Abstract
Mouse tibial growth plates were examined for the presence of adhesion molecules using immunohistochemistry and RT-PCR. All of the components of the classical cadherin/catenin complex (cadherin, α-, β-, and γ-catenin), as well as a heavy presence of p120, were identified in the murine growth plate. All of the major cadherins (1-5, 11, 13, and 15) were, for the first time, identified and localized in the murine growth plate. We have demonstrated that most of the cadherins and catenins reside in the zone of hypertrophy. Only α-catenin and E-, P-, R-, and VE-cadherin were found in all regions of the growth plate. The results for T-cadherin were inconclusive.
A
Most cadherins, except for T-cadherin, are transmembrane and have a cytoplasmic domain that interacts with catenins and frequently with the actin cytoskeleton (Ivanov et al. 2001; Wheelock and Johnson 2003). The N-terminal is extracellular and contains five cadherin domains, the most distal of which is responsible for recognition of other cadherin molecules (Ivanov et al. 2001). The C-terminal is intracellular and is attached to an α-catenin by a β- or γ-catenin (Ivanov et al. 2001). β-Catenin has a dual role in stabilizing cell–cell adhesion and transducing canonical Wnt signaling, as reviewed by Nelson and Nusse (2004), and a reciprocal relationship exists between the two functions (Stains and Civitelli 2005a; Mbalaviele et al. 2006).
In neuroepithelial tissues, more than 20 different cadherins are involved at various stages of embryogenesis and in different locations (Ivanov et al. 2001). In vitro studies have demonstrated a requirement for N-cadherin in the early phases of embryonic limb development. Cadherin-11 can be substituted for N-cadherin, but its persistence prevents progression from precartilage to chondrocyte development (Stains and Civitelli 2005a). Osteogenic differentiation is accompanied by downregulation of R-cadherin and upregulation of cadherin-11 (Stains and Civitelli 2005a). Because the growth plate is considered to reflect the stages of chondrogenesis, this non-quantitative project was undertaken to determine which cadherins and catenins might actually be functioning in the mammalian growth plate and at which stage.
Materials and Methods
Immunohistochemistry
Mouse tibias (from the GEMcore C57/BL6 maintained colony at Texas A&M University, College Station, TX) were dissected free from soft tissue and fixed in Carson's modified Millonig's phosphate-buffered formalin for 48 hr, and decalcified and sectioned, and every fifth section was placed, three to a slide, on Silant-coated slides (Surgipath; Richmond, IL). All procedures performed in this experiment were in accordance with the Institutional Animal Care and Use Committee guidelines (Texas A&M University). Three random slides revealing the full width of the growth plate were examined from each group. Immunohistochemistry analysis was performed using antibodies at the concentrations shown in Table 1. All antibodies used in this study were either anti-mouse or were confirmed reactive with mouse on Western blots by the manufacturer. Four-μm sections were cut and mounted on coated slides. Slides were deparaffinized in a 56C oven overnight, followed by xylene and rehydration in a graded alcohol series. Heat-induced epitope retrieval was performed for 1 hr at 70C in a Biocare (Concord, CA) Medical Decloaking Chamber using Reveal Antigen Retrieval Solution (Biocare). The slides were allowed to cool, and were rinsed in running tap water. Endogenous peroxidase activity was blocked by 3% hydrogen peroxide for 5 min. Slides were rinsed in 1 × TBS buffer and subjected for 5 min to a casein background Sniper and a 20 min Avidin-Biotin blocker (Biocare) to reduce nonspecific background and endogenous biotin staining, respectively. Following a buffer rinse, the slides were incubated with primary antibody (or buffer only, in the case of the negative controls) for 1 hr at room temperature. After rinsing with buffer for 5 min, the slides were incubated in a biotinylated secondary antibody, as specified in Table 1, for 15 min. Following a buffer wash, the slides were incubated in conjugated streptavidin horseradish peroxidase for 10 min and betazoid 3, 3′ diaminobenzidine for 5 min. All slides were counterstained briefly with hematoxylin and dehydrated through a series of alcohol and xylene before coverslipping.
Incubation conditions for the antibodies used
RT-PCR
Tibias were removed and rapidly frozen in liquid nitrogen and stored at −80C until analysis. The growth plates were dissected free from the remainder of the tibia, and the samples were pulverized in Trizol solution under liquid nitrogen in a Spex Freezer Mill (Metuchen, NJ). RNA was isolated according to the manufacturer's instructions, with slight modifications for bone. After isolation of the RNA, 50 μl of RNA was DNase treated using an RNAqueous-4 PCR kit (Ambion, Inc.; Austin, TX). Five μl of 10 × DNase buffer plus 1 μl of DNase I were added to the total RNA for 30 min at 37C, then 5 μl of DNase inactivation reagent was added for 2 min at room temperature. The solution was centrifuged at 10,000 × g for 1 min and collected. The DNase-treated RNA was then diluted 50:50 with RNase- and DNase-free water. One μl of diluted RNA was then reverse transcribed using Superscript III One-Step RT-PCR with Platinum Taq DNA Polymerase (Invitrogen; Eugene, OR) according to the manufacturer's protocol, with slight modifications in annealing temperature and cycles, as shown in Table 2. The Oligo primers used are listed in Table 2. The resultant PCR products were resolved on a 1% agarose gel containing ethidium bromide.
Results
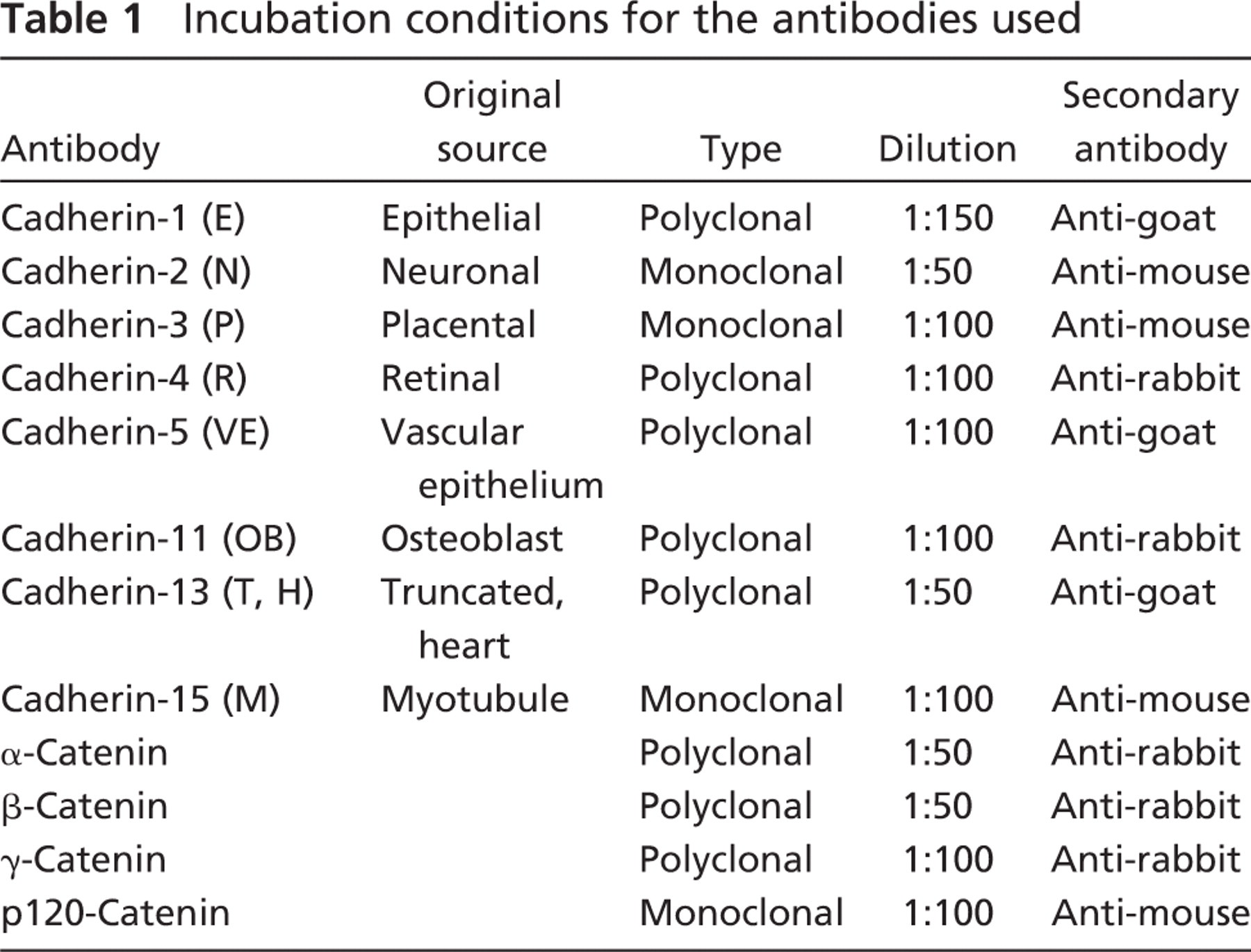
All of the catenin components of the classical cadherin/catenin complex were identified in the mouse growth plate, both histochemically and by RT-PCR. Table 3 is a semi-quantitation of the number of cells reacting in the various regions of the growth plate for each cadherin and catenin. α-Catenin had a moderate reaction in all of the zones of the growth plate (Figure 1), and the β-catenin reaction was moderate in the hypertrophic zone and the resting zone. Several repetitions of the PCR for β-catenin were all very faint, but always present. γ-Catenin reaction was much stronger in both the hypertrophic and resting zones. The reaction for p120 was very strong in the hypertrophic and resting zones of the growth plate. All of the catenin reactions appeared to be cytoplasmic, but their exact localization will require more-precise techniques.
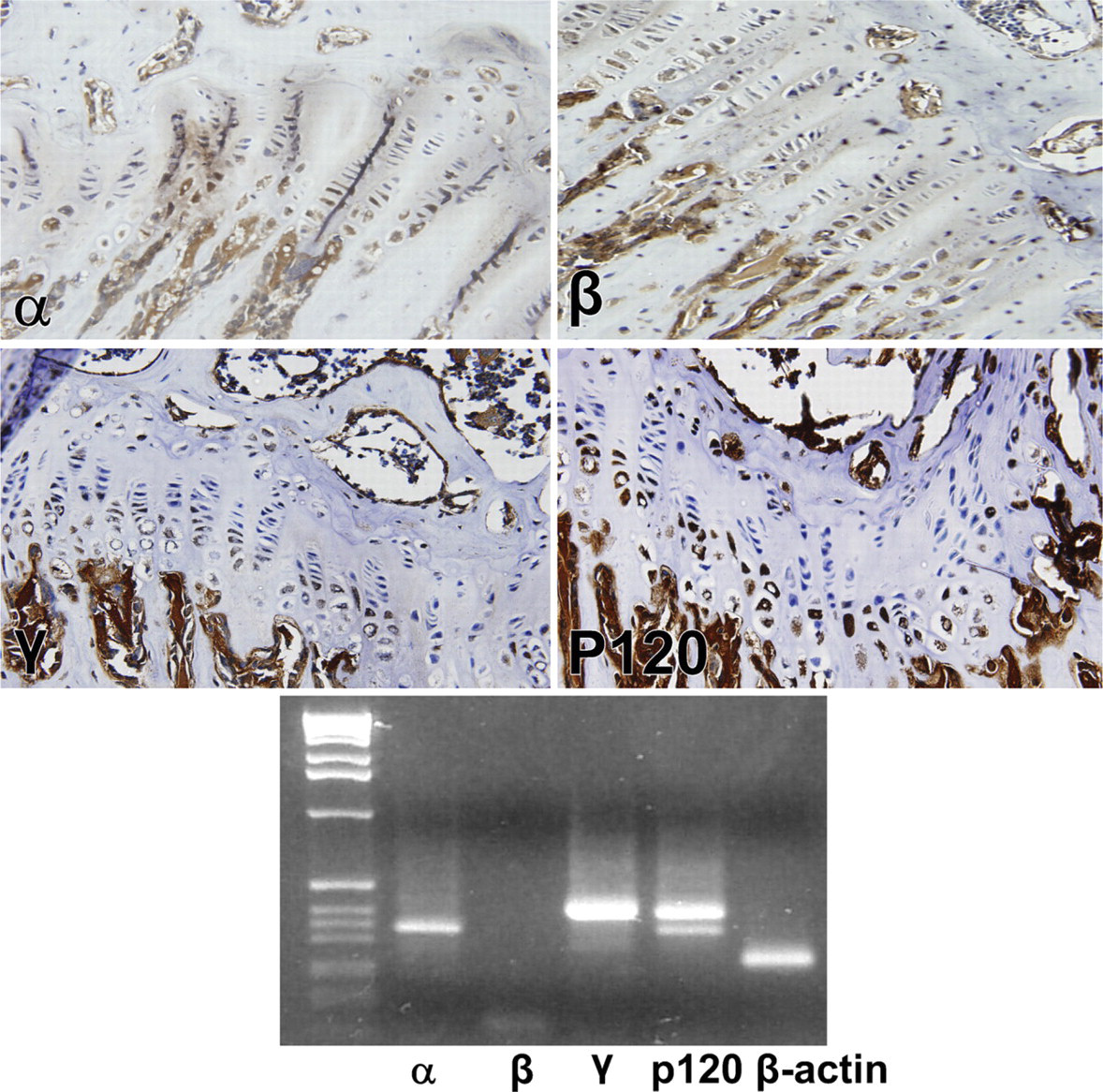
Except for T-cadherin, which was always weak to absent by PCR, the rodent growth plate was positive for all cadherins tested, both histochemically and by RT-PCR, but in slightly different distributions. N-cadherin reacted only at the upper hypertrophic zone or zone of maturation (Figure 2), whereas E-cadherin yielded reaction product in essentially all zones of the growth plate, as did P-, R-cadherin (Figure 2), and VE-, T-cadherin (Figure 3). T-cadherins' reactions were very weak by immunohistochemistry and essentially absent by RT-PCR, and its distribution, as depicted in Table 3, must be evaluated with caution. Reaction product for cadherins OB and M were limited to the zone of hypertrophy (Figure 3). All cadherin reactions were strongest in the zone of hypertrophy.
Primers and conditions used for RT-PCR

Discussion
Kawaguchi et al. (2001b) demonstrated that each mesenchymal lineage cell line has a characteristic cadherin profile expression pattern, with chondrocyte and osteoblast lineage cells expressing N-cadherin consistently and osteoblastic cell lines also expressing cadherin-11 (Cheng et al. 1998; Shin et al. 2000; Kawaguchi et al. 2001b). As these cells grew in culture, they also began to express P-cadherin. E-cadherin was not expressed in any of the mesenchymal tissue. Interestingly, many cadherins were found in early calveria primary cultures, but more-mature bone cells expressed only N-, P-, and 11-cadherin (Kawaguchi et al. 2001b). Kawaguchi et al. (2001b) explained the variable expression levels as due to either brain or blood vessel contamination or to osteoblasts at various stages of development. Our study would lend support to the latter, inasmuch as the immunohistochemistry data preclude similar contamination and demonstrate stages of development in a confined area.
Kii et al. (2004) demonstrated in teratoma formation that cadherin-11 stimulated osteogenesis in a different manner from N-cadherin and induced Runx2 and Osterix, which, like our localization for cadherin-11, are expressed in the hypertrophic zones (Nakashima and de Crombrugghe 2003; Colnot 2005). Others have demonstrated the requirement for cadherin-11 for nodule formation in MMR-14, a primary growth plate–derived chondrocyte line, which they described as late hypertrophic zone (Matsusaki et al. 2006) and in newborn calvarial cells (Kawaguchi et al. 2001a).
Cadherins mediate transmembrane signaling by their intracellular domain, which has been reported to interact with the actin cytoskeleton via the catenins α, β, and γ, possibly regulating cadherin-mediated cell adhesion (Woodward and Tuan 1999). All of the components of this classical cadherin/catenin complex were identified in the mouse growth plate, and because there has been little cell–cell contact reported in the growth plate, it is assumed that communication involves primarily cell–extracellular matrix communication. E-, P-, N- and VE-cadherins and α-and β-catenin are rapidly degraded in the absence of p120 (Davis et al. 2003), which we have found to be abundant in the growth plate. β-Catenin is known to exist in three subcellular pools: membrane bound; cytoplasmic, in association with adenomatous polyposis coli tumor suppressor protein; and a nuclear pool, in association with LEF and T-cell factors, as reviewed by DeLise et al. (2000). It appears to be involved in cytoskeletal changes, important in early chondrogenesis, through the regulation of an N-cadherin, BMP-2, MAPK (ERK1/2, P38)- (Modarresi et al. 2005) to-Sox9 pathway, creating a feedback loop. Separate, highly conserved binding sites for β-catenin and p120 on the cadherin cytoplasmic domain suggest critical and indispensable roles for both proteins in cadherin function (Thoreson et al. 2000). It is interesting that the greatest presence of the cadherins and catenins was found in the zone of hypertrophy, which might attest to its importance as a regulatory center for development in the growth plate.
Percent of cells that are positive for the reaction product
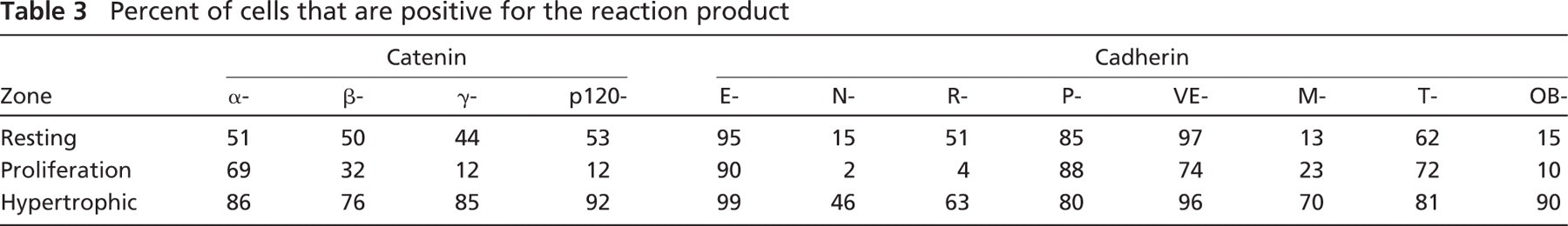
Immunohistochemical reaction of α-, β-, γ -, and p120-catenin in the mouse growth plate. α-Catenin was uniformly distributed throughout the growth plate, whereas β-, γ-, and p120-catenins were found only in the resting and hypertrophic zones. Lower panel depicts RT-PCR gel of the respective catenins.
β-Catenin binds to a distinct site on the COOH terminus of the cadherin molecule, inhibits the differentiation of chondroprogenitor cell progression, and accelerates hypertrophic chondrocyte differentiation or bone formation. So when the PTHrP/Ihh loop inhibits Sox9 through the Rock/RhoA pathway, β-catenin is also influenced through the action of the Wnt signaling cascade, which ultimately regulates Runx-2 and Osterix (Akiyama et al. 2004; Hill et al. 2005). Osterix is an absolute requirement for bone formation and is inhibitory for Sox9, the major gene for chondrogenesis (Nakashima et al. 2002; Nakashima and de Crombrugghe 2003; Hill et al. 2005).
Immunohistochemical reaction of E-, N-, R- and P-cadherin in the mouse growth plate. E-cadherin yielded reaction product in essentially all zones of the growth plate, as did P- and R-. N-cadherin reacted only at the upper hypertrophic zone or zone of maturation. Lower panel depicts RT-PCR gel of the respective cadherins.
P120 is a member of the armadillo supergene family, which has been reported to bind VE-, N-, and E-cadherins at the juxtamembrane domain and regulates their cell surface trafficking (Anastasiadis and Reynolds 2000; Thoreson et al. 2000; Ireton et al. 2002; Chen et al. 2003; Davis et al. 2003; Xiao et al. 2003), possibly through the association and recruitment of the microtubule protein kinesin (Chen et al. 2003). It has been suggested that p120 functions as a gatekeeper, determining whether cadherins are stably retained at the cell surface, or are endocytosed for degradation (Peifer and Yap 2003). In this study, p120′s pattern of distribution was more like that of the N-, OB-, or VE-cadherins, but much stronger than that of the cadherins. It has no interactions with α-catenin or actin and when overexpressed, exists in a soluble cytoplasmic pool (Thoreson et al. 2000). RhoA organizes actin stress fibers and is necessary for cell transformation by oncogenes such as src and ras (Anastasiadis et al. 2000). Cytoplasmic p120 regulates Rho-GTPase activity with high levels inhibiting Rho (Anastasiadis et al. 2000) and low p120 levels activating Rac and Cdc42 (Noren et al. 2000; Mehta 2004). Thus, p120 may function in the maintenance of cell shape and enhanced cell motility. Cadherins function as ligand-activated receptors that modulate Rac and Rho GTPase activity upon adhesive ligation, and may influence the precise sites at the plasma membrane where Rho signaling occurs (Yap and Kovacs 2003). Noren et al. (2000) demonstrated that free cytoplasmic p120 forms a complex with Vav2, a guanine nucleotide exchange factor that has activity for Rho, Cdc42, and Rac and is required for p120-induced cytoskeletal changes when it is in the cytoplasmic pool. This elevates the migratory activity of the cell (Anastasiadis and Reynolds 2001). Cadherin activates Rac and PI3 kinase signaling control processes such as proliferation and apoptosis (Yap and Kovacs 2003). Anastasiadis and Reynolds (2000) reviewed the evidence for p120 entering the nucleus, interacting with Kaiso, and modifying its transcriptional activity.
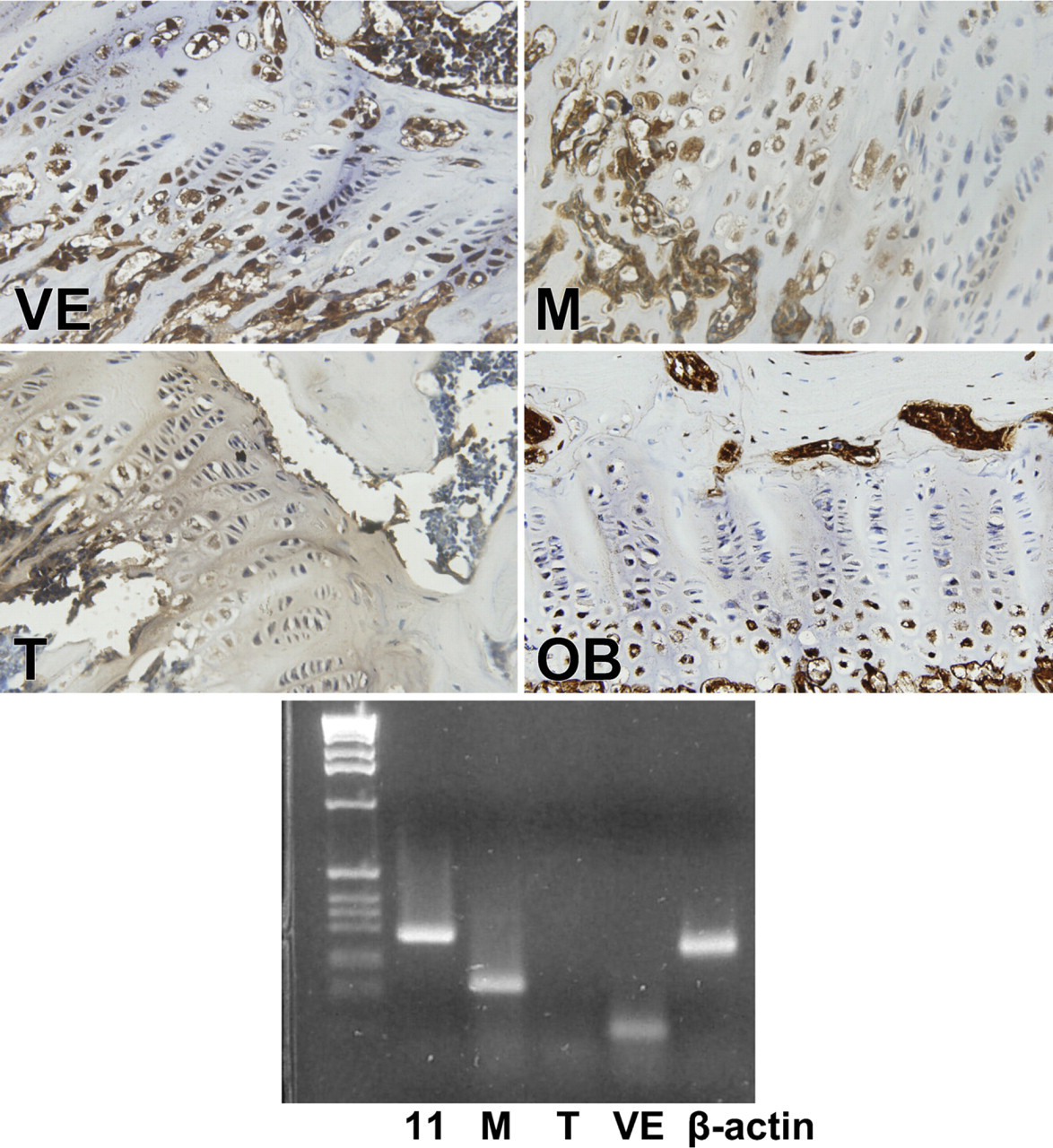
Immunohistochemical reaction of VE-, M-, T- and OB-cadherin in the mouse growth plate. VE- and T- cadherin yielded reaction product in essentially all zones of the growth plate, but the reaction of T- was very faint and inconclusive. Reaction products for OB- and M- cadherins were limited to the zone of hypertrophy. Lower panel depicts RT-PCR gel of the respective cadherins.
T-cadherin has been implicated in suppressing proliferation in nervous tissue development and tumorigenesis (Ivanov et al. 2001) and has been identified in arterial pericytes, which seem to have the ability to form osteocytes, but its appearance in the growth plate is questionable.
M-cadherin is usually associated with muscle, and Kawaguchi et al. (2001b) found it to be expressed only in mesenchymal-lineage cell lines that are myoblastic. He also demonstrated that inducing osteogenesis in muscle tissue with the introduction of BMP-2 caused a major reduction in M-cadherin expression. We were surprised to see the localization of M-cadherin in the growth plate and can only attribute it to changes in myotubule organization during the developmental process (Charrasse et al. 2006).
In conclusion: (1) We have identified, by immunohistochemistry and RT-PCR, all of the components of the classical cadherin/catenin complex in the murine growth plate; (2) we have identified, for the first time, the almost universal presence of the major cadherins in the murine growth plate; (3) we have identified a heavy presence of p120 in the mouse growth plate; (4) we have demonstrated that most of the cadherins and catenins reside in the zone of hypertrophy, implicating it as a pivotal sensor of the cells' environment and as having a highly significant role in cell signaling. Only α-catenin and E-, P-, R- and VE-cadherin were found in all regions of the growth plate. The presence of T-cadherin is inconclusive.
Footnotes
Acknowledgements
This work was supported in part by National Institutes of Health Grant RO1-CA-095608 (WEZ), grant P30 ES09106 from the Center for Environmental and Rural Health (ARP, WEZ), and the Center for Bone, Joint and Spine Research, Temple Texas (HWS).
