covered. Negative controls were performed by replacing the primary antibody with normal horse serum. Immunohistochemical staining was performed using an appropriate positive control. Positive cells showed a brownish color, and negative controls as well as unstained cells were blue.
Immunocytochemistry and Controls
Cells were grown on quadriperm test slides (Langenbrinck), dried, wrapped, and stored at −80C. After thawing, cells were briefly fixed with formalin (Merck; 5% in PBS, 5 min). Biotinylated galectin-1 used for the experiments was diluted to 0.3 μg/ml with PBS and incubated with the slides overnight at 4C in the absence and presence of up to 40 μg/ml TF-α disaccharide coupled to polyacrylamide (TF-PAA; Lectinity Holdings, Moscow, Russia). After washing, Cy3-labeled streptavidin, diluted 1:200, served as fluorochrome. The slides were finally embedded in mounting buffer. For controls, human oocytes were incubated with 2 μg/ml mouse IgM in PBS and used as isotype control antibodies.
Immunofluorescence and Confocal Laser Microscopy
For the immunohistochemical characterization, cryosections were examined as previously described (Jeschke et al. 2002). Antibodies are anti-mucin 1 (mouse IgG) and anti-TF (Nemod TF 2, mouse IgM; Glycotope). For galectin-1 expression, anti-galectin-1 (clone 201002; Rat IgG2b, R&D Systems, Wiesbaden, Germany) antibody was used. Briefly, all samples were fixed in 5% buffered formalin. The antibodies were diluted to 2 μg/ml with PBS and incubated with the slides overnight at 4C. After washing, Cy2-labeled goat anti-mouse IgG/IgM or Cy3-labeled goat anti-mouse IgG/IgM or anti-rat IgG, diluted 1:200, served as second antibody. The slides were finally embedded in mounting buffer containing 4,6-diamino-2-phenylindole, resulting in blue staining of the nucleus. Slides were examined with a Zeiss (Jena, Germany) Axiophot photomicroscope. Digital images were obtained with a digital camera system (Axiocam; Zeiss) and saved on computer.
Imaging was also performed using a confocal laser microscope (LSM 510; Zeiss), and images were processed using Adobe Photoshop for adjustment of contrast and size.
Evaluation and Statistical Analysis
The intensity and distribution patterns of specific immunohistochemical staining were evaluated using a semi-quantitative assay, as previously described (Remmele and Stegner 1987), and used to assess the expression patterns of other antigens, like CA-125 and cathepsin D (Mylonas et al. 2003c,d). The International Remmele Score was calculated by multiplication of optical staining intensity (graded as 0 = no, 1 = weak, 2 = moderate, and 3 = strong staining) and the percentage of positive stained cells (0 = no staining, 1 = <10% of the cells, 2 = 11–50% of the cells, 3 = 51–80% of the cells, and 4 = >81% of the cells). The slides were examined by two independent observers, including a gynecological pathologist (N.S.). Sections were examined using a 3CCD color camera (JVC; Victor Company of Japan, Yokohama, Japan) and a Leitz (Wetzlar, Germany) microscope. The results were evaluated using the non-parametric Mann-Whitney U rank-sum test, and the non-parametric Spearman correlation coefficient was used for estimating correlations between MUC1, TF, and galectin-1 (SPSS; Chigaco, IL). Significance was assumed at p<0.05.
Results
Immunohistochemical Expression of MUC1 in Glandular Epithelial Tissue Throughout the Menstrual Cycle
We identified a moderate staining of MUC1 in glandular epithelial cells of the proliferative phase of the human endometrium (Figure 1A). In addition, a strong staining of MUC1 was found in glandular epithelial cells of the early secretory phase of the human endometrium (Figure 1B). In the late secretory phase, MUC1 is significantly upregulated in glandular epithelial cells (Figure 1C, p=0.017) compared with the proliferative phase. A summary of the staining results of MUC1 in glandular epithelial cells is presented in Figure 2.
Expression of the TF Epitope in Secretory Glandular Epithelial Tissue
We identified a moderate staining of TF in glandular epithelial cells of the proliferative phase of the human endometrium (Figure 3A). In addition, a significantly enhanced staining of TF was found in glandular epithelial cells of the early secretory phase of the human endometrium (Figure 3B, p=0.006). In the late secretory phase, TF is also significantly upregulated in glandular epithelial cells (Figure 3C, p=0.018) compared with the proliferative phase. A summary of the staining results of TF in glandular epithelial cells is presented in Figure 4.
Binding of Exogenous Biotinylated Galectin-1 to Secretory Glandular Epithelial Tissue
We identified no binding of exogenous biotinylated galectin-1 to glandular epithelial cells in the proliferative phase of the human endometrium (Figure 5A). In addition, a significantly enhanced binding of galectin-1 to glandular epithelial cells was found in the early secretory phase of the human endometrium (Figure 5B, p=0.016). In the late secretory phase, galectin-1 binding to glandular epithelial cells is also significantly upregulated (Figure 5C, p=0.004) compared with the proliferative phase. A summary of the staining results of galectin-1 binding to glandular epithelial cells is presented in Figure 6.
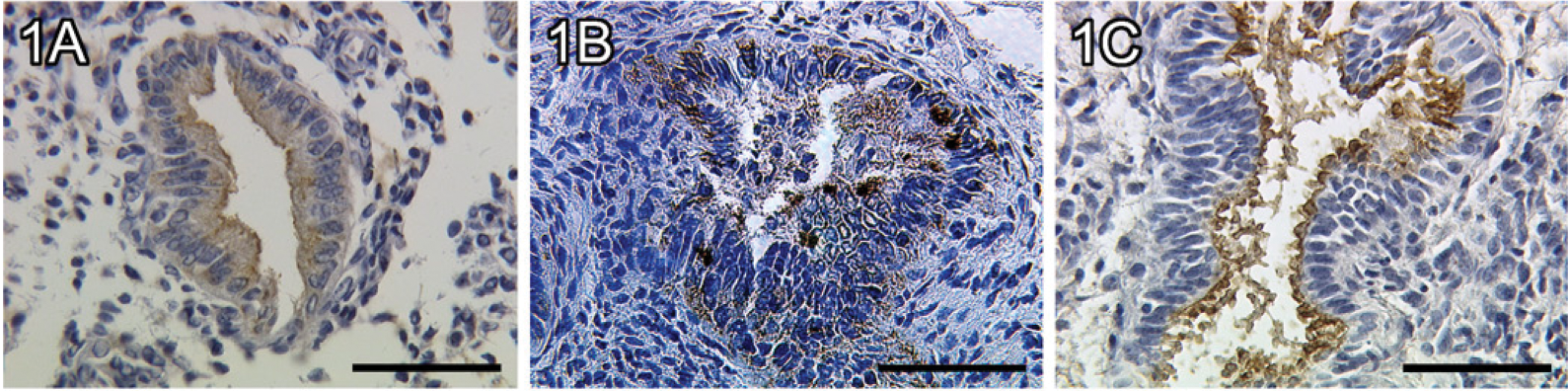
Expression of mucin-1 (MUC1) in endometrial glandular epithelial tissue of a proliferative endometrium (
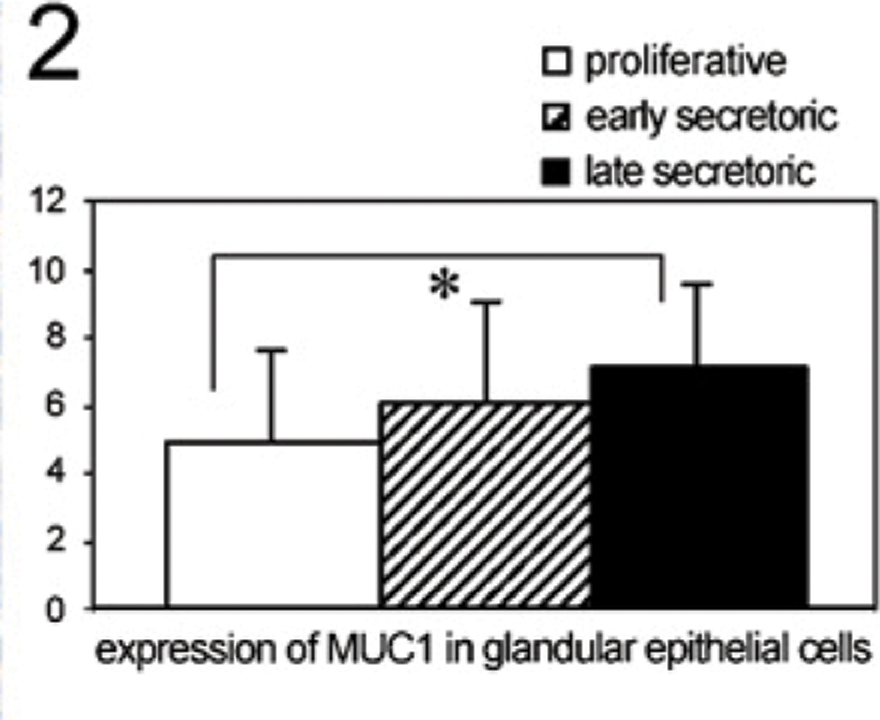
Staining intensity of MUC1 in glandular epithelial tissue throughout the menstrual cycle determined by a semiquantitative immunohistochemical score on the different tissue slides. Data shown represent means ± SEM. Asterisk indicates significant differences (p<0.05).
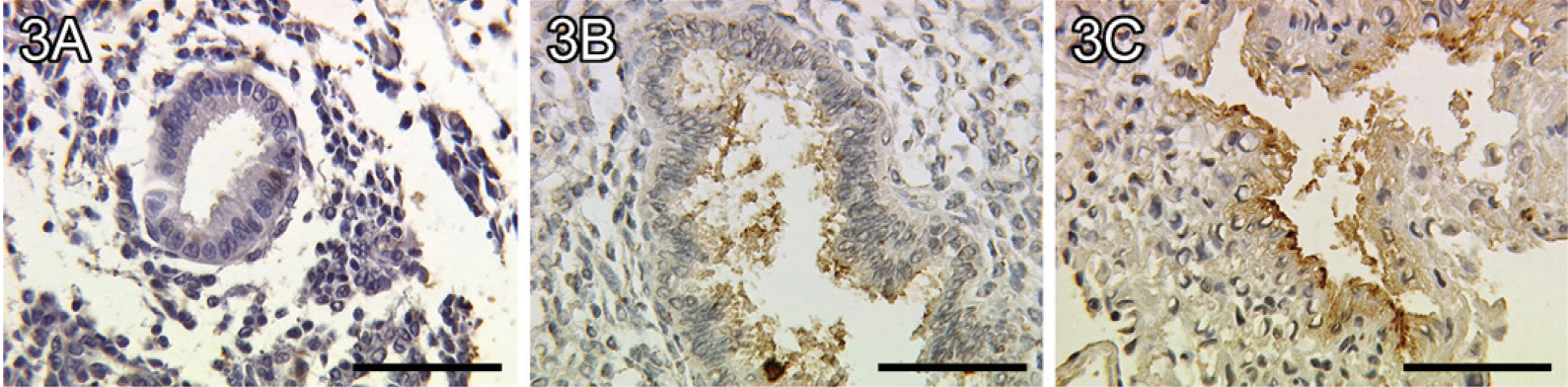
Faint expression of the Thomsen-Friedenreich (TF) epitope in endometrial glandular epithelial tissue of a proliferative endometrium (
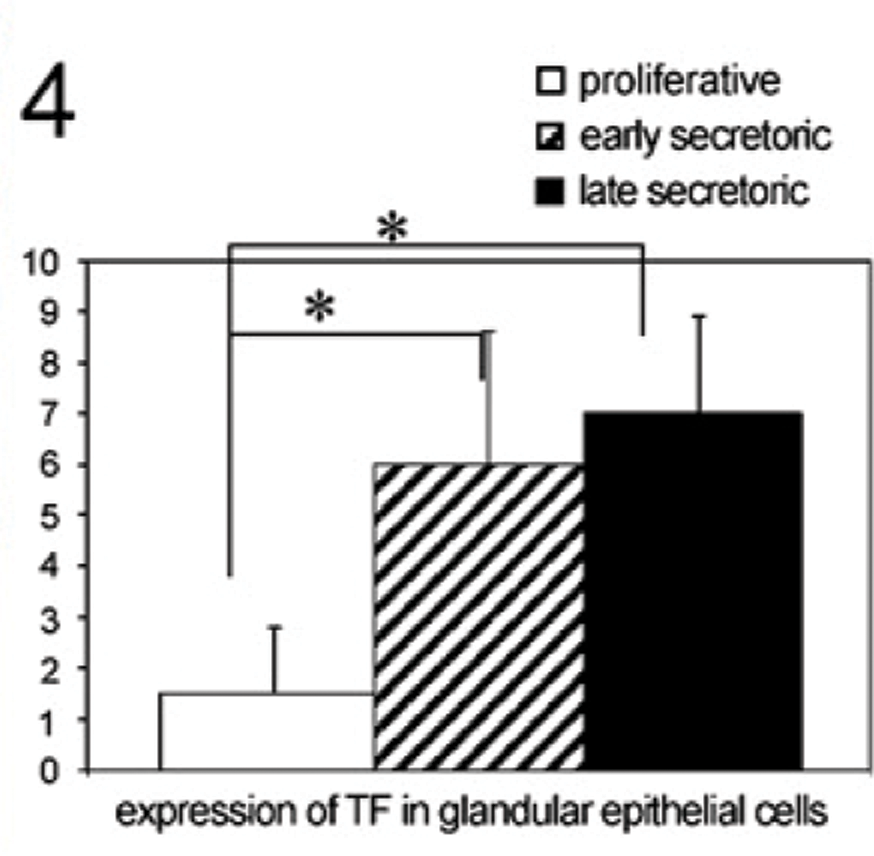
Staining intensity of TF in glandular epithelial tissue throughout the menstrual cycle determined by a semiquantitative immunohistochemical score on the different tissue slides. Data shown represent means ± SEM. Asterisk indicates significant differences (p<0.05).
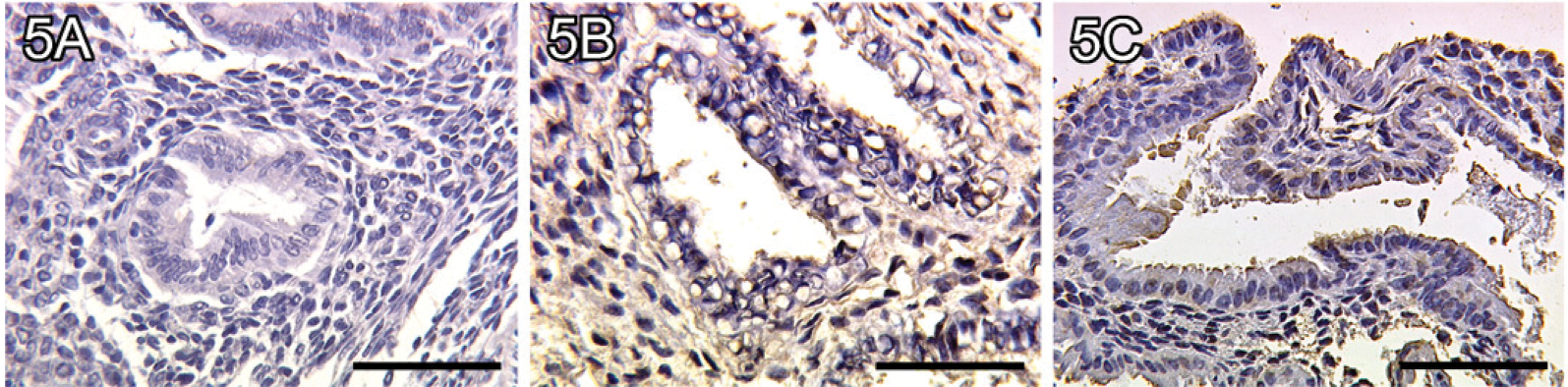
No binding of galectin-1 to endometrial glandular epithelial tissue of a proliferative endometrium (
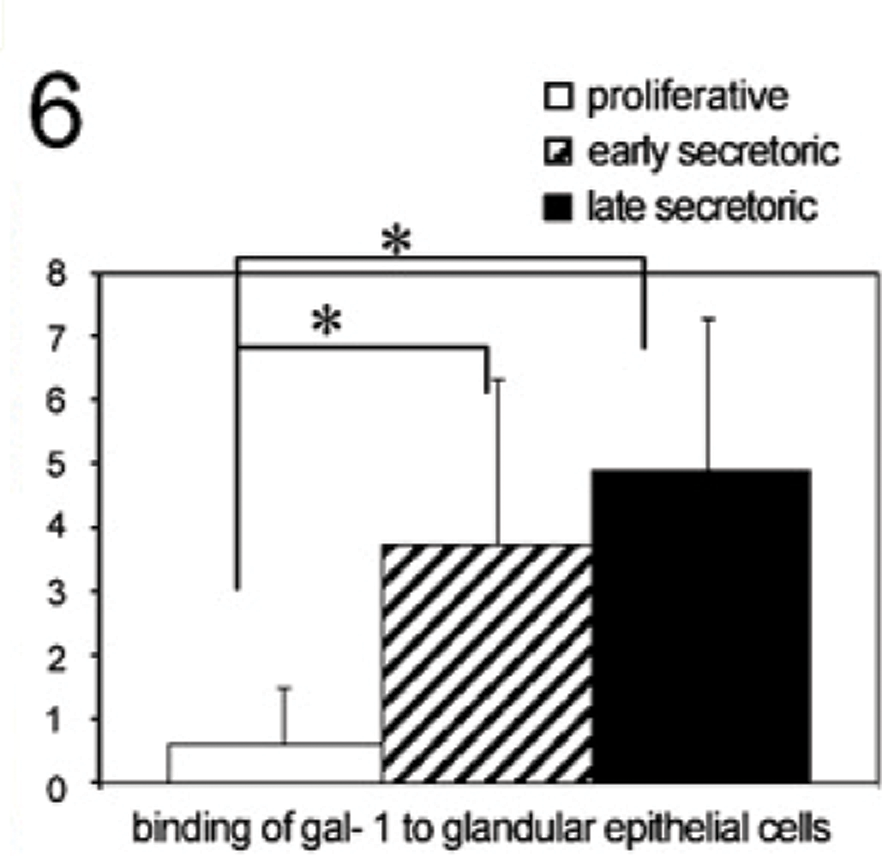
Staining intensity of galectin-1 binding to glandular epithelial tissue throughout the menstrual cycle determined by a semiquantitative immunohistochemical score on the different tissue slides. Data shown represent means ± SEM. Asterisk indicates significant differences (p<0.05).
Coexpression of MUC1 and TF, and Binding of Exogenous Biotinylated Galectin-1 to Endometrial Epithelial Apical Surface Tissue in the Early Secretory Phase
We identified a strong expression of MUC1 in endometrial epithelial apical surface tissue of an early secretory endometrium (Figure 7A). A strong expression of TF epitope was also found in endometrial epithelial apical surface tissue of an early secretory endometrium (Figure 7B). Binding of galectin-1 to endometrial epithelial apical surface tissue was observed in an early secretory endometrium (Figure 7C). In addition, we found coexpression of MUC1 and TF on endometrial epithelial apical surface tissue only in the early secretory phase (Figure 7D–7F). In the early secretory phase, we also show colocalization of galectin-1 binding (Figure 7H) and TF epitope (Figure 7G).
Correlation Between MUC1, TF, and Galectin-1
Comparison of MUC1 and TF staining (Figures 2 and 4) and, in addition, galectin-1 binding analysis (Figure 6) in proliferative (n=32), early secretory (n=10), and late secretory phase (n=12) endometrial tissue slides used in this study revealed a positive correlation for TF epitope expression and galectin-1 binding (coefficient of correlation: 0.633, p=0.01), and in addition, galectin-1 binding and MUC1 expression (coefficient of correlation: 0.355, p=0.034) throughout the menstrual cycle.
Expression of the TF Epitope on the Human Oocyte and on Irregularly Fertilized Oocytes
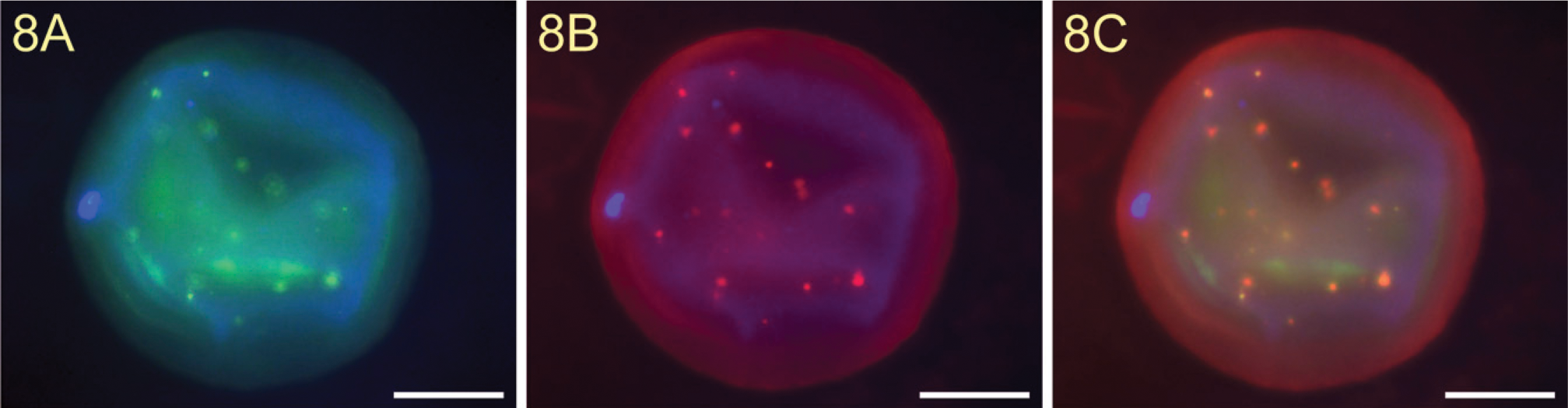
We showed a strong staining of the TF epitope on the surface of a normal unfertilized human oocyte (Figure 8A) and on the surface of an irregularly fertilized oocyte (Figure 9A). Incubation of mouse IgM isotype control antibodies showed no staining of TF (Figure 10A).
Binding of Exogenous Biotinylated Galectin-1 to a Human Oocyte
In addition, we also found a strong binding of galectin-1 to the surface of these cells (Figures 8B and 9B). Pre-incubation of TF-PAA conjugates with biotinylated galectin-1 did not result in galectin-1 binding (Figure 10B).
Cocultivation of Irregularly Fertilized Oocytes and Human Uterine Epithelial Ishikawa Cells
Irregularly fertilized oocytes attached to human uterine epithelial Ishikawa cells after 24 hr of cultivation. TF staining is shown in Figure 11A and galectin-1 staining in Figure 11B. TF and galectin-1 double staining showed simultaneous expression of both antigens (Figure 11C), which were examined with confocal laser microscopy at the upper surface of Ishikawa cells and the undersurface of the oocyte. The exact localization of the human oocyte on Ishikawa cells was determined by inverse microscopy (Figure 11D) before staining.
Discussion
Changes in the epithelium of the endometrium are characterized by alterations in cell surface molecules and the appearance of pinopodes. This coincides with the window of endometrial receptivity in the menstrual cycle (Horne et al. 2005). In this study, we identified an upregulation of MUC1, TF epitope, and, in addition, galectin-1 binding, in glandular epithelium and epithelial apical surface tissue from the proliferative to the late secretory phase. In addition, we identified TF epitope and galectin-1 expression plus binding on the human oocyte and irregularly fertilized oocytes.
Human endometrial epithelium undergoes progesterone-modulated differentiation during the menstrual cycle (Smith et al. 1989; Hoadley et al. 1990). MUC1 expression in the female reproductive tissues is dependent on the stage of the menstrual cycle (Dockery et al. 1988a,b,1993). Earlier studies have shown that MUC1 expression in endometrial tissues is at the highest in secretory phases (Hey et al. 1994), when embryo implantation occurs. MUC1 is associated with the apical surface of epithelial cells and is also secreted, detectable in uterine fluid at elevated levels in the implantation phase (Hey and Aplin 1996). However, its physiological role is uncertain; it may either inhibit intercellular adhesion by steric hindrance or carry carbohydrate recognition structures capable of mediating cell–cell interaction (Aplin 1997). Hey and Aplin (1996) showed that endometrial epithelium expresses both Sialyl-Lewis × (SLex) and Sialyl-Lewis a (SLea), with a distribution and pattern of menstrual cycle regulation similar to that of MUC1. In addition, Genbacev et al. (2003) found that on the maternal side, human uterine epithelial cells upregulated selectin oligosaccharide-based ligands like SLex during the window of receptivity. On the fetal side, human trophoblasts expressed L-selectin, which binds to SLex.
Endometrial MUC1 contains ∼40% glycan by weight (Hey et al. 1994; Aplin and Hey 1995). Several MUC1-associated glycans show strong hormonal regulation in endometrium (Hoadley et al. 1990). Keratane sulfate chains are increased in the secretory phase of the cycle (Aplin et al. 1994). A high density of MUC1 at the cell surface can inhibit cell adhesion. This raises the possibility of the existence of a uterine barrier to implantation that might allow maternal rejection of poorer quality embryos (Aplin 1999). Hatched blastocysts were found to attach readily to the epithelial surface. MUC1 was lost from epithelial cells beneath and near to the attached embryo, whereas normal expression persisted in neighboring cells (Aplin et al. 2001). In addition, the endometrial mucin MUC1 is upregulated by progesterone and downregulated in vitro by the human blastocyst (Meseguer et al. 2001).
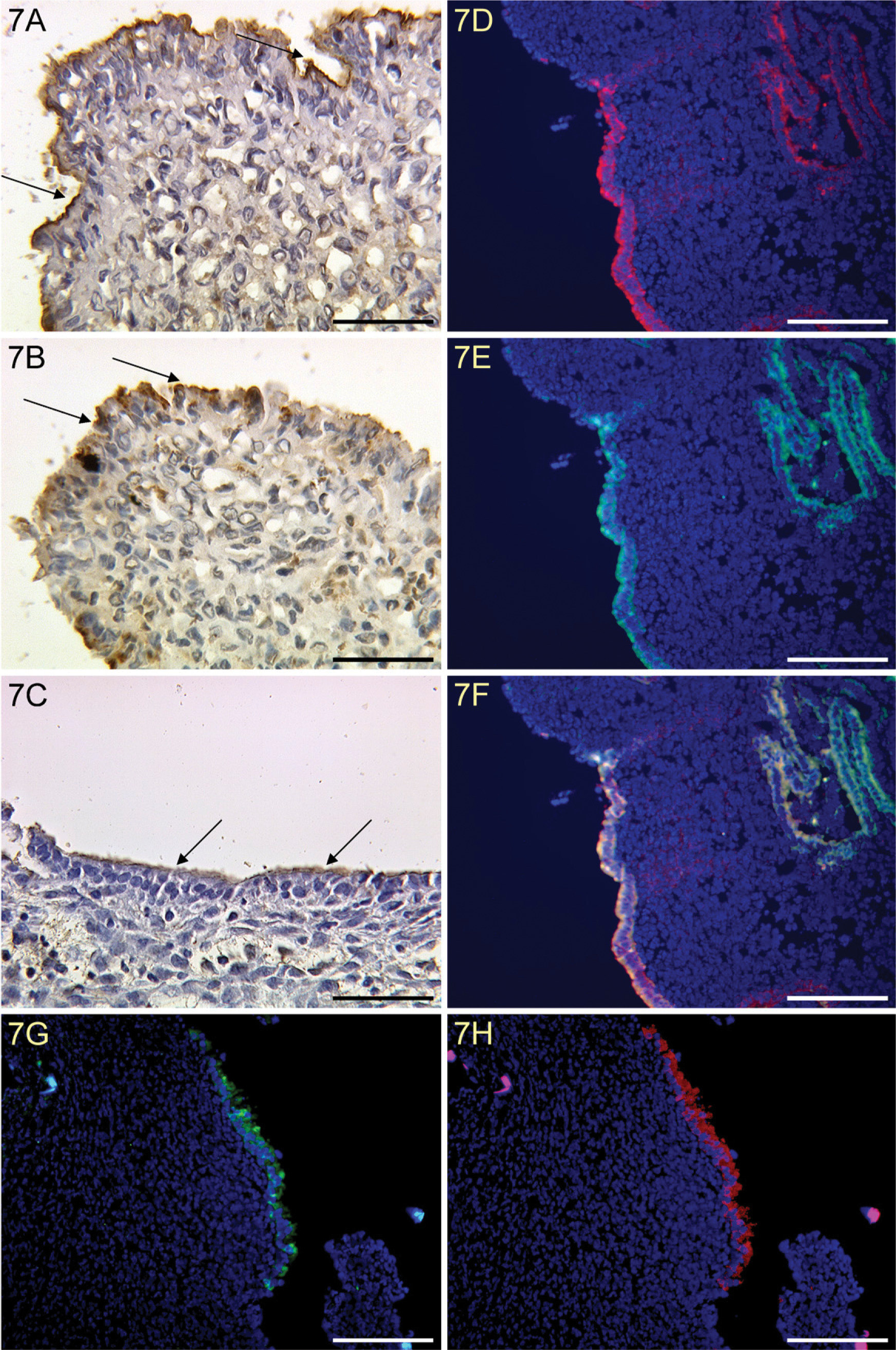
Strong expression of MUC1 in endometrial epithelial apical surface tissue of an early secretory endometrium (
Expression of TF antigen on a human oocyte (
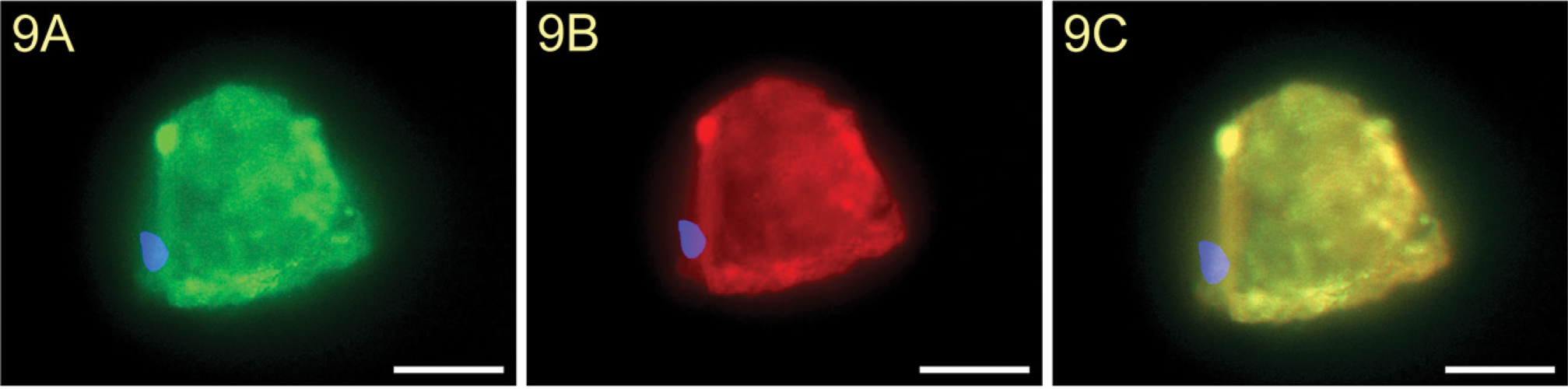
Expression of TF antigen on an irregularly fertilized human oocyte (
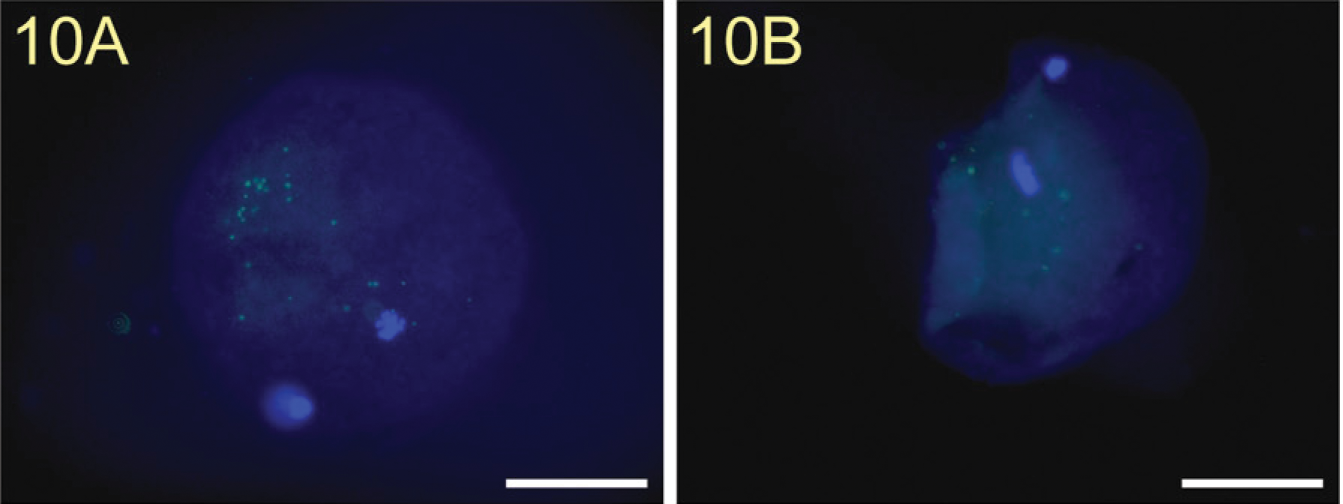
TF antigen isotype control on an irregularly fertilized human oocyte showed no staining (
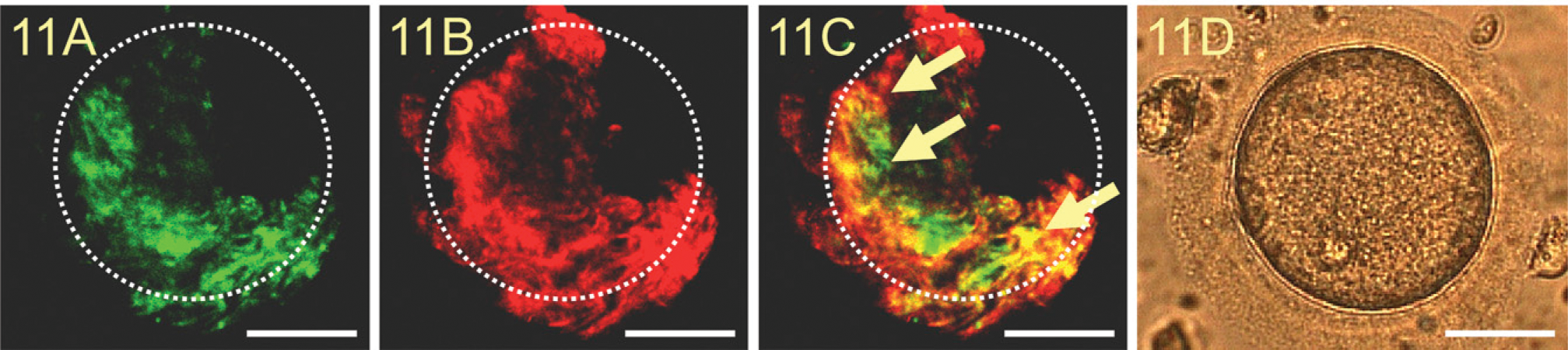
Binding of a human oocyte to human uterine epithelial Ishikawa cells. TF expression appears in green (
Here we show that endometrial MUC1 carries an additional glycosylation type (the TF epitope). The TF glycotope (Galβ1–3GalNAc-) is expressed in more than 85% of human carcinomas (Springer 1984; Goletz et al. 2003). In normal adult human tissues, TF epitopes are only expressed in limited amounts and are restricted to a few immunologically privileged sites (Cao et al. 1996). The TF epitope is, however, expressed on fetal epithelia and mesothelia (Barr et al. 1989), on transferrin from human amniotic fluid (van Rooijen et al. 1998), and on trophoblast cells (Richter et al. 2000). The TF epitope and its carrier protein MUC1 are expressed by syncytiotrophoblasts at the materno-fetal interphase and by extravillous trophoblast cells invading the decidua (Jeschke et al. 2007). It is also found on BeWo trophoblastic tumor cells forming a syncytium in vitro (Jeschke et al. 2002). In a former study, we showed that galectin-1 recognizes appropriate glycoepitopes on the syncytiotrophoblast and on chorionic carcinoma cells (BeWo) (Jeschke et al. 2004). Results demonstrate further that ligation of galectin-1 to galbeta1–4GlcNAc and galbeta1–3GalNAc (TF) epitopes on BeWo cells has regulatory effects on hCG and progesterone production and on apoptosis of these cells (Wiest et al. 2005; Jeschke et al. 2006). In this study, we showed that TF expression is significantly upregulated in glandular epithelium and epithelial apical surface tissue from the proliferative to the secretory phase. TF expression in endometrial tissues is at the highest in the secretory phases, when embryo implantation occurs. We also found that galectin-1 binding to glandular epithelium and epithelial apical surface tissue is significantly upregulated from the proliferative to the secretory phase. In addition, there is a positive correlation of TF expression and galectin-1 binding in the human endometrium throughout the menstrual cycle and a positive correlation of MUC1 expression and galectin-1 binding throughout the menstrual cycle. This finding supports the thesis that expression of the TF epitope on MUC1 itself changes during the menstrual cycle.
Galectin-1 expression by the bovine blastocyst was described previously by Mohan et al. (2004). Here we show that human oocytes and irregularly fertilized human oocytes also express galectin-1, TF, and, in addition, bind galectin-1 on their surfaces. Because galectin-1 has two carbohydrate recognition domains, it is able to mediate either intramolecular or intermolecular cross-linking by binding to more than one sugar residue (Perillo et al. 1998) and to support cell adhesion (Martinez et al. 2004). In addition, expression of galectin-1 in the human endometrium increased significantly in the late secretory phase and in decidua of the sixth to ninth weeks of pregnancy (von Wolff et al. 2005). For this reason, we performed binding experiments with Ishikawa cells as a model for the endometrium and the oocyte to show that galectin-1 is colocalized with TF staining. These findings suggest that TF and galectin-1 expression may be regulated by both the blastocyst and the endometrium.
On the basis of the results of our studies, we are aware that colocalization of galectin-1 and TF epitope in oocytes and endometrium may not necessarily be an indication for the functional involvement of galectin-1 in implantation. Moreover, Fowlis et al. (1995) and Colnot et al. (1996) reported galectin-1 expression during pre- and postimplantation development in mice. They found that galectin-1 and galectin-3 are first expressed in trophectoderm cells of the implanting mouse embryo. Both galectins may be implicated in the process of implantation. However, they also showed that the lack of galectin-1 in galectin-1–null mutant mice is compatible with implantation (Colnot et al. 1998). Therefore, additional systems of attachment and implantation like the SLea/x-Selectin-system might, at least in part, explain why these double-mutant mice (gal1−/- and gal3−/-) were still able to implant.
In summary, we identified a significant upregulation of MUC1, TF epitope, and galectin-1 binding in glandular epithelium and epithelial apical surface tissue from the proliferative to the late secretory phase. In addition, we identified TF epitope and galectin-1 expression plus binding on the human oocyte and irregularly fertilized oocytes. Upregulation of the TF epitope on the glandular epithelium and epithelial apical surface tissue in the secretory phase and binding of galectin-1 during the same phase suggest the possibility of galectin-1–mediated trophectoderm binding to the endometrium during the window of implantation.
Footnotes
Acknowledgements
Supported by the Deutsche Forschungsgemeinschaft (UJ, HW) and in part by the FöFoLe project of the Ludwig-Maximilians-University Munich (IM).
We thank the 1st Department of Obstetrics and Gynaecology for assistance in obtaining the material and S. Kunze for assistance with histochemistry.
