Abstract
Nephrin, a major intercellular junction (ICJ) molecule of mammalian podocytes in the renal glomerulus, is absent in the avian genome. We hypothesized that birds use ICJ molecules other than nephrin in their podocytes. Therefore, in the present study, we examined the possible involvement of adherens junction (AJ) proteins in the ICJs of avian podocytes. We found the AJ proteins N-cadherin and α- and β-catenins in podocytes of quail and chickens but not in those of rats, pigs or humans. The AJ proteins were prominent in avian glomerulus-rich fractions in immunoblot analyses, and in immunofluorescence microscopy analyses, they were localized along glomerular capillary walls appearing in at least two staining patterns: weakly diffuse and distinctly granular. Immunoelectron microscopy demonstrated that the significant accumulation of immunogold particles for the AJ proteins were especially evident in avian slit diaphragms and AJs. Furthermore, N-cadherin was found to be expressed in all nephron cells in the early developmental stage but became confined to podocytes during maturation. These results indicate that avian slit diaphragms clearly express AJ proteins as compared with that in the mammal—where AJ proteins are suppressed to an extremely low level—and that avian podocytes are interconnected by AJs per se in addition to slit diaphragms.
Introduction
Podocytes are epithelial cells that form the outermost layer of the glomerular capillary tuft in the kidney. They exhibit a very elaborate morphology with several primary processes radiating from the cell body to embrace glomerular capillaries. The cellular processes ramify into thinner ones resulting in numerous fine processes, or foot processes, which cover the glomerular basement membrane and exhibit interdigitation with adjacent cells. The paracellular spaces between foot processes are bridged by slit diaphragms, which are intercellular junctions (ICJs) unique to podocytes. Slit diaphragms play a critical role in the glomerular filtration barrier. The basic configuration of podocytes with slit diaphragms and foot processes is highly conserved throughout all classes of vertebrates (Takahashi-Iwanaga 2002; Ichimura et al. 2007).
Avian podocytes also have foot processes and slit diaphragms that are very similar to those of mammals. Specifically, the actin filament organization of avian podocyte foot processes is identical to that in mammals but not reptiles and amphibians, probably due to the distinctively high glomerular filtration rate in birds and mammals (Braun and Dantzler 1997; Ichimura et al. 2007). In spite of this morphological similarity, there is a crucial difference between birds and mammals: the avian genome has been reported to lack nephrin (Miner 2012; Völker et al. 2012). Nephrin is the first identified component of the slit diaphragm and a transmembrane protein of the immunoglobulin family of cell adhesion molecules (Kestilä et al. 1998). In addition to mammals, the expression of nephrin is highly conserved among several lower vertebrates, including zebrafish (Kramer-Zucker et al. 2005), medaka fish (Ichimura et al. 2013) and Xenopus (Gerth et al. 2005). It has been reported that nephrin promotes cell–cell adhesion through homophilic interactions, forming a pore structure that is capable of restricting the passage of albumin-sized molecules; this indicates that nephrin is a required structural component of the slit diaphragm (Khoshnoodi et al. 2003; Wartiovaara et al. 2004). Thus, we hypothesized that birds use ICJ molecules other than nephrin in their podocytes.
In preliminary experiments conducted to search for junctional proteins in quail glomeruli by immunohistochemistry, we found significant staining for α-catenin, a cytoplasmic protein indispensable in adherens junction (AJ), along the capillary wall. However, as per our previous report (Yaoita et al. 2002), α-catenin was undetectable in the mature podocytes of rats. The AJ is a type of cell–cell adhesion structure, in which cadherin molecules bridge the neighboring plasma membranes through their homophilic interactions (Meng and Takeichi 2009). Cadherins associate with cytoplasmic proteins, α-, β-, γ- and p120-catenins, which in turn bind to the actin cytoskeleton. In the present study, we examined cadherin and catenins in the podocytes of quail and chickens and contrasted their expression profiles with those of rats, pigs and humans by immunofluorescence and immunoelectron microscopy. We found avian podocytes to express AJ proteins significantly, and to be connected by AJs in addition to slit diaphragms that contained AJ proteins.
Materials & Methods
Animals
The fertilized eggs of Japanese quail, Coturnix japonica, were purchased from a commercial hatchery (Ebihara Farm; Tochigi, Japan). The eggs were hatched and kept as previously described (Nishimura et al. 2007). Female Wistar rats were purchased from Charles River Japan (Atsugi, Japan) and were used in these experiments at the ages of 11 to 12 weeks. The kidneys of 2-year-old female chickens and 6-month-old pigs were obtained from the Niigata Poultry Cooperative Association (Niigata, Japan) and the Niigata meat inspection center (Niigata, Japan), respectively. Human kidney tissues were obtained from patients who had undergone nephrectomies due to urological cancers and had not received any chemotherapy or radiotherapy, and the renal cortical tissues that were obtained were most distant from the tumors.
The procedures for the present study were approved by the Animal Committee at Niigata University School of Medicine (Approval Number: 52 on 5/16/2005, 44-5 on 4/27/2012, 26 on 11/7/2014) and by the Ethics Committees of Niigata University Faculty of Medicine (Approval Number: 675 on 10/28/2014). All animals were treated according to Niigata University’s guidelines for animal experimentation.
Antibodies
Mouse monoclonal anti-α-catenin (clone αCAT-7A4; Invitrogen, Carslbad, CA) was raised against the C-terminus of mouse α-catenin and reacted with human, mouse, rat, chicken and Xenopus α-catenin. Rabbit anti-β-catenin (Sigma-Aldrich; St Louis, MO) was raised against the peptide corresponding to amino acids 768–781 of human or mouse β-catenin. Mouse monoclonal anti-N-cadherin (clone 3B9; Invitrogen) was raised against the intracellular domain of chicken N-cadherin. This antibody does not cross-react with other cadherin family members, including P- and E-cadherins. Rabbit anti-podocin antibody (kindly provided by Dr. H. Tsukaguchi, Kansai Medical University, Osaka, Japan) was raised against a synthetic peptide comprising the 17 carboxy-terminal amino acids of human podocin. Rabbit anti-laminin antibody (Sigma-Aldrich) was raised against laminin from EHS mouse sarcoma and reacted with laminin of human, mammal, avian, reptilian and amphibian sources. Mouse monoclonal anti-β-actin (clone AC-15) and anti-KLH antibodies (clone 11711) were obtained from Sigma-Aldrich and R&D Systems, Inc. (Minneapolis, MN), respectively. Anti-β-actin antibody cross-reacts with β-actin-expressing cells in a wide variety of species, including chickens.
Isolation of Glomeruli
Glomeruli were isolated from the kidneys of quail at the ages of 11 and 12 weeks, and chickens at the ages of 2 years using the serial sieving method. The kidneys were cut into small pieces with a surgical blade. The minced tissue was pressed through a stainless screen mesh with 150-µm pore openings and rinsed with phosphate-buffered saline (PBS) through screen meshes with smaller-sized pores in succession (75 µm, 46 µm and 25 µm for quail; 106 µm, 75 µm and 46 µm for chickens). Samples rich in glomeruli on the 25-µm screen mesh for quails and on 46-µm screen mesh for chickens were collected and used for western blot analysis and classified as glomerulus-rich samples. Approximately 60% of tissue fragments in these samples were comprised of isolated glomeruli, and the rest were tubular fragments.
Western Blotting
The glomerulus-rich samples and superficial areas of adult quail and chicken kidneys described above were homogenized in lysis buffer (8M Urea, 1 mM DTT, 1 mM EDTA, 50 mM Tris-HCl, pH8.0) on ice. Western blotting was performed as described previously (Koda et al. 2011). Briefly, a 20-µg sample of each protein was loaded into the wells of a 10% sodium dodecyl sulfate-polyacrylamide gel, and the separated proteins transferred onto a polyvinylidene difluoride membrane. The membranes were pre-incubated with 5% nonfat milk in PBS for 2 hr, and incubated with primary antibodies overnight. The primary antibodies were diluted with 5% nonfat milk in PBS: 0.25 µg/ml mouse anti-N-cadherin, 0.1 µg/ml mouse anti-α-catenin, 0.1 µg/ml rabbit anti-podocin and 1:10,000 diluted rabbit anti-β-actin antibodies. After washing in 0.05% Tween-20 in PBS, the membranes were then incubated with a 1:1000 diluted goat anti-mouse or -rabbit immunoglobulin conjugated to peroxidase-labeled dextran polymer (mouse EnVision, rabbit EnVision; DAKO, Carpinteria, CA), and the immunoreactivity was visualized using ECL Prime Western Blotting Detection Regent (GE Healthcare; Buckinghamshire, UK).
Immunofluorescence microscopy
Indirect immunofluorescence was applied to frozen kidney sections as described previously (Koda et al. 2011). In brief, frozen kidneys were sectioned at a thickness of 3 µm in a cryostat, fixed in 2% paraformaldehyde (PFA) for 10 min, washed with PBS thrice, treated with 10% normal goat serum for 30 min, and then incubated overnight with respective primary antibodies. For double-label immunostaining, primary mouse and rabbit antibodies were mixed as follows and applied simultaneously: anti-α-catenin antibody (2 µg/ml) or anti-N-cadherin antibody (2 µg/ml) (mouse antibodies), and anti-β-catenin antibody (1:1000 dilution), anti-podocin antibody (1 µg/ml) or anti-laminin antibody (1:100 dilution) (rabbit antibody). After washing with PBS, the sections were stained with fluorescein isothiocyanate (FITC)-conjugated or tetramethylrhodamine isothiocyanate (TRITC)-conjugated anti-rabbit IgG, washed with PBS, and subsequently reacted with TRITC-conjugated or FITC-conjugated anti-mouse IgG. DAPI (4’,6-Diamidino-2-Phenylindole, Dihydrochloride) was used for nuclear staining. PBS and normal rabbit serum were used as negative controls for the primary antibodies. Immunofluorescence was observed with a laser scanning confocal microscope (A1Rsi; Nikon, Tokyo, Japan).
Immunoelectron Microscopy
Immunoelectron microscopic observations of rat kidneys were carried out as reported previously (Koda et al. 2011). In brief, 1 mm3 tissue blocks from periodate lysine PFA (PLP)-perfused kidneys were placed in the PLP fixative for 4 hr at 4°C, dehydrated, and then embedded in hydrophilic methacrylate resin. Ultrathin sections of the glycol methacrylate-embedded tissues were collected on nickel grids, stained with mouse monoclonal antibodies against N-cadherin or α-catenin, or a rabbit antibody against β-catenin, and detected with respective anti-mouse or anti-rabbit IgG coupled to 15-nm gold (GE Healthcare). Tissues were then counterstained with 5% aqueous uranyl acetate and 1% lead citrate for observation by electron microscopy (H600A; Hitachi, Tokyo, Japan). Murine monoclonal anti-KLH antibody was used as a negative control for the primary antibody.
Histological Examination
Following quail decapitation, kidneys from embryos were quickly excised and immersed in 4% PFA (pH 7.2) for 3 days, dehydrated in ethanol, and embedded in paraffin. Left and right kidneys were collected from E15 embryos. Tissues were sliced (longitudinal sections) to a thickness of 3–4 µm and stained with periodic acid-Schiff (PAS) for morphological examination. Histological examination was conducted with an Olympus model BX51 microscope (Olympus Optical Co., Ltd; Tokyo, Japan).
Results
Western Blot Analysis using Avian Kidney
Western blot analysis was performed to confirm the specificity of the antibodies used in this study and to elucidate the distribution of AJ proteins in the kidney by comparing between lysates from glomerulus-rich samples and superficial areas of adult kidneys (Fig. 1). Each band was located at the position corresponding to the expected molecular weight of the antigen. Antibodies against N-cadherin and podocin detected a significant-sized band in the glomerulus-rich samples but did not—or did at only a low level—detect a band in samples corresponding to superficial areas, indicating glomerular predominance in the kidney. In contrast, antibodies against α-catenin, β-catenin (data not shown), and β-actin revealed nearly identical signals in both samples, and showed no difference in intensity.
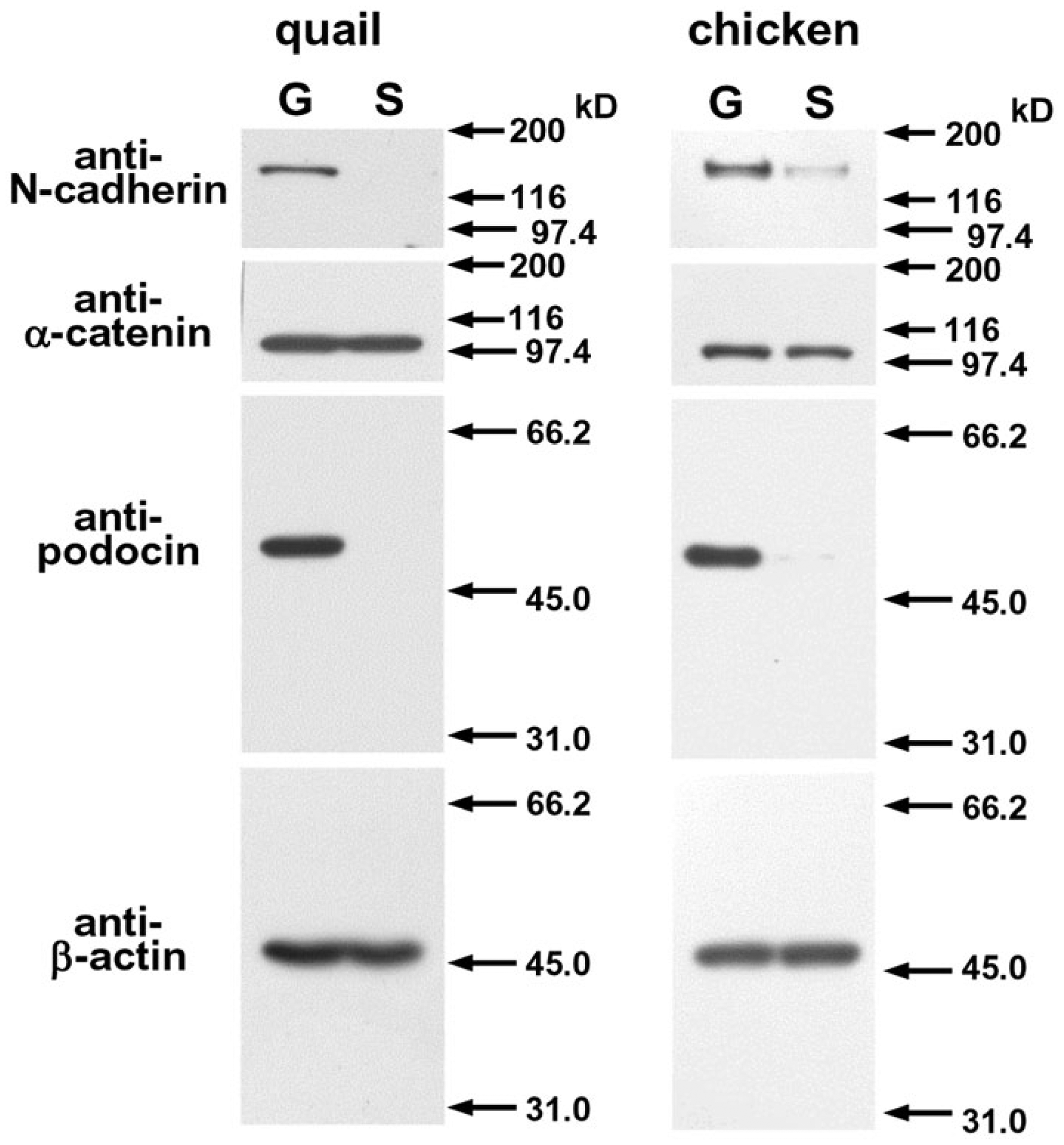
Western blot analysis of N-cadherin, α-catenin, podocin and β-actin in lysates from glomerulus-rich samples (G) and superficial areas (S) of adult quail and chicken.
Immunolocalization of α-Catenin in the Glomerulus
α-Catenin staining in the glomerulus was compared among quail, chickens, pigs, rats and humans; this is because α-catenin is shared by all AJs, whereas cadherin types vary among different types of cells (Fig. 2). Podocin staining was used as a specific marker for the ICJs of podocytes including slit diaphragms and tight junctions (Roselli et al. 2002; Kawachi et al. 2003; Shono et al. 2007). Double-label immunofluorescence microscopy revealed colocalization of α-catenin and podocin in quail and chicken glomeruli (Fig. 2A–2B”). In contrast, human glomeruli did not show α-catenin staining to be colocalized with podocin (Fig. 2C–2C”). Glomeruli from the rat or pig also showed no colocalization (data not shown), as reported previously (Yaoita et al. 2002). All animals showed distinct α-catenin staining of parietal epithelial cells of Bowman’s capsule, glomerular endothelial cells, and tubular epithelial cells. β-Catenin staining (data not shown) was identical to that of α-catenin staining.
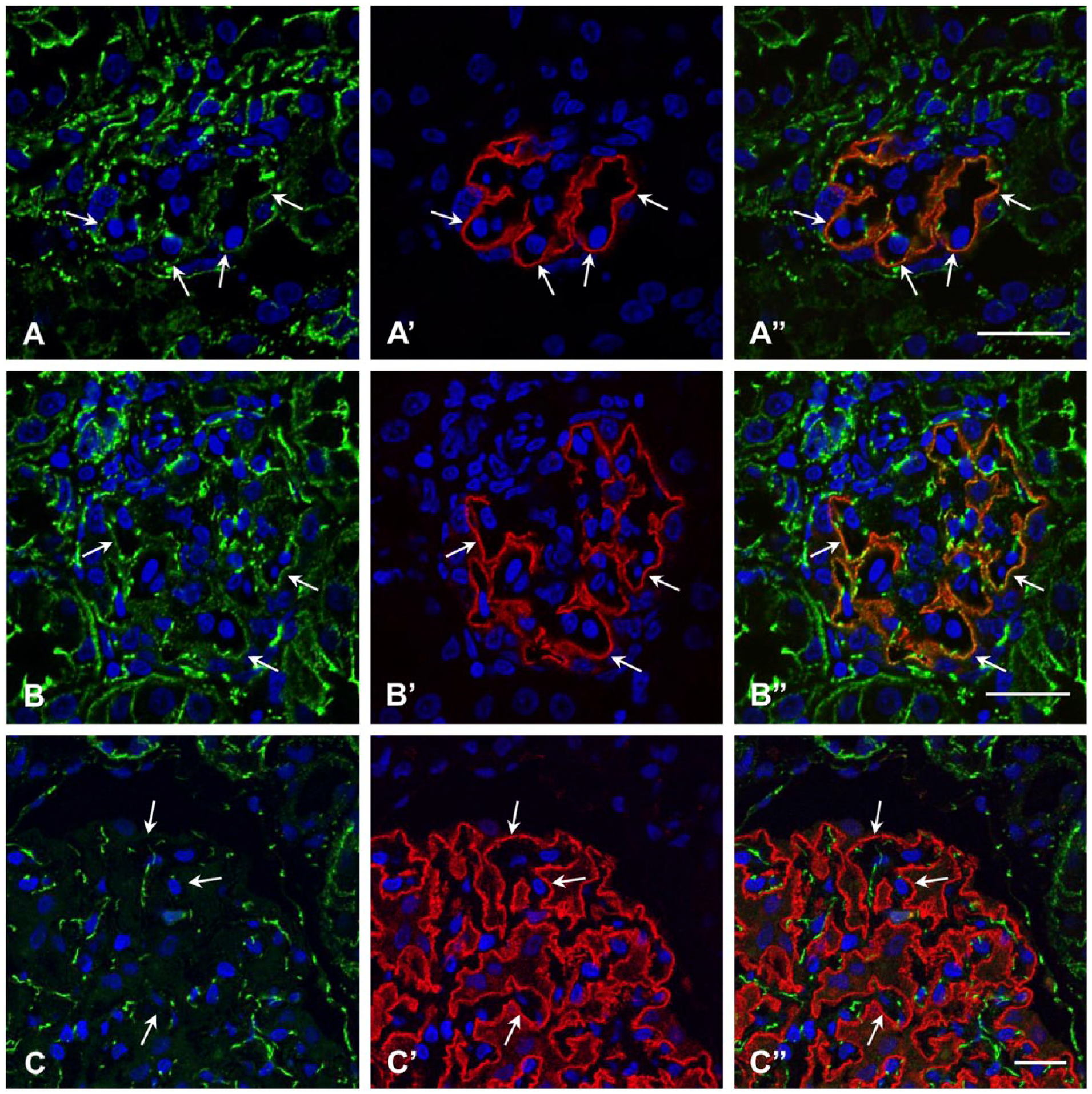
Indirect immunofluorescence microscopy for α-catenin in the glomeruli of quail (A–A”), chicken (B–B”) and human (C–C”). α-catenin (A–C, A”–C”) is stained green; podocin (A’–C’, A”–C”) is red. Podocin is colocalized with α-catenin in quail and chicken (arrows in A–B”) but not in human (arrows in C–C”). α-Catenin is also seen in glomerular endothelial cells, parietal epithelial cells of Bowman’s capsule, and tubular cells. Scale, 20 µm.
Immunolocalization of N-cadherin in the Glomerulus
N-cadherin staining was observed significantly in glomeruli of the avian kidney (Fig. 3A). Similar to α-catenin, N-cadherin colocalized with podocin (Fig. 3B, 3B’, 3E, 3E’), and was located on the outer side of the basement membrane stained with anti-laminin antibody (Fig. 3C–3D’, 3F, 3F’); this indicated localization of N-cadherin in podocytes. The staining showed at least two types, weakly diffuse and distinctly granular (Fig. 3D, 3D’, 3F, 3). N-cadherin colocalizing with podocin was undetectable in rat, pig or human glomeruli (data not shown), as reported previously (Nouwen at al. 1993; Nameta et al. 2009).
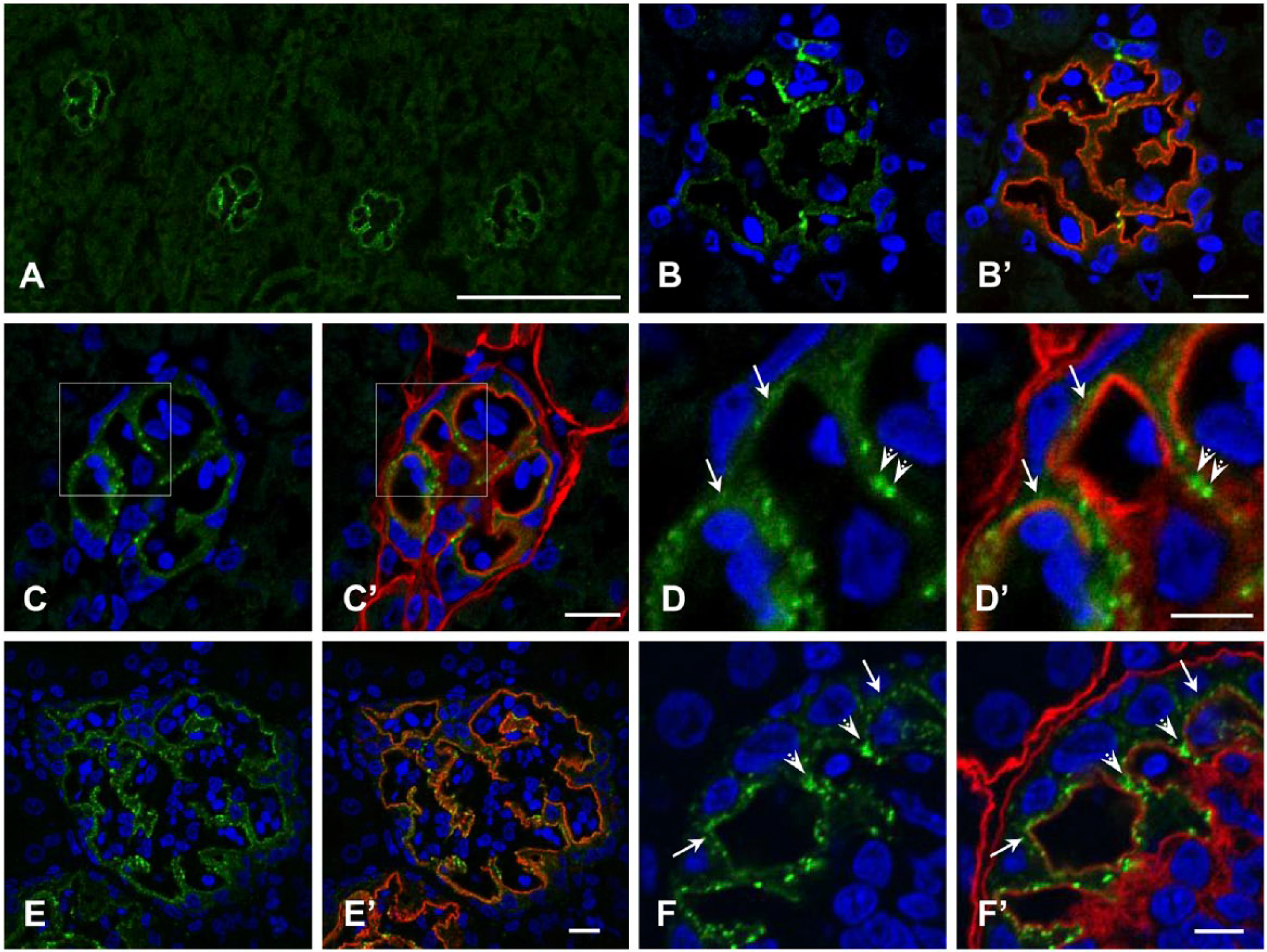
Indirect immunofluorescence microscopy for N-cadherin in glomeruli of quail (A–D’), chicken (E–F’). N-cadherin (A–F’) is stained green; podocin (B’, E’) and laminin (C’, D’, F’) are red in double-labeled immunofluorescence. N-cadherin is seen along the glomerular capillary wall in both birds. At least two types of staining pattern, weakly diffuse (arrows in D, D’, F, F’) and distinctly granular (arrowheads in D, D’, F, F’), are observed. (D) and (D’) are enlargements of the boxed areas in (C) and (C’), respectively. N-cadherin colocalizes with podocin (B, B’, E, E’). Scale (A) 100 µm; (B’, C’, E’) 10 µm; (D’, F’) 5 µm.
Localization of AJ Proteins in the Podocyte
Immunoelectron microscopy using the immunogold technique for N-cadherin and α- and β-catenins in avian podocytes was carried out to examine which podocyte structures were labeled with the antibodies (Fig. 4). Gold particles for these three AJ proteins were mainly observed at the base of slit diaphragms, which were labeled with one gold particle in most cases (Figs. 4A, 4C, 4E). In contrast, the assemblage of several particles was detected at ICJs, different from the slit diaphragms. The ICJs were characterized by intercellular bridges and intercellular spaces that were narrower than those of slit diaphragms (Figs. 4B, 4D, 4F). Some ICJs were located on the apical side of foot processes, and were observed to connect primary processes or cell bodies of adjacent podocytes.
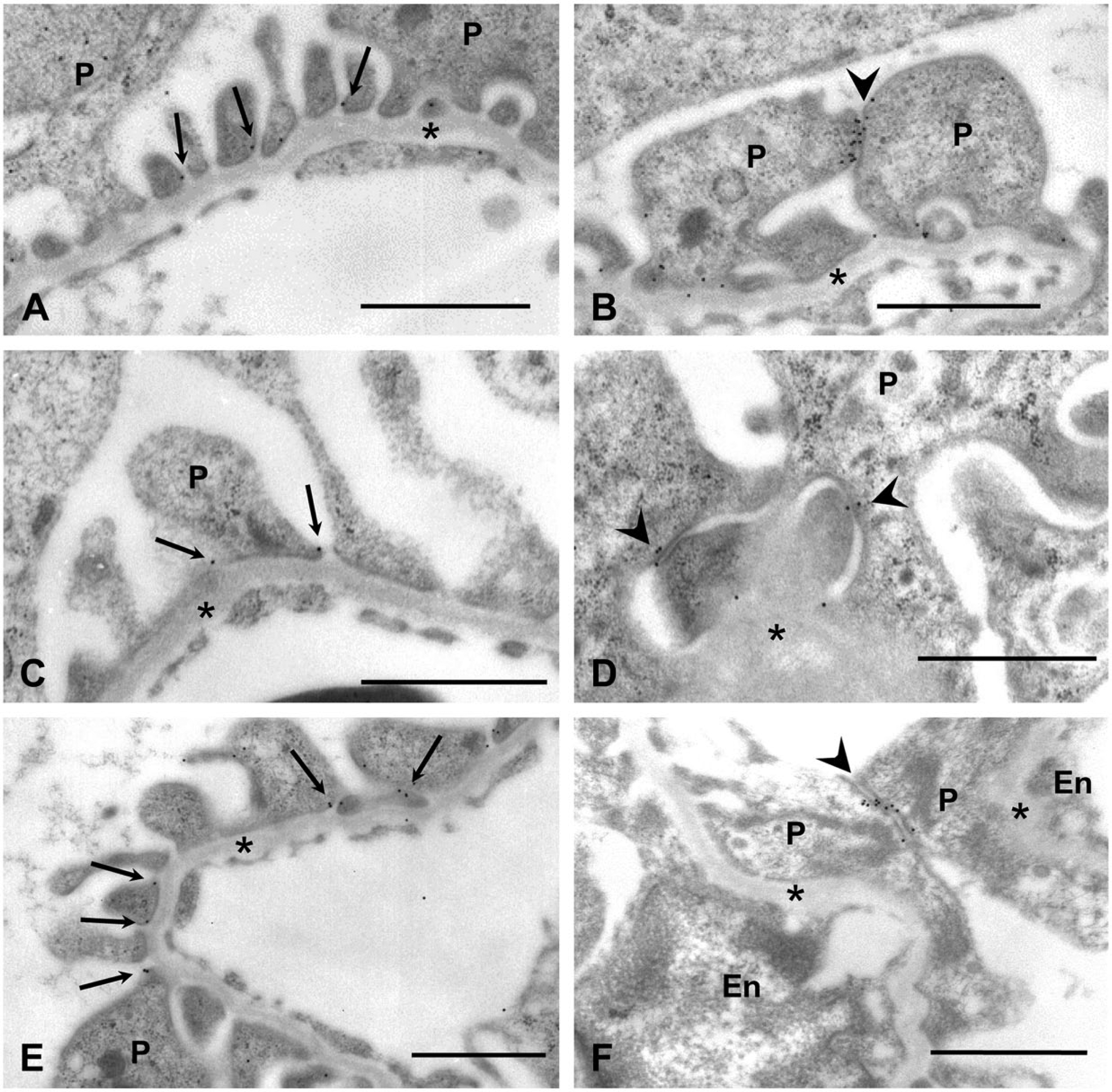
Immunoelectron microscopy for N-cadherin (A, B, F), α-catenin (C, D), and β-catenin (E) in ICJs of quail (A–E) and chicken (F) podocytes. Immunogold particles are seen at the base of slit diaphragms (arrows in A, C, E). Accumulations of particles are detected at ICJs, different from slit diaphragms (arrowheads in B, D, F). Some ICJs connect primary processes of adjacent podocytes. P, podocyte; En, endothelial cell; asterisk, glomerular basement membrane. Scale, 1 µm.
N-cadherin Localization in the Embryonic kidney
N-cadherin expression during podocyte differentiation was examined in quail kidneys at E15, when every stage of the developing nephron can be observed in the kidney (Nishimura et al. 2007) (Fig. 5). The nephrogenic zones, consisting of cells with a high proportion of nucleus, were located near the surface area of kidneys (Fig. 5A). Nephron development started with the condensation of mesenchymal cells around the tips of ureteric buds, and this was followed by various stages of glomeruli from the S-shaped body stage to the maturing stage (Fig. 5B–5E). Immature glomeruli were located in the superficial region of the kidney, with more mature glomeruli found in the deeper region.
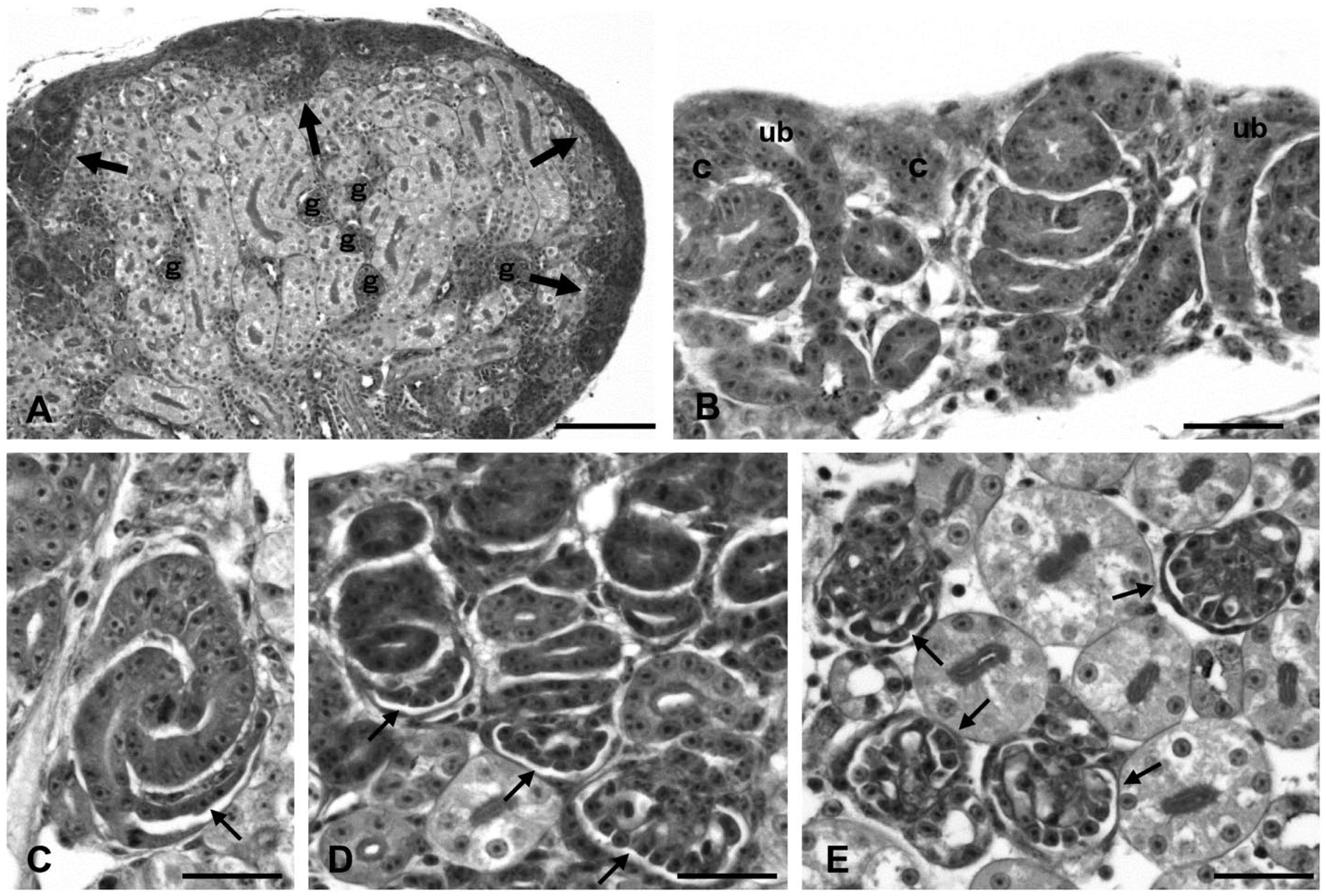
Kidney at embryonic day 15 (periodic acid-Schiff staining). Nephrogenic zones (arrows) are observed near the surface area of the kidney, and glomeruli (g) are found in the deeper region (A). In the nephrogenic zone, the mesenchymal condensate (c) appears around the terminal ampulla of the ureteric bud (ub) (B). Glomeruli at various stages, including the S-shaped body, capillary loop, and maturing stages (arrows), are found in the superficial region of the cortex (C–E). Scale (A) 80 µm; (B–E) 20 µm.
In E15 kidneys, N-cadherin was detected in the nephrogenic zones in addition to the developing glomeruli and in cells lining the surface of the kidney (Fig. 6A). Double-labeling with anti-podocin antibody showed that N-cadherin is expressed by developing nephron cells before presumptive podocytes are recognized (Fig. 6B). Cells in the condensation around the tips of ureteric buds were stained with anti- N-cadherin antibody (Fig. 6C). In the S-shaped body stage, all nephron cells were positive for N-cadherin (Fig. 6D). With maturation, all cells except podocytes lose N-cadherin staining (Figs. 6E–6G).
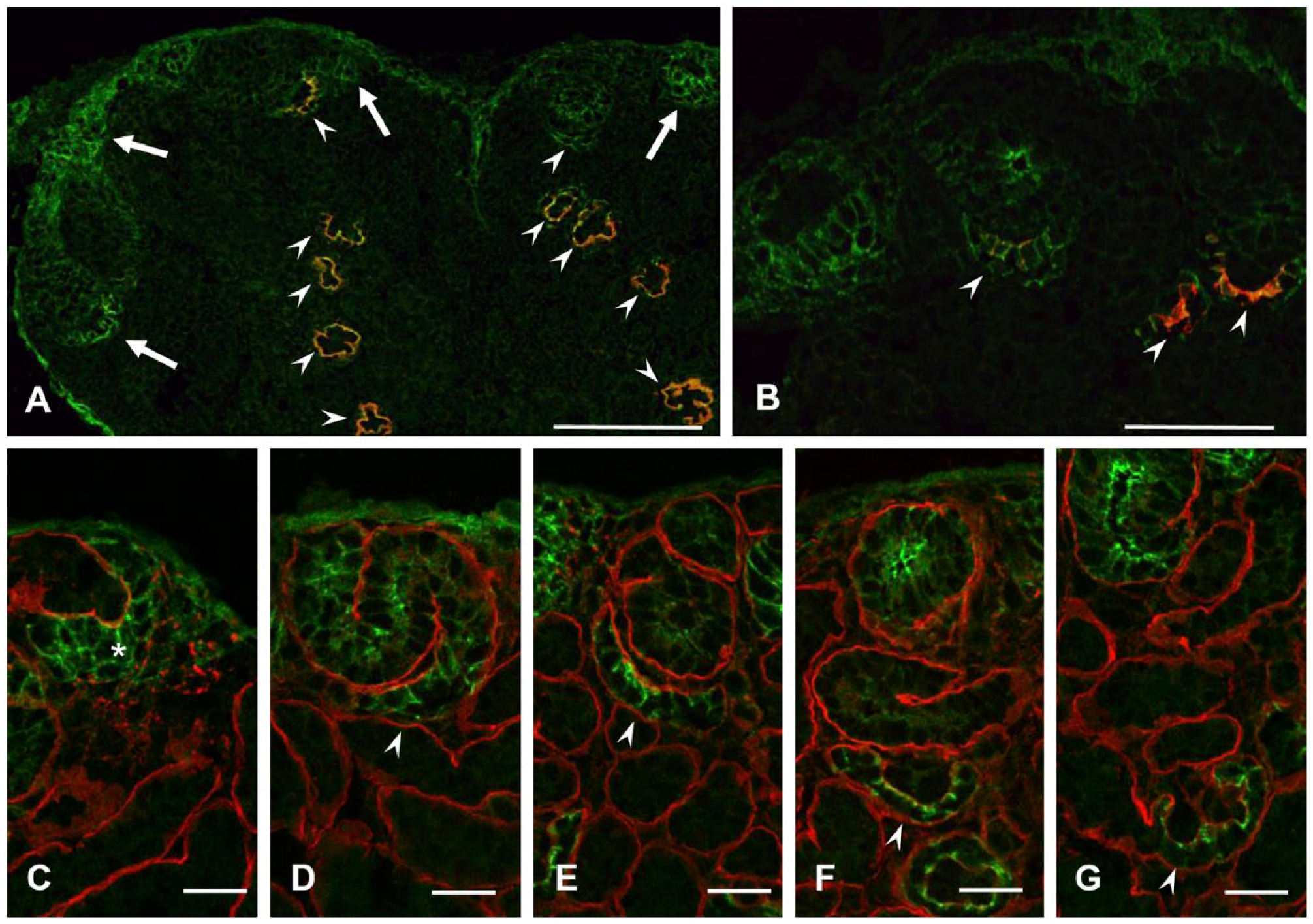
Indirect immunofluorescence microscopy of quail embryonic kidney for N-cadherin. N-cadherin is stained green (A–G); podocin (A, B) and laminin (C-G) are stained red. Podocin staining shows N-cadherin in nephrogenic zones near the surface of the kidney in addition to podocin-positive glomeruli and cells lining the surface of the kidney (A, B). Condensation (asterisk) around the tip of the ureteric bud is positive for N-cadherin (C). With maturation of glomeruli, N-cadherin staining is localized to podocytes (D–G). Scale (A) 100 µm; (B) 50 µm, (C–G) 20 µm.
Discussion
In the present study, the distribution of AJ proteins was examined in the glomeruli of quail and chickens by contrasting them with those of mammals by immunofluorescence and immunoelectron microscopy. Reactivity and specificity of antibodies are critical in immunostaining. The antibodies used in this study have been shown to react with antigens in a variety of species including chickens and mammals. The rabbit antibody against podocin was raised against the human antigen, and its cross-reactivity with avian podocin has not been examined. Western blot analysis, therefore, was performed to confirm the reactivity and specificity of the antibodies using quail and chicken samples. The position of the specific bands in the western blot was compatible with the molecular weight of each protein. In addition, the amino acid sequences of the antigen peptide of podocin are the same as that in chicken podocin. Thus, it is appropriate to assume that these antibodies react with each of the avian antigens.
In this study, immunofluorescence and immunoelectron microscopy yielded consistent findings with regard to the localization of AJ proteins in avian podocytes. Weakly diffuse and distinctly granular fluorescence corresponded to low-density immunogold particles on slit diaphragms and high-density ones on ICJs, with narrower intercellular spaces than those of slit diaphragms, respectively. The AJ was originally defined using ultrastructural analysis (Farquhar and Palade 1963). It is characterized as a region at the interface of two adjacent cells with opposing plasma membranes, 10–20 nm apart. The AJ’s intercellular space is occupied by rod-shaped molecules bridging the plasma membranes (Hirokawa and Heuser 1981; Miyaguchi 2000), which is apparently different from a slit diaphragm with a wider intercellular space of 35–40 nm (Rodewald and Karnovsky 1974). Our immunolocalization study revealed that the ICJs decorated with immunogold particles for AJ proteins had the aforementioned morphological features, and that slit diaphragms also contained AJ proteins. The findings indicate that avian podocytes are connected with AJs per se in addition to slit diaphragms containing AJ proteins. AJs have not been reported in the adult podocytes of mammals, even by ultrastructural studies.
Both quail and chicken podocytes were stained with anti-N-cadherin antibody. Although multiple cadherin types are co-expressed in many types of cells, we could not examine the presence of the other cadherin, because, with the exception of N-cadherin, reliable antibodies with cross-reactivity to avian cadherin were not available. It is likely that the other types of cadherin are expressed in avian podocytes. N-cadherin staining was observed in the early stages of quail metanephric development. All cells were positive for N-cadherin in the mesenchymal condensate around the terminal ampulla of the ureteric buds and in the S-shaped body stage. In the later stages, staining was restricted to podocytes during maturation. In the developing mammalian kidney, cadherins are expressed only in the S-shaped body stage and capillary loop stage, and disappear during the maturing stage (Nouwen et al. 1993; Tassin et al. 1994; Cho et al. 1998; Goto et al. 1998; Usui et al. 2003). P-cadherin was reported in rat podocytes in the adult (Reiser et al. 2000), but we have not been able to succeed in reproducing these experiments. Catenins in mammalian podocytes have also been shown to be expressed in the early stages of nephron development and shown to disappear in parallel with the appearance of slit diaphragms (Tassin et al. 1994; Goto et al. 1998; Usui et al. 2003). Thus, AJ proteins in mature podocytes are suppressed to extremely low levels in mammals, which is in clear contrast to their persistent staining in avian podocytes.
The N-cadherin expression of podocytes shared by two different genera of birds suggests that cadherins may play a significant role in podocyte function. Like nephrin, cadherin molecules are transmembrane junctional proteins and connect cells by homophilic interactions. However, further investigation is required to elucidate whether N-cadherin is a substitute for nephrin in birds lacking nephrin. In general, AJs composed of cadherin–catenin complexes are the main cell–cell adhesion structure and whose cytoplasmic face is linked to the actin cytoskeleton (Baum and Georgiou 2011; Yonemura 2011). Their disruption causes loosening of cell–cell contacts, leading to the disorganization of tissue architecture. To maintain a high glomerular filtration rate, the glomerular capillary wall endures a high transmural pressure gradient, which favors capillary expansion. In addition, the blood pressure of fowl is significantly higher than that of mammals (Grubb 1983; Nishimura et al. 2001); although, glomerular capillary pressure in birds has not been determined. Podocytes forming the outermost layer of the glomerulus must stabilize their structure, including ICJs and the cytoskeleton, to counteract the expansile forces (Ichimura et al. 2007). Thus, it is plausible that the existence of AJs and AJ proteins in slit diaphragms may provide structural strength to the ICJs of avian podocytes.
The present study indicates that some AJs were located at the apical side of foot processes. Like tight junctions, slit diaphragms contain Par3, Par6, and atypical protein kinase C cell polarity proteins (Hartleben et al. 2008; Hirose et al. 2009). Slit diaphragms also serve as a fence maintaining the polarized distribution of membrane proteins by restricting the intermixing of distinct apical and basolateral membrane proteins, as shown by the distribution of podocalyxin, which is the major sialoprotein of the glomerulus (Schnabel et al. 1989). The finding of AJ localization in this study indicates that AJs in quail podocytes localize in the apical domain of the plasma membrane. This is intriguing because the AJs of typical epithelial cells lie at the basal side of tight junctions. AJs above slit diaphragms are also observed in the immature podocytes of mice; although AJs appear transiently during development (Goto et al. 1998). The intriguing distribution of AJs suggests a unique polarity phenotype of podocytes. We previously demonstrated in rat podocytes that a tight junction protein, claudin-5, was not only located at tight junctions but also present on both the apical and basolateral domains of the plasma membrane (Koda et al. 2011). These findings suggest that the polarity of podocytes may be different from that of general epithelial cells.
In summary, in this study, we showed that AJ proteins were present in slit diaphragms and that AJs interconnected avian podocytes, which is quite in contrast to that observed for mammalian podocytes. These features indicate different properties among avian podocytes from those of mammals.
Footnotes
Acknowledgements
We thank Dr. Hiroshi Tsukaguchi, Kansai Medical University, Osaka, Japan, for his kind gift of rabbit anti-podocin antibody.
Author Contributions
YZ, BX, TO, HK and FN performed the immunofluorescence microscopy using frozen tissues. MN and TY accomplished the immunoelectron microscopy. HT, SM and HF carried out the Western blot analysis. EY, HN and YY designed the study and drafted the manuscript. All authors have read and approved the final manuscript.
Competing Interests
The authors declared no potential competinng interests with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant-in-Aid for Scientific Research (C) from the Ministry of Education, Culture, Sports, Science and Technology (No. 24591190 to E.Y. and No. 21390307 to H.N.).
