Abstract
Our recent studies demonstrated the localization of protein 4.1B, a member of the 4.1 skeletal membrane proteins, to the basolateral membranes of the S1-S2 renal proximal tubules. In the present studies, we investigated the presence of binding partners that could form a molecular complex with the 4.1B protein. Immunohistochemistry revealed the localization of p55, a membrane-associated guanylate kinase, and the sodium bicarbonate cotransporter1 (NBC1), to the basolateral membrane domain of S1-S2 in mouse renal proximal tubules. Using immunoprecipitation of kidney lysates with anti-p55 antibody, a positive band was blotted with anti-4.1B antibody. GST fusion proteins including the NBC1 and 4.1B regions were confirmed to bind with each other by electrophoresis after mixing. Both NBC1- and 4.1B-specific bands were detected in renal protein mixtures immunoprecipated by either anti-4.1B- or NBC1-specific antibodies. It is likely that NBC1, 4.1B, and p55 form a molecular complex in the basolateral membrane of the kidney S1-S2 proximal tubules. We propose that the 4.1B-containing membrane skeleton may play a role in regulating the Na+ and HCO3 - reabsorption in S1-S2 proximal tubules.
T
Erythrocyte membrane skeletal 4.1R was reported to interact with a membrane-associated guanylate kinase (MAGUK) protein, p55 (Ruff et al. 1991; Alloisio et al. 1993). MAGUKs have also been reported to localize in and to be required for the formation of several types of cell junctions, including epithelial tight and septate junctions as well as synaptic and neuromuscular junctions (Dimitratos et al. 1999), suggesting their function as regulators of cell polarity (Caruana 2002). They function by binding to the transmembranous proteins as well as to other signal transduction proteins, maintaining the structural specialization of the cell membranes (Montgomery et al. 2004). p55 was also reported to be enriched in the synaptic plasma membrane and postsynaptic density fractions of the forebrain (Jing-Ping et al. 2005).
In addition to the formation of molecular complexes inside the cell membrane, an important point might be what kind of transmembranous proteins interact with 4.1B in the renal proximal tubules. In S1-S2 renal proximal tubules, sodium bicarbonate cotransporter1 (NBC1) is well known to be a major transporter for HCO3 - reabsorption across the basolateral membranes, and contributes significantly to Na+ reabsorption (Soleimani et al. 1991). NBC1 is a member of the SLC4 family of bicarbonate transporters, which comprises at least eight genes (Romero et al. 2004). Functionally, eight bicarbonate-transporter family proteins fall into two major groups (Soleimani 2001; Romero et al. 2004; Pushkin and Kurtz 2006): three Cl-/HCO3 - anion exchangers (AE1-3) and five NBCs. The AE1-3 part of the family appears to have branched off from a common ancestor nearly simultaneously, whereas NBC1 and NBC4 were derived from a form that was intermediate between the AEs and NBC2 and NBC3. Interestingly, a previous report showed that 4.1R bound with AE1 in inside-out vesicles prepared from erythrocyte membranes (Jons and Drenckhahn 1992), and binding studies between 4.1R and AE1 also indicated their interaction. Ankyrin, a membrane skeletal component, is also thought to interact with AE1, resulting in functional changes of the interaction of AE1 with 4.1R (An et al. 1996).
Owing to the fact that the NBC family proteins share homologous structures with AE family proteins, we focused on NBC1 to evaluate the possibility of its molecular interaction with 4.1B in the mouse kidney. Further, following the immunohistochemical studies, which revealed that the localization of p55 was identical to that of 4.1B in the kidney proximal tubule, we examined possible molecular interactions of 4.1B with p55.
Materials and Methods
Antibodies
An affinity-purified rabbit polyclonal anti-rat 4.1B antibody was raised against a specific domain of the protein, and verified to specifically bind rodent 4.1B and to have no immunoreaction with other members of the 4.1 family, as described previously (Yamakawa and Ohara 2000; Terada et al. 2004a; Ohno et al. 2004). Affinity-purified rabbit polyclonal anti-p55 antibody (Jing-Ping et al. 2005) and anti-NBC1 antibody (Amlal et al. 2001) have also been well characterized and have been shown to be useful for immunohistochemistry and immunoprecipitation analysis.
Animals and Tissue Preparations
All animals used in the present study were handled in compliance with the University of Yamanashi Guidelines for Experiments on Animals. Adult (3-4 months old) or postnatal day 2, 4, 10, or 20 (P2, P4, P10, P20) C57BL/6 mice were anesthetized with sodium pentobarbital and perfused via the heart with 4% paraformaldehyde in 0.1 M phosphate buffer (PB), pH 7.4. Their kidneys were removed and immersed in the same fixative at 4C for 1–2 hr. After rinsing in phosphate-buffered saline (PBS), they were immersed in 30% sucrose in PB at 4C overnight, then embedded in OCT compound and sectioned at 4-6-μm thickness in a cryostat (HM505E; Carl Zeiss, Jena, Germany). Some fixed tissues were dehydrated in a graded series of alcohol and xylene, embedded in paraffin, and sectioned.
Immunohistochemistry for Light Microscopy
The cryosections or deparaffinized sections were first treated with 0.1% Triton X-100 in PBS and 10% goat serum for 30 min each. They were incubated with the rabbit anti-p55 antibody or anti-4.1B antibody at 4C overnight, and then with biotin-conjugated (Vector Lab.; Burlingame, CA) or AlexaFluor 488-conjugated (Molecular Probes; Eugene, OR) anti-rabbit IgG antibody at room temperature for 1 hr. For the biotinylated primary antibody, sections were then stained with horseradish peroxidase (HRP)-avidin-biotin complex (Vector), and visualized with metal-enhanced diaminobenzidine (DAB) (Pierce; Rockford, IL). Serial sections were immunostained to analyze the relationship between the localization of protein 4.1B and that of p55, because both the anti-4.1B and anti-p55 antibodies originated from rabbits.
Immunoelectron Microscopy
Conventional pre-embedding immunoelectron microscopy was performed as described previously (Terada et al. 2005). Briefly, 6-μm-thick cryosections, which had been immunostained with the primary anti-p55 antibody, were subsequently incubated with biotinylated goat anti-rabbit IgG antibody and then with HRP-avidin-biotin complex (Vector). After being rinsed in PBS, they were fixed again with 0.25% glutaraldehyde in PB for 10 min, signals were visualized using the DAB method (Pierce), and the sections were treated with 1% osmium tetroxide in PB for 5 min. After routine dehydration in a graded series of ethanol, the sections were embedded in epoxy resin by the inverted gelatin capsule method. Ultrathin sections at 70-nm thickness were cut on an ultramicrotome (Ultracut E; Reichert, Germany) and collected on grids. They were stained only with uranyl acetate to identify the localization of DAB reaction products. Finally, they were observed at an acceleration voltage of 75 kV in an electron microscope (H-7500; Hitachi, Tokyo, Japan).
Plasmids, Recombinant Proteins, GST Fusion Protein Pull-down
Rat 4.1B (KIAA0987) (Yamakawa and Ohara 2000) and human NBC1 (Satohetal. 2003) cDNA fragments were cloned in pSPORT (Invitrogen; San Diego, CA) and pcDNA3.1 (Invitrogen), respectively. The fragments, including sequences to produce peptides NBC1 (147NLLEKDY152 in GenBank accession number AF007216) and 4.1B (406IKRKAPFF413 in GenBank accession number AB032827), were produced by polymerase chain reaction (PCR) with LA-Taq DNA polymerase (#RR002A; TaKaRa, Tokyo, Japan), the PCR products were directly cloned into pCR-XL-TOPO (Invitrogen; Carlsbad, CA), and preservation of the target sequences was confirmed. The primers were 5′-GTTCAGATGAATGGGCATACG-3′ (NBC1, sense) and 5′-ATTCAAGCTC TTTCGGCAATT-3′ (NBC1, antisense), corresponding to nucleotides 1254-1274 and 1416-1436 in human kNBC1 mRNA; and 5′-AAAAGGCCTAAAAGCATGCAG-3′ (4.1B, sense) and 5′-AACTGGTTGGACCCTGCTAAG-3′ (4.1B, antisense), corresponding to nucleotides 402-422 and 576-596 in rat 4.1B mRNA. The sequence of BamHI or EcoRI was added to that of the sense and antisense strand, respectively. Then the cloned vectors were cleaved with the restriction enzymes BamHI and EcoRI and cloned into the bacterial expression vector pGEX-6P-1 (GE Healthcare Bio-Science Corp.; Piscataway, NJ) to produce GST-fused NBC1 (V369-L430 in GenBank accession number AF007216) or GST-fused 4.1B (K108-K172 in GenBank accession number AB032827) peptides. GST fusion proteins were expressed in
The pull-down assay was performed as follows. Recombinant NBC1 protein immobilized on glutathione-Sepharose 4B beads (GE Healthcare) was combined with the produced 4.1B, which was cleaved with a protease (ProScission Protease; GE Healthcare) and incubated overnight at 4C. The beads were washed with TENT buffer containing 1% Triton X-100, 140 mM NaCl, 2 mM EDTA, and 20 mM Tris (pH 8.0) three times for 10 min each at 4C. As a control, the cleaved 4.1B protein was mixed with glutathione-Sepharose 4B beads and washed in the same way. The proteins bound to beads were eluted by boiling in Laemmli sample buffer, and subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The gel was then stained with Coomassie brilliant blue.
Immunoprecipitation Experiments
For the immunoprecipitation study for 4.1B-p55 interaction, kidney tissue lysate from nine adult C57BL/6 mice (eighteen kidneys) was incubated overnight at 4C with anti-p55 antibody. The lysate was the supernatant from a mouse kidney homogenized with TENT buffer containing a protease inhibitor cocktail (Sigma; St. Louis, MO) and centrifuged at 10,000 × g for 30 min at 4C. For the immunoprecipitation study to examine the 4.1B-NBC1 interaction, two mice were perfused with PBS via the heart, and the kidney cortex tissues were collected by cutting with a razor blade. The tissues were homogenized in Tris buffer containing 20 mM Tris (pH 7.4), 1 mM EDTA, 150 mM NaCl, and 0.25 M sucrose, and centrifuged at 500 × g, and the supernatant was centrifuged at 105,000 × g. Then the pellet was collected and solubilized in TENT buffer, and incubated overnight at 4C with anti-4.1B or anti-NBC1 antibody.
The following day, immunocomplexes were separated using protein G-Sepharose (GE Healthcare) and the washed beads were boiled in Laemmli sample buffer. Proteins eluted from the beads, together with aliquots of the starting extract, were subjected to SDS-PAGE and Western blotting analysis using anti-NBC1 or anti-4.1B antibody. After incubation with HRP-conjugated anti-rabbit IgG antibody (Vector), the blots were visualized using a chemiluminescent system (Pierce).
Results
p55 Protein Immunolocalization Along the Basolateral Membrane of the Proximal Tubules
Using a specific antibody against p55, an immunohistochemical study was performed in adult mouse kidney (Figure 1). p55 was immunolocalized in the renal cortex, but not in the medulla (Figures 1A-1F). The renal tubules having prominent apical microvilli were immunostained, indicating that they were proximal convoluted tubules (Figures 1G and 1H). The immunostaining was localized along the basolateral membranes (Figure 1I). By immunoelectron microscopy, the immunoreaction products were shown to be located in the basal infoldings, as well as the basolateral plasma membranes (Figure 1J). These results indicate that p55 is predominantly located on the basolateral membrane and its submembrane domain in the proximal tubules.
p55 Localization in Postnatal Mouse Renal Development
In the next series of experiments, we investigated the ontogeny of p55 expression and localization. Accordingly, immunostaining was performed in postnatal day 2 (P2), P4, P10, and P20 mouse kidneys (Figure 2), because it was reported that the proximal tubules mature after birth, especially near the renal capsule (Iino et al. 2001; Bonnici and Wagner 2004). In the P2 mouse renal cortex, p55 immunostaining was weakly observed in some tubules (Figure 2A), and the staining pattern was diffuse in the cytoplasm. In the P4 and P10 renal cortex, the intensity of the staining was increased compared with that on P2 (Figures 2B and 2C). The staining was still localized in the cytoplasm. The immunostaining pattern of p55 in the P20 renal cortex was basolateral (Figure 2D), which was similar to that in adult mice, as shown in Figure 1. These findings indicate that the p55 distribution in the basolateral membrane correlates with the maturation of the renal proximal tubules.
p55 and 4.1B Colocalize and Form a Complex in the S1-S2 Proximal Tubules
We previously reported that 4.1B is immunolocalized in the S1-S2 proximal convoluted tubules (Terada et al. 2004b). To evaluate the relative distribution of 4.1B and p55, immunostaining for 4.1B and p55 was performed in adjacent serial sections (Figure 3). The tubules stained with the anti-p55 antibody (Figures 3A and 3B) had a staining pattern similar to those stained with the anti-4.1B antibody (Figures 3C and 3D). This indicates that 4.1B and p55 are localized on the basolateral membranes of the same S1-S2 proximal convoluted tubules.
To further evaluate the molecular interaction of 4.1B and p55, immunoprecipitation studies were performed (Figure 3E). As indicated, renal tissue lysate immunoprecipitated with the anti-p55 antibody was positively blotted with the anti-4.1B antibody (arrow in Figure 3E). This suggests that p55 and 4.1B form a molecular complex in the S1-S2 renal proximal tubules.
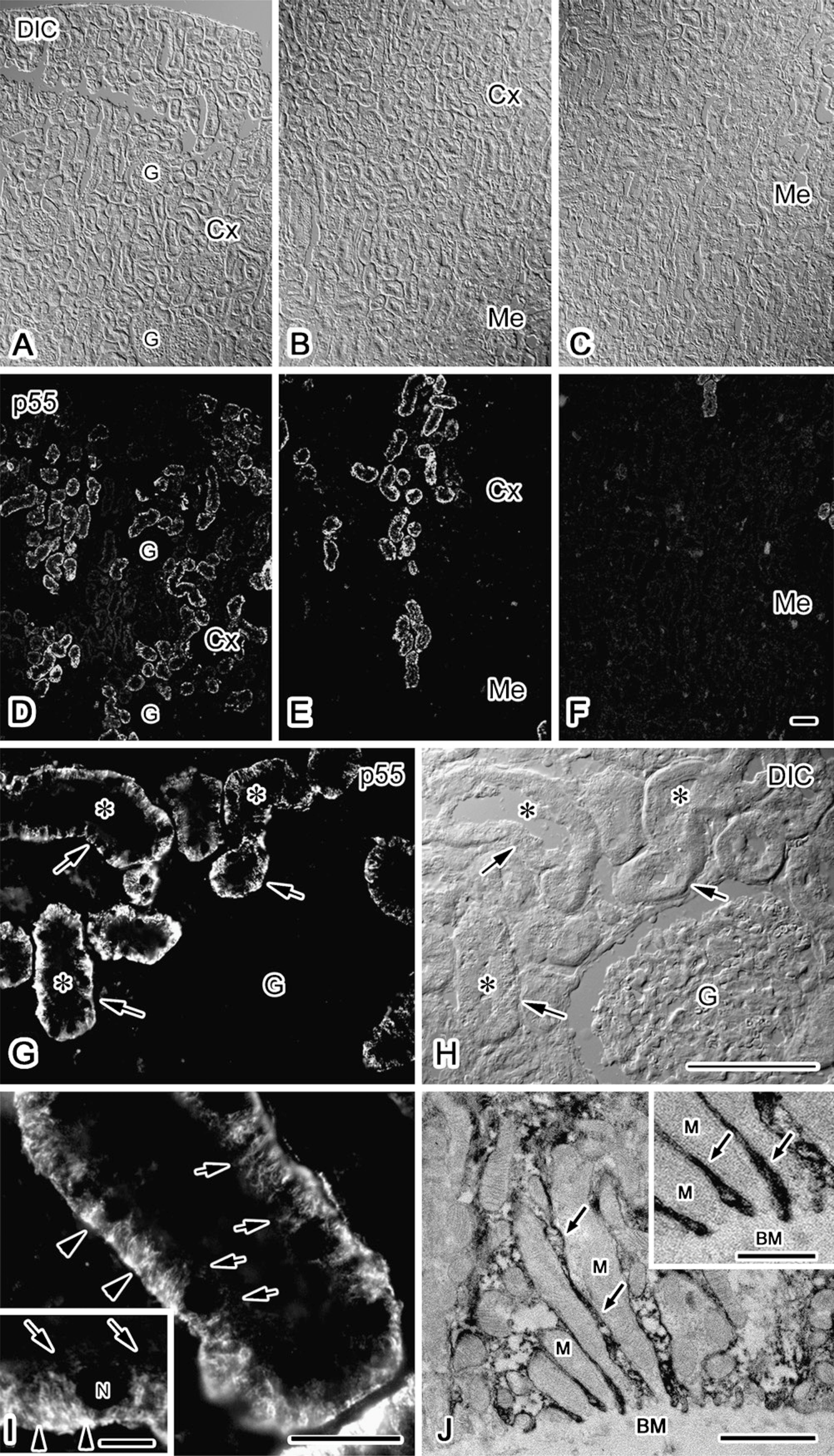
Immunolocalization of p55 in the adult mouse kidney. p55 is immunostained in the renal tubules in the cortex (Cx in
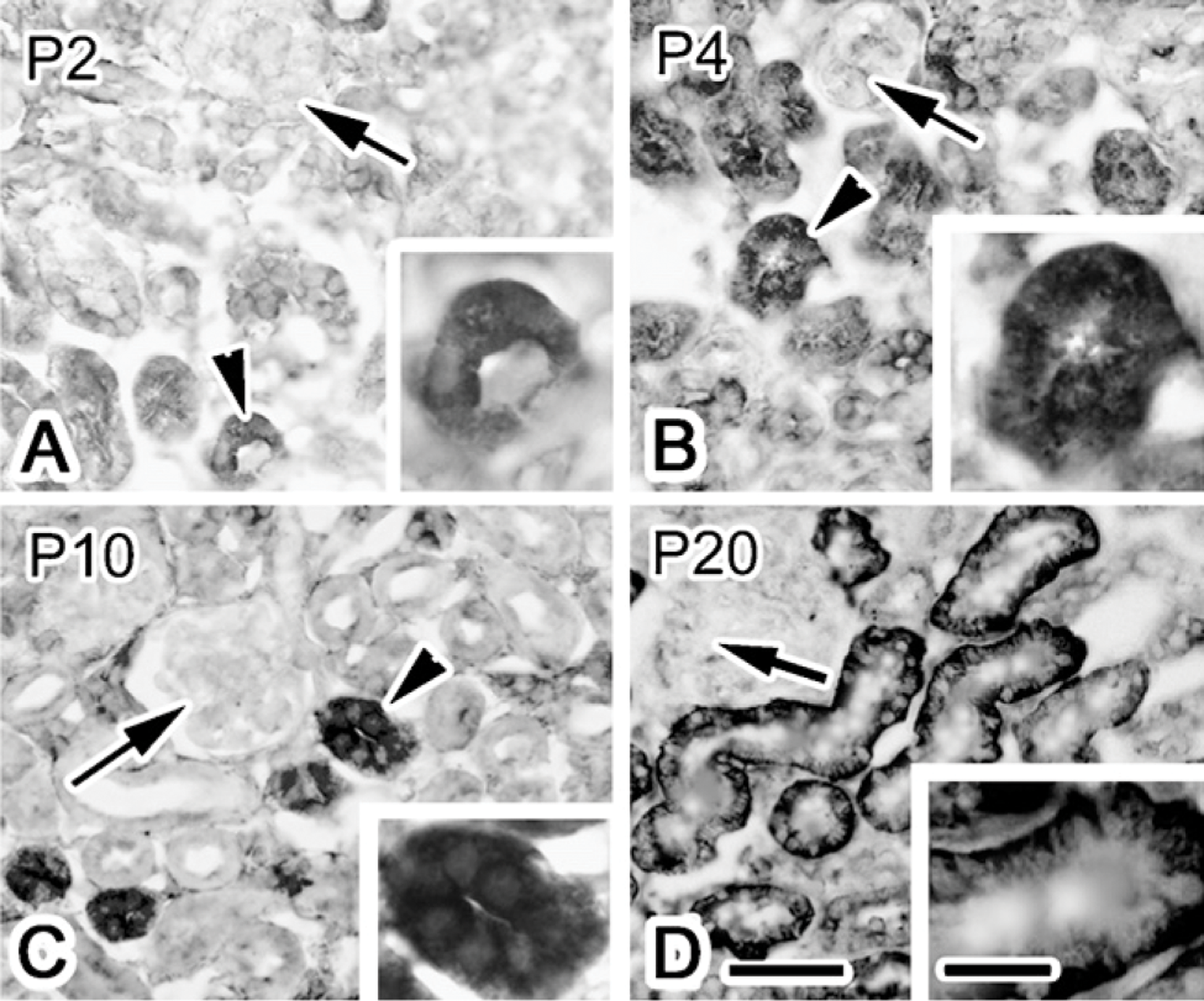
Immunolocalization of p55 in the renal cortex of postnatal day 2, 4, 10, and 20 (P2, P4, P10, and P20) mice, visualized with DAB detection. On day 2, 4, and 10, p55 is immunostained in the cytoplasm (arrowheads in
Molecular Complex of Protein 4.1B and NBC1 in the Kidney
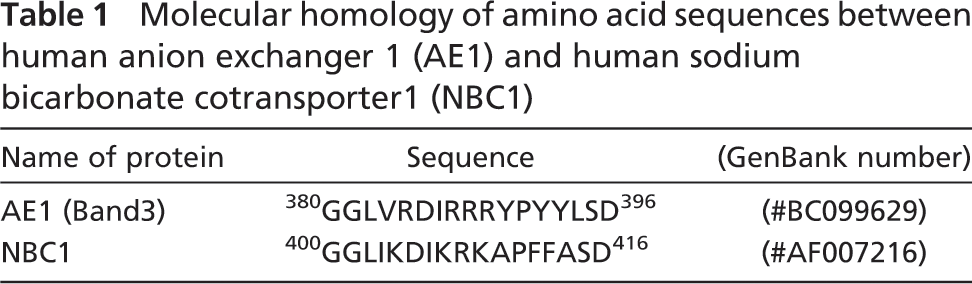
NBC1 is known to localize in the S1-S2 proximal tubules (Soleimani 2001), and displays an immunostaining pattern that is identical to that of 4.1B (Terada et al. 2004b). It was reported that AE1 interacts with 4.1R (Jons and Drenckhahn 1992), and that both AE1 and NBC1 belong to the superfamily of SLC4 bicarbonate transporters (Soleimani 2001; Alper et al. 2002; Romero et al. 2004; Pushkin and Kurtz 2006). They display significant overall structural homology, with highly similar stretches of amino acid fragments, as shown in Table 1. We therefore synthesized GST fusion proteins for NBC1 and 4.1B, as described in Materials and Methods. NBC1-GST fusion protein was immobilized on glutathione-Sepharose beads and mixed with 4.1B protein that had been cleaved from 4.1B-GST fusion protein. The results demonstrate that the immobilized NBC1 protein binds with the 4.1B peptide [arrow with “4.1B (Pro)” in Figures 4A and 4B].
To further elucidate whether 4.1B and NBC1 interact in situ, solubilized mouse kidney cortex proteins were analyzed (Figures 5A and 5B). The tissue lysates were prepared, immunoprecipitated with anti-4.1B and anti-NBC1 antibodies, and stained with antibodies directed against NBC1 and 4.1B, respectively. As shown, both immunoprecipitants were blotted with anti-NBC1 and anti-4.1B antibodies (Figures 5A and 5B, respectively). Coupled with a previous report demonstrating the co-localization of NBC1 and 4.1B on the basolateral membrane of the kidney proximal tubule (Terada et al. 2004b), these results indicate that NBC1 and 4.1B form a molecular complex in the S1-S2 renal proximal tubules.
Discussion
The kidney proximal tubule plays an essential role in HCO3 - reabsorption and systemic acid-base homeostasis (Soleimani 2001; Romero et al. 2004). It is estimated that the proximal tubule reabsorbs ∼90% of the filtered HCO3 - and 70% of the filtered Na+. These functions are achieved through coordinated function of several ion transporters (Boron et al. 1997). In the S3 segment, the efflux of HCO3 - across the basolateral membrane is mediated mainly by AEs (Boron et al. 1997). However, in the S2 and S1 segments of the proximal tubule, the relative contribution of AEs decreases and that of NBC1 increases (Kondo and Fromter 1990). In this report, we presented data that protein 4.1B and NBC1 form molecular complexes in the S1-S2 renal proximal tubules. This indicates that membrane skeletal proteins, including 4.1B, may play a role in the regulation of NBC1. Previous studies have demonstrated that synthetic IRRRY displaced the binding of 4.1R to AE1 (Jons and Drenckhahn 1992); one of the NBC1 predicted binding sites for 4.1B is the homologous amino acid sequence “IKRKA,” as indicated in Table 1.
Published reports indicate the presence of regulatory factors in renal NBC1 activity. Angiotensin-II and acute acidosis were reported to increase the NBC1 activity by Src family kinase and proline-rich tyrosine kinase 2 (Pyk2) coupling (Espiritu et al. 2002), indicating a role for Pyk2 in acid-base homeostasis (Gluck 2004). Interestingly, Pyk2 has the FERM domain, which was originally identified in 4.1 (four-one), ezrin, radixin, and moesin proteins (Lipinski et al. 2006). It has also been shown that carbonic anhydrase-II binds in vitro to the C terminus of kidney NBC1, indicating its function as a transport metabolon (Pushkin et al. 2004). On the other hand, dopamine was reported to be an inhibitor of renal NBC1 (Kunimi et al. 2000; Robey et al. 2001). Molecular interaction between dopamine receptor and 4.1N, a member of the 4.1 family, at the neuronal plasma membrane was recently reported (Binda et al. 2002). Although the interaction of dopamine receptors with 4.1B has not been examined in the renal proximal tubule, it is plausible that a large molecular complex of membrane skeleton proteins exists that acts to localize such transmembrane proteins. An intriguing question, of course, pertains to the role of membrane skeleton in the regulation of ion transporters in the basolateral membrane of the proximal tubule. In skate erythrocytes, volume expansion modulated 4.1R binding (Musch and Goldstein 2001). In that experiment, microsomal membranes isolated from erythrocytes under volume-expanded conditions demonstrated a significant decrease in the amount of 4.1R that was bound to AE1, probably through competitive attachment to ankyrin. Although membrane skeletal molecules such as ankyrin, spectrin, and/or actin have not been convincingly identified in the basolateral domain of the renal proximal tubules, membrane skeletal molecular complexes might sense the cell volume as well as ion gradients, as reported in erythrocytes. Mass spectrometry of proteins that are immunoprecipitated by NBC1, p55, or 4.1B may yield new insight into the identity of other proteins that are involved in the signaling/transport complex.
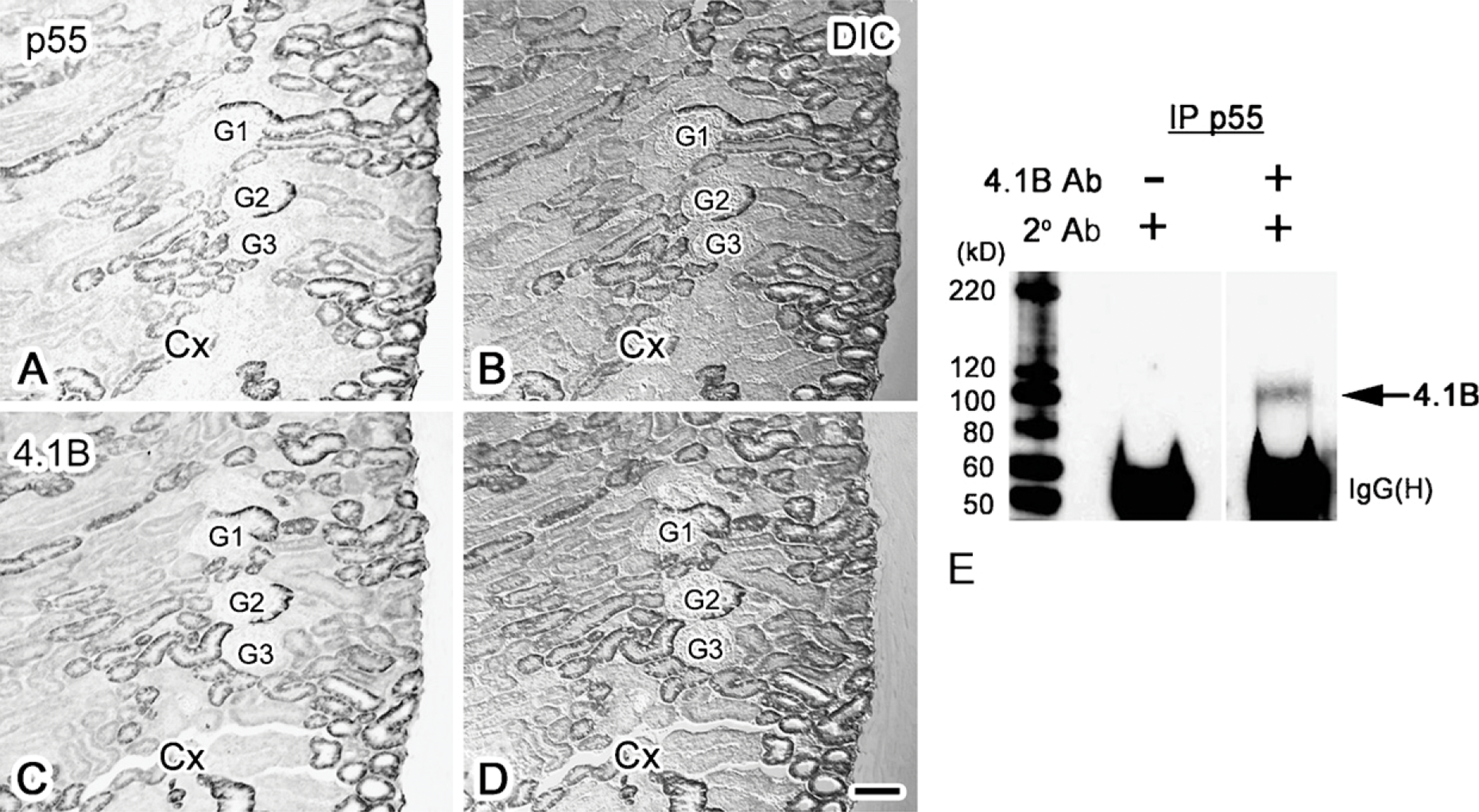
Immunolocalization of p55 (
Molecular homology of amino acid sequences between human anion exchanger 1 (AE1) and human sodium bicarbonate cotransporter1 (NBC1)
Our results demonstrated that p55, a MAGUK protein, was localized in the basolateral membrane of S1-S2 proximal tubules, and interacted with 4.1B. The MAGUKs are thought to localize in and be required for the formation of several types of cell junctions (Dimitratos et al. 1999). They are also presumed to be related to trafficking of signaling molecules by kinesin motors (Schnapp 2003). p55 was first characterized in erythrocytes (Ruff et al. 1991); however, it is shown to be present in other tissues, such as the postsynaptic density (PSD), where it colocalizes with various PSD proteins, such as PSD-95 and calmodulin-associated serine/threonine kinase (CASK) (Jing-Ping et al. 2005). A member of the MAGUKs is the discs large-1 (Dlg-1), which was recently reported to be localized in the basolateral membranes of the developing mouse kidney (Naim et al. 2005). Dlg-1 was shown to be expressed in ureteric and mesenchyme-derived epithelial cells during kidney development, suggesting its role in the mesenchyme-to-epithelial transition. Another MAGUK protein, mlin-7, was also reported to be localized to the basolateral membrane with a broad nephron distribution in the inner and outer medulla (Straight et al. 2000). It is reported that the main bicarbonate-absorbing transporters in the kidney proximal tubule, namely NHE3 and NBC1, display progressive postnatal maturation, with almost fully developed capacity at 20 days after birth (Bonnici and Wagner 2004). In the present studies, p55 was found to be expressed in the cytoplasm in renal proximal tubules immediately after birth, but was detected in the basolateral membrane at postnatal day 20, suggesting that p55 may play a role in the maturation of the proximal tubule. Alternatively, it is plausible that coordinated maturation of NBC1 and p55 reflects the development of acidifying mechanisms in the proximal tubule. With the association of several MAGUKs, such as hCASK and hDlg, reported in certain epithelial cells (Nix et al. 2000), it will be interesting to examine the ontogeny and role of MAGUKs, such as p55 and Dlg-1, in renal proximal tubule maturation and function.
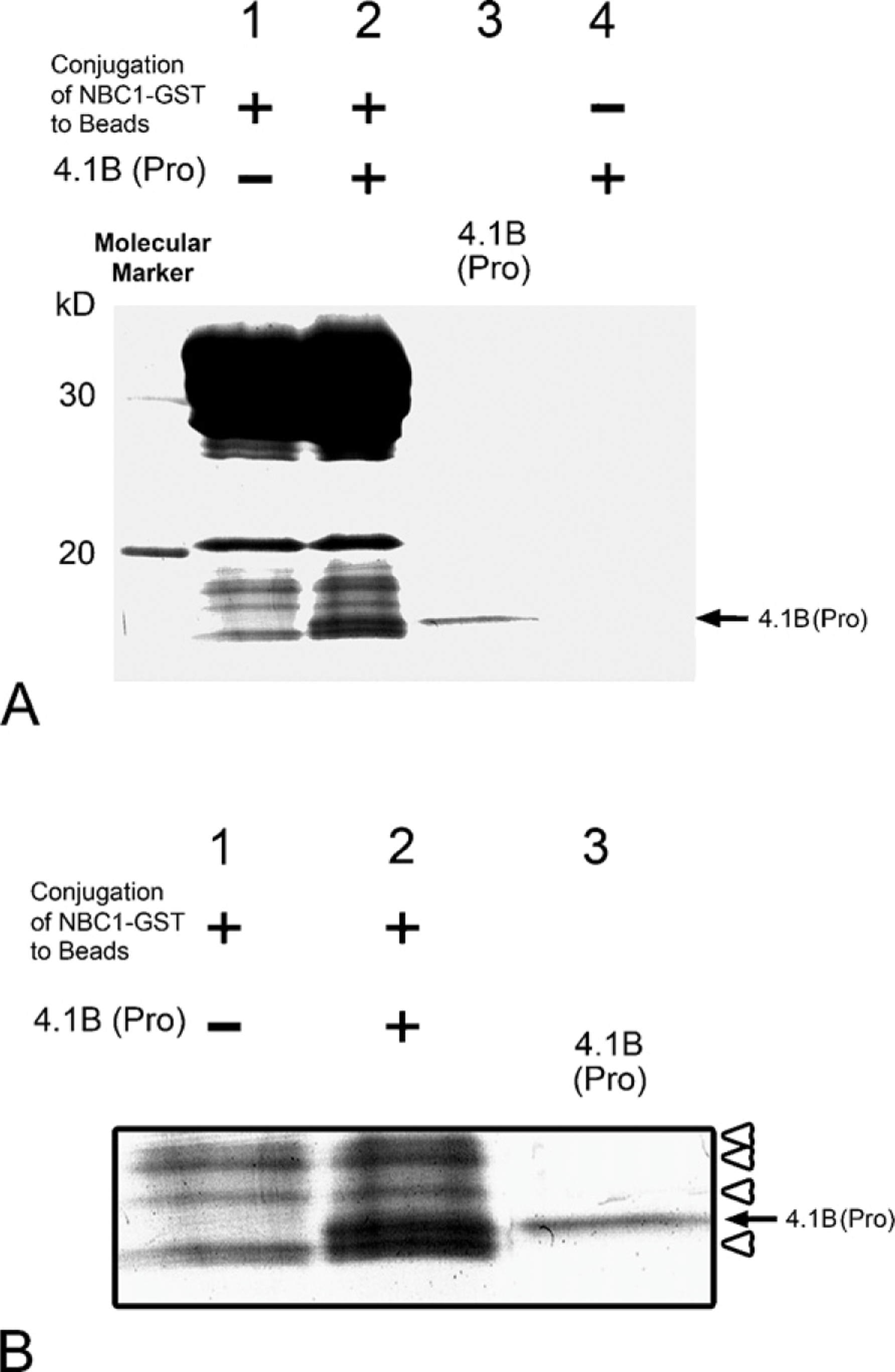
Interaction of 4.1B and NBC1. (
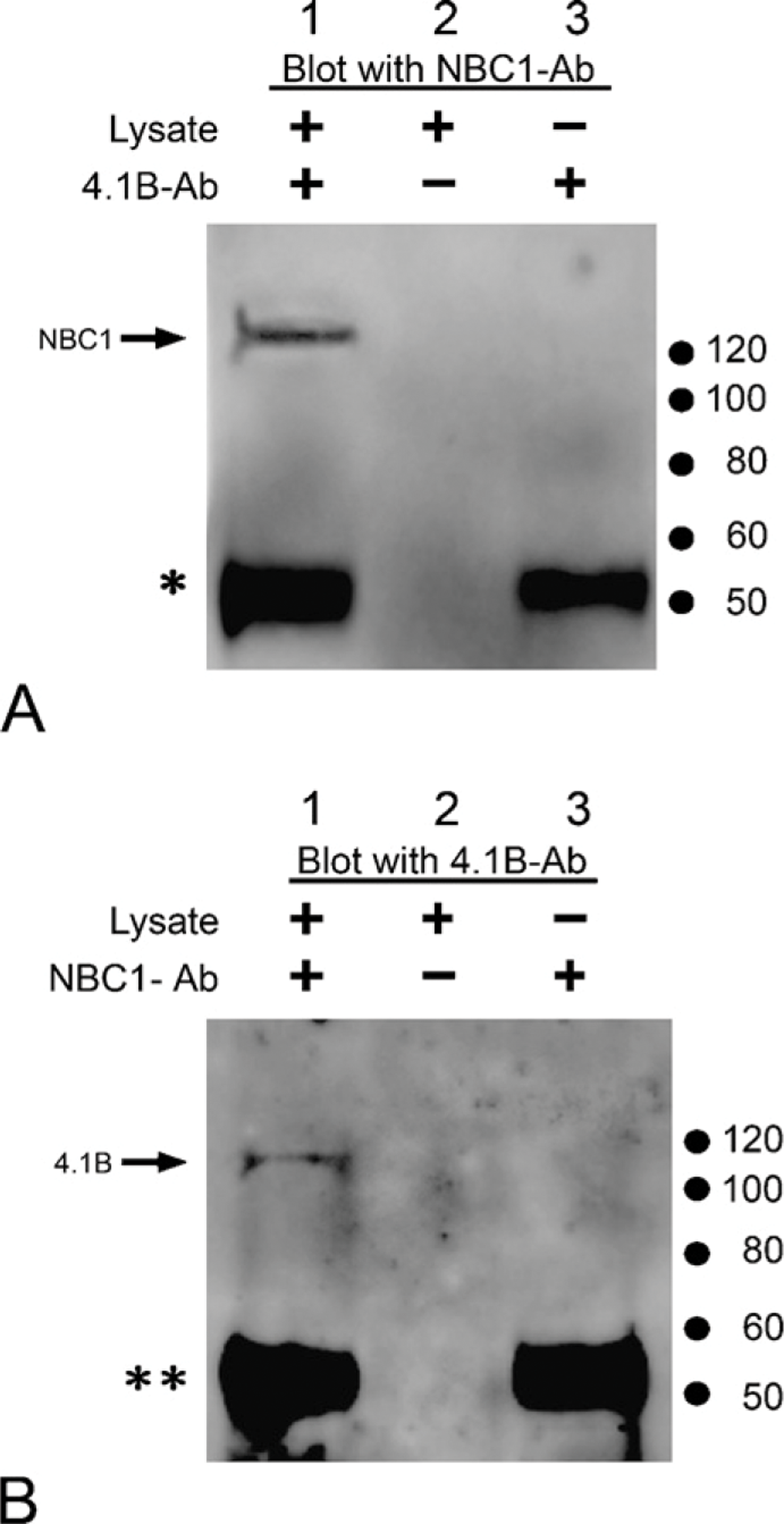
Immunoprecipitation of mouse kidney lysate using anti-4.1B antibody (4.1B-Ab) (
Footnotes
Acknowledgments
This work was partially supported by a grant from the Ministry of Education, Science, Sports and Culture of Japan (No.18590181) to N.T.
