Abstract
Introduction
MicroRNAs (miRs) exhibit the potential to act as therapeutic targets for the management of human cancers including ovarian cancer. The role of microRNA-30 (miR-30) via modulation of RAB32 expression has not been studied in ovarian cancer. Consistently, the present study was designed to characterize the molecular role of miR-30/RAB32 axis in human ovarian cancer.
Methods
Cell viability was determined by MTT assay. Expression analysis was carried out by qRT-PCR. Dual luciferase assay was used to confirm the interaction between miR-30 and RAB32. Scratch-heal and transwell chamber assays were used to monitor the cell migration and invasion. Western blotting and immunofluorescence assays were used to determine the protein expression.
Results
The results revealed significant (p < 0.05) downregulation of miR-30 in human ovarian cancer cell lines. Overexpression of miR-30 in ovarian SK-OV-3 and A2780 cancer cells significantly (p < 0.05) inhibited their proliferation. Besides, ovarian cancer cells overexpressing miR-30 showed significantly (p < 0.05) lower migration and invasion. The miR-30 upregulation also altered the expression pattern of marker proteins of epithelial–mesenchymal transition in ovarian cancer cells. In silico analysis predicted RAB32 as the molecular target of miR-30 at post-transcriptional level. The silencing of RAB32 mimicked the tumor-suppressive effects of miR-30 overexpression in ovarian cancer cells. Nonetheless, overexpression of RAB32 could prevent the tumor-suppressive effects of miR-30 on SK-OV-3 and A2780 cancer cells.
Conclusion
Taken together, the results suggest the tumor-suppressive role of miR-30 and point towards the therapeutic utility of miR-30/RAB32 molecular axis in the management of ovarian cancer
Introduction
Ovarian cancer is the seventh most prevalent type of cancer in women. It has poor prognosis and imposes tremendous disease burden globally.1,2 Ovarian cancer can be subdivided into different histological subtypes that have different identifiable risk factors, cells of origin, molecular compositions, clinical features, and treatments. These histological subtypes include epithelial cancers that account for around 90% of ovarian cancers and include serous, endometrioid, clear-cell, and mucinous carcinomas. 3 Most of the cases are reported with the advanced disease stages limiting the applicability of the current treatment strategies and thus adding to the severity of ovarian cancer. 4 The statistical data suggests that around 3,00,000 cases of ovarian cancer were reported in 2018 alone along with approximately 1,85,000 deaths. 5 To better curb this malignancy, the pathogenesis of this disease needs to be studied at molecular level. During recent times, the non-coding RNAs have attained tremendous research attention for their dominant role in human tumorogenesis.6,7 Micro-RNAs (miRs), a heterogeneous group of short, long non-coding RNAs have been deduced to exhibit profound regulatory role in diverse human diseases. 8 Studies have envisaged that miRs control crucial molecular events in human cancer progression involving the tumor progression and metastasis. 9 MicroRNA-30 (miR-30) family of miRs has been elucidated to display significant level of dysregulation in human cancers like gastric cancer, breast cancer, head and neck cancer, and hepatocellular carcinoma.10–13 MiR-30a-5p (now onwards mentioned as miR-30) has been shown to exhibit marked downregulation in ovarian cancer and promotes the growth and metastasis of ovarian cancer cells. 14 The miR-30 has also been shown to regulate the metastasis of human breast cancer. 15 In yet another study, miR-30 has been shown to regulate the epithelial to mesenchymal transition of non-small cell lung cancer cells. 16 Nonetheless, the role of miR-30 via modulation of RAB32 expression has not been studied in any type of cancer including ovarian cancer. Against this backdrop, the present study was undertaken to investigate the role and to explore therapeutic implications of miR-30/RAB32 axis in ovarian cancer.
Materials and methods
Culture and transfection of cell lines
Four different human ovarian cancer cell lines (SK-OV-3, A2780, Caov3, and OVAC433) as well and the epithelial non-cancerous HOSE-6-3 cells were purchased from the American Type Collection Center (ATCC, USA). The cells were cultured using RPMI-1640 medium (Thermo Fisher Scientific) with a supplementation of 10% fetal bovine serum (FBS) at 37°C with 5% CO2. For in vitro culturing, the cell lines were incubated at 37°C with 5% CO2. The miR-30 mimics (overexpression), miR-NC (negative control), miR-30 inhibitor (silencing), inhibitor-NC (negative control), si-RNAs (si-RAB32#1 and si-RAB32#2), and si-NC (negative control) oligos were purchased from GenePharma (Shanghai, China). For the overexpression of RAB32, its ORF was cloned into pcDNA3.1 overexpression vector, while vector alone–transfected cells served as negative control. The transfection of cancer cells with suitable construct(s) was performed using Lipofectamine 2000 (Invitrogen) as per the manufacturer’s protocol.
Expression analysis
List of primers used in the study.

Note: miR-30: MicroRNA-30.
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay
The stably transfected SK-OV-3 and A2780 cells along with the corresponding negative controls were plated into 96-well plates with a cell density of 3 × 104 per well. MTT (Sigma, USA) reagent was then added to the wells at 0, 12, 24, 36, 48, or 96 h of culture duration. As per the manufacturer guidelines, the absorbance (OD570) value was recorded for each well using a microplate reader (Bio-Rad, USA) for the determination of cell viability.
Online bioinformatics and luciferase reporter assay
The molecular target of miR-30 was identified by TargetScan (www.targetscan.org/) online software. The in silico predication was validated using dual luciferase assay. Here, the pGL3-RAB32 3′UTR (WT or MUT) reporter plasmid was generated containing the putative miR-30 binding site, either native or mutated. Following this, each reporter plasmid was co-transfected into SK-OV-3 cells. Following 24 h incubation at 37oC, the transfected cells were analyzed for relative luciferase activity using a Dual Luciferase Reporter System Assay kit (Promega Corporation). The luciferase activity was normalized as firefly luciferase activity to Renilla luciferase activity.
Western blotting
After being extracted from cell lines through RIPA buffer treatment, approximately 35 μg of total proteins were subjected to separation using the sodium dodecyl sulfate polyacrylamide gel electrophoresis. Next, the proteins separated were transferred to the polyvinylidene fluoride (PVDF) membranes which were blocked with 5% non-fat milk. This was followed by the subsequent incubation of nitrocellulose membranes with the specific primary antibodies overnight at 4oC. Finally, an IRDye 800CW-conjugated secondary antibody was used to visualize the proteins specifically bound to their respective primary antibodies. The protein bands were analyzed with the help of an LI-COR imaging system (LI-COR Biosciences). Human β-actin served as internal control in protein expression study.
Scratch-heal assay
The stably transfected SK-OV-3 and A2780 cancer cells were cultured in 12-well plates till confluent monolayers were obtained. The cell layers were scratched mechanically with the help of a 200 μL pipette tip to carve a linear scratch. The cell surface was thrice washed with phosphate buffered saline (PBS), and the plate was incubated for 24 h at 37oC to allow the scratch-healing. The scratches were photographed at 0 h and 24 h to estimate the relative cell migration using a light microscope (magnification, × 100).
Transwell assay
The invasion of stably transfected SK-OV-3 and A2780 cancer cells was determined using transwell chamber assay. Around 2 × 104 transfected cells were cultured in upper well of transwell chamber carrying Matrigel (BD Biosciences, San Jose, CA, USA) coating at 37oC. The lower chamber was filled with serum-free culture medium. After 24 h incubation, the cells from the upper side of intervening membrane were carefully removed using a cotton swab while those sticking to the basal side of the membrane were fixed with 70% ethanol for 10 min, stained using 0.2% crystal violet (Sigma), and counted under a light microscope (Olympus, Tokyo, Japan). Five visual fields were chosen at random for relative quantification of cell invasion.
Immunofluorescence
The transfected SK-OV-3 cells were cultured for 24 h, and subsequently, washed and fixed in 4% paraformaldehyde for 15 min. The cells were then rinsed in PBS three times for 5 min each and blocked with 5% BSA in PBS for 30 min at 25°C. Primary antibody was added and incubated for 1 h at 37°C and then detected using FITC conjugated secondary antibody. Images were captured using a fluorescence microscope.
Statistical analysis
The SPSS22.0 software was used for all the statistical analyses. Results were presented as mean ± standard deviation (SD) calculated from three experimental replicates. Comparison between treatment parameters was made using independent t-test where p < 0.05 was marked to represent statistically significant difference.
Results
miR-30 downregulation in ovarian cancer
To gain insights about the miR-30 expression profile, the miR-30 transcript levels were examined in the ovarian cancer cell lines (SK-OV-3, A2780, Caov3 and OVAC433) using its expression level in epithelial ovarian cells (HOSE-6–3) as a reference. All the cancer cell lines were found to express significantly (p < 0.05) lower miR-30 transcript levels with reference to normal epithelial cells (Figure 1(a)). Least expression of miR-30 was confirmed from SK-OV-3 and A2780 cancer cell lines and were thus selected for further study. MicroRNA-30 suppresses the proliferation of the human ovarian cancer cells. (a) Expression of miR-30 in normal and ovarian cancer cells (b) Expression of miR-30 in miR-NC and miR-30 mimics transfected SK-OV-3 and A2780 ovarian cancer cells (c) Cell viability of miR-NC and miR-30 mimics transfected SK-OV-3 and A2780 cancer cells. The experiments were carried out in triplicate, and the data is shown as mean ± SD (*p < 0.05). Note: miR-30: MicroRNA-30.
Inhibition of ovarian cancer proliferation by miR-30 overexpression
To decipher the role of miR-30 in regulating ovarian cancer cell proliferation, miR-30 was overexpressed in SK-OV-3 and A2780 cancer cells by transfecting them with miR-30 mimics. The qRT-PCR was used to confirm the overexpression of miR-30 in both the cell lines (Figure 1(b)). Assessment of the proliferation of cancer cells was made along with the respective negative control cells at different culture time points using MTT assay. It was found that both SK-OV-3 and A2780 cancer cell lines overexpressing miR-30 exhibited significantly (p < 0.05) lower proliferation rate in comparison to the corresponding negative control cells (Figure 1(c)). The results are thus indicative of the tumor-suppressive role of miR-30 in ovarian cancer.
RAB32 as post-transcriptional target of miR-30 in ovarian cancer
In silico analysis was used to distinctively make a prediction about the regulatory target of miR-30 in ovarian cancer. Ras-related protein RAB32 was predicted as the specific molecular target of miR-30, and the latter was found to interact with a specific binding site in RAB32 3′-UTR (Figure 2(a)). To investigate the same, dual luciferase reporter assay was performed using miR-30 binding site as native (WT) or mutated (MUT). SK-OV-3 ovarian cancer cells, carrying co-transfection constructs of miR-30 and WT 3′-UTR stretch of RAB32, exhibited significant (p < 0.05) decline in luciferase activity than ones carrying the reporter construct of MUT RAB32 3′-UTR (Figure 2(b)). The ovarian cancer cell lines showed significantly (p < 0.05) higher expression levels of RAB32 as compared to that of the normal epithelial cells (Figure 2(c)). In addition, the post-transcriptional targeting of RAB32 by miR-30 was also evidenced from its considerably reduced protein levels in miR-30 overexpressing SK-OV-3 and A2780 cancer cells in comparison to negative control transfected cells (Figure 2(d)). In contrary, inhibition of miR-30 expression caused a considerable increase in the expression of RAB32 in SK-OV-3 and A2780 cells (Figure 2(e)). Collectively, the results are suggestive of post-transcriptional silencing of RAB32 by miR-30 in ovarian cancer. MicroRNA-30 targets RAB32 in ovarian cancer (a) TargetScan analysis showing RAB32 as the target of miR-30 (b) Dual luciferase assay showing interaction between miR-NC and miR-30 mimics in SK-OV-3 cells (c) qRT-PCR analysis showing expression of RAB32 in normal and ovarian cancer cells (d) western blots showing the expression of RAB32 in miR-NC and miR-30 mimics transfected SK-OV-3 and A2780 cells (e) western blots showing the expression of RAB32 in inhibitor-NC and miR-30 inhibitor transfected SK-OV-3 and A2780 cells. The experiments were carried out in triplicate, and the data is expressed as mean ± SD (*p < 0.05). Note: miR-30: MicroRNA-30.
Modulation of miR-30 regulatory role via miR-30/RAB32 axis
To understand whether miR-30 exerts its regulatory role via RAB32 targeting in ovarian cancer, RAB32 was silenced in SK-OV-3 and A2780 ovarian cancer cells. Silencing of RAB32 with two siRNA constructs, si-RAB32#1, and si-RAB32#2 was confirmed by qRT-PCR (Figure 3(a)) as well as by western blotting (Figure 3(b)). The inhibition of RAB32 was higher with RAB32#1, and hence, this silencing construct was used in subsequent experiments. The cancer cell proliferation was analyzed using MTT assay. It was shown that RAB32 silencing mimicked the tumor-suppressive effect of miR-30 overexpression in SK-OV-3 and A2780 ovarian cancer cells and the cancer cells proliferated at significantly (p < 0.05) lower rates (Figure 3(c)). In contrast, overexpression of RAB32 in SK-OV-3 and A2780 ovarian cancer cells overexpressing miR-30 mitigated the tumor-suppressive effect of miR-30 (Figure 3(d)).
Regulation of ovarian cancer cell migration, invasion and epithelial-to-mesenchymal transition by miR-30/RAB32 axis
Interestingly, the scratch-heal assay showed that miR-30 overexpression in SK-OV-3 and A2780 ovarian cancer cells inhibited their migration significantly (p < 0.05) (Figure 4(a)). However, the decline in ovarian cancer cell migration by miR-30 upregulation was reversed when RAB32 was overexpressed. The miR-30 overexpression similarly inhibited the invasion of ovarian cancer cells and the same was rescued by RAB32 overexpression (Figure 4(b)). Collectively, the results indicate that miR-30/RAB32 molecular axis modulates the regulatory role of miR-30 in SK-OV-3 and A2780 ovarian cancer migration and invasion. MicroRNA-30 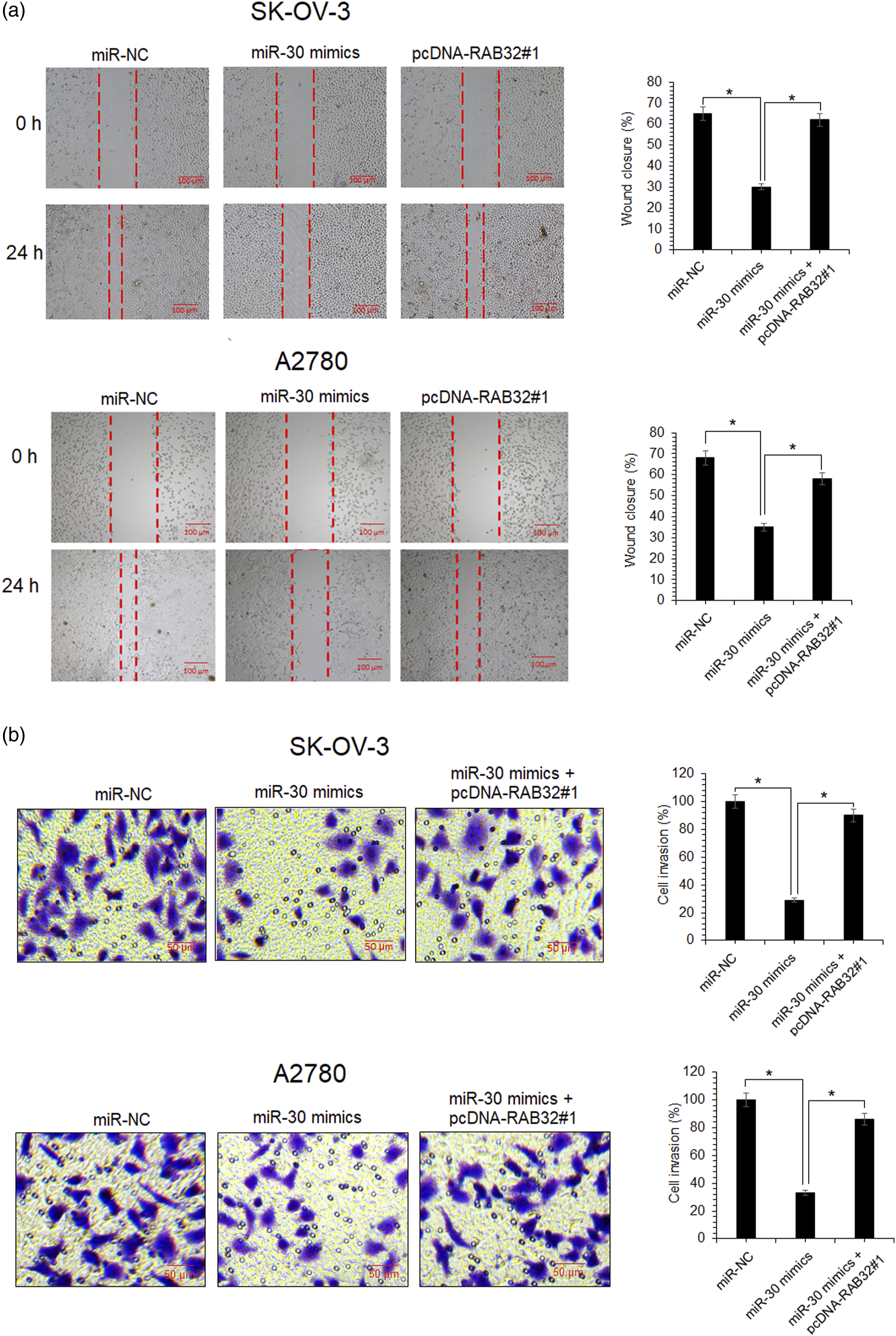
To investigate the regulatory role of miR-30 in controlling the epithelial-to-mesenchymal transition of ovarian cancer cells, western blotting of E-cadherin and α-catenin (epithelial markers) and vimentin and snail (mesenchymal markers) proteins was performed. Overexpression of miR-30 was shown to enhance the expression of epithelial marker proteins while mesenchymal marker protein expression levels were seen to considerably decrease under miR-30 overexpression (Figure 5(a)). Nonetheless, overexpression of RAB32 was shown to abolish the effect of miR-30 overexpression on epithelial and mesenchymal marker protein expression (Figure 5(a)). For further confirmation, we carried the immunofluorescence analysis of one epithelial marker (E-cadherin) and one mesenchymal marker (vimentin). The results showed that miR-30 overexpression increased the expression of E-cadherin and inhibited the expression of vimentin. However, overexpression of RAB32 could prevent these effects of miR-30 on E-cadherin and vimentin expression (Figure 5(b)). The results thus suggest that the SK-OV-3 ovarian cancer cell epithelial-to-mesenchymal transition is negatively regulated by miR-30 via its action through miR-30/RAB32 axis. MicroRNA-30 
Discussion
Human carcinogenesis is very intricate and is driven through multiple phenomena at molecular level. Past two decades have witnessed an inclination in the research interest in non-coding regulatory RNAs. MiRs in particular have gained tremendous scientific attention for their huge importance in regulating the growth and progression of human cancers. 9 The pathogenesis of ovarian cancer, accounting for significant mortality, is yet to be fully explored. A number of studies have put forward that miRs exhibit dysregulation in ovarian cancer.17,18 There are also reports that few members of miR-30 family are repressed in ovarian cancer cell lines. 14 Consistent with these findings, the results of present study proved that miR-30 is significantly downregulated in ovarian cancer. Previously, miR-30 has been shown to play a tumor-suppressive role in several types of human cancers.19–21 Similar tumor-suppressive regulatory function of miR-30 was confirmed against the ovarian cancer using in vitro cell line system. Each miR may modulate the expression of several target genes, for instance, miR-30 has been shown to target TCF21, Snai1, bone metastasis–associated genes, and Rab18 to name a few.16,22–24 However, there is not a single study which reports RAB32 as the target of miR-30. In the present study, RAB32 was selected as target of miR-30 for investigation. It was found that miR-30 interacts with RAB32 at post-transcriptional level in ovarian cancer to mediate its growth regulatory role in ovarian cancer. RAB32 belongs to Ras-oncogene protein family and has significant upregulation in human cancers.25,26 RAB32 was shown to act as the regulatory target of tumor suppressive miR-141-5p in chronic myeloid leukemia. 27 The miR-141-5p/RAB32 axis regulated the proliferation, migration, and apoptosis of myeloid leukemia cells. 27 Interestingly, the results of present study suggested miR-30 downregulation as a crucial factor responsible for the metastasis of ovarian cancer cells. Overexpression of miR-30 was shown to inhibit the migration and invasion of ovarian cancer cells through post-transcriptional silencing of RAB32. Similar role of miR-30 has been elucidated against the non-small lung cancer cells, previously. 22 Moreover, miR-30 has also been shown to inhibit the invasion of the breast cancer cells. 15 Further, the ovarian cancer cells overexpressing miR-30 showed a tendency towards reversal of epithelial-to-mesenchymal transition. The epithelial-to-mesenchymal transition enhances the proliferation of cancer cells and weakens the intrinsic pathways of programmed cell death. 28 This is in agreement with a previous study wherein miR-30 was found to inhibit epithelial-to-mesenchymal transition of non-small cell lung cancer cells. 16 Expression of proteins like E-cadherin and α-catenin (epithelial markers) drastically falls while that of vimentin and snail (mesenchymal markers) is greatly increased during epithelial-to-mesenchymal transition. 29 Epithelial-to-mesenchymal transition also enables cancer cells to evade the cell adhesion and to invade neighboring tissues, thereby promoting the process of metastasis. 25 The metastasis of ovarian cancer is largely responsible for its lethality and treatment failure. 30 Although the present study revealed the tumor-suppressive effects of miR-30 in ovarian cancer cells via suppression of RBA32 expression, in vivo studies are required for further confirmation. Moreover, expression profiling of miR-30 in different types of human ovarian cancer tissues including those arising from fallopian tubes may provide better insights about the therapeutic potential of miR-30.
Conclusion
Collectively, ovarian cancer exhibits significant downregulation of miR-30. The miR-30 overexpression inhibited growth, migration, and invasion of ovarian cancer cells via post-transcriptional targeting of RAB32. The results were suggestive of the therapeutic implications of miR-30/RAB32 axis in the management of ovarian cancer cells.
Footnotes
Declaration of conflicting interests
The authors declare that there are no conflicts of interest
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Shandong Provincial Science and Technology Development Program of Medicine and Health (No. 2017WS269) and the Science and Technology Program of Shandong Academy of Medical Sciences (No. 2017–19)
