Abstract
The mechanism of osteosclerosis associated with myelofibrosis in megakaryocyte (MK)-related myeloproliferative disorders is largely unknown. However, growth factors released from the bone marrow cells, including from MKs, have been implicated in myelofibrosis, osteosclerosis, and angiogenesis. GATA-1 is a transcription factor required for normal MK development. GATA-1 deficiency in mice (GATA-1low) leads to increased megakaryocytic proliferation, followed by osteosclerosis and myelofibrosis. In this study we investigated the expression of bone morphogenetic proteins (BMPs) and BMP receptors and their possible role in the development of osteosclerosis in the MKs of 12-month-old GATA-1low mice by immunohistochemistry, cytomorphometry, and quantitative real-time PCR. Marrow MKs from both wild-type and GATA-1low mice showed moderate to intense staining for BMP-2, −4, and −6 and BMPR-IA and BMPR-II, whereas splenic MKs showed no BMP immunostaining. Presence of BMP protein in the bone marrow of GATA-1low mice was more than that seen in controls, owing to an increased number of MKs and osteoblasts. The osteosclerosis seen in GATA-1low mice appeared not to be due to a reduced number of functional osteoclasts because the number of tartrate-resistant acid phosphatase-positive osteoclasts was greater in GATA-1low mice than in controls. Our findings demonstrate the presence of significant amounts of BMP-2, −4, and −6 along with their receptors in bone marrow MKs of WT and GATA-1low mice. The increased levels of BMPs appear to be a result of increased numbers of MKs in GATA-1low mice and may, in part, account for the stimulation of osteoblastic activity and resulting osteosclerosis.
B
MKs arise from pluripotential stem cells through a series of identifiable stages of differentiation, culminating in terminally differentiated MKs, whose major function is to form and release platelets. As was recently reviewed (Kacena et al. 2006a), the molecular dissection of the MK differentiation pathway has been greatly facilitated by the identification of transcription factors required for the cells' successful advance from one stage to another. Loss of these specific factors precludes the cells from full maturation and results in the accumulation of cells at the latest stage of differentiation prior to the arrest. In brief, FOG (Friend of GATA) is required for the multipotential progenitor to become the MK progenitor. FOG is also required for the MK progenitor to develop into an immature MK. GATA-1 is required for the immature MK to become a mature MK, and NF-E2 and its heterodimer maf are required for the mature MK to become a terminally mature MK. GATA-1low mice exhibit a phenotype characterized by a marked megakaryocytosis and thrombocyto-penia (Shivdasani et al. 1997; Vyas et al. 1999). The thrombocytopenia in GATA-1low mice results from maturational arrest of MK differentiation at a stage between immature and mature MK development. Interestingly, although these mice develop myelofibrosis after 12 months of age (Vannucchi et al. 2002), osteosclerosis is evident by 3 to 5 months of age (Kacena et al. 2004). In addition to the increase in MKs in the bone marrow of GATA-1low mice, these mice have elevated levels of TGF-β1, PDGF, VEGF, and OC in their femurs as compared with normal mice (Vannucchi et al. 2002). Because BMPs are well known for their role in bone induction and repair (Urist 1997; Wozney and Rosen 1998), we wanted to test whether increased BMP expression in bone marrow MKs might be at least partly responsible for the osteosclerosis typically seen in GATA-1low mice. The hypotheses for our current study are that (1) MKs in the bone marrow of GATA-1low mice contribute to osteosclerosis by stimulating bone formation via increased release of BMPs, which up-regulate osteoblast proliferation and differentiation and (2) osteosclerosis seen in 12-month-old GATA-1low mice is not due to lower osteoclast number and/or reduced activity.
Materials and Methods
Generation and Maintenance of GATA-1low Mice
The generation and maintenance of mutant mice with low expression of GATA-1 transcription factor in MKs was carried out as described previously (McDevitt et al. 1997; Shivdasani et al. 1997; Kacena et al. 2004).
Tissue Preparation
Tibias and spleens were dissected from 12-month-old wild-type (WT) C57BL/6 mice and GATA-1low mice and fixed for 24 hr in 4% paraformaldehyde at 4C. After fixation, tissues were decalcified in 10% EDTA for 7 days at room temperature and then embedded in low-melting temperature paraffin and sectioned at 5-μm thickness. Hematoxylin and eosin (H-E) staining was performed, and the number of MKs and degree of osteosclerosis were compared between WT and GATA-1low splenic and tibial sections using histomorphometry.
Tartrate-resistant Acid Phosphatase (TRAP) Staining and Osteoclast Histomorphometry
Tibial sections from GATA-1low and WT mice were incubated in 1 mM ferrous sulfate and 10 mM ascorbic acid prepared in TBS for 1 hr in the dark at room temperature (protocol kindly provided by Dr. Lovitch and Dr. Rios; University of Missouri, Kansas City, MO) and stained for TRAP, using a Sigma TRAP kit (no. 386-A; Sigma Chemical Co., St Louis, MO). Cytomorphometric parameters, namely, osteoclast number/mm2 and osteoclast surface/bone surface, were analyzed by Osteomeasure software (OsteoMetrics; Atlanta, GA).
Immunohistochemistry
Deparaffinized, rehydrated sections were preincubated in 0.3% Triton X-100 in PBS at room temperature for 15 min. After Triton X-100 pretreatment, the sections were washed several times in deionized Millipore (Bedford, MA) filtered H2O and briefly incubated with 3% H2O2 for 10 min at room temperature to block endogenous peroxidase activity. Sections were washed with deionized water and PBS before blocking with 1.5% blocking serum for 1 hr followed by overnight incubation in primary antibody solution at 4C. Primary antibodies (polyclonal goat anti-human BMP-2, −4, and −6; BMPR-IA and BMPR-II) were diluted (1:50) in PBS. After incubation in primary antibody, sections were washed in PBS three times. PBS-washed sections were incubated in appropriately diluted, biotinylated secondary antibody (biotinylated anti-goat immunoglobulins) followed by incubation in streptavidin-peroxidase and buffered substrate solution (pH 7.5) containing hydrogen peroxide and 3,3′ DAB chromogen solution. Sections were counterstained in Meyer's hematoxylin for 5 min and then mounted with Cytoseal-60 (Richard-Allan Scientific; Kalamazoo, MI) before viewing microscopically.
Primary Antibodies
Anti-BMP-2, −4, and −6 and anti-BMPR-IA and anti-BMPR-II were polyclonal goat antibodies obtained from Santa Cruz Biotechnology (Santa Cruz, CA).
Preparation of MKs From Fetal Liver Cells
Preparation of MKs from fetal liver cells was done according to the method previously described by Kacena et al. (2006b). Fetuses were dissected from pregnant GATA-1low or WT mice at E13.5–15.5. Livers from fetuses were removed, and single cell suspensions were made by serially passaging of whole liver through 18-, 20-, and 22- or 23-gauge needles. Cells from five fetal livers were seeded in 100-mm culture dishes containing DMEM medium + 10% fetal calf serum + 1% murine thrombopoietin (TPO) derived from the supernatant of the TPO-producing fibroblast cell line GP 122 (~0.1 μg/ml purified recombinant murine TPO) (Villeval et al. 1997). Thrombopoietin is a major MK growth factor and is required for MK proliferation and maturation (Bartley et al. 1994; de Sauvage et al. 1994; Wendling et al. 1994; Zeigler et al. 1994). Upon confluence, MKs were isolated by density gradient separation using a single-step albumin gradient in which the bottom layer was 3% albumin in PBS and the middle layer was 1.5% albumin in PBS (Drachman et al. 1997). The MK fraction was collected from the bottom of the tube, washed twice, and then subjected to RNA isolation as described below. The MK fraction obtained from albumin density gradient separation was ~90% pure (Drachman et al. 1997), whereas the remaining cells or the non-MK fraction was comprised of non-MKs, MK precursors, and a small number of MKs.
Real-time RT-PCR Analysis
Real-time PCR was performed in a two-step discontinuous procedure as follows: (a) reverse transcription step leading to the synthesis of cDNA from total RNA (including the synthesis of BMP-2, −4, and −6 cDNA) and (b) quantitative realtime PCR leading to the relative quantitation of BMP mRNA expression levels in MKs vs. osteoblasts.
Reverse Transcription
Total RNA was extracted from MKs obtained from fetal liver cells from pregnant WT or GATA-1low mice using RNeasy Mini Kits (Qiagen; Santa Clara, CA). Total RNA was also extracted from mouse calvarial primary osteoblasts and served as positive control. Reverse transcription reactions were performed in a 20-μl reaction volume containing 10X PCR buffer, MgCl2, dNTPs, murine leukemia virus reverse transcriptase, oligo dT, and total RNA (1 μg) using the reaction conditions specified for the reverse transcription kit (Promega; Madison, WI). cDNA was kept at 220C until used for PCR.
Real-time quantitative PCR was performed in a 20-μl reaction volume using the standard protocols of Applied Biosystems 7500 Sequence Detection System and software (Applied Biosystems; Foster City, CA) using 1:10 diluted cDNA, Universal PCR Master Mix kit, SYBR green, 300–900 nM of each primer. Primer pairs of BMP-2, −4, and −6 were obtained using a search in Prime Bank (BMP-2 ID #6680794a1; BMP-4 ID #6680796a2; BMP-6 ID #6680798a2) (Wang and Seed 2003) and for GAPDH as endogenous control (forward: 5′ AAC GAC CCC TTC ATT GAC-3′, reverse: 5′ TCC ACG ACA TAC TCA GCA C-3′) were identified using BLAST program www.ncbi.nlm.nih.gov)). Relative quantitation of target mRNA expression, normalized to an endogenous control and relative to a calibrator, was calculated using the mathematical expression for fold change i.e., 2−ΔΔCt (fold) as described by Livak and Schmittgen (2001), where ΔCt = Ct of the target gene - Ct of the endogenous control gene (GAPDH), and ΔΔ Ct = ΔCt of the samples for target gene - ΔCt of the calibrator for the target gene.
Statistical Analyses
Quantitative data are presented as mean ± SEM. Statistical significance between groups was determined by ANOVA.
Results
Increased Numbers of MKs Expressing BMPs and Their Receptors in the Bone Marrow of GATA-1low Mice
An abnormally low level of expression of GATA-1 (GATA-1low) in mice resulted in excessive accumulation of immature MKs in the bone marrow (Figure 1) and spleen (data not shown). In addition to increased MK numbers (Figure 2), MKs in the bone marrow of 12-month-old GATA-1low mice were more immature as evidenced by the presence of larger nuclei with more intense chromatin staining (Figure 1B vs Figure 1A). Histomorphometric counts of MKs confirmed the increased number of MKs in the tibial marrow and spleen of 12-month-old GATA-1low mice (Figure 2). There was a striking increase in the number and size of bone trabecula per unit area in GATA-1low vs WT medullary cancellous bone (Figure 3B vs Figure 3A).
Increased Numbers of TRAP+ Osteoclasts in GATA-1low Mice
To ascertain that the osteosclerosis seen in 12-month-old GATA-1low mice was not due to reduced osteoclast number, we compared the number of TRAP-staining osteoclasts in GATA-1low vs WT metaphysis. Microscopic examination of TRAP staining (Figure 4A) showed an increase in the number of TRAP+ osteoclasts in GATA-1low mice vs WT bone marrow. Morphometry confirmed this to be a statistically significant increase in the number of osteoclasts/mm2 of bone in GATA-1low vs WT mice (Figure 4B). Like MKs, individual osteoclasts from both WT and GATA-1low bone marrow demonstrated similar immunolocalization of BMPs (data not shown).
Fetal Liver MKs Show Expression of BMP mRNA
To detect the presence of mRNA for BMPs in fetal liver MKs of WT and GATA-1low mice, we performed realtime quantitative PCR using primers specifically designed for BMP-2, −4, and −6. Results showed the presence of mRNA for BMP-2, −4, and −6 in both WT and GATA-1low MKs (Figures 5A and 5B). Expression of BMP-2 mRNA in the MKs of GATA-1low and WT mice was greater when compared with calvarial osteoblast samples. Expression of BMP-2 mRNA was greater in WT than in GATA-1low MKs (Figure 5A). Expression of BMP-4 mRNA was greater in WT than in GATA-1low MKs and control osteoblast sample (Figure 5A). Expression of BMP-6 mRNA was strikingly higher in GATA-1low MKs as compared with WT MKs and osteoblast RNA samples (Figure 5B).
Marrow MKs Show Immunocytochemical Localization and Expression of BMPs
Marrow MKs of both WT and GATA-1low mice showed moderate to intense staining for BMP-2, −4, and −6 and for BMP receptors IA and II (BMPR-IA BMPR-II) (Figure 6). BMP-7 stained variably in MKs of both WT and GATA-1low bone marrow. Interestingly, splenic MKs showed no BMP staining in either WT or GATA-1low mice (data not shown). Overall, immuno-histochemistry showed that the BMP expression per unit area of the bone marrow appeared to be greater in the bone marrow of GATA-1low mice than in WT mice, owing to an increased number of MKs/mm2 of bone marrow.
Thus, the increased number of BMP-staining MKs, osteoblasts, and osteoclasts in the marrow of GATA-1low mice probably contributed to the impression of more BMP staining overall in GATA-1low mice bone marrow.
Discussion
The presence of increased numbers of BMP-expressing MKs in the bone marrow of GATA-1low mice suggests that increased BMP secretion by MKs in 12-month-old GATA-1low mice may cause an abnormal stimulation of marrow osteoprogenitor cells, with a resulting overgrowth of cancellous bone (osteosclerosis) (Figure 3B). In this study, expression of mRNA for BMP-2, −4, and −6 was detected in the MKs of WT and GATA-1low mice, and the presence of BMP-2, −4, and −6 proteins and their receptors was confirmed by immunocytochemistry. The relatively increased expression of BMP-6 mRNA and protein observed in the MKs of GATA-1low mice might have had a proportionately greater role in stimulating osteoblastic precursors to undergo osteogenic differentiation. Previous studies have revealed that among the osteogenic BMPs (BMP-2, −4, −6, and −7), BMP-6 is the most effective inducer of osteogenic differentiation of hematopoietic mesenchymal stem cells (Friedman et al. 2006). BMP-6 influences proliferation and osteoblastic lineage differentiation of hematopoietic stem cells via increased release of soluble gp130 (Ahmed et al. 2001). A recent study demonstrated that gp130 knock-in mice displayed an osteosclerotic phenotype (Itoh et al. 2006). The mechanism of BMP-6-induced osteosclerosis in the GATA-1low mice via gp130 or other stimulation is not completely understood as yet and needs to be studied in detail. A proposed schematic illustration of BMP-mediated osteosclerosis in GATA-1low mice is shown in Figure 7. This mechanism could help to explain the osteosclerosis typically present in patients with megakaryocytic leukemia and myelofibrosis with myeloid metaplasia in which the number of immature MKs and/or megakaryoblasts is greatly increased in the marrow (Reilly 1992; Tefferi 2006).
It has been previously reported that wild-type MKs can directly stimulate osteoblast expression of procollagen (Bord et al. 2004). Earlier studies have shown that platelet-released supernatants increased migration and proliferation of bone marrow-derived mesenchymal progenitor cells (Grubear et al. 2004; Kark et al. 2006) but suppressed osteogenic differentiation induced by BMPs under in vitro conditions (Gruber et al. 2006).
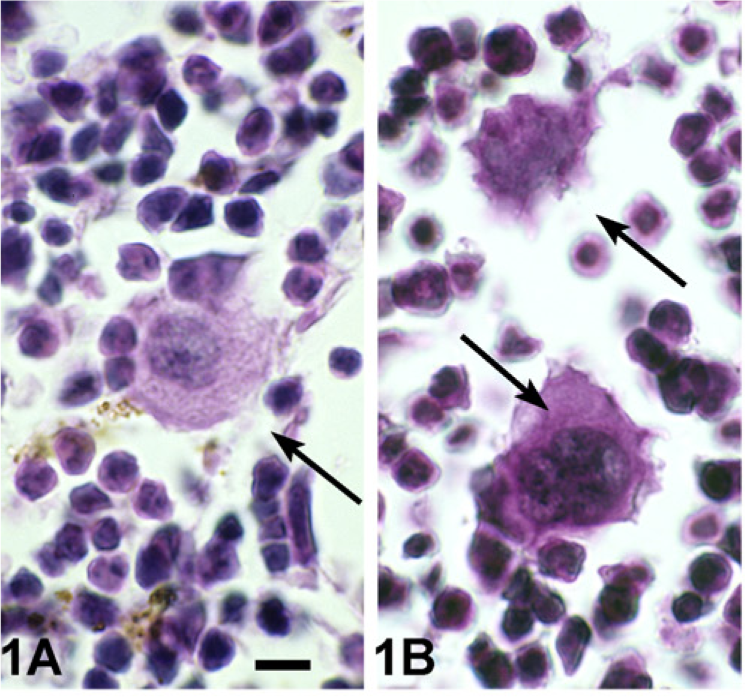
Hematoxylin and eosin (H-E)-stained sections of wild-type (
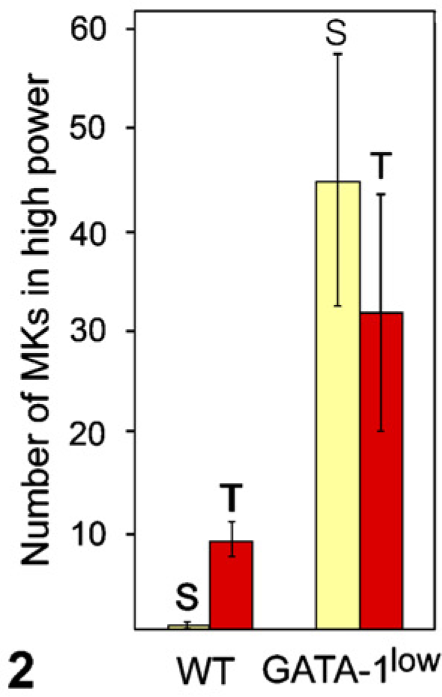
Comparison of the cytomorphometric count of the number of MKs in high power in WT vs GATA-1low spleen (S, yellow bar) and bone marrow in the tibia (T, red bar) at 12 months of age. MKs are significantly more numerous in GATA-1low than in WT spleen and bone marrow (p<0.05).
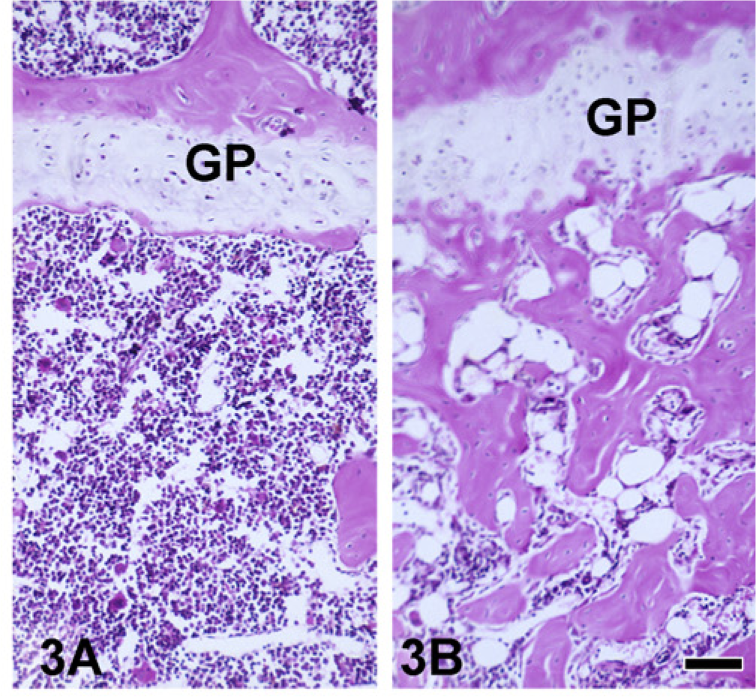
H-E-stained sections of wild-type (
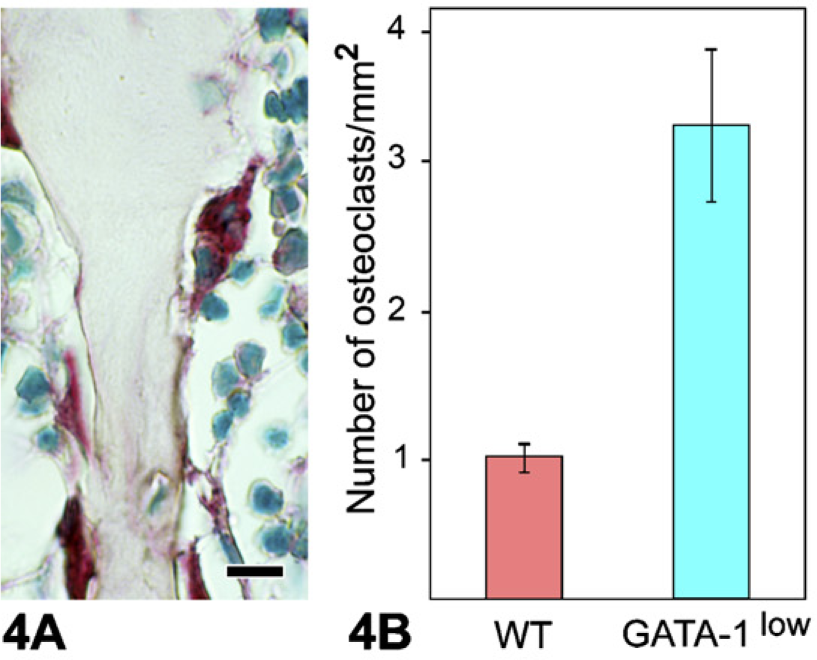
(
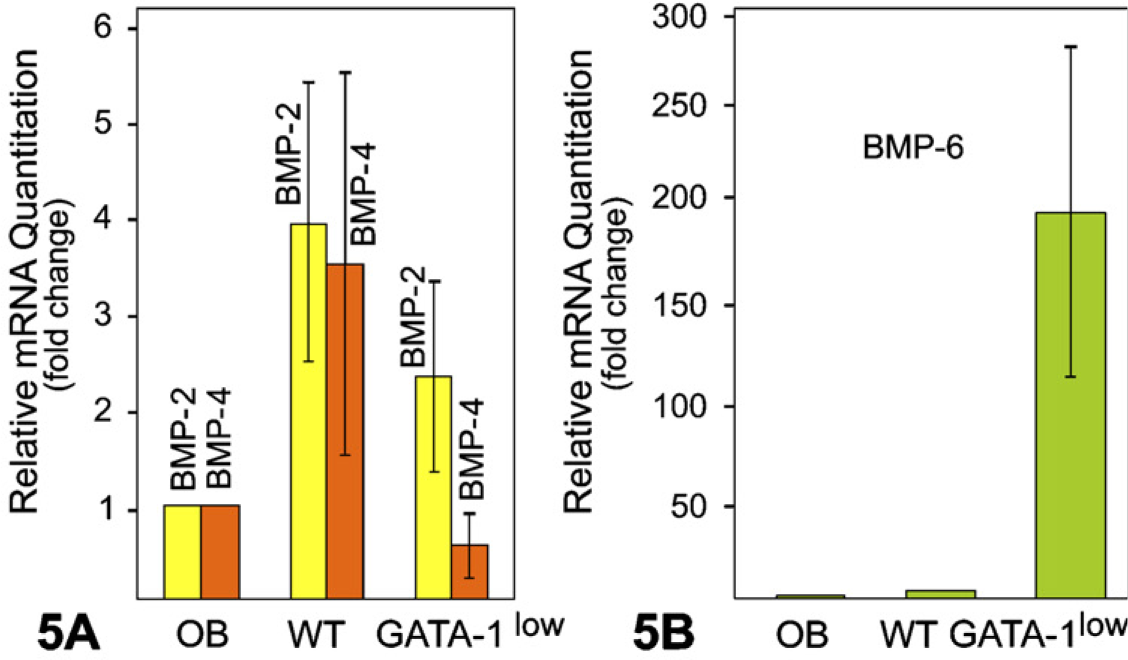
Real-time quantitative PCR analyses show the expression of bone morphogenetic proteins (BMP)-2, −4 (A), and −6 mRNA (B) in MKs of GATA-1 WT and GATA-1low mice. Results are reported as fold change or relative quantitation of target mRNA expression (2−ΔΔCt method) normalized to an endogenous control (GAPDH) and relative to a calibrator (osteoblast RNA sample). Error bars represent the standard error of the mean associated with the ΔΔCt value.
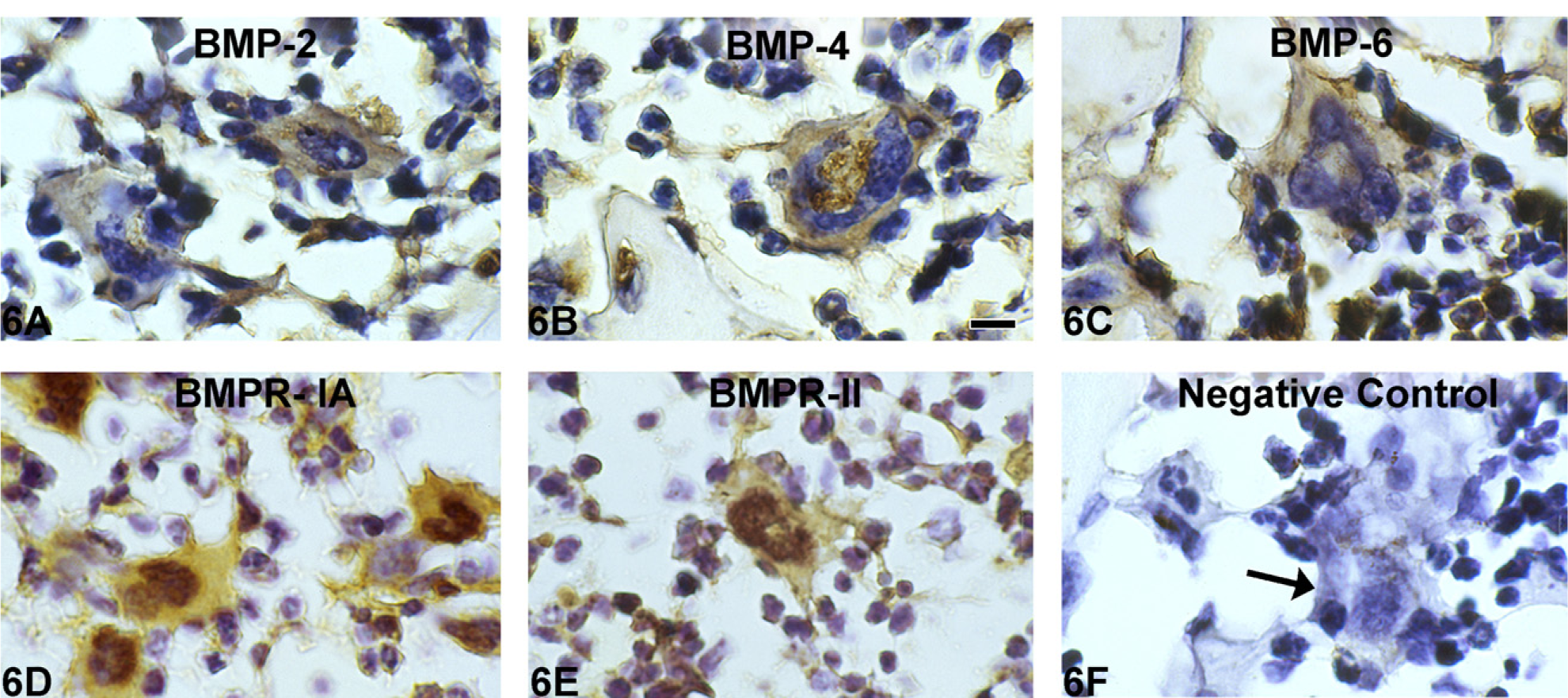
Immunohistochemistry for BMPs and BMP receptors shows positive staining for BMP-2, −4, and −6 and BMP receptors BMPR-IA and BMPR-II in GATA-1low bone marrow MKs (
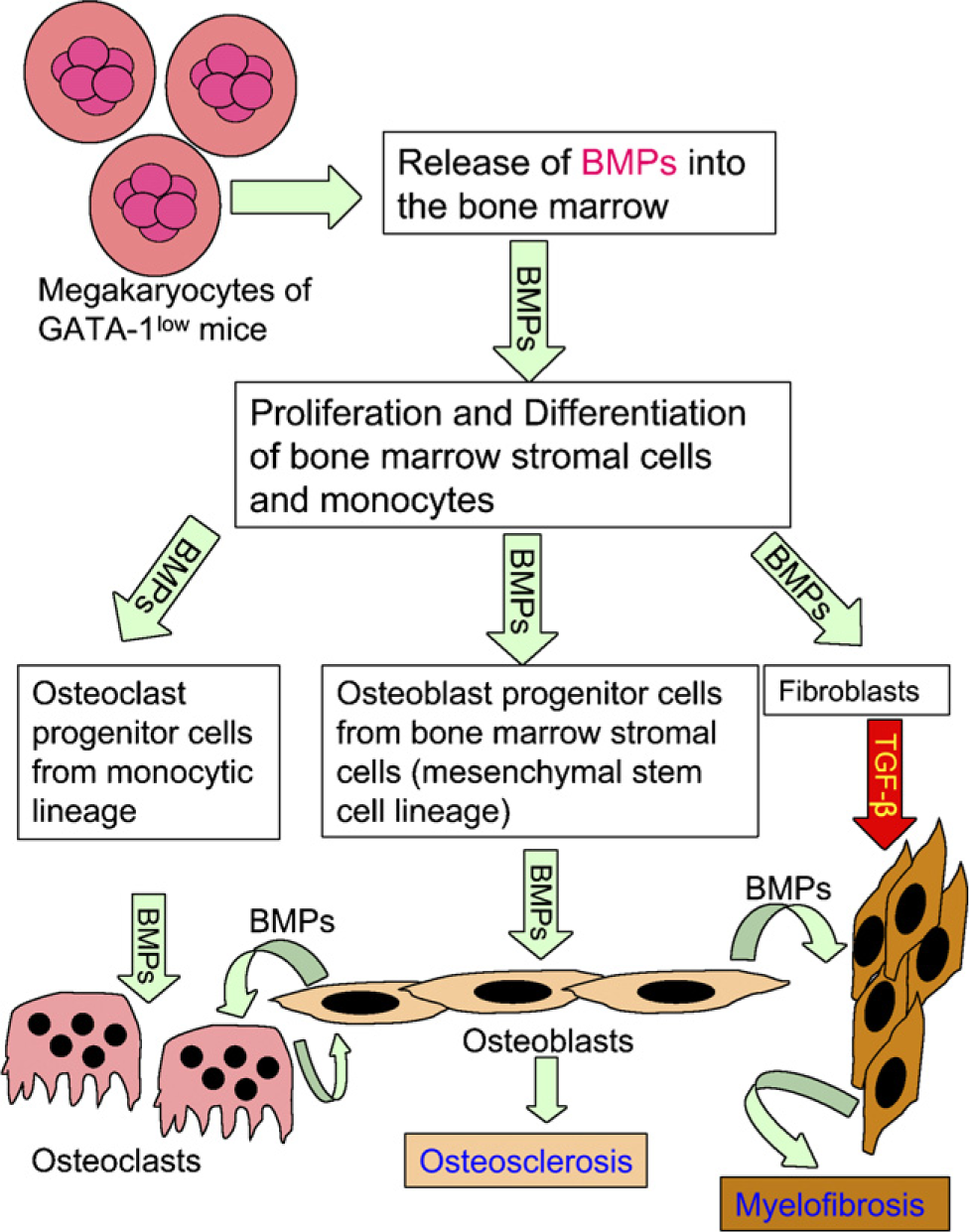
Schematic illustration showing a possible mechanism for BMP-mediated osteosclerosis in GATA-1low mice. MKs of GATA-1low mice release BMPs into the bone marrow. In the bone marrow, BMPs may probably stimulate the proliferation and differentiation of stromal cells toward osteoblastogenesis and fibroblastogenesis originating from mesenchymal stem cells and/or osteoclastogenesis from osteoclast precursors of monocytic origin. Thus, proliferation and differentiation of osteoblasts under the influence of BMPs could result in osteosclerosis.
At present, the mechanism of BMP secretion by MKs to stimulate osteoblast proliferation and differentiation has not yet been clearly defined. Secretion could be related to the release of BMPs into bone marrow from BMP-carrying MK granules similar to the neutrophil-mediated mechanism suggested for the release of TGF-β from MKs (Vannucchi et al. 2005). Alternatively, BMP release could be via MK generation of blood platelets because the latter have also been shown to be carriers of BMPs-2, −4, and −6 (Sipe et al. 2004). Another not unlikely possibility is that the BMPs contained within the MK cytoplasm are secreted through the plasma membrane in a relatively soluble form as probably occurs with BMP secretion by osteoblasts.
Alternatively, the osteosclerosis seen in 12-month-old GATA-1low mice could result from osteoclastic bone resorption being suppressed in these mice, leading to unopposed, osteoblastic bone formation. This hypothesis is supported by previous reports demonstrating that coculture of MKs with osteoblasts leads to increased expression of OPG, an osteoclast differentiation inhibitor, along with decreased RANK-ligand (RANK-L) expression (Bord et al. 2004, 2005). RANK-L is an osteoclast stimulator (Suda et al. 1992). The combined effect of these changes could result in reduced osteoclast number and activity and reduced bone resorption. Beeton et al. (2006) and Kacena et al. (2006b) recently reported that MKs inhibited the number and function of osteoclasts in vitro. However, our present data show that, in vivo, in GATA-1low mice there is a significant increase in TRAP-positive osteoclast number, accompanied by an increase in resorptive surface (Kacena et al. 2004). Thus, these latter data suggest that the stimulation of osteogenesis in vivo in GATA-1low bone marrow is more likely due to the direct stimulation of osteoblastic activity rather than being caused by decreased osteoclastic activity.
In this connection, it is interesting to note that normal osteoclasts have been shown to express significant levels of BMPs (Nishimura et al. 1997; Onishi et al. 1998; Dhanyamraju et al. 2003; Garimella et al. 2006). In the present study we also observed BMP immuno-localization in osteoclasts of GATA-1low and WT mice (data not shown). Thus, the increase in osteoclast number seen in GATA-1low mice (Figure 4) may also contribute to the induction of osteosclerosis (Figure 3) in these mice via BMPs from osteoclasts. BMPs expressed and secreted by normal osteoclasts may constitute an important component of the intercellular “cross-talk” that leads to osteoblast recruitment and differentiation by osteoclasts at bone remodeling sites. This theory is supported by the work reported by Aubin (1999), demonstrating that marrow monocytes (osteoclast precursors) are capable of stimulating osteoblast differentiation.
To summarize, our study demonstrates that (1) immunostainable levels of BMP-2, −4, and −6 are similar in marrow MKs of 12-month-old GATA-1low and WT mice; (2) overall, BMP levels in the bone marrow of GATA-1low mice exceeded those of WT mice, owing to an increased number of MKs and osteoclasts staining positively for BMP-2, −4, and −6/mm2 of bone marrow; (3) osteosclerosis seen in 12-month-old GATA-1low mice apparently was not due to reduced osteoclast-mediated resorptive activity as the number of TRAP+ osteoclasts was greater in GATA-1low mice than in controls.
In conclusion, release of BMPs by increased numbers of MKs may, in part, be responsible for the stimulation of osteoblastic activity and resulting osteosclerosis in GATA-1low mice. The cross-talk among the BMP-expressing MKs, osteoblasts, and other cells of the bone marrow might also contribute to the underlying mechanism of osteosclerosis as seen in MK-related, clinical myeloproliferative disorders.
Footnotes
Acknowledgements
This research was supported by National Institutes of Health Grants DE-05262 (to HCA), AR-052088 (to JW), and AR-049190 (to MCH) and a pilot and feasibility award from the Yale Core Center for Musculoskeletal Disorders/NIH AR-46032 (to MAK).
We thank Ms. Eileen Roach for help in the preparation of figures for the manuscript.
