Abstract
It has been believed that epithelial cells maintain tight junctions at all times, including during cell division, to provide a continuous epithelial seal. However, changes in localization of integral tight junction proteins during cell division have not been examined. In this study, using SV40-immortalized mouse hepatocytes transfected with human Cx32 cDNA, in which tight junction strands and the endogenous tight junction proteins occludin, claudin-1, ZO-1, and ZO-2 were induced, we examined changes in localization of the tight junction proteins at all stages of cell division. All tight junction proteins were present between mitotic cells and neighboring cells throughout cell division. In late telophase, the integral tight junction proteins occludin and claudin-1, but not the cytoplasmic proteins ZO-1 and ZO-2, were concentrated in the midbody between the daughter cells and were observed at cell borders between the daugher and neighboring cells. These results indicate that the integral tight junction proteins are regulated in a different manner from the cytoplasmic proteins ZO-1 and ZO-2 during cytokinesis.
T
Previously, we established cell lines that stably expressed Cx32 (Kojima et al. 1999) by introduction of human Cx32 cDNA into an immortalized mouse hepatocyte cell line (CHST8 cells) that lacked expression of Cx26 and Cx32. When we examined changes in expression and function of tight junctions in the transfectants compared to parental cells, the integral tight junction proteins occludin and claudin-1 and −2 were induced and the number of tight junction strands increased (Kojima et al. 1999). Therefore, these transfectants appear to be useful for studies on regulation of the endogenous tight junctions.
In this study, to elucidate the mechanisms involved in maintenance of the epithelial barrier during cell division, we immunocytochemically examined changes in localization of occludin, claudin-1, ZO-1, and ZO-2 during cell division of immortalized mouse hepatocytes transfected with human Cx32 cDNA. To frequently observe cells at the M-phase of the cell cycle, the cells were treated with Taxol (Taxus brevifolia), which reduces the rate of cytokinesis at low concentrations by kinetically stabilizing spindle microtubules (Jordan et al. 1993; Rowinsky and Donehower 1995; Snyder and McLelland 1996). By immunocytochemistry on the transfectants treated with Taxol, all tight junction proteins were present between mitotic cells and neighboring cells throughout all phases of mitosis. In the late telophase, however, the integral tight junction proteins occludin and claudin-1, but not the cytoplasmic proteins ZO-1 and ZO-2, were concentrated in the midbody between the daughter cells.
Materials and Methods
Cell Culture, cDNA Construction, and Transfection
CHST8 is a clonal cell line derived from female C3H/HeJ mouse hepatocytes conditionally immortalized with the temperature-sensitive SV40 large T-antigen gene as described previously (Lee et al. 1995). The cells were incubated with DMEM medium (GIBCO BRL; Gaithersburg, MD) with 4% fetal bovine serum (Sigma; St Louis, MO), 20 mM HEPES (Sigma), 0.5 mg/liter insulin (Sigma), 10–7 M dexamethasone (Sigma), 1 g/liter galactose (Sigma), 30 mg/liter proline (Sigma), 1 mM sodium pyruvate (Sigma), and antibiotics at 33C.
A fragment containing nucleotides 1–1558 of human connexin32 (Cx32) cDNA (Kumar and Gilula 1986) was subcloned into the expression vector pREP9 (Invitrogen; Carlsbad, CA) at the KpnI-BamHI restriction site. CHST8 cells were transfected with 2 μg of Cx32 cDNA using the LIPOfectamine regent (GIBCO). After 48 hr, the cells were transferred to selection medium containing 2 μg/ml puromycin (Sigma). When surviving colonies were large enough to detect visually, they were individually picked and separately propagated. After initial screening of five clones for Cx32 expression by immunocytochemistry, we chose highly expressing clones and expression of human Cx32 and its mRNA in the stable cell lines was confirmed by immunocytochemistry and by Western and Northern blotting analyses, as previously reported (Kojima et al. 1999).
It is known that Taxol at a low concentration (10 nM) reduces the rate of cytokinesis (Snyder and McLelland 1996) and induces many mitotic cells (Jordan et al. 1993). To examine changes in localization of tight junctional proteins in many mitotic cells, the transfectants were treated with 10 nM Taxol (Sigma) for 24 hr.
Immunofluorescence Microscopy
Cells grown on glass coverslips were fixed with cold absolute acetone for 10 min. Double immunocytochemistry with monoclonal anti-α-tubulin (Amersham; Poole, UK) and with polyclonal anti-occludin (Zymed Laboratories; San Francisco, CA), polyclonal anti-claudin-1, polyclonal anti-ZO-1 (Zymed), polyclonal anti-ZO-2 (Zymed) antibodies, and rhodamine–phalloidin (Molecular Probes; Eugene, OR) was performed. Rabbit polyclonal anti-claudin-1 antibody was raised against the synthetic polypeptide SYPTPRPYPKPTPSSGKD, which corresponds to the COOH-terminal cytoplasmic domains of mouse claudin-1 (amino acids 192–209) (Immuno-Biological Laboratories; Fuzioka, Japan). α-Tubulin was visualized using Alexia 594 (red)-conjugated anti-mouse IgG (Molecular Probes) and occludin, ZO-1, ZO-2 by Alexia 488 (green)-conjugated anti-rabbit IgG (Molecular Probes). α-Tubulin was visualized using Alexia 488 (green)-conjugated anti-mouse IgG (Molecular Probes) and actin by rhodamine–phalloidin (red). The specimens were examined with an epifluorescence microscope (Nikon; Tokyo, Japan) and a laser scanning confocal microscope (MRC 1024; Bio-Rad, Hercules, CA).
Results
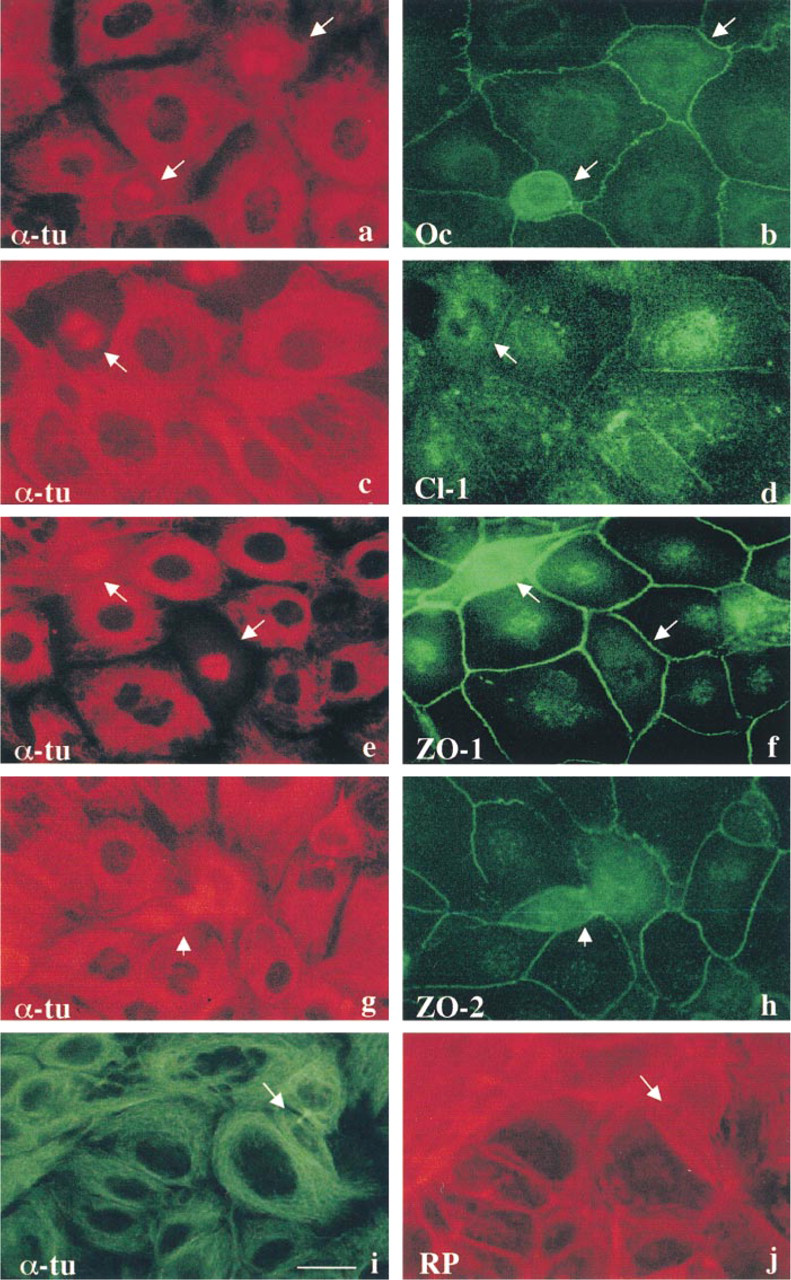
In immortalized mouse hepatocytes transfected with human Cx32 cDNA, occludin, claudin-1, ZO-1, and ZO-2 were clearly localized at cell borders (Figure 1). When the transfectants were treated with 10 nM Taxol for 24 hr, many mitotic cells (~20%) at various stages of cell division were observed. However, no conspicuous changes of polymerized microtubules in cytoplasm were observed. To examine changes in localization of tight junction proteins during cell division, double immunocytochemistry with α-tubulin and either occludin, claudin-1, ZO-1, or ZO-2 was performed. Localization of α-tubulin indicated the phases of mitotic cells: the metaphase, anaphase, telophase, and cytokinesis (Figures 1a, 1c, 1e, and 1g). All tight junction proteins, occludin, claudin-1, ZO-1, and ZO-2, were present between mitotic cells and neighboring cells throughout all phases of mitosis (Figures 1b, 1d, 1f, and 1g).
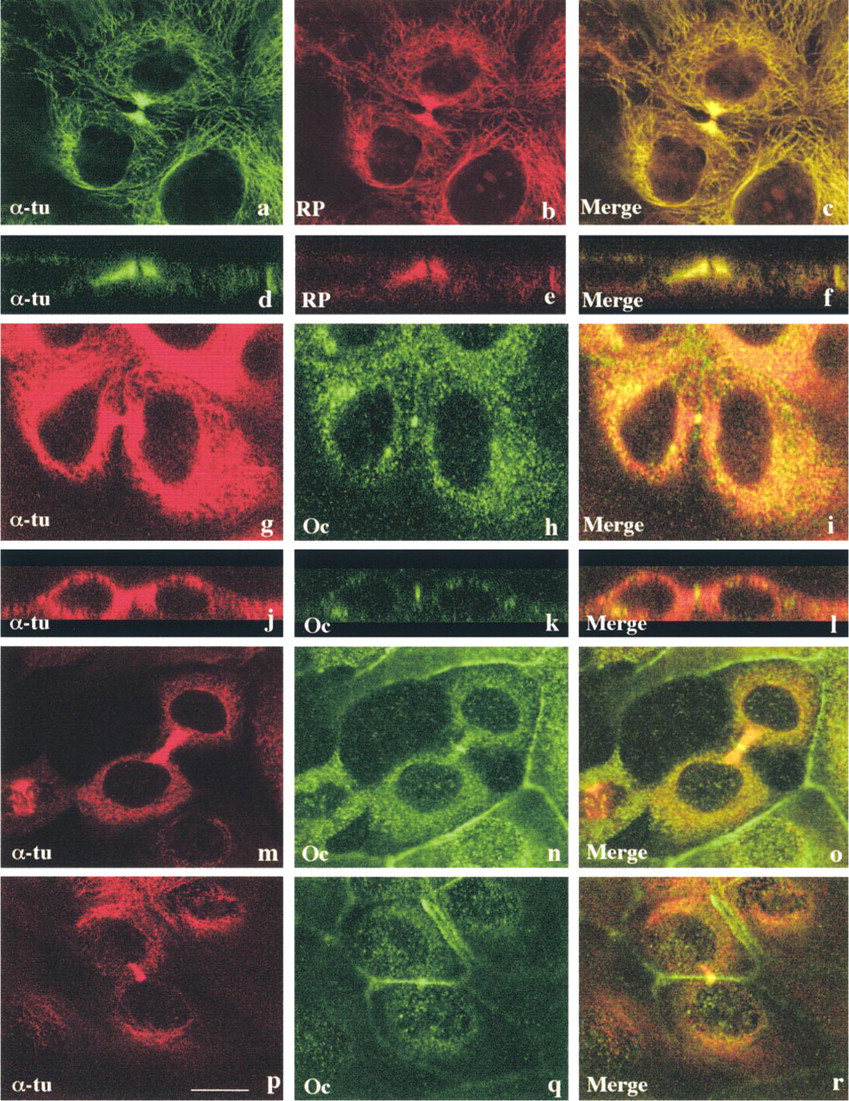
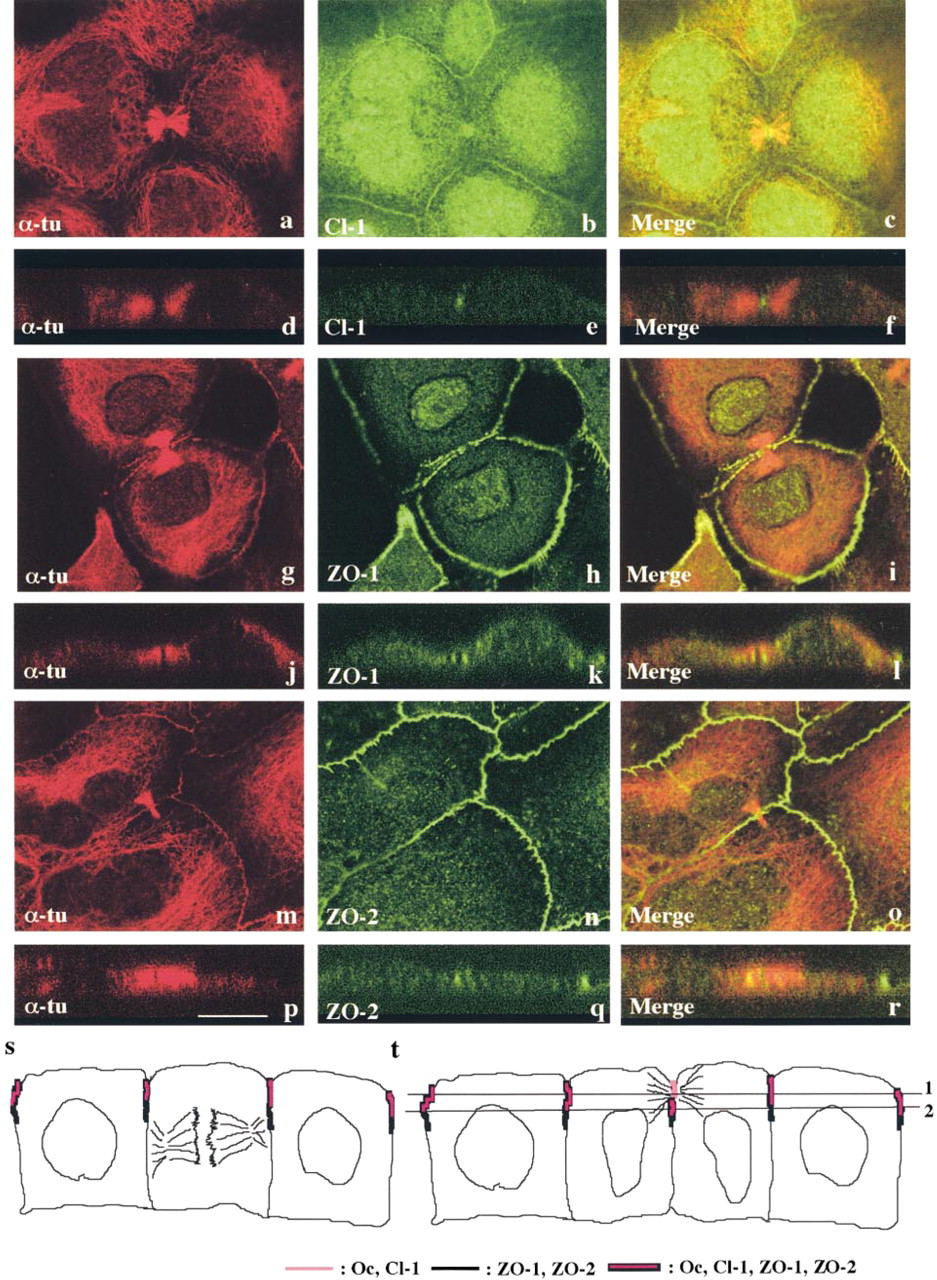
Next, attempting to elucidate changes in tight junctions during cell division, we further focused on changes in localization of tight junction proteins between the two daughter cells of mitosis. As shown in Figures 2a–2f, staining of α-tubulin and actin clearly demonstrated a tiny area not stained by either one, between two thick bundles of tubulin and actin in the middle of the midbody. Double immunostaining with α-tubulin and occludin demonstrated that occludin was concentrated in the midbody (Figures 2g–2r). In Z-sections, occludin was clearly observed in the midbody on the most apical site between the two daughter cells (Figures 2j–2l). Furthermore, occludin was observed not only in the midbody but also at cell borders between daughter cells and neighboring cells (Figures 2m–2o). Occludin was also observed at cell borders beneath the midbody between the daughter cells (Figures 2p–2r). Double immunostaining with α-tubulin and claudin-1 demonstrated that claudin-1 was also present in the midbody and between daughter cells and neighboring cells, like occludin (Figures 3a–3f). Double immunostaining with α-tubulin and tight junction-associated cytoplasmic proteins ZO-1 or ZO-2 demonstrated that both ZO-1 and ZO-2 were localized just beneath the midbody, not in it (Figures 3g–3r). Figures 3s and 3t show schematic figures of localization of tight junction proteins during epithelial cell division. All tight junction proteins, occludin, claudin-1, ZO-1, and ZO-2, were present between mitotic cells and neighboring cells, regardless of the phase of mitosis, but only occludin and claudin-1 were localized in the midbody between the daughter cells. Line 1 of Figure 3t corresponds to Figures 2j–2l and Figures 3d–3f (occludin, claudin-1), and Line 2 corresponds to Figures 3j–3l, and Figures 3p–3r (ZO-1, ZO-2). Between the daughter cells at late telophase, the most apical part formed the midbody, in which only occludin and claudin-1 were localized, while all tight junction proteins were detected at the cell borders beneath the midbody.
Double immunocytochemistry with α-tubulin (
Confocal laser microscopy after double staining with α-tubulin (
Confocal laser microscopy after double staining for α-tubulin (
Discussion
In immortalized mouse hepatocytes transfected with human Cx32 cDNA, increases in the number of tight junction strands and in the amount of integral tight junction proteins occludin and claudin-1 and −2 were observed, as we previously reported (Kojima et al. 1999). Therefore, this transfectant appears to be useful for studies on regulation of tight junction proteins, because the expression of the tight junction proteins is endogenously upregulated. In this study we focused on changes of localization of tight junction proteins of the epithelial cells during cell division to minimize the focal deficit of the continuous epithelial seal caused by mitosis.
It is well known that tight junction structures in epithelial cells are maintained during mitosis (Jinguji and Ishikawa 1992; Baker and Garrod 1993; Fishkind and Wang 1995). Furthermore, it has also been reported that the tight junction-associated protein ZO-1 is observed at all stages of mitosis (Reinsch and Karsenti 1994). In this study the tight junction proteins, occludin, claudin-1, ZO-1, and ZO-2 were present between mitotic cells and neighboring cells throughout all phases of mitosis. These findings strongly suggest that the paracellular barrier between mitotic cells and neighboring cells is maintained even during cell division.
In ultrastructural studies, intestinal epithelial cells at late telophase still show tight junction structures between the midbody and neighboring cells (Jinguji and Ishikawa 1992). Consistently, the present study demonstrated that the tight junction proteins occludin, claudin-1, ZO-1, and ZO-2 were localized at cell borders between mitotic cells and neighboring cells throughout all stages of mitosis, whereas occludin and claudin-1, but not ZO-1 and ZO-2, were located in the midbody as well as at cell borders. These results indicated that the integral tight junction proteins were regulated in a different manner from the cytoplasmic proteins ZO-1 and ZO-2 during cytokinesis. Because ZO-1 and ZO-2 may be required for tight junctions to perform the barrier function (Schneeberger and Lynch 1992; Gumbiner 1993), the paracellular pathway around the midbody might be permeable. Just beneath the midbody, however, the integral and cytoplasmic tight junction proteins were localized throughout the cell borders, including between the daughter cells. Thus, the continuous epithelial seal was maintained even during cytokinesis.
It is known that many cytoskeletal proteins, including ERM, are located in the midbody (Sandig and Kalnins 1990; Sato et al. 1991). Furthermore, small GTPase RhoA, which controls the formation of actin structures (Hall 1998) and the assembly of tight junctions (Jou et al. 1998), is also concentrated in the midbody (Madaule et al. 1998). In the present experiments, however, the scaffold protein ZO-1 that links actin and tight junction proteins was not detected in the midbody, althought occludin and claudin-1 were localized in it. Taken together, these results suggest that there may be other scaffold proteins that bind to occludin and claudins or another mechanism of interaction between the integral tight junction proteins and actin filaments. Alternatively, it may be very difficult to detect ZO-1 and ZO-2 by immunocytochemistry, probably because these cytoplasmic proteins are too condensed in the midbody, since the midbody was negative for both tubulin and actin by immunocytochemistry in our study despite the observation of microtubules in it by electron microscopy.
Footnotes
Acknowledgements
Supported in part by Grants-in-Aid from the Ministry of Education, Culture, Sports and Science, and the Ministry of Welfare of Japan.
