Abstract
Canine idiopathic lymphocytic-plasmacytic colitis (LPC) is a well-recognized clinical and pathological entity in the dog, associated with altered immune cell populations and cytokine expression profiles. Clinical and experimental data indicate that alterations in the permeability of the intestinal epithelium contribute to the pathogenesis of a range of related conditions. The apical junction complex plays a significant role in regulating epithelial paracellular permeability, and we have characterized the distribution of a number of its component tight junction (ZO-1, occludin, claudin-2) and adherens junction (E-cadherin and β-catenin) proteins in normal colon and colon from dogs with idiopathic LPC. ZO-1, occludin, E-cadherin, and β-catenin exhibited a distribution in normal canine colon similar to that described previously in humans and rodents. In contrast to the situation in humans, claudin-2-specific labeling was observed in the normal canine colonic crypt epithelium, decreasing in intensity from the distal to the proximal crypt and becoming barely detectable at the luminal surface of the colon. There was little evidence for significant changes in ZO-1, occludin, E-cadherin, or β-catenin expression in dogs affected by idiopathic LPC. However, claudin-2 expression markedly increased in the proximal crypt and luminal colonic epithelium in affected dogs, suggesting a role in the pathogenesis of canine LPC.
Keywords
I
It has been widely hypothesized that the intestinal inflammatory response seen in chronic idiopathic IBD, both in humans and in dogs, occurs as a consequence of a deranged or inappropriate immunological response to bacterial- and/or food-derived antigens (Bhan et al. 1999; Garside 1999; Jergens 1999; Madsen et al. 2000; German et al. 2003). The role of food-derived antigens in the pathogenesis of canine IBD is supported by the observation that dietary manipulation on its own is associated with clinical improvement in some dogs with clinical and histopathological features of IBD (Luckschander et al. 2006). The role of luminal bacteria is supported by the observation that manipulation of bacterial flora, with either antibiotics or probiotics, is effective in ameliorating intestinal inflammation both in humans with spontaneous IBD and in experimental models (Sartor 2001).
It has been suggested that epithelial barrier dysfunction, and more specifically, derangements of the apical junction complex (AJC) of the intestinal epithelium, may be involved in the generation of an aberrant immune response, due, for example, to a loss of epithelial cell polarity or an abnormal delivery of antigens via a paracellular pathway (Hershberg and Mayer 2000; Soderholm et al. 2002; German et al. 2003). In support of this hypothesis, alterations in intestinal permeability, both paracellular and transcellular, are known to occur in naturally occurring and experimental models of IBD (Schmitz et al. 1999; Soderholm et al. 2002). In particular, patients with Crohn's disease (CD) and 10–20% of their clinically unaffected relatives have been shown to have increased intestinal permeability, implicating altered intestinal permeability in the pathogenesis of IBD (Hollander et al. 1986; Katz et al. 1989). Similarly, heightened paracellular permeability, as demonstrated by measurement of lactulose-to-rhamnose ratios (Quigg et al. 1993; Allenspach et al. 2006) and by measurement of transepithelial electrical resistance (Ulrich et al. 2004), has been demonstrated in some dogs with chronic idiopathic enteropathy (lymphocytic-plasmacytic enteritis and/or colitis) and idiopathic IBD, respectively.
The paracellular permeability of the intestinal epithelium is regulated by intercellular tight junctions (TJs), which are composed of a number of transmembrane proteins, including occludin and members of the claudin family, which are linked to the actin cytoskeleton by the cytoplasmic protein ZO-1 (Mitic et al. 2000). The adherens junction (AJ), which is immediately subjacent to the TJ, comprises transmembrane protein E-cadherin and the associated cytoplasmic proteins, the catenins, and plays an important role in the formation of TJs (Cereijido et al. 2000). Recent work in human IBD has identified numerous derangements in both TJs and AJs of the intestinal epithelium. Kucharzik et al. (2001) demonstrated decreased expression of occludin in both inflamed and non-inflamed colonic mucosa from ulcerative colitis (UC) and CD patients. This study also showed downregulation of ZO-1, E-cadherin, and β-catenin in epithelial cells immediately adjacent to transmigrating neutrophils. Prasad et al. (2005) observed reduced expression and re-distribution of claudin-3 and claudin-4 at the surface epithelium in both UC and CD, while claudin-2 expression was increased. Several studies have also demonstrated reduced expression of E-cadherin in the colonic mucosa from patients with IBD (Dogan et al. 1995; Hanby et al. 1996; Karayiannakis et al. 1998; Gassler et al. 2001), particularly adjacent to the site of epithelial ulceration. It remains to be determined whether these alterations are part of the cause or merely a consequence of IBD (Hollander 2002).
Given the lack of data pertaining to the expression of these intercellular junctional proteins in the normal canine colon, the initial aim of this study was to characterize the normal distribution and localization of TJ proteins ZO-1, occludin, claudin-2, and AJ proteins E-cadherin and β-catenin in the canine colon. Building on this information, we then aimed to investigate any alterations in the distribution or localization of these junctional proteins in the colonic epithelium of dogs with clinical and histopathological evidence of canine idiopathic colitis.
Materials and Methods
Patients and Tissue Collection
Archived formalin-fixed, paraffin wax–embedded (FFPE) colonic tissues from five dogs with no history or clinical signs of intestinal disease were retrieved from the University of Edinburgh Veterinary Pathology Unit archives. These samples had been obtained immediately postmortem from dogs that were presented for euthanasia for behavioral reasons (n=2), old age (n=1), chronic pulmonary disease (n=1), and urinary incontinence (n=1). Ages ranged from 18 months to 13 years (one was unknown). Four dogs were male and one was female (Table 1). Fecal analysis was negative for known pathogens. Histopathological examination of full-thickness colonic biopsies was normal in all cases.
FFPE colonic tissue specimens from five dogs with LPC were also retrieved. All dogs had been referred to the Royal (Dick) School of Veterinary Studies Hospital for Small Animals for the investigation of chronic large-intestinal disease. Clinical signs (Table 1) had been present for between 6 weeks and 6 years and included the following: diarrhea (n=5); tenesmus (n=3); hematochezia (n=5); increased frequency of defecation (n=4); excess fecal mucus (n=5); abdominal discomfort (n=2); and intermittent vomiting (n=2). Ages ranged from 4 to 9 years old. All five dogs were male, one of which was neutered. Following informed owner consent, all dogs had undergone colonoscopy, and multiple biopsies (four to eight) had been collected from the colon using non-serrated pinch biopsy forceps. Diagnosis of idiopathic LPC was made on the basis of clinical presentation, exclusion of known causes of large-intestinal diarrhea, and typical histopathological characteristics. On the basis of clinical evaluation, four of the five dogs had inflammation restricted to the colon, where as in the fifth dog, in addition to moderate-to-severe LPC, inflammation was also present in the duodenum (Table 1). To minimize the heterogeneity of the clinical material, cases were selected on the basis of histopathological changes consistent with moderate-to-severe LPC.
Ethical aspects of the study design were appraised by the University of Edinburgh Veterinary Ethical Review Committee and were found to meet accepted standards.
Antibodies
Polyclonal rabbit anti-claudin-2 (PAD: MH44) and anti-occludin (PAD: Z-T22) antibodies were purchased from Invitrogen, Ltd. (Paisley, UK). Monoclonal mouse anti-β-catenin IgG1 (clone: 14/beta-catenin) and anti-E-cadherin IgG2a (clone: 36) antibodies were purchased from BD Biosciences (Oxford, UK). Monoclonal rat anti-ZO-1 IgG1 (clone: R26.4C) was prepared in-house (Scudamore et al. 1998). Control immunoglobulins were purchased from Sigma-Aldrich (Poole, UK). The secondary F(ab′)2 conjugates donkey anti-rabbit-Rhodamine Red X (RRX), goat anti-mouse-RRX, and donkey anti-rat-Cyanine-5 (Cy5) were purchased from Stratech Scientific (Newmarket, UK).
Breed, age, sex, clinical signs, and duration of clinical signs and histopathological diagnosis
M, male; F, female; MN, male neutered; LPC, lymphocytic-plasmacytic colitis. Severity of clinical sign graded subjectively as mild/occasional (+), moderate (++), and severe/frequent (+++); n/o, not observed; n/a, not applicable.
Immunocytochemistry
Four-μm FFPE tissue sections were mounted on poly-L-lysine-coated slides (BDH; Poole, UK), dewaxed in xylene, and rehydrated through graded ethanol series to water. Antigen retrieval was achieved by microwave treatment in 0.01 M Tris-EDTA buffer (0.01 M Tris-base, 0.001 M EDTA, 0.05% Tween 20, pH 9.0), for 20 min at 800 W. Slides were then washed in distilled water, followed by PBS, then PBS/0.5% Tween 80 (PBS/T80) before loading into a Sequenza immunostaining center (Shandon; Runcorn, UK). Sections were blocked with PBS/T80 + 10% normal goat serum (NGS) for 30 min at room temperature. All antibodies and conjugates were diluted in PBS/T80 + 10% NGS (2 μg/ml for purified antibodies, 1:10 dilution of tissue culture supernatants). For single labeling, sections were incubated with primary/control antibodies for 1 hr at room temperature. For dual labeling, sections were incubated with primary antibody and anti-ZO-1, control IgG and anti-ZO-1, or primary antibody and control rat IgG1 for 1 hr at room temperature. Slides were washed with PBS and incubated in the dark with secondary F(ab′)2 conjugates at 2 μg/ml for 30 min at room temperature. Sections were counterstained with SYBRgreen (Invitrogen, Ltd.) for 15 min at room temperature, washed in PBS, and mounted with Mowiol mounting medium (Calbiochem-Novabiochem; San Diego, CA).
Image Acquisition
Confocal images were acquired using an MRC-600 confocal laser-scanning microscope (Bio-Rad Laboratories; Hemel Hempstead, UK) mounted on an Axiovert-100 inverted microscope equipped with Plan-Apochromat objective lenses (Carl Zeiss Ltd; Welwyn Garden City, UK). Fluorophores were excited and imaged sequentially using the 488-nm (SYBRgreen), 568-nm (RRX), and 647-nm (Cy-5) lines from a 15-mW Kr/Ar laser (Bio-Rad Laboratories).
Examination of Sections
Sections were evaluated at ×200 and ×630 (oil immersion) magnification to identify areas of consistent immunofluorescent staining and acceptable orientation. Immunofluorescence was evaluated along the length of multiple colonic crypts and in areas of intact luminal epithelium, and representative images were acquired. Fluorescence intensity was subjectively graded as absent (−), weak (+), moderate (++), or strong (+++), and the localization and distribution of fluorescence were noted. Dual-labeled sections were evaluated for colocalization of ZO-1 and claudin-2. For purposes of evaluation, the colonic epithelium was divided into luminal, proximal, and distal crypt regions, and the intercellular junction was divided into apical and basolateral compartments.
Results
Distribution and Localization of Intercellular Junctional Proteins in the Normal Canine Colon
Representative images of sections of colon from normal dogs labeled with AJC-specific antibodies are shown in Figure 1; expression of all AJC proteins was detected in the colonic mucosa of all healthy dogs. ZO-1-specific labeling (Figures 1A and 1B) was restricted to the AJC of intestinal epithelial cells (arrow) and lamina propria endothelial cells (arrowhead), with limited evidence for basolateral or cytoplasmic labeling. ZO-1 appeared to be uniformly expressed along the length of the crypt axis and at the luminal surface of normal canine colon. Occludin-specific labeling (Figures 1C and 1D) was most intense at the epithelial cell AJC (arrow), with fainter labeling along the basolateral membranes (arrowhead). As described for ZO-1, occludin appeared to be uniformly expressed throughout the epithelium of normal canine colon. Weaker, occludin-specific labeling of the endothelial cell AJC was also observed in the lamina propria. Claudin-2-specific labeling (Figures 1E and 1F) was largely restricted to the epithelial cell AJC (arrow), with some punctate basolateral labeling (arrowhead). Claudin-2 was readily detectable in normal canine colonic crypt epithelium, decreasing in intensity from the distal to the proximal crypt and becoming barely detectable at the luminal surface of the colon. E-cadherin (Figures 1G and 1H) and β-catenin (Figures 1I and 1J) -specific labeling was restricted to the AJC and basolateral membranes of normal canine colonic epithelial cells, with little evidence of specific labeling outside the epithelium. For both E-cadherin and β-catenin, the overall intensity of staining decreased from the luminal epithelium to the distal crypts. At the luminal epithelium, labeling was uniform along the length of the intercellular junction, with expression becoming polarized toward the AJC in the distal crypts.
Upregulation of Claudin-2 Expression in the Superficial Colonic Epithelium of Dogs With Idiopathic Colitis
The expression and localization of ZO-1, occludin, claudin-2, E-cadherin, and β-catenin was subsequently evaluated in colonic mucosal biopsies from five dogs with a clinical and histopathological diagnosis of idiopathic colitis, representative images of which are shown in Figure 2. As with the healthy dogs, expression of all AJC proteins was detected in the colonic mucosa of all five dogs with idiopathic colitis. No discernable differences in the distribution or staining intensity of ZO-1, occludin, E-cadherin, or β-catenin were observed between normal (Figure 1) and affected dogs (Figure 2). However, claudin-2 expression was increased in the proximal crypt and luminal epithelium in all five affected dogs (Figures 2E and 2F, arrow; Table 2), and sporadic cytoplasmic expression was also observed (Figure 2F, arrowhead). Dual-immunofluorescent labeling of claudin-2 (green) and ZO-1 (red) demonstrated colocalization of these proteins (yellow) to the apical intercellular junction in all cases (Figure 3). Increased expression of claudin-2 within the proximal crypt epithelium and luminal surface of affected dogs is clearly identified (Figure 3D, 3F, and 3H). In addition to this apparent increase in claudin-2 expression at the luminal surface of the colon, sporadic cytoplasmic expression in crypt epithelial cells was observed in three of five affected cases (Figure 3C, arrow).
Discussion
In this study, we have established the baseline distribution and localization of a number of key TJ and AJ proteins in the canine colon. ZO-1 and occludin appear to have a distribution and localization similar to those described in the human colonic mucosa (Kucharzik et al. 2001). Similarly, the distribution and localization of the AJ proteins E-cadherin and β-catenin parallel those described previously in the dog (McEntee and Brenneman 1999) and in humans (Karayiannakis et al. 1998). Although the distribution and localization of claudin-2 appear to be similar to those seen in the rat colon (Rahner et al. 2001), the presence of claudin-2-specific labeling in the normal colonic crypt epithelium differs from the reported expression in humans, where claudin-2 is undetectable in normal colonic tissue (Escaffit et al. 2005; Prasad et al. 2005). In addition, we have demonstrated an alteration in the distribution of claudin-2 in the colonic mucosa of dogs with idiopathic LPC, with increased expression in proximal crypt and luminal epithelium.
Claudin-2 is one of a multi-gene family of proteins, consisting of more than 20 members, which form both structural and functional components of TJs (Furuse et al. 2001). Differential expression of these proteins at the TJ confers tissue-specific variation in both electrical resistance and paracellular permeability. In cell culture studies using MDCK monolayers, upregulation/forced expression of claudin-2 has been shown to result in a “leaky” TJ phenotype, with a decrease in transepithelial electrical resistance (TEER) (Furuse et al. 2001) and a cation-selective increase in permeability (Amasheh et al. 2002). The differential expression of claudin-2 along the crypt–luminal epithelium axis in the normal colon correlates with differing permeability of the crypts (leaky) compared with the upper compartments (tight epithelium) in the colon (Madara and Marcial 1984). Although the biological significance of the upregulation of claudin-2 expression at the luminal epithelium observed in this study requires further clarification, it is tempting to speculate that these changes result in increased epithelial permeability and/or alteration in epithelial polarity. The observation by Ulrich et al. (2004) that the colonic mucosa of dogs with IBD showed low TEER is consistent with this hypothesis.
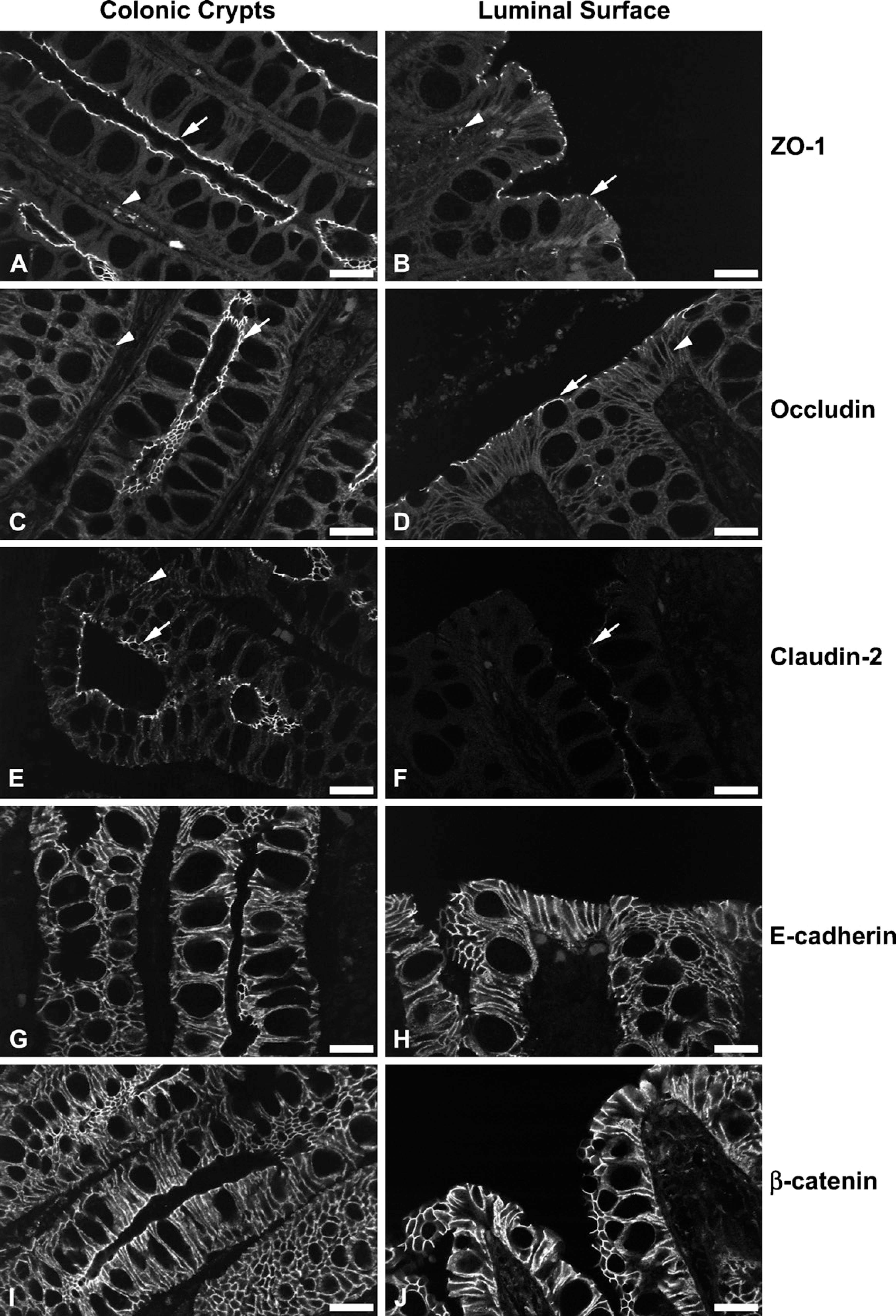
Representative micrographs mapping the distribution and localization of tight junction (TJ) and adherens junction (AJ) proteins. (
Semi-quantitative assessment of claudin-2 expression in the colonic mucosa

Fluorescence intensity was graded as absent (−), weak (+), moderate (++), or strong (+++) and is presented as a range derived from evaluation of multiple crypt units. Claudin-2 expression is increased at the luminal epithelium and in the proximal crypts in dogs with idiopathic colitis.
The observation that the distribution of claudin-2 expression is altered in canine idiopathic colitis may provide some insight into the aetiopathogenesis of canine IBD. Similar changes in the distribution of claudin-2 expression have been observed in active UC, where claudin-2 was detected at the surface epithelium (Prasad et al. 2005). Using a cell culture model of the colonic epithelium, Prasad et al. (2005) extended the study to demonstrate an increase in claudin-2 expression following incubation with IL-13 (a signature Th2-type cytokine), but interestingly, claudin-2 expression was decreased by incubation with and TNF-α and IFN-γ. Heller et al. (2005) have subsequently demonstrated/hypothesized that IL-13 produced by CD1d-positive natural killer T cells in the colonic mucosa of patients with UC is likely to induce upregulation of claudin-2, thus contributing to the pathogenesis of UC. This is of particular interest because canine idiopathic colitis has been shown to have a T-helper 1–polarized cytokine profile with upregulation of IL-2 and TNF-α mRNA expression in colonic mucosal biopsies (Ridyard et al. 2002), and these cytokines are purported, as in human IBD, to play an important role in the pathogenesis and perpetuation of intestinal inflammation. Our results may support the hypothesis that there is an evolution in the inflammatory process and cytokine profile with chronicity, and that the divergent cytokine patterns may be observed at different stages of disease, as proposed by Desreumaux et al. (1997). Although colonic IL-13 mRNA expression has been evaluated in dogs with food-responsive idiopathic colitis, both prior to and following treatment, with no significant change, it is not clear whether the expression is elevated in comparison to healthy dogs (Sauter et al. 2006). These observations indicate that further evaluation of IL-13 expression in the colon of dogs with idiopathic colitis is warranted.
Downregulation of occludin is observed in colonic mucosa of patients with both UC and CD (Gassler et al. 2001; Kucharzik et al. 2001), and it is hypothesized that this is secondary to the effects of IFN-γ and TNF-α at both the transcriptional and protein level (Mankertz et al. 2000; Kucharzik et al. 2001). Evidence for this hypothesis was provided by Bruewer et al. (2003) who demonstrated dramatic redistribution of occludin in intestinal epithelial cell monolayers following incubation with IFN-γ and TNF-α. Because TNF-α is also thought to contribute to the pathogenesis of canine idiopathic colitis (Ridyard et al. 2002), the absence of any demonstrable changes in the distribution or localization of occludin expression in this study is of note. One could speculate that the cytokine-induced changes may be dose dependent.
Reduction in E-cadherin expression has been demonstrated in human IBD, both in UC and CD (Dogan et al. 1995; Hanby et al. 1996; Karayiannakis et al. 1998; Gassler et al. 2001), but only at the site of ulceration of the luminal epithelium. In these studies, E-cadherin expression was normal in non-inflamed areas of intestine and in the epithelial cells lining the crypts in areas of active inflammation. Ulcerative lesions of the luminal epithelium are uncommon in dogs with idiopathic colitis, and this may account for the absence of observed changes in the present study; in common with human IBD (Dogan et al. 1995; Hanby et al. 1996; Karayiannakis et al. 1998; Gassler et al. 2001), there were no qualitative changes in E-cadherin expression in the epithelial cells lining the colonic crypts.
In addition to its contribution to cell–cell adhesion, E-cadherin has been shown to play a central role in the control of epithelial cell proliferation, migration, and differentiation, and thus in normal epithelial homeostasis (Hermiston and Gordon 1995). Escaffit et al. (2005) recently demonstrated differential expression of E-cadherin within the normal intestinal mucosa, with lower expression in the lower crypts and, more specifically, the pluripotential stem cells within the crypts, which are responsible for maintenance of normal intestinal morphology. Because crypt hyperplasia is one of the earliest histopathological changes seen in canine chronic idiopathic colitis (Guildford 1996), thought to reflect more-rapid epithelial turnover, we anticipated downregulation of E-cadherin in colonic tissue from dogs with colitis. The absence of any alteration in E-cadherin expression was therefore of note, although this may reflect the qualitative nature of our study; further evaluation of E-cadherin using a more quantitative methodology may be useful.
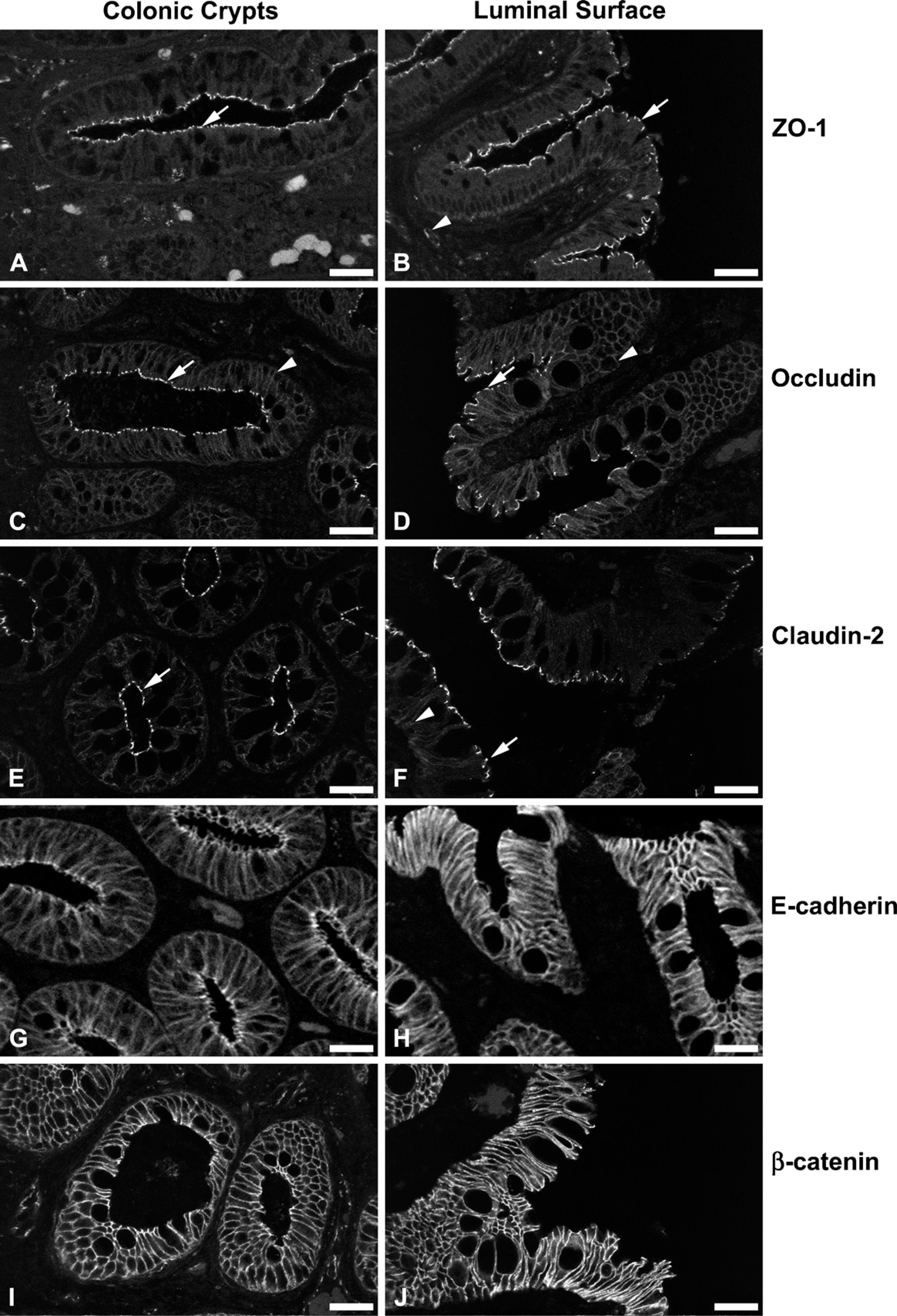
Representative micrographs of colonic mucosal biopsies from dogs with idiopathic colitis labeled with TJ and AJ protein-specific antibodies. (
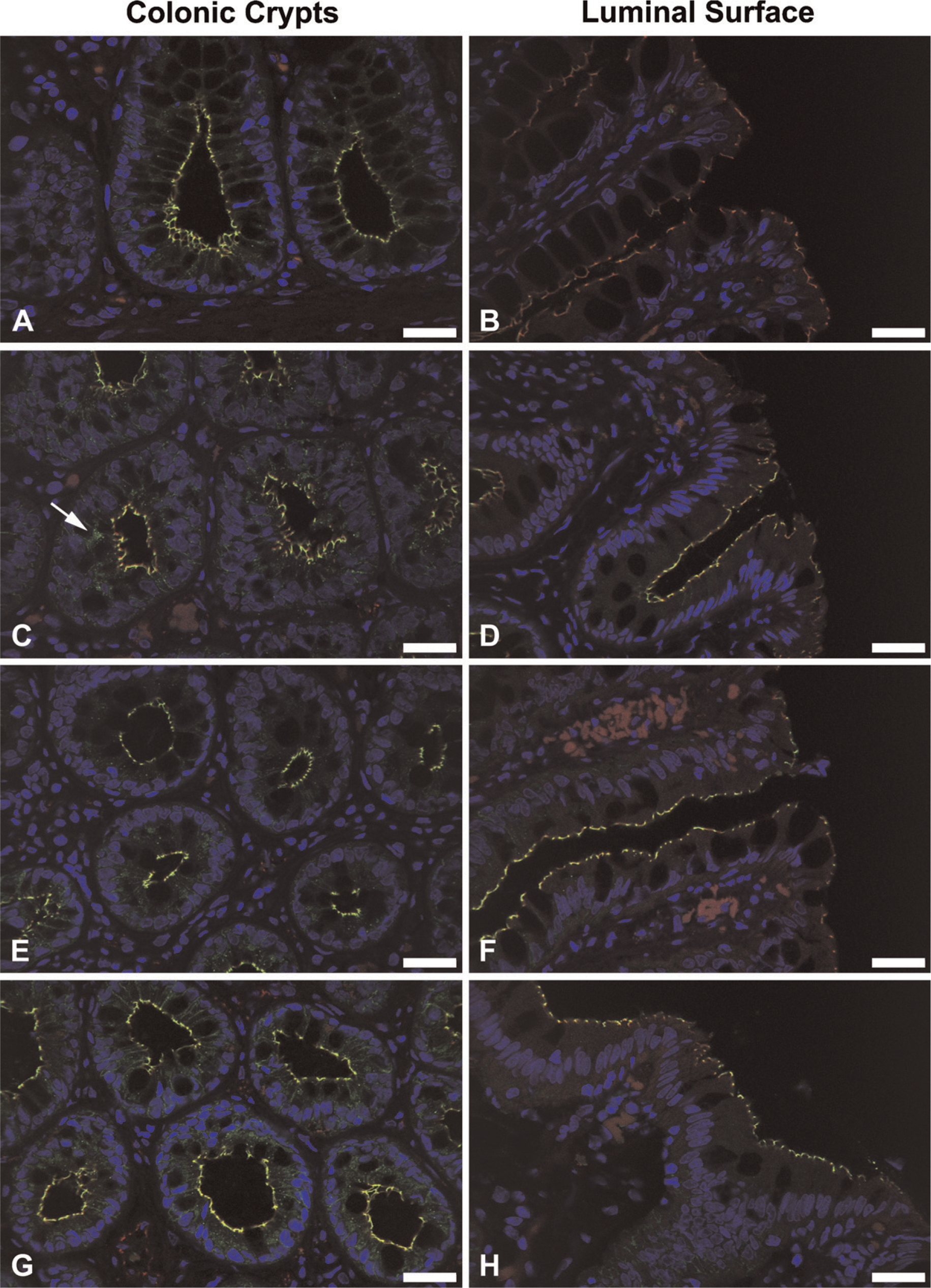
Dual-immunofluorescent labeling of claudin-2 (green) and ZO-1 (red) demonstrating colocalization of these proteins (yellow) in the normal canine colon (
In conclusion, this work has established baseline data for the expression of a number of important AJC proteins in the canine colon. Further, we have, for the first time, recorded alterations in the expression of claudin-2 in association within canine idiopathic colitis, opening further avenues for exploring the aetiopathogenesis of this poorly understood clinicopathological entity.
Footnotes
Acknowledgements
This work was supported by the Pet Plan Charitable Trust.
