Abstract
Single antigen-targeted intraperitoneal radioimmunotherapy for ovarian cancer has shown limited success. Due to the heterogeneous expression of tumor antigens on cancer cells, a multi-antigen targeting approach appears logical to augment the therapeutic efficacy of antibody-guided therapy. In the interest of developing this novel approach, ovarian cancer tissue microarray slides containing cancer and benign/non-neoplastic tissue samples (n = 92) were processed for single-, double-, and triple-antigen labeling using antibodies for the tumor-associated antigens TAG-72, MUC1, and CA125. Among all ovarian cancer types, 72%, 61%, and 50% of the samples showed immunolabeling for TAG-72, MUC1, and CA125, respectively. Expression level of these antigens was significantly (p<0.005) higher in advanced stage carcinomas compared with early stage. Of the 48 epithelial ovarian cancer samples, individual anti-TAG-72, MUC1, and CA125 antibody probing showed labeling in 89.5%, 87.5%, and 73.0% of the cases, respectively. In the majority of the cancer samples (>70%), a heterogeneous labeling pattern was observed (only 30–40% of the cancer cells within the sample were labeled). However, upon combining the three antigens (triple-antigen labeling), 98% of the epithelial ovarian cancer samples were labeled and >95% of the cancer cells within each sample were labeled. Our data indicate that the heterogeneous expression of cancer antigens appears to be a major obstacle in antibody-guided therapy, and this can be overcome by multiple antigen targeting. Therapeutic efficacy of antibody-guided therapy for ovarian cancer treatment will be enhanced by the combined targeting of TAG-72, MUC1, and CA125.
O
Radioimmunotherapy (RIT), the use of radioisotopes combined with monoclonal antibodies (MAbs), has shown promising results in the effective treatment of hematological malignancies, especially non-Hodgkin's lymphomas (NHL) (DeNardo et al. 1999, 2001). Due to its success in the last few years and the increasing interest of clinical oncologists, RIT is a rapidly emerging therapeutic modality for the treatment of a wide variety of carcinomas (Sharkey et al. 1993; DeNardo et al. 1999). The targeted nature of RIT offers an edge over non-targeted therapies and holds promise of a greater efficacy with less normal cell/organ toxicity (Goldenberg 2002; Milenic et al. 2004a). This approach combines the advantages of specific cancer cell targeting by the use of MAbs against tumor-associated antigens (TAAs) with the cytotoxic properties of therapeutic radionuclides. In addition to targeting both known and esoteric malignant lesions, RIT permits the maximum eradication of cancer cells through a bystander radiation effect and may be more effective in drug-resistant tumors (Batra et al. 2002; Goldenberg 2002; Milenic et al. 2004b). Intraperitoneal radioimmunotherapy (IRIT) directed against single antigens has shown only limited success in ovarian cancer treatment (Goldenberg 2002; Milenic et al. 2004a, b, 2005), largely due to the heterogeneous expression of the targeted TAAs by ovarian cancer cells. Focal and heterogeneous expression of target antigens on individual cells within ovarian tumors may be the main reason for the suboptimal response of single-antigen IRIT because a large number of cancer cells remain untargeted, allowing for disease relapse soon after treatment. To prevail over the heterogenic expression of TAAs by ovarian cancer cells, developing a multi-antigen targeting approach using antibodies against different tumor antigens will be a necessary choice.
CA125, which is in clinical use as an ovarian cancer marker, lacks sensitivity in 20% of ovarian cancers, and Rosen et al. (2005) recently documented the need for additional complementary markers to increase sensitivity of ovarian cancer screening. Likewise, successful IRIT will also require the use of multiple antigen targets. TAAs TAG-72, MUC1, and CA125 are known to be overexpressed in ovarian cancer (Thor et al. 1986, 1987; Giuntoli et al. 1998; Bast et al. 2005; Rosen et al. 2005) and may prove to be useful targets for a multi-targeted IRIT. In this study, we analyzed the individual and combined targeting potential of TAG-72, MUC1, and CA125 in ovarian cancer. To determine their combined targeting potential, we performed double and triple staining on ovarian cancer tissues. Our study clearly demonstrates that a combination of TAG-72, MUC1, and CA125 labeled 98% of ovarian cancer samples with almost 100% labeling of cancer cells within each cancer sample. These data suggest the requirement of a multi-targeting approach to improve the efficacy of antibody-guided therapy. To the best of our knowledge, this is the first report to study the combined expression of TAG-72, MUC1, and CA125 with multi-antigen staining procedures to show that a multi-targeting approach may improve the efficacy of IRIT for ovarian cancer treatment.
Materials and Methods
Clinical Samples
Tissue microarray slides (AccuMax Array, ISU ABXIS Co.; Seoul, Korea) contained 72 malignant (48 epithelial ovarian cancer and 24 non-epithelial ovarian cancer) and 20 non-neoplastic (5 normal and 15 benign) ovarian tissue samples. Histological diagnoses of the ovarian cancer samples were provided along with the tissue microarray slides.
Immunohistochemistry (IHC)
Microarray slides were stained with anti-human MUC1, TAG 72, and CA125 MAbs by heat-induced epitope retrieval IHC technique using a Vector ABC kit (Vector Laboratories; Burlingame, CA) as described earlier (Chauhan et al. 2006). Briefly, tumor tissue sections on the slide were deparaffinized using EZ-DeWax (Bio Genex; San Ramon, CA) and rehydrated with graded alcohols. Heat-induced antigen retrieval was performed in hot citrate buffer (pH 6.0) while heated in a microwave oven at 700 W for 15 min. Tissue samples were washed and incubated with 0.3% H2O2 methanol: PBS solution for 30 min to quench endogenous peroxidase activity. Tissue sections were incubated in diluted Vectastain normal serum (provided in the kit) for 30 min followed by primary antibody [1:500 dilution of anti-MUC1 (PAb) or 1:200 dilution of anti-TAG-72 (CC49); Lab Vision Corporation, Fremont, CA or 1:200 dilution of anti-CA125 (OC125); Axxora Platform, San Diego, CA] incubation at room temperature for 1 hr. After washing with PBS containing 0.05% Tween-20 (PBS-T), samples were incubated with secondary antibody for 30 min and washed again with PBS-T before incubation with ABC solution. IHC reaction color was developed by treating tissue sections with 3,3′-diaminobenzidine substrate (DAB substrate kit; Vector Laboratories) according to the manufacturer's instructions. A reddish-brown precipitate indicated positive immunoreactivity. Slides were washed with water, counterstained with hematoxylin, dehydrated, and mounted with Vectamount permanent mounting media (Vector Laboratories).
Double- and Triple-antigen Labeling of Tissue Microarray Slides
Microarray slides were deparaffinized using EZ-DeWax (Bio-Genex) and rehydrated using graded alcohols. Slides were washed in deionized water for 10 min, rinsed in 1X TBS, incubated in freshly prepared 0.3% H2O2, and washed again in 1X TBS. Heat-induced antigen retrieval was performed in hot citrate buffer (pH 6.0) while heated in a microwave oven at 700 W for 15 min. After cooling, slides were washed with TBS, and tissue sections were incubated in Protein Blocking SNIPER (Biocare Double Stain Kit; Biocare Medical, Concord, CA) for 10 min to block nonspecific antigen–antibody immunoreactivity. Slides were then incubated with a primary antibody cocktail containing a 1:500 dilution of MUC1 antibody (PAb) and 1:200 dilution of TAG-72 antibody (CC49) at room temperature for 1 hr. After washing with 1X TBS, slides were incubated with the secondary antibody cocktail containing anti-mouse–HRP and anti-rabbit-ALP (both included with the Biocare Double stain kit) at room temperature for 45 min and then washed in 1X TBS. HRP reaction color was developed by incubating the slides for 5 min with the DAB substrate (Betazoid DAB kit; Biocare Medical) according to the manufacturer's instructions. Following a wash in water, the slides were incubated with Vulcan Fast Red (Biocare Medical) for 5 min to develop the reaction color from the ALP and then washed in water. To prepare for the third label, the slides were washed again in 1X TBS, incubated for 4 min in denaturing solution (Biocare Medical), washed in 1X TBS, and incubated in Protein Blocking SNIPER for 10 min. After that, slides were incubated with anti-CA125 (OC125) antibody for 1 hr at room temperature, washed in 1X TBS, and incubated with the secondary antibody (anti-mouse-HRP; Biocare Medical) for 1 hr. Following washing with TBS-T, Bajoran purple (Biocare Medical) was applied for 8–12 min. The slides were rinsed with water, counterstained with hematoxylin, dehydrated, and mounted with Vectamount (Vector Laboratories) permanent mounting media.
Scoring Criteria
All slides were analyzed using an Olympus BX 41 microscope (Olympus; Tokyo, Japan). Intensity of immunoreactivity of the MUC1, TAG-72, and CA125 was independently scored by an experienced pathologist (MDK). Staining intensity was graded on a scale from 0–3 (i.e., 0 for no staining, 1+ for weak immunoreactivity, 2+ for moderate immunoreactivity, and 3+ for strong immunoreactivity). Labeling extent was scored as follows: <25% of tumor cells labeled (1); 26–50% of the tumor cells labeled (2), 51–75% of the tumor cells labeled (3), and >75% of the tumor cells labeled (4). Intensity and extent of labeling scores were multiplied to obtain the composite score with the maximum value being 12.
Immunolabeling profiles of TAG-72, MUC1, and CA125 in all ovarian tissue samples (n = 92) a
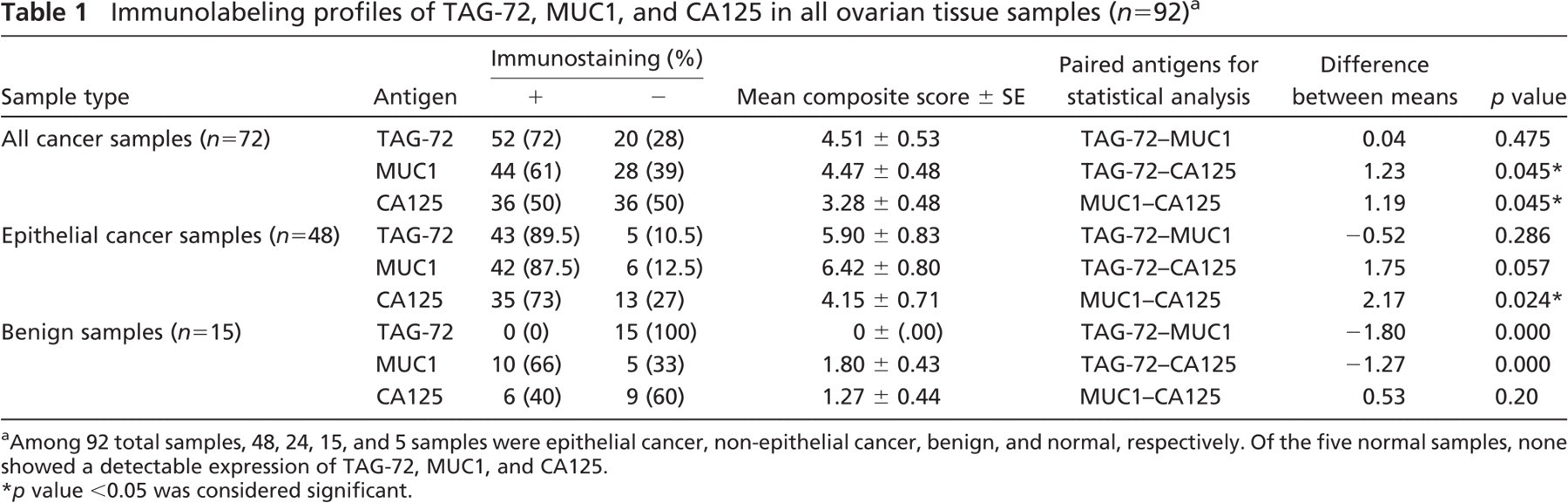
Among 92 total samples, 48, 24, 15, and 5 samples were epithelial cancer, non-epithelial cancer, benign, and normal, respectively. Of the five normal samples, none showed a detectable expression of TAG-72, MUC1, and CA125.
p value <0.05 was considered significant.
Statistical Analysis
Paired t-tests were used to analyze the labeling sensitivity of TAG-72, MUC1, and CA125 in early and advanced stage ovarian cancer samples; p values <0.05 were considered to be statistically significant.
Results
Higher Immunolabeling of TAG-72, MUC1, and CA125 in Ovarian Cancer as Compared With Benign/Normal Ovary
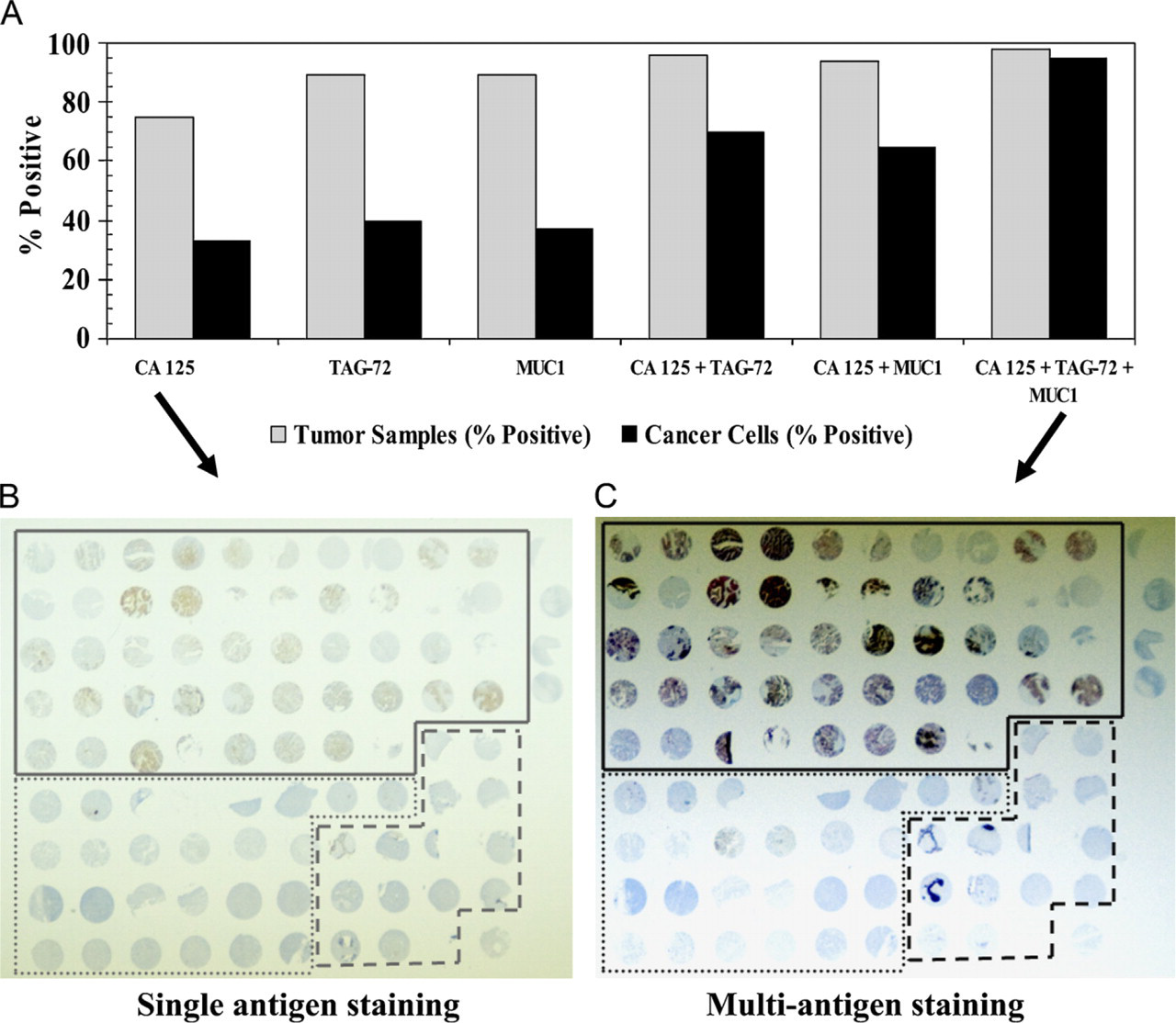
Labeling sensitivity of TAG-72, MUC1, and CA125 was analyzed on tissue microarray slides using IHC. Of 92 ovarian tissue samples, 72 spots were malignant tissues, 15 spots were benign, and 5 spots were normal tissue samples. TAG-72, MUC1, and CA125 labeling were observed in 72% (n = 52), 61% (n = 44), and 50% (n = 36) of the malignant samples, respectively, whereas none of the normal samples was labeled by TAG-72, MUC1, or CA125 (n = 5). Intensity and percentage of stained cancer cells was scored by an experienced pathologist (MDK) to generate mean composite staining scores. As expected, the mean composite staining scores for all targets were much higher in malignant samples than scores for normal or benign samples. TAG-72 was not detected in any of the benign ovarian tissue samples (n = 15). However, MUC1 and CA125 expression was observed at low levels in benign ovarian tissue samples (Table 1). In comparing extent and intensity of labeling between targets, mean composite staining scores of TAG-72 (4.51 ± 0.53) and MUC1 (4.47 ± 0.48) were significantly higher compared with CA125 (3.28 ± 0.48, p<0.05; Table 1).
Tissue microarray slides contained 48 epithelial (mucinous, clear cell, transitional cell, and endometroid) and 24 non-epithelial types of carcinomas. Among the 48 epithelial ovarian cancer samples, TAG-72, MUC1, and CA125 labeled 89.5%, 87.5%, and 73.0% of the cases, respectively. Additionally, mean composite staining scores of the epithelial cancer samples were higher than the scores attained for all types of ovarian cancer. For the ovarian epithelial cancers, mean composite staining score of MUC1 (6.42 ± 0.80) was significantly higher than CA125 (4.15 ± 0.71, p<0.05; Table 1). Of the 24 non-epithelial cancers (Brenner, yolk sac, granulosa, and dysgerminoma, n = 6 of each type), TAG-72 showed a positive reactivity in 100% of the dysgerminoma (n = 6) and 16.6% of the Brenner samples (n = 1), whereas MUC1 showed a detectable labeling in only 66.6% of the Brenner samples (n = 4). CA125 labeling was detectable in only 16.6% of each Brenner (n = 1) and granulosa (n = 1) tumor samples.
Prominent Immunolabeling of TAG-72, MUC1, and CA125 in Advanced Cancer Stage
Immunolabeling of TAG-72, MUC1, and CA125 was comparatively analyzed in early and advanced stage ovarian cancer samples (Table 2). In early stage samples, TAG-72, MUC1, and CA125 labeling was detected in 53%, 53%, and 37% of the samples, respectively. However, in advanced stage samples, TAG-72, MUC1, and CA125 immunolabeling was observed in a higher number of the samples (94%, 71%, and 65%, respectively) (Table 2). Mean composite labeling scores of TAG-72, MUC1, and CA125 were significantly higher (p<0.005) in advanced stage samples as compared with the early stage samples (Table 2). Additionally, in early stage samples the mean composite labeling scores of MUC1 and TAG-72 were markedly higher as compared with CA125. These data suggest a positive correlation between high immunolabeling of TAG-72, MUC1, and CA125 with advanced stage ovarian cancer and indicate that MUC1 and TAG-72 label early stage samples more extensively and intensely than CA125.
Immunolabeling of TAG-72, MUC1, and CA125 in Epithelial Ovarian Cancer Samples
In 90% of cases, ovarian cancer originates from the ovarian surface epithelium; therefore, epithelial types of ovarian carcinoma (EOC) are most common. Based on the cellular morphology, epithelial cancer samples were grouped into five major histological types: serous, mucinous, clear cell, transitional cell, and endometroid. To be an effective target antigen for radioimmunotherapy, protein expression level should be high enough to result in +2 or +3 labeling intensity. Among EOCs, TAG-72, MUC1, and CA125 showed +2 and +3 labeling intensity in 89.5%, 87.5%, and 73% of the samples, respectively (Table 3). Among different histological types of EOCs, TAG-72 showed immunolabeling in 100% of serous and transitional types of samples. MUC1 was detected in 100% of transitional and endometroid types of samples. CA125 showed maximum (90%) labeling in clear cell samples and labeled only 40% of the mucinous samples (Table 3).
Comparative immunolabeling profiles of TAG-72, MUC1, and CA125 in early (stages I–II) vs advanced (stages III–IV) stage ovarian cancer samples (n = 72) a
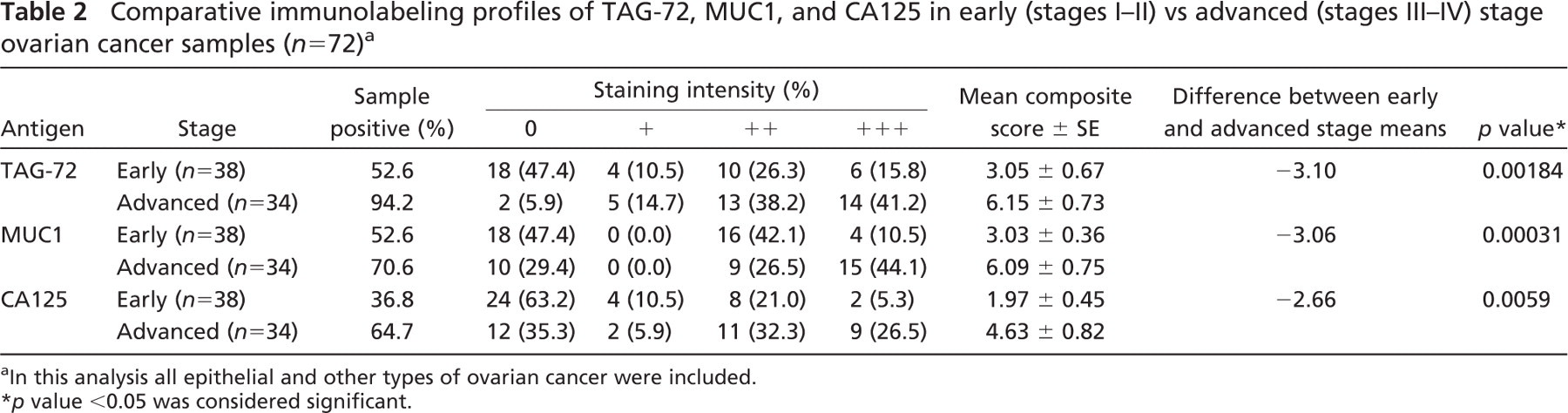
In this analysis all epithelial and other types of ovarian cancer were included.
p value <0.05 was considered significant.
Heterogeneous Labeling of TAG-72, MUC1, and CA125 in Ovarian Cancer
We have observed labeling of TAG-72, MUC1, and CA125 in 75–89% of the EOC samples (Table 3). Additionally, only 30–40% of the cancer cells were immunolabeled within the majority of the cancer samples (>70%). Figure 1 shows representative examples of the heterogeneous immunolabeling pattern of TAG-72, MUC1, and CA125 in different epithelial ovarian cancer types. None of the cancer antigens showed a homogeneous expression pattern in >20% of the cancer samples. These data clearly suggest that an antibody-guided therapy using only a single target antigen will have limited therapeutic efficacy because only a fraction of cancer cells in the tumor are targeted.
Multi-antigen Staining Procedure Labeled a Markedly Higher Percentage of Cancer Cells in Tumors
IRIT studies for ovarian cancer treatment have shown limited therapeutic benefit, possibly due to heterogeneous expression of the single target antigen by ovarian cancer cells. Therefore, single antigen-targeted IRIT fails to target a significant number of ovarian cancer cells. A multi-antigen targeting approach for IRIT procedures appears to be logical to overcome the heterogeneous expression of the targeting antigen. Accordingly, we sought to determine the combined immunolabeling of TAG-72, MUC1, and CA125 in ovarian cancer tissues. An irregular and heterogeneous labeling of CA125, MUC1, and TAG-72 is clearly evident in double and triple immunolabeling (Figure 2). In double-staining procedures, a combination of MUC1 + TAG-72 and MUC1 + CA125 labeled a markedly higher percentage of cells than the single antigen-staining procedure (Figures 2A and 2B and Figure 3). Moreover, triple staining (labeling three antigens on the same sample) achieved >95% labeling of ovarian cancer cells in 98% of epithelial ovarian cancer samples (Figures 2C–2E and Figure 3; Table 3). Multi-antigen labeling certainly stained a higher percentage of ovarian cancer cells, and these data clearly suggest the requirement of multi-antigen targeting approaches to effectively target and obliterate the majority of cells within a cancerous tumor.
Labeling efficacy of TAG-72, MUC1, and CA125 in different histological types of epithelial ovarian cancer samples (n = 48), alone and in combination a
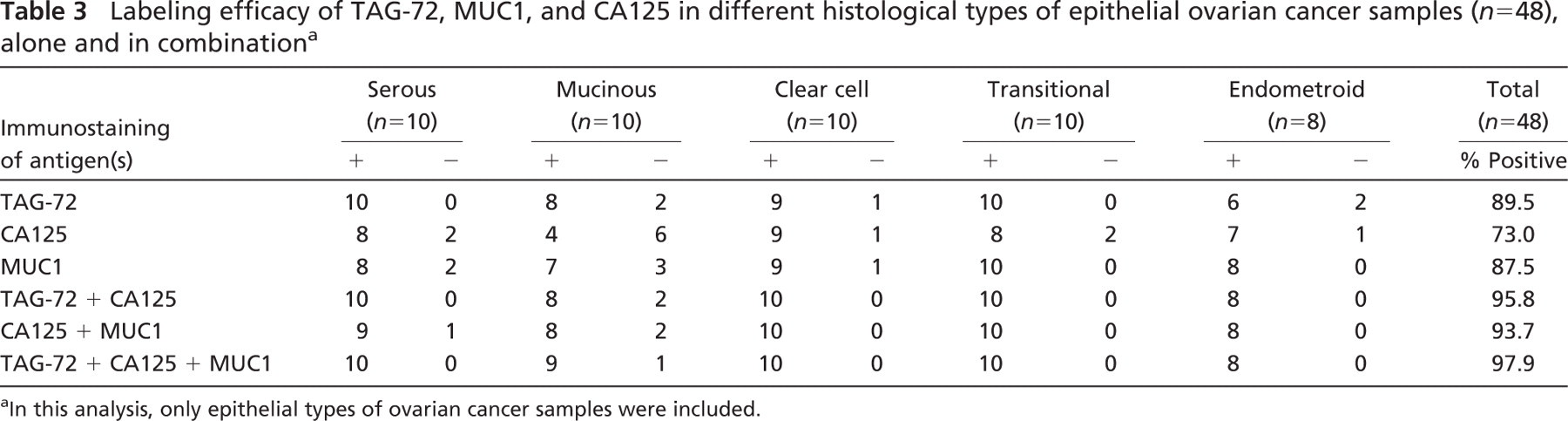
In this analysis, only epithelial types of ovarian cancer samples were included.
Discussion
Significant advancements have been achieved in the field of cancer therapeutics in recent years; however, the survival of ovarian cancer patients has increased only moderately during the past several decades (Jemal et al. 2006). This is primarily due to the inability to diagnose ovarian cancer at an early stage and to the lack of an effective therapeutic modality for advanced stage ovarian cancer treatment (Bast et al. 2005). Due to aggressive intraperitoneal metastatic seeding and the development of chemoresistance by the ovarian cancer cells, the efficacy of standard treatments (debulking surgery and chemotherapy) is very limited in its ability to provide complete disease remission and cancer-free survival (Armstrong 2002). Development of antibody-guided molecular therapies is the current trend in cancer therapeutics, including treatment for ovarian cancer. IRIT has shown encouraging results for ovarian cancer treatment in clinical trials but is suboptimal for providing a significant improvement in patient survival (Goldenberg 2002). Therefore, in the present study we have determined if a multi-antigen targeting approach can improve the labeling sensitivity of ovarian cancer cells in ovarian tumors.
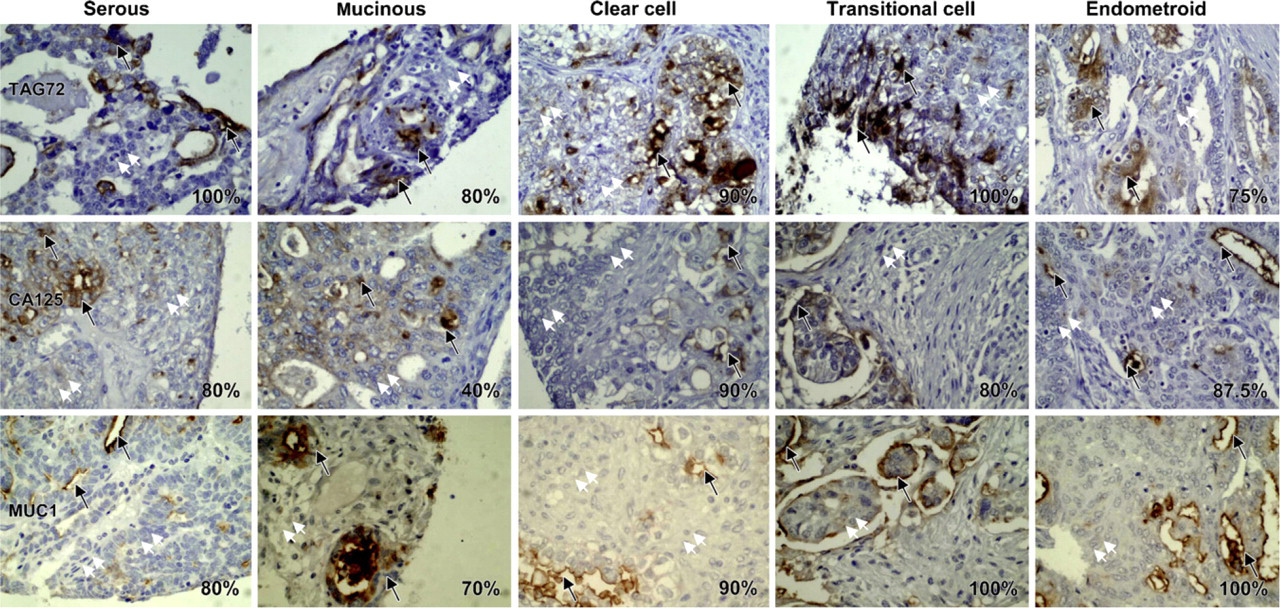
Heterogeneous expression pattern of TAG-72, MUC1, and CA125 in different histological types of epithelial ovarian cancer tissue samples. Upper, middle, and lower panels contain representative figures of TAG-72, MUC1, and CA125 immunostaining in serous, mucinous, clear cell, transitional, and endometroid types of epithelial ovarian cancers. In the majority of samples, only 30–40% of the cancerous cells express the target antigen (brown color, representative areas indicated by black arrows). Many areas containing cancerous cells are not labeled (representative areas indicated by white arrows).
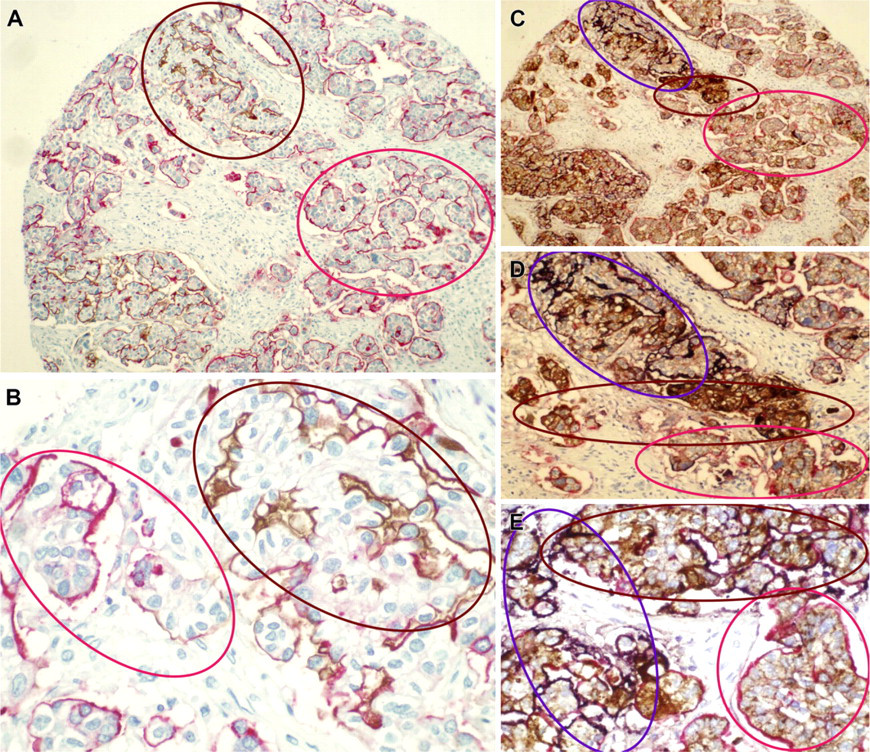
Multi-antigen labeling of ovarian cancer samples. (
Labeling sensitivity of three most commonly used targeting antigens for ovarian cancer treatment, TAG-72, MUC1, and CA125, was analyzed in situ by IHC as a direct measure of antigen labeling. Ovarian tissue microarray slides were used to avoid discrepancies while performing IHC procedures with a large number of samples. TAG-72, MUC1, and CA125 were over-expressed in ovarian cancer samples as compared with benign/normal ovarian samples (Table 1). Additionally, expression of these antigens was significantly higher in advanced stage cancer samples compared with early stage cancer samples (Table 2). However, none of the single cancer antigens showed expression in 100% of the epithelial ovarian cancer samples (Table 3). Nonetheless, in combination TAG-72, MUC1, and CA125 labeled a considerably higher number (98%) of ovarian tissue samples in each of the categories compared with any of the single antigen-labeling procedures (Table 3).
Increased percentage of stained cells with multi-antigen labeling. (
Our results show that the cells within a cancerous tumor heterogeneously express TAG-72, MUC1, and CA125 in the majority (>70%) of ovarian cancer samples (Figure 1). Heterogeneous expression of a tumor-targeting antigen may limit the therapeutic efficacy of a single antigen-targeted antibody therapy because a significant number of cells will not be targeted. The most promising way to improve the therapeutic efficacy of antibody-guided therapy will be the utilization of an antibody cocktail that can target multiple cancer antigens and thereby can label a higher number of cancer cells in a tumor. To determine the efficacy of this approach, double- and triple-antigen labeling were performed on ovarian cancer tissue microarray slides. In these experiments, the same ovarian cancer tissue arrays were probed with a cocktail of antibodies, i.e., anti-MUC1 + anti-CA125 and anti-MUC1 + anti-TAG-72 for double-antigen staining and a cocktail of anti-MUC1 + anti-TAG-72 + anti-CA125 for triple-antigen staining. Results of these experiments clearly demonstrate that in double- and triple-staining procedures the cocktail of antibodies labeled a markedly higher percentage of cancer cells within a tumor as compared with any of the single antigen-labeling procedures (Figure 2 and Figure 3).
Ovarian cancer is a highly metastatic disease. The poor outcome of this disease is associated with the high frequency of peritoneal metastatic seeding of the EOCs and their growth as ascites in the abdominal cavity (Auersperg et al. 1998, 2001). Because ovarian cancer primarily grows as ascites in the peritoneal cavity, IRIT may be a very useful and effective therapy for ovarian cancer. At present, several RIT formulations based on targeting antigens such as TAG-72, MUC1, and CA125 are under preclinical and clinical investigations. IRIT based on 90Y-labeled anti-MUC1 antibody showed a significantly prolonged duration of survival in clinical trials (Epenetos et al. 1987; Janssen et al. 2003). This IRIT was relatively more effective in patients with smaller tumor nodules (2 cm) compared with patients with larger tumors (Epenetos et al. 1987; Janssen et al. 2003). 131I-labeled anti-OC125 (CA125) murine antibody F(ab)2 and MOv18 chimeric antibodies are also being evaluated in ovarian cancer patients. In addition to these formulations, 177Lu- and 90Y-labeled murine anti-TAG-72 antibodies (CC49 and B72.3) are also under examination for ovarian cancer treatment (Meredith et al. 1996; Alvarez et al. 2002). IP administration of 177Lu-labeled CC49 radioimmunoconjugates showed promising results in initial clinical trials (Alvarez et al. 1997; Meredith et al. 2001). These formulations showed therapeutic advantage after IP administration in ovarian cancer patients (Schneider-Gadicke et al. 1992; Crippa et al. 1995; Behr et al. 1997; Mahe et al. 1999). Results of these studies are encouraging but are suboptimal for achieving maximal therapeutic benefit. The suboptimal response of the above-mentioned RIT formulations occurs primarily because ovarian cancer cells heterogeneously express the targeting antigen. Therefore, single antigen-targeted RIT remains refractory for a significant number of untargeted cancer cells. In addition, metastatic seedlings grow in different sizes of tumor nodules, and one particular radionuclide may be limited in its ability to effectively destroy different sizes of tumor nodules because of its specific linear energy transmission (LET) properties (Behr et al. 1999).
The aforementioned studies clearly demonstrate that antibody-guided RIT may be an effective alternative therapeutic modality for ovarian cancer treatment if properly modified and optimized. To prevail over the heterogeneic expression of TAAs by ovarian cancer cells and to target different sizes of tumor nodules, a multi-antigen targeting approach using therapeutic radionuclides of different LET properties will be an enviable alternative. However, a potential problem of IRIT using MUC1 is normal organ toxicity because it is expressed in some normal organs. Therefore, MUC1 targeting antibodies against the tandem repeat region will be ideal because this region is differentially glycosylated in normal (hyperglycosylated) vs cancer cells (hypoglycosylated) (Hollingsworth and Swanson 2004). These antibodies will potentially target ovarian cancer cells but not normal cells expressing MUC1. Expression of TAG-72 and CA125 is, however, usually limited to cancer tissues. Therefore, combining these three antigens ideally should not cause normal organ toxicity.
In the future, nanoparticle therapeutics will also be based on molecular targeting, which can be achieved by using nanoparticles linked to ligands such as MAbs/antibody fragments that are directed against cancer-associated antigens. Immunonanoparticles will combine antibody-mediated tumor recognition with nanoparticle-mediated anti-cancer drug delivery. An immunoliposome consisting of novel anti-HER2 scFv F5 conjugated to phospholipase D, currently in development, is selectively bound to and internalized by HER2-overexpressing tumor cells (Park et al. 1995, 1997a, b, 2001; Hong et al. 1999). In addition, paclitaxel-loaded nanoparticles targeted to the transferrin receptor have also shown an enhanced efficacy in animal models (Kang et al. 2004; Sahoo et al. 2004; Sahoo and Labhasetwar 2005; Vasir and Labhasetwar 2005). These studies confirm the proof of principle for the development of targeted nanopharmaceuticals.
In our double- and triple-staining experiments, we show for the first time that, in combination, TAG-72, MUC1, and CA125 label a broad spectrum of ovarian cancer cells within tumor samples. This study suggests that these tumor antigens are highly expressed in advanced stage samples and may be target antigens of interest for developing multi-antigen IRIT for the effective treatment of advanced stage ovarian cancer.
Footnotes
Acknowledgements
This research was supported by a Sanford Research/ USD grant.
The authors acknowledge Kelley Vannatta for technical support. We also thank Dr. Keith Miskimins, Director, Cancer Biology Research Institute, for critical review and suggestions.
