Abstract
We assessed the distribution and relative immunohistochemical staining intensity of the bone morphogenetic protein-7, osteogenic protein-1 (OP-1), in its pro- and mature forms, and four of its receptors, type I (ALK-2, ALK-3, and ALK-6) and type II in normal adolescent New Zealand White rabbit articular cartilage. Expression of the protein and its receptors was also examined in cartilage from joints that had been previously subjected to cartilage matrix degradation. Pro-OP-1 was moderately expressed in chondrocytes of the superficial, middle, and deep cartilage zones and in the osteocytes. The expression of mature OP-1 was similar, with the exception of less staining in the superficial zone of cartilage. Expression of these two forms of OP-1 was enhanced in the middle and deep cartilage zones after catabolic challenge. The type I receptor, ALK-6, displayed the strongest staining of the receptors in both cartilage and bone, whereas ALK-2 displayed the weakest staining. No differences were observed in the receptor staining levels after catabolic challenge. This study shows that OP-1 and its receptors have been identified in rabbit articular cartilage and bone, suggesting a possible role for this pathway in cartilage and bone homeostasis.
B
Although the potential role of exogenous OP-1 in the homeostasis and repair of human articular cartilage had been previously shown by our department (Flechtenmacher et al. 1996; Huch et al. 1997; Koepp et al. 1999; Nishida et al. 2000), it was only recently that both message and protein for endogenous OP-1 were shown to be expressed in human adult articular chondrocytes (Chubinskaya et al. 2000).
Articular hyaline cartilage provides a smooth gliding surface for the opposing articular surfaces of synovial joints. It consists of four zones, superficial, middle, deep, and calcified, each of these zones being distinguished by the shape of their cells, arrangement of collagen fibers and, in the case of the calcified zone, the presence of a calcified matrix. Although articular cartilage is a highly metabolic tissue, it has little or no ability to repair itself, with catabolic processes exceeding anabolic processes such as those in which TGF-β is involved (Morales and Hascall 1989). The presence or absence of OP-1 in articular chondrocytes and its differential expression within the individual zones may provide important information concerning the relative metabolic/reparative status of the regions at any given time during normal or pathological processes.
OP-1 is present in human adult articular cartilage in two forms, inactive (pro-) and active (mature) (Chubinskaya et al. 2000). It was previously shown in our laboratory that, in normal cartilage, protein for pro-OP-1 was found primarily in the deep layer, whereas the mature form was detected in the superficial layer (Chubinskaya et al. 2000). Furthermore, the expression of OP-1 mRNA was upregulated in knee cartilage from patients undergoing knee replacement due to diagnosed osteoarthritis (OA) compared to normal tissue from organ donors.
Like other BMPs, OP-1 binds and initiates a cellular signaling cascade via a complex of trans-membrane serine/threonine kinase receptors, termed type II and type I receptors (Massagué 1998; ten Dijke et al. 2000). Type II receptors are constitutively active kinases, which transphosphorylate type I receptors. Type I receptors then activate intracellular substrates such as Smad proteins, and thus determine the specificity of the intracellular signals (Miyazono et al. 2000). OP-1 transduces the signal through a number of type II receptors [the BMP receptor type II and activin receptor type II (ActR-II)], and type I receptors [activin-receptor-like kinsases (ALK) 2, 3, and 6]. Alk-3 is a BMP receptor type IA, and ALK-6 is a BMP receptor type IB. ALK-2 has been shown to bind activin, but the idea was expressed that it is mainly a type I receptor for certain BMPs, e.g., BMP-6 and BMP-7 (ten Dijke et al. 1994; Miyazono et al. 2000).
The purpose of our study was to investigate whether endogenous OP-1 and its receptors could be detected in normal intact rabbit articular cartilage and, if so, if their protein expression is modified in a model of enzymatically induced cartilage matrix damage. In the present study we compare the localization and distribution of both forms of OP-1 (pro- and mature) and its receptors, type II and type I (ALK-2, ALK-3, and ALK-6).
Materials and Methods
Animal Treatments
Fourteen adolescent male New Zealand White rabbits (3.5 kg) were assigned to one of two groups: untreated controls (n=7) and chymopapain injury (n=7).
Chymopapain (Chymodiactin; a gift of Boots Pharmaceuticals, Lincolnshire, IL) injury in the left knee joint was carried out by intra-articular injection of a sterile solution of 2.0 mg of the enzyme activated by sodium
All rabbits underwent a 5-day acclimation period before any treatment. The treatment protocol was approved by the Institutional Animal Care and Use Committee of Rush-Presbyterian-St. Luke's Medical Center, (Chicago, IL). Rabbits were sacrificed 28 days after the intra-articular chymopapain injection or after a comparable time for the control rabbits.
Collection of Serum Samples
Blood was obtained by venipuncture on each of the 2 days before, on the day of (but before chymopapain injection), 24 and 48 hr after chymopapain injection, and once per week thereafter. Blood samples were allowed to clot at 4C and centrifuged, after which the serum was obtained and analyzed for content of antigenic keratan sulfate (agKS) to ensure that degradation of articular cartilage PG occurred in all rabbits injected with chymopapain.
Measurement of Serum Levels of agKS
To ensure cartilage damage after chymopapain injection into the knee joint, agKS was measured by a modified ELISA as previously described (Williams et al. 1988a, b, 1993; Manicourt et al. 1991). All determinations were done in duplicate. Values are reported as equivalents of the International Standard of agKS purified from human costal cartilage (a gift from Drs. M.B. Mathews and A.L. Horwitz; University of Chicago, IL).
Tissue Collection and Grading
At sacrifice, femoral and tibial articular surfaces were utilized in another study. Patellar articular surfaces from all 14 animals were examined and graded blindly on a 5-point gross visual scale (Muehleman et al. 1997). Briefly, grade 0 = no signs of cartilage damage, grade 1 = early fibrillation (shallow pits or grooves affecting the cartilage surface in the absence of changes in articular surface geometry), grade 2 = deep fibrillation and fissuring or pitting and possibly small osteophytes, grade 3 = extensive fibrillation and fissuring and 30% or less of the articular cartilage surface eroded down to the subchondral bone and the presence of osteophytes, and grade 4 = greater than 30% of the articular surface eroded down to the subchondral bone and prominent osteophytes.
Histological Preparation
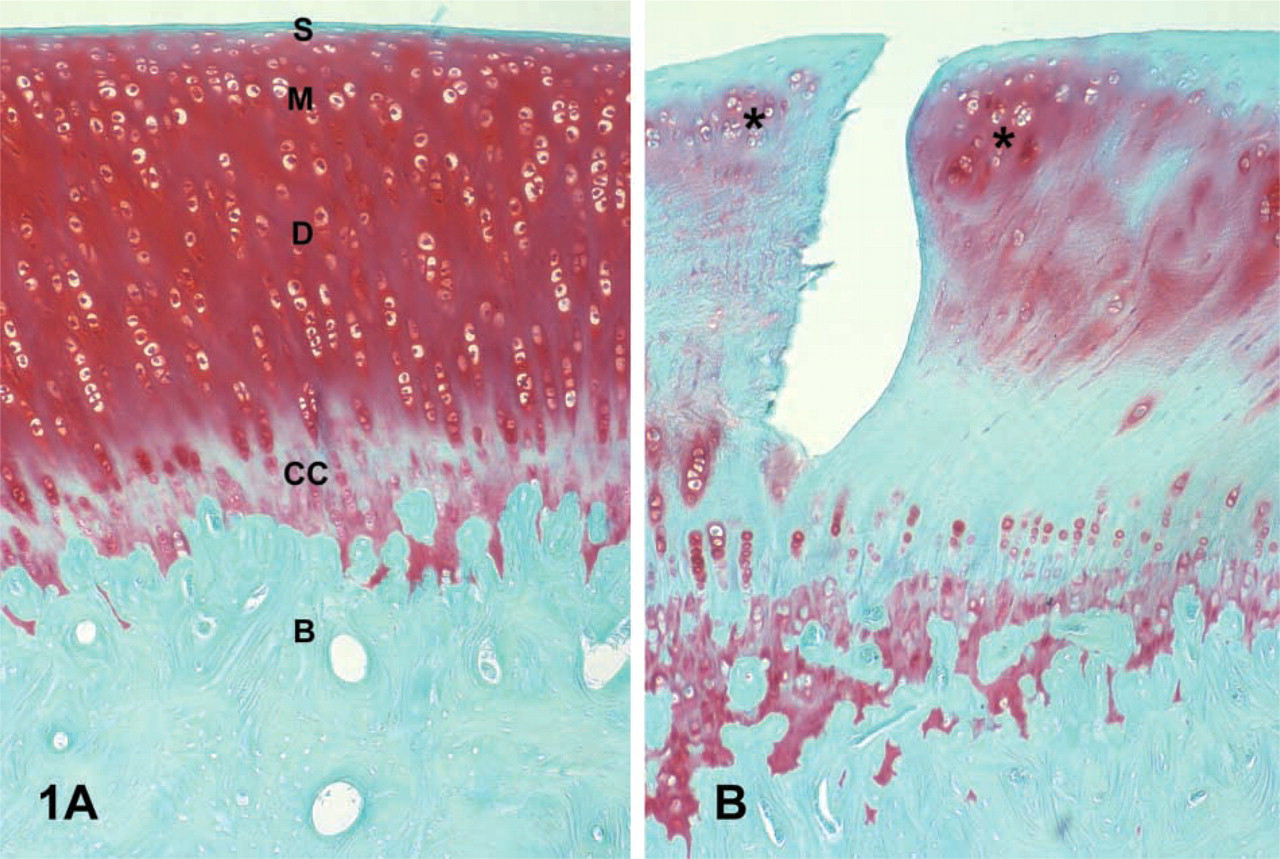
Whole patellae from both knee joints of all 14 animals were fixed in 10% neutral buffered formalin, decalcified in aqueous formic acid:sodium citrate (50:50), paraffin-embedded, and sectioned to 5-μm thickness. Sets of six alternate sections from each knee joint of all animals were either immunostained as described below or stained with Safranin O and fast green (Rosenberg 1971). Cartilage damage on Safranin O/fast green sections was graded blindly on a 20-point scale for articular surface integrity, Safranin O staining, clone formation, and cellularity as previously described (Uebelhart et al. 1993).
Immunohistochemistry
Before incubation with primary antibodies, tissue sections were digested with keratanase [Pseudomonas sp; EC 3.2.1.103 (0.01 U/ml)], keratanase II [Bacillus sp. KS 36 (0.0001 U/ml)], and chondroitinase ABC [Proteus vulgaris; EC 4.2.2.2 (0.01 U/ml)] in 100 mM Tris/50 mM Na-acetate buffer (pH 6.5) at 37C for 90 min to increase the penetration of antibodies into the cartilage. All three types proteinases were obtained from Seikagaku (Tokyo, Japan). Six types of primary antibodies (provided by Stryker Biotech and Ludwig Cancer Institute) were used: anti-pro-OP-1 polyclonal antibody, anti-mature OP-1 monoclonal antibody, anti-type II BMP receptor polyclonal antibody, and three polyclonal antibodies to type I BMP receptor (ALK-2, ALK-3, and ALK-6). Primary antibodies were applied at a 1:100 dilution. For negative controls, the primary antibodies were replaced with either normal serum or secondary antibody alone. For anti-OP-1 antibodies as an additional control, primary antibodies were preabsorbed with human recombinant pro- and mature OP-1. The ImmunoPure ABC Alkaline Phosphatase Staining Kit (Pierce; Rockford, IL) was utilized with biotinylated rabbit anti-goat IgG or horse anti-mouse IgG as secondary antibodies. The specificity of all antibodies was tested previously (ten Dijke et al. 1994; Yonemori et al. 1997; Onishi et al. 1998; Chubinskaya et al. 2000). The colored reaction product was developed with alkaline phosphatase-based one-step NBT/BCIP substrate (ImmunoPure ABC kit; Pierce). To inhibit endogenous alkaline phosphatase activity, ImmunoPure Phosphatase Suppressor (levamisole) was used.
Grading of the Immunostained Sections
Each of the immunostained sections was graded blinded and in agreement by two investigators. Scores for intensity and percentage of cells stained are as follows: ± represents little or no staining in the majority of cells; ++ represents moderate to intense staining in approximately 40–75% of cells; +++ represents intense staining in which 75–100% of cells were well filled with stain.
Results
agKS Levels
Pre-injection serum levels of agKS were 107.43 ng/ml ± 15.0 for the untreated control rabbits and 90.43 ng/ml ± 15.43 for the chymopapain-injected rabbits. For each rabbit, the change in the serum level of agKS at different time points was calculated by deducting the pre-injection baseline level from each post-injection level. The pre- and post-injection serum levels for the rabbits not receiving chymopapain injections were calculated from the same time points as for the chymopapain-injected rabbits. Serum agKS levels from all rabbits receiving chymopapain injection increased sharply (mean increase 181.43 ng/ml ± 33.07) compared to baseline values taken before injection, thereby substantiating cartilage matrix degradation (p<0.001) (Thonar et al. 1985; Williams et al. 1988a, b). This increase occurred in every rabbit receiving a chymopapain injection. AgKS values returned to pre-chymopapain injection values within 48 hr (data not shown). Serum agKS levels from time-matched control rabbits that did not receive chymopapain injection did not increase significantly at any time.
Gross Visual Inspection
No gross changes were observed on the patellar articular surfaces of any of the control rabbits or on the patellar surfaces of the control limbs of the chymopapain-injected rabbits. Each of the chymopapain-injected joints displayed slight fibrillation on the central portion of the patellar articular surface.
Histology: Safranin O Staining
Microscopic inspection of Safranin O-stained sections revealed that the articular cartilage of all control patellae (those receiving no chymopapain injection, from both rabbit groups) had a smooth surface, normal cellularity, and intense staining for PGs (Figure 1A). The patellar cartilage of all chymopapain-injected knees displayed various degrees of surface fibrillation, cell loss, cell cloning, and pronounced loss of Safranin O staining (Figure 1B).
Expression of OP-1
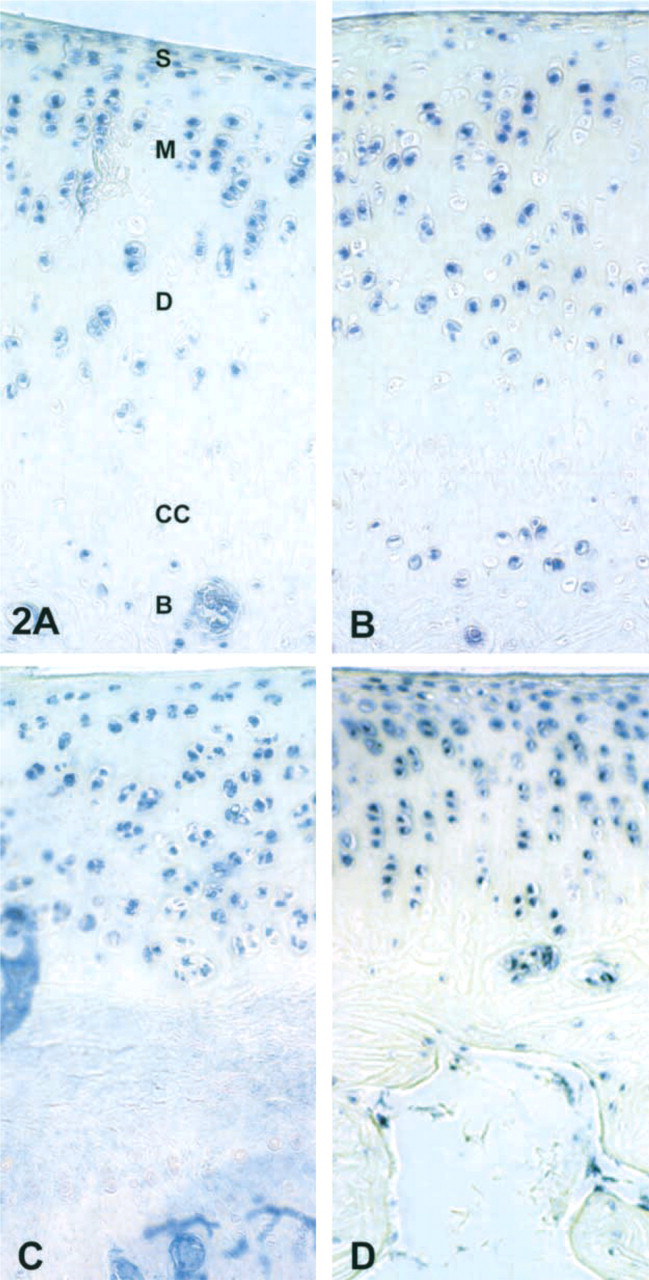
A summary of immunohistochemical staining for OP-1 and its receptors is shown in Table 1. Both forms of OP-1 (pro- and mature) were identified in chondrocytes and osteocytes of rabbits from both control and CP-injected joints. However, there were detectable differences in the intensity of staining and the cartilage layer distribution of pro- and mature OP-1. Pro-OP-1 had a strong presence in the superficial, middle, and deep zones of cartilage from both groups, but with slightly higher intensity of staining in the chymopapain-injected compared to the control uninjected knees (Figures 2A and 2B). Osteocytes were also positive for pro-OP-1 throughout the patellae in both groups of rabbits. There was little or no detectable pro-OP-1 in the chrondrocytes of the calcified cartilage, in the cartilage matrix, or in the bone matrix.
Staining for mature OP-1 was detected in cells of both uninjected and CP-injected tissues. In control joints, positive staining for mature OP-1 was found at low levels in the superficial zone of cartilage and at moderate levels in the middle zone (Figure 2C). In CP-injected joints, higher levels of mature OP-1 were primarily localized in cells of the middle and deep zones of articular cartilage (Figure 2D), whereas in the superficial zone mature OP-1 was barely detectable. In contrast to mature OP-1, the superficial zone of cartilage in CP-injected joints was strongly positive for pro-OP-1. Overall staining for pro-OP-1 was more intense in all cartilage zones of the patellae of the CP-injected knees compared to the uninjected knees (Figure 2D). In both uninjected and CP-injected knees, mature OP-1 was also detected in the osteocytes throughout the bone, but little or no staining was found in the cartilage matrix, calcified cartilage, or bone matrix.
(
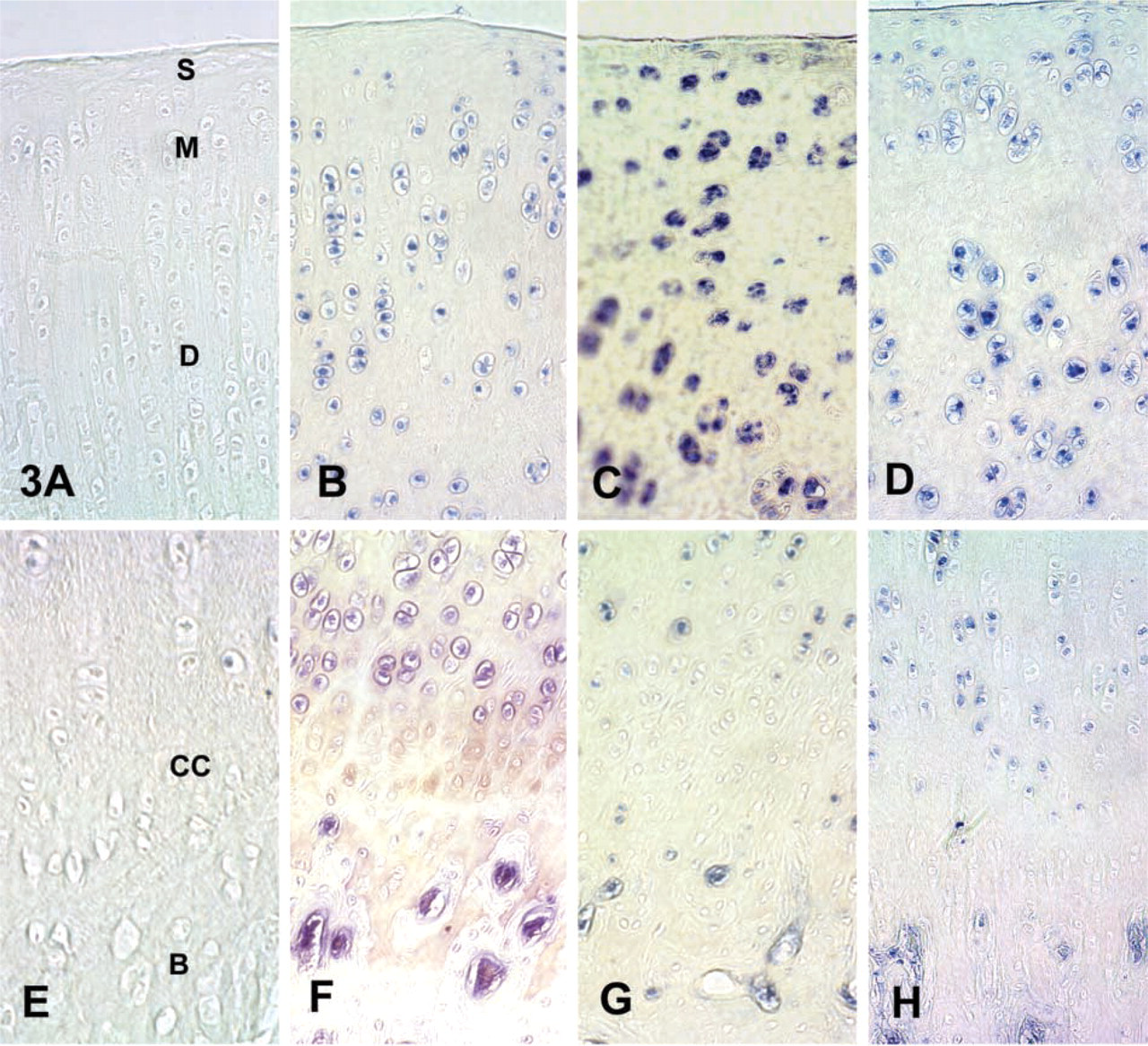
Concerning OP-1 receptors, the presence and distribution of each specific BMP receptor were similar in all joints of both rabbit groups. Type II BMP receptor and type I BMP receptor, ALK-3, were weakly present in chondrocytes of the superficial zone, but at moderate to strong levels in chondrocytes of the middle and deep cartilage zones and in osteocytes throughout the bone (Figures 3A and 3B). Staining for these receptors was either not detected or only slightly detected in the calcified cartilage zone. Staining was never detected in the cartilage matrix or bone matrix.
Expression of pro- and mature forms of OP-1 and three of its receptors in the articular cartilage of rabbit patellae a

not present; +, minimal expression; ++, moderate expression; +++, maximal expression.
Immunostaining of cartilage with (
Anti-ALK-2 stain was either absent or appeared in the middle and deep articular cartilage zones with much less intensity than anti-ALK-3 or anti-type II receptor staining (Figure 3C). As with the other receptors, ALK-2 was either not detectable or just barely detectable in chondrocytes of the calcified cartilage. No matrix staining for ALK-2 was detected.
Type I BMP receptor, ALK-6, was moderately expressed in the superficial zone chondrocytes and maximally expressed in the chondrocytes of the middle and deep cartilage zones as well as in the osteocytes of the bone. ALK-6 was either not detectable or only weakly detectable in the chondrocytes of the calcified cartilage zone and in the cartilage and bone matrix.
Immunostaining of the uncalcified zones of cartilage with (
Discussion
The present study was a continuation of ongoing investigations on the understanding of the biology of OP-1 and its receptors in articular cartilage under physiological and pathophysiological conditions. In addition to the study of untreated New Zealand White rabbit knee joint cartilage, the chymopapain model of cartilage matrix damage was used in this species because it was previously shown to mimic the cartilage matrix pathology seen in early stages of OA (Williams et al. 1988a, b) thus enabling us to observe changes in endogenous OP-1 and its receptors after catabolic injury. We found that both pro- and mature OP-1 and four of its receptors, type I (ALK-2, ALK-3, and ALK-6) and type II, are expressed in rabbit articular cartilage chondrocytes and bone osteocytes of New Zealand White rabbits.
The differential zonal distribution of the staining for OP-1 was similar to that reported in the weanling rat by Anderson et al. (2000). They found that immunostaining for OP-1, as well as for other BMPs, was most intense in mid-zone (maturing) and mid- to deep-zone (“hypertrophic”) chondrocytes of articular cartilage where most apoptosis is occurring during the process of endochondral bone development. In the present study in the adolescent rabbit, we also found this to be the case for both forms of OP- 1 and three of its receptors and we found little or no staining in the calcified cartilage and no staining in the cartilage matrix or bone matrix. The distribution of OP-1 and its receptors in rabbit cartilage seems to be in contrast to that found for human adult cartilage in our previous studies (Chubinskaya et al. 2000, 2001), in which mature OP-1 and all receptors were primarily localized in the superficial layer of normal articular cartilage and pro-OP-1 was mainly found in the middle and deep zones. Cellular localization of OP-1 in rabbit cartilage may indicate that in this tissue the majority of OP-1 is stored intracellularly or within the vicinity of the cell membrane and is not released into the matrix.
We have also found that experimentally induced matrix destruction through chymopapain injection into the knee joint increases the detectable levels of pro- and mature OP-1, possibly indicating a reparative response by the chondrocytes, i.e., the elevation in OP-1 mRNA expression and synthesis. Our recent findings that demonstrated an upregulation of OP-1 mRNA expression in human chondrocytes as a response to treatment with IL-1 support this hypothesis (Merrihew et al. 2002). The increase in mature OP-1 under these experimental conditions could also be due, in part, to the activation of different intracellular and extracellular proteases that may be responsible for the transformation of pro-OP-1 into the mature form. Among these factors, attention should be given to at least two classes of enzymes: furin-type convertases and serine proteases (Piek et al. 1999). Furin convertases involved in the proteolytic cleavage of the precursor molecules of TGF-β and BMP-4, while plasmin and cathepsins control the activation of latent TGF-β into biologically active mature protein. Importantly, elevated levels of furin were reported in OA cartilage compared to normal cartilage (Moldovan et al. 2000).
We cannot rule out the possibility that chymopapain induces an activation or degradation of OP-1 as well as other proteinases involved in cartilage degradation. However, chymopapain, as a cystein proteinase, would probably not be involved in the known proteolytic cleavage of the precursor molecules of the BMPs as by serine proteinases or furin-type convertases. Concerning degradation, it is known from the unpublished results of Stryker Biotech and our own laboratory that OP-1 is very resistant to degradation. The only known protease that leads to autodegradation of OP-1 by removing a segment of 20 amino acids from the N-terminus is trypsin. However, the unavailability of recombinant pro-OP-1 does not allow a direct in vitro test.
Concerning OP-1 receptors, we found ALK-3, ALK-6 (type I receptors), and type II receptor at moderate to maximal levels in chondrocytes of all cartilage zones except the calcified zone. This was the case for both uninjected control and chymopapain-injected joints. It appears from these findings that OP-1 and its receptors are found to a far lesser extent in cartilage zones that exhibit a lower metabolic rate and are not concerned with the overall maintenance of cartilage integrity.
Type I receptor, ALK 2, was either not detectable or just barely detectable in chondrocytes of each of the zones of cartilage and in the osteocytes. As it was shown previously (ten Dijke et al. 1994, 2000), OP-1 exhibits a higher affinity to BMP receptor IA (ALK-3) and BMP receptor IB (ALK-6) and a much lower affinity to ALK-2, which is an activin type I receptor. The lack of ALK-2 in rabbit cartilage and the presence of OP-1 and its other signaling molecules might indicate that the representatives of this BMP signaling pathway are the primary factors (or specific factors) responsible for the anabolic processes in this tissue. Further work on activin and its pathway will provide more details on which factors are the key players in cartilage repair in rabbit models. However, this is beyond the scope of the current project.
A study on the epiphyseal plate of growing rats (Sakou 1998) found that TGF-β1 and BMP receptors IA and II were particularly expressed in chondrocytes undergoing proliferation and maturation. However, hypertrophied chondrocytes displayed decreased expression. The authors suggested that this decrease may simply be the result of the receptors spreading over a larger region of the hypertrophied chondrocyte membrane. A related study (Yazaki et al. 1998) showed that OP-1 immunostaining was more intense in the 12-week-old rat than in the 2-week-old rat but was hardly detectable at 24 weeks, suggesting a role for OP-1 in the maturation of epiphyseal growth plate. Furthermore, BMP receptors IA, IB, and II were more intense in 12-week than in 24-week rats, and there was decreased expression in the hypertrophic cartilage.
The calcified cartilage is an important transitional zone between the uncalcified cartilage and the underlying subchondral bone for force transmission (Mente and Lewis 1994), attachment of the two tissue types, and for limiting diffusion from bone to cartilage (Oegema et al. 1997). Because PGs remain in the matrix after mineralization of the calcified cartilage zone and because mineralization does not extend to the cell membrane of the chondrocytes (Oegema and Thompson 1995), these cells are not completely walled off from their matrix. In adult human tissue the calcified cartilage is quiescent but not inactive (Oegema et al. 1997). It is likely that the lack of immunostaining for both forms of OP-1 or its receptors found in the calcified cartilage in the present study is a reflection of the metabolic state of the chondrocytes in this zone. Although the viability of calcified zone chondrocytes remains questionable, they are at least viable before and at the time of calcification (Oegema and Thompson 1995). Furthermore, they are still capable of synthesizing PGs, although to a lower level than in the middle and deep zones (Marles et al. 1991). The question of whether or not chondrocytes of this zone can, and do, respond to damage remains. Therefore, because this zone does not exhibit the same degree of metabolic activity, it is not totally surprising that we could not detect OP-1 or its receptors here.
We also cannot rule out an artifactual cause for this lack of staining due to the decalcification of the tissue, although stain for both forms of OP-1 and two of its receptors could be detected in the osteocytes of decalcified tissue.
Surprisingly, as detected by immunohistochemistry, the receptor levels were not increased in the chymopapain-injected knee joints. Because OP-1 is not the only member of the BMP family that binds to these receptors, it is a possibile that much higher levels of the BMP receptors are present on chondrocyte membranes than are needed for OP-1 signaling alone. The fact that all tissues were subjected to chondroitinase before immunostaining rules out the possibility that the chymopapain of the injected rabbits simply rendered the tissue more susceptible to staining with the OP-1 antibodies.
The idea of a reparative/anabolic response of pro- and mature OP-1 after chymopapain-induced cartilage damage is supported by the finding of Urist (1997) that endogenous bone morphogenetic protein was observed in cartilage repair and subsequent heterotopic bone formation after experimental hole punch in the rabbit ear.
In conclusion, we have demonstrated that endogenous OP-1 and its receptors could be detected in adolescent rabbit articular cartilage. Interestingly, as in human cartilage, both forms of OP-1 (pro- and mature) are found in rabbit cartilage. However, the distribution of these forms is different from those described in humans and is different among experimental groups. In rabbits, whereas pro-OP-1 was present in all cartilage layers, mature OP-1 was detectable in the middle and deep layers but only weakly detectable in the superficial layer, which might indicate a distinct processing of OP-1 by superficial layer chondrocytes in rabbits. The present study shows that OP-1 and its receptors have been identified in rabbit articular cartilage and bone, suggesting a possible role for this pathway in cartilage and bone homeostasis.
Footnotes
Acknowledgments
Supported by NIH grants AR45301–01, 2P50–AR-39239–11, Stryker Biotech grant KK-001, and by the Dutch Organization for Scientific Research (ALW 809.67.024).
