Abstract
The ELR+ chemokine CXCL15, which recruits neutrophils during pulmonary inflammation, is also known as lungkine due to its reported exclusive expression in the lung. We now report that CXCL15 mRNA and protein are also expressed in other mucosal and endocrine organs including the gastrointestinal and urogenital tracts and the adrenal gland. Our results indicate that CXCL15 is expressed throughout the gastrointestinal tract, with the exception of the cecum. Gastric CXCL15 protein expression is approximately 10-fold lower than pulmonary expression and primarily occurs in a specific lineage of gastric epithelial cells, the prezymogenic and zymogenic cell. Similar to the increased expression of CXCL15 during pulmonary inflammation, gastric inflammation induced by infection with Helicobacter felis caused an increase in gastric CXCL15 expression. However, colonic CXCL15 expression was not altered in two different models of colonic inflammation, the Helicobacter hepaticus T-cell transfer model and the mdr1a −/− model of colitis. These findings clearly demonstrate that CXCL15, previously reported to be the only lung-specific chemokine, is also highly expressed in other murine mucosal and endocrine organs. The functional role of CXCL15 in mucosal disease remains to be elucidated. This manuscript contains online supplemental material at http://www.jhc.org. Please visit this article online to view these materials.
C
CXCL15 has been reported to be strongly expressed in the adult lung of the inbred mouse strains BALB/c and C57BL/6 but does not appear to be expressed in lymphoid organs such as the spleen (Rossi et al. 1999; Ohneda et al. 2000). CXCL15 is upregulated in response to multiple inflammatory stimuli including an ovalbumin-induced model of asthma and in Nippostrongylus brasiliensis or Aspergillus infection models (Rossi et al. 1999). In these models, CXCL15 is released by bronchoepithelial cells into the airways where it functions to increase the migration of neutrophils into the airway spaces (Rossi et al. 1999). Additional support for a role of CXCL15 in pulmonary defense comes from studies using Klebsiella pneumoniae-infected mice deficient in CXCL15 (Rossi et al. 1999; Chen et al. 2001). Compared with infected wild-type controls, CXCL15−/− mice had an increased pulmonary bacterial load and decreased survival.
In contrast to the role of CXCL15 in murine models of asthma and pulmonary infections, murine intestinal injury after ischemia-reperfusion resulted in no change in CXCL15 serum levels, although expression of other ELR+ chemokines, KC and MIP2, was dramatically increased (Maheshwari et al. 2004). Although CXCL15 serum levels did not change, baseline serum level of CXCL15 was 10 times greater than the serum concentrations of KC and MIP-2. Because it was believed that pulmonary CXCL15 was secreted only into the airspaces, we decided to more fully investigate the possibility that secretion of CXCL15 could occur in other mucosal or endocrine organs.
Materials and Methods
Mice
Five inbred mouse strains were used in this study. All mice were between 6 and 10 weeks old at the time of baseline analysis or initial infection, unless otherwise indicated. C57BL/6J, C57BL/6J-Rag-1tm1Mom (B6.Rag-1 −/−), and BALB/cJ were obtained from The Jackson Laboratory (Bar Harbor, ME), and FVB/N mice and FVB.mdr1a −/− mice were obtained from Taconic (Hudson, NY). Mice were maintained on a 12:12-hr light-dark schedule and fed standard laboratory mouse chow. Animal procedures and protocols were conducted in accordance with the Institutional Animal Care and Use Committee at the University of Alabama at Birmingham.
Antibodies and Reagents
Primary antibodies used in this study include mouse anti-H+/K+-ATPase (clone 2G11; Sigma, St Louis, MO), rabbit anti-intrinsic factor (IF, 1:2000; generous gift of David Alpers, Washington University Medical School, St Louis, MO), rabbit anti-pepsinogen (1:2000; generous gift of Michael Samloff, UCLA, Los Angeles, CA), and biotinylated goat anti-mouse CXCL15 that showed no cross-reactivity with other chemokines and cytokines as tested by R&D Systems (0.5 μg/ml, DuoSet Part 840950; Minneapolis, MN). Fluorescently labeled lectins used in this study were rhodamine cholera toxin B (4 μg/ml, CTB; List Biological, Campbell, CA) and Texas Red Griffonia simplicifolia (1:200, TX R GSII; EY Laboratories, San Mateo, CA). Secondary and detection antibodies used included Cy3 goat anti-mouse Fab fragment (Jackson ImmunoResearch; West Grove, PA), biotin donkey anti-rabbit IgG (5.6 μg/ml; Jackson ImmunoResearch), streptavidin-HRP (1 μg/ml; Jackson ImmunoResearch), and FITC or Cy3-tyramide diluted per manufacturer's instructions (1:100 and 1:1000, respectively; PerkinElmer, Boston, MA). Nuclei were visualized by Hoechst dye (2 μg/ml; Sigma).
Tissue Preparation
Immediately after sacrifice by isoflurane inhalation and cervical dislocation, tissues were removed for total RNA isolation and immunohistochemistry. All tissues removed were divided in half with one half immersed in liquid nitrogen for total RNA isolation, whereas the other half was submerged in Bouin's Fixative Liquid (Fisher Scientific; Pittsburgh, PA). Tissues were immersion fixed for 18-24 hr at 4C, changed to 70% ethanol, placed in cassettes, and embedded in paraffin. Five-μm sections were prepared on a microtome and attached to precleaned microscope slides (Snowcoat X-tra, Surgipath; Richmond, IL).
Total RNA was isolated by the Trizol method (Invitrogen; Carlsbad, CA), which uses phenol and guanidine isothiocyanate for total RNA extraction (Chomczynski and Sacchi 1987). Prior to cDNA synthesis, genomic DNA was removed from the extracted total RNA using the Turbo DNase kit (Ambion; Austin, TX). Using equivalent amounts of mRNA (2 μg), cDNA was made utilizing reverse transcription with the Transcriptor First Strand cDNA Synthesis Kit (Roche; Pensberg, Germany). Quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR) was performed using Applied Biosystems Assays-On-Demand primer/probe sets and TaqMan Universal PCR Mix (PE Applied Biosystems; Foster City, CA) combined with the Stratagene MX3000P real-time PCR machine. The following Applied Biosystems Assays-On-Demand primer/probe sets were used: 18S housekeeping gene (Hs99999901_s1; GeneBank ID #X03205), CXCL15 (Mm00441263_m1; GeneBank ID #NM 011339.1), CXCL1/KC (Mm00433859_m1; GeneBank ID #NM 008176), CXCL5/LIX (Mm00436451_g1; GeneBankID #NM 009141), and CXCL2/MIP2 (Mm00436450_m1; GeneBankID #NM 009140). Gene expression was calculated using the “delta-delta Ct” relative quantitation method per manufacturer's instructions (Applied Biosystems). Briefly, the relative expression (ΔCt) of each target gene is determined by comparing its crossing threshold (Ct) to the Ct for reference or housekeeping gene. For our studies, we chose 18S as the reference gene because our pilot study (unpublished data), combined with other published results, indicates that several commonly utilized reference genes such as GAPDH, HPRT, and β-actin are altered during inflammatory states in the gastrointestinal tract (Bas et al. 2004). The ΔCt from each sample was then compared with an experimentally defined control to determine the ΔΔCt, which is used in the formula 2ΔΔCt to determine the fold change in mRNA expression (Heid et al. 1996; Bas et al. 2004).
To quantify the amount of CXCL15 protein in the various organs, protein was isolated from multiple C57BL/6 tissues. Briefly, tissues were placed in a buffer of 500 mM NaCl/50 mM Hepes, pH 7.4, containing 0.1% Triton X-100, 0.02% NaN3 (Fisher Scientific), and protease inhibitor cocktail for mammalian tissues (p8340; Sigma). Tissues were then homogenized followed by an overnight freeze at −20C. After thawing, tissue extracts were spun at 6000 x g for 20 min at 4C (McLoughlin et al. 2006). The supernatant protein concentration was determined by the Bio-Rad Protein Concentration Assay following the DC Protein Assay Instruction Manual (cat. #500-0116; Bio-Rad Laboratories, Hercules, CA). To determine the amount of CXCL15 expressed in each tissue, routine ELISAs were performed on triplicate samples according to the manufacturer's protocol (R&D Systems). Optical density of the samples was read on a VERSAmax microplate reader (Molecular Devices; Sunnyvale, CA), and data were analyzed with SoftMax Pro 4.7 (Molecular Devices Corporation). Limit of detection for this assay was 200 pg/ml.
Immunofluorescence Staining
Immunofluorescence staining for CXCL15 was performed on paraffin-embedded tissues. Briefly, tissues were deparaffinized with Citrosolv (Fisher Scientific) and isopropanol and rehydrated with PBS. The tissues then went through a 3% hydrogen peroxidase step for 5 min to block endogenous peroxidases and an antigen-retrieval step (0.1 M citric acid, 0.1 M Na citrate boiled for 17 min) to unmask epitopes. Next, tissues were blocked with avidin and biotin (catalog #SP-2001; Vector Laboratories, Burlingame, CA) each for 15 min. The last blocking step was a PBS blocking buffer (which consists of 1% bovine serum albumin and 0.3% Triton) for 15 min to block nonspecifically binding proteins and to provide antibodies access to cell surface and internal antigens. After the above blocking steps, slides were incubated with biotinylated anti-mouse CXCL15 (R&D Systems) primary antibody (overnight at 4C), followed by streptavidin-HRP, with detection by FITC-tyramide.
Mouse anti-H+/K+-ATPase was detected using a “mouse on mouse” protocol with detection by Cy3 goat anti-mouse Fab fragment (Brown et al. 2004). Briefly, slides are blocked with PBS blocking buffer for 1 hr. During this hour the primary and secondary antibodies are incubated together at a 1:2 ratio for 40 min at room temperature and then blocked with excess serum to block the unbound Fab fragments for 10 min before being placed on the slide. Detection of the primary rabbit anti-IF and -pepsinogen was by a biotin donkey anti-rabbit IgG, followed by a streptavidin-HRP step and, finally, a Cy3-tyramide step. For the IF, pepsinogen, CTB, and TX R-GSII costaining experiments with CXCL15, primary antibodies were added together and the CXCL15 staining was taken through to detection (FITC-tyramide), followed by an additional 3% hydrogen peroxidase step before detection of the other primary antibody. For the H+/K+-ATPase and CXCL15 costaining experiment, the CXCL15 staining protocol was taken through to detection, slides were then blocked for the “mouse on mouse” protocol, and the H+/K+-ATPase/Cy3 Fab fragment complex was then added.
To determine the specificity of the CXCL15 immunostaining pattern, a preabsorption control was performed (Burry 2000). Briefly, a 2-fold excess (on a weight basis) of the recombinant mouse CXCL15 (100 μl of 1 μg/ml; R&D Systems) was preincubated with the biotin goat anti-CXCL15 (1 μl of 50 μg/ml) for 1 hr at 37C. This preincubation mixture was then placed on the slide in place of the primary antibody and the remaining steps were carried out as described above. For all staining protocols, a slide was done with no primary antibodies to confirm that the staining seen was real.
Helicobacter felis Infections
C57BL/6J mice were infected with H. felis or mock infected as described previously (Roth et al. 1999). Briefly, mice were infected orally with 5 × 107 CFU H. felis (ATCC 49179) per inoculation in 25 μl of brain-heart infusion broth (BHI)/glycerol three times over a 1-week period. This protocol results in 100% infection efficiency in our laboratory. Mock-infected mice were inoculated with freezing medium, a sterile BHI/glycerol mixture.
Colitis Models
Colitis was induced in Helicobacter hepaticus-infected B6.Rag-1 −/− mice using a previously described model with minor modifications (Kullberg et al. 2002). Adult B6.Rag-1 −/− recipient mice, certified free from Helicobacter by the supplier, were infected orally three times over a 7-day period with 5 × 107 CFU H. hepaticus (ATCC 51488) resuspended in 25 μl freezing medium. H. hepaticus was cultured on Brucella blood agar (Difco; Kansas City, MO) and inoculated into Brucella broth containing 5% fetal calf serum for infections as described previously (Burich et al. 2001). Lymphocytes were isolated from spleens and mesenteric lymph nodes of uninfected C57BL/6J mice by mechanical dissociation followed by lysis of erythrocytes and purification for CD4+ T cells using magnetic microbeads coated with rat anti-mouse CD4 (L3T4; Miltenyi Biotec, Auburn, CA). CD4+ T cells (4 × 105) from uninfected C57BL/6J mice were injected IP into 2-week H. hepaticus-infected or mock-infected B6.Rag-1 −/− mice. Animals were followed for an additional 6 weeks by closely monitoring for weight loss and diarrhea. At sacrifice, the colons of recipient mice were excised, processed, and stained with hematoxylin and eosin; portions of each tissue were also stored in LN2 for subsequent mRNA analysis as described above.
The second model of colitis utilized in this study was the FVB.mdr1a −/− mouse (Panwala et al. 1998). In the FVB. mdr1a −/− mouse model, absence of intestinal P-glycoprotein leads to severe colonic inflammation (Panwala et al. 1998). To assess the involvement of CXCL15 in this model of IBD, FVB.mdr1a −/− and FVB control mice were sacrificed at 5 months of age. Their colons were excised, processed, and stained with hematoxylin and eosin; portions of each tissue were also stored in LN2 for subsequent mRNA analysis as described above.
Graphic and Statistical Analysis
Graphs of the calculated mean fold change from the RT-PCR were created using GraphPad Prism 4 (GraphPad Software; San Diego, CA). All qRT-PCR graphs are organized horizontally and with the y-axis at one to represent the baseline expression of each gene for the comparison, except for Figure 1A, which is normalized to lung. Error bars represent the standard error between fold changes. Statistics were performed for all RT-PCR and ELISA experiments using Sigma Stat v. 2.03, and results were graphed with GraphPad Prism 4. Student's t-test was performed to determine statistical significance defined as p<0.05.
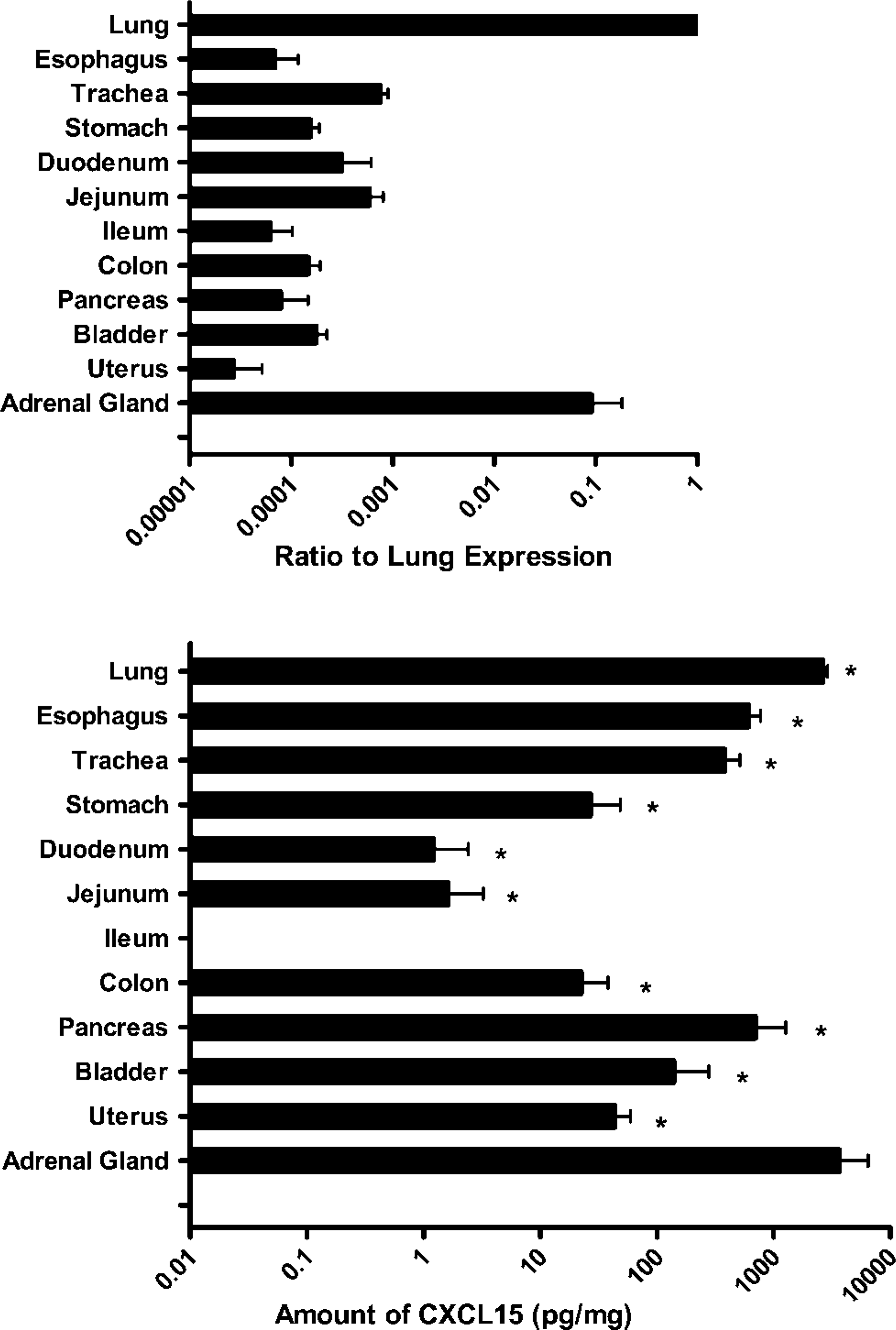
Expression of CXCL15 in multiple murine tissues. Top: mRNA expression of CXCL15 in mucosal and endocrine tissues of 12-week-old C57BL/6 mice. Expression is graphed as a ratio of lung CXCL15 expression. Expression was similar for FVB/N and BALB/c mice (data not shown). As previously reported, the lung has a high level of expression of CXCL15, but here we also demonstrate significant levels of expression in other mucosal and glandular organs including trachea, stomach, jejunum, and adrenal gland. Bottom: CXCL15 protein expression was determined by ELISA. High protein expression is seen in the lung, adrenal gland, and other mucosal tissues (n = 3). ∗ p<0.05 as compared with lung.
Results
CXCL15 Expression in Murine Mucosal and Endocrine Tissues
We examined the level of CXCL15 mRNA expression in mucosal and endocrine organ systems of the C57BL/6J mouse. Figure 1(top) shows the mRNA expression levels of CXCL15, graphed as a ratio against the expression seen in the lung. Although the highest expression of CXCL15 is found in the lung, there is significant expression in adrenal glands and in multiple mucosal organs including the respiratory, gastrointestinal, and urogenital tracts. There was minimal to no expression of CXCL15 detected in the cecum, testes, or spleen (Figure 1, top, and data not shown). Expression of CXCL15 by RT-PCR was tested on the same tissues from BALB/cJ and FVB/N mice, and expression level was similar to those shown for the C57BL/6J mice (data not shown).
To validate qRT-PCR expression, total protein was isolated from C57BL/6J tissues, and CXCL15 protein expression was determined by ELISA. As predicted by mRNA analysis, the lung expressed high levels of CXCL15 protein; however, surprisingly on a per milligram basis, the adrenal gland actually expressed the highest level of CXCL15 protein (Figure 1, bottom). CXCL15 protein was not observed in the ileum and cecum.
To determine the pattern of CXCL15 protein expression, several tissues were analyzed by immunofluorescent staining. As can be seen in Figures 2A-2D, tissues expressing CXCL15 mRNA all demonstrated CXCL15 immunoreactivity as detected by a biotin goat anti-mouse CXCL15 antibody. This included the adrenal gland, trachea, colon, and uterus. In the adrenal gland, staining for CXCL15 was concentrated in the pancortical region, with increased intensity in the zona glomerulosa (Figure 2). In the trachea, CXCL15 is located in the pseudostratified epithelial cells (Figure 2). In Figure 2C, CXCL15 can be found in the upper part of the colonic gland as well as in the colonic goblet cells. In the uterus, CXCL15 is observed in the endometrium (Figure 2D). The spleen did not show positive immunostaining for CXCL15, confirming lack of mRNA expression (data not shown).
Specificity of CXCL15 Immunofluorescence in Pulmonary Tissue
Because expression of CXCL15 protein was previously demonstrated only in BALB/c lung bronchoepithelial cells, we wanted to determine if our biotinylated goat anti-mouse CXCL15 detected a similar staining pattern in C57BL/6J lung tissue (Rossi et al. 1999). As can be seen in Figure 2E, there is expression of CXCL15 in the C57BL/6J lung in cells in both the lung parenchyma and bronchoepithelial cells. Expression of CXCL15 was also analyzed in BALB/cJ and FVB/N mouse lungs, and a similar pattern of expression was seen (data not shown). To determine if the lung parenchymal cells stained by CXCL15 were bone marrow derived, cells were costained with antibodies to CD45+; there was no colocalization of the two stains (data not shown). Antibody-blocking studies were performed to determine the specificity of the immunofluorescence. After preincubation of the CXCL15 antibody with CXCL15 peptide, no immunofluorescence was detected in the lung, indicating that the immunofluorescence was specific for CXCL15 (Figure 2F).
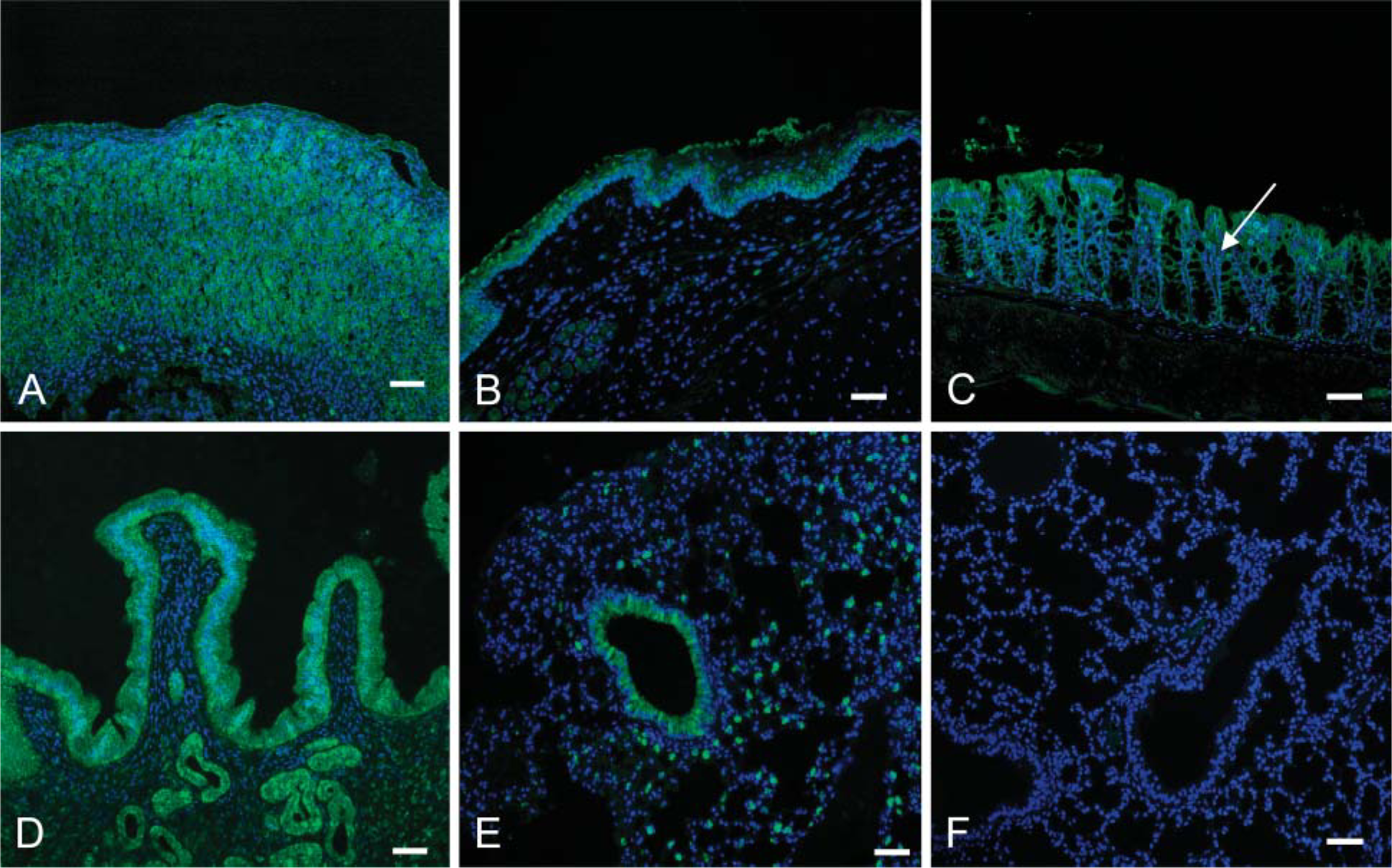
Immunofluorescence analysis of CXCL15 protein expression. (
CXCL15 Expression in Gastric Epithelial Cells
Because our laboratory has extensive expertise in the study of gastric epithelial biology, we next performed double-immunofluorescent studies to determine the exact expression pattern of CXCL15 in the stomach (Lorenz and Gordon 1993). The two mucus-producing cell types—surface mucus cells and mucus neck cells—produce mucin and trefoil factors that form the mucus layer, which acts to protect the surface of the stomach from the acidic luminal environment (Ito 1987). These cells are identified by their expression of mucin glycoproteins that react with the cholera toxin B (CTB) subunit (Figure 3) and the lectin GSII (Figure 3G), respectively (Lorenz and Gordon 1993). Parietal cells, found mostly in the zymogenic zone and mucoparietal zone, express H+/K+-ATPase, which is responsible for the production of the acidic environment in the lumen (Figure 3D). Zymogenic (chief) cells found in the zymogenic zone secrete proteins such as pepsinogen (Figure 3J) and intrinsic factor (Figure 3M), needed for digestion and absorption of vitamin B12, respectively. Based on Figures 3C, 3F, 3I, 3L, and 3O, it can be seen that CXCL15 immunostaining colocalized with a subset of cells expressing pepsinogen and intrinsic factor. This staining pattern identifies the CXCL15 expression gastric cell type as the prezymogenic and zymogenic cell. CXCL15 does not appear to be expressed by parietal cells, surface mucus cells, or the pure mucus neck cell population.
Expression of CXCL15 in H. felis-infected Mice
Because chemokines play a role in the movement of inflammatory cells, expression of ELR+ CXC chemokines was analyzed in our H. felis model of gastric inflammation. This model develops a severe chronic active gastritis responsible for a series of epithelial alterations including atrophy, metaplasia, and dysplasia, which ultimately result over 12-15 months in gastric adenocarcinoma formation (Roth et al. 1999; Cai et al. 2005; McCracken et al. 2005). Figure 4A shows the normal stomach glandular zone of an 8-week mock-infected C57BL/6 stomach, whereas Figure 4B demonstrates the dramatically altered epithelial glandular architecture and inflammatory infiltrate in the stomach of a C57BL/6J mouse infected with H. felis for 8 weeks. Figure 4G shows that CXCL15 mRNA expression is increased in 8-week H. felis-infected stomachs when compared with the CXCL15 expression in 8-week mock C57BL/6 stomachs (the mock experimental control was assigned a fold change of 1). Supplemental Figure 1 shows CXCL15 immunofluorescent staining in a mock-(Supplemental Figure 1) and H. felis-infected stomach (Supplemental Figure 1). At this timepoint there is a loss of zymogenic cells and a massive expansion of the dysplastic mucus neck cell population. This dysplastic population shows a diffuse staining of CXCL15, which correlates with the increase in mRNA expression shown in Figure 4G.
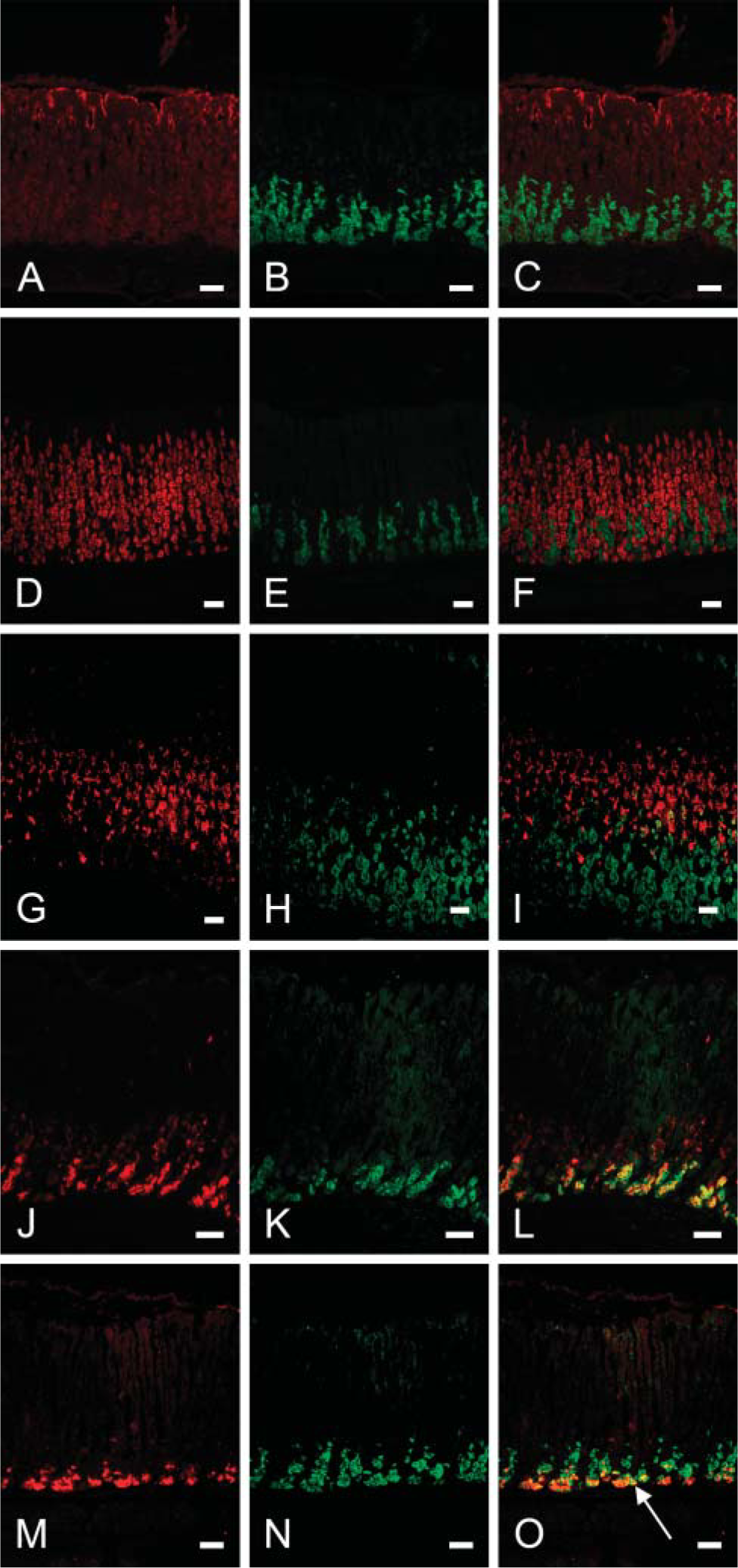
CXCL15 expression in gastric prezymogenic cells. (
Expression of CXCL15 in Two Murine Models of Colitis
Currently, our laboratory investigates two mouse models of inflammatory bowel disease (IBD). One model utilizes the FVB.mdr1a −/− mouse (multidrug-resistance gene), which spontaneously develops intestinal inflammation if housed under specific pathogen-free conditions (Panwala et al. 1998). The mdr1a gene encodes a 170-kDa transmembrane transporter protein known as P-glycoprotein, which is part of the adenosine triphosphate binding transporter family and found in the murine distal small intestine and colon (Stephens et al. 2002; Elson et al. 2005). The second model of IBD involves the transfer of regulatory T cell-deficient lymphocyte subsets into immunodeficient B6.Rag-1 −/− mice. Different permutations of this model include transfer of CD45RBhi- or CD62L-enriched T cells, as well as T cells from regulatory cytokine-deficient mice (Powrie et al. 1994; Asseman et al. 1999; Mudter et al. 2002). In these studies we used a previously reported model of colitis induction in which transfer of CD4+ T cells from uninfected C57BL/6 donor mice induces colitis upon transfer to Helicobacter hepaticus-infected, immunodeficient B6.Rag −/− mice (Kullberg et al. 2002).
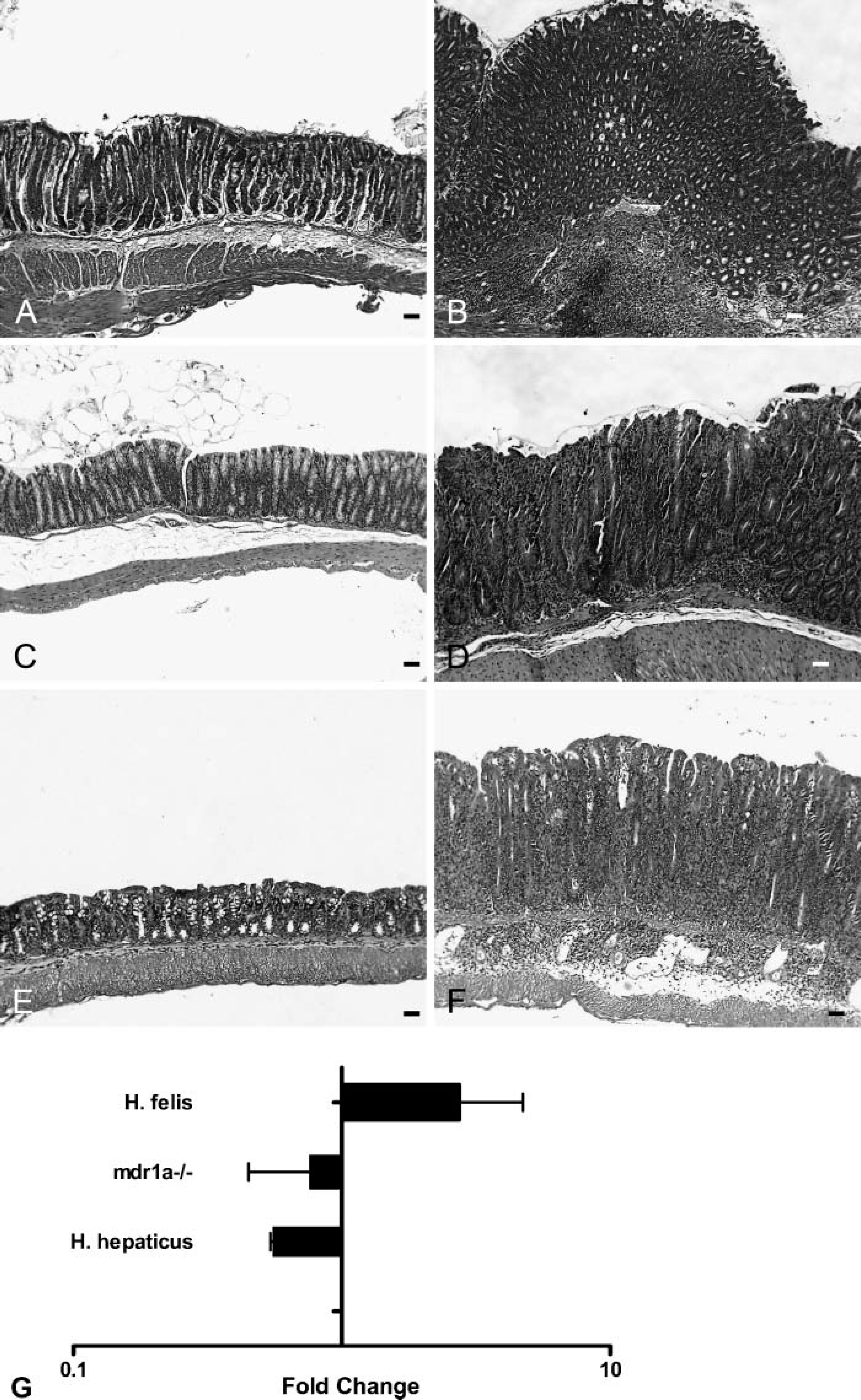
Histology and CXCL15 expression in gastric (
Colonic expression of CXCL15 was tested in both of these models and, surprisingly, the expression was decreased in mice with severe colitis, the opposite of our results in the gastritis model (Figure 4G). Figure 4C shows the normal uninflamed colon from an FVB/N mouse, and Figure 4D demonstrates the extensive inflammatory infiltrate and gland thickening that spontaneously occurs in a 6-month-old FVB.mdr1a −/− mouse. Figure 4E shows an absence of inflammation in a mock-infected B6.Rag-1 −/− recipient of CD4+ T cells that do not develop diarrhea or colitis. However, the colon from an H. hepaticus-infected B6.Rag-1 −/− recipient of CD4+ T cells that does develop diarrhea demonstrates severe intestinal inflammation (Figure 4F). Supplemental Figure 1 shows the expression of CXCL15 by immunofluorescence in these two colitis models. Unlike the gastritis model, it can be seen that the protein level of CXCL15 is unchanged or slightly decreased with colitis, correlating with the mRNA data in Figure 4G.
ELR+ CXC Chemokine Expression in Gastric and Intestinal Models of Inflammation
Because CXCL15 is a member of the ELR+ CXC chemokine family, we contrasted its expression in our gastric model and in one of our intestinal models of inflammation with the other members of the murine ELR+ CXC chemokine family: KC, LIX, and MIP2. As can be seen in Figure 5A, gastric expression of only two of these ELR+ CXC chemokines, CXCL15 and LIX, is increased in C57BL/6 mice infected with H. felis for 8 weeks, as compared with the expression levels in mock-infected animals. Additionally, mdr1a −/− mice on the FVB background develop severe colitis and have increased colonic expression of MIP-2, LIX, and KC without an increase in CXCL15, as can be seen in Figure 5B. All three models of inflammation have a significant component of neutrophils in the inflammatory infiltrate (Supplementary Figure 2). Because CXCL15 is not increased in the two models of colitis but is increased in the gastritis model, it is unclear if it plays any role in this neutrophil chemotaxis.
Discussion
Our findings clearly demonstrate that CXCL15 expression at both the mRNA and protein level is not restricted to the adult lung. The original report of CXCL15 expression analyzed its expression in tissues by Northern blot. In a study by Mocharla et al. (1990), it was shown that reverse transcriptase PCR is ∼1000-fold more sensitive than Northern blot. Our qRT-PCR data indicate that the level of expression in all tissues, with the exception of the adrenal gland, is ∼1000-fold less than the extremely high levels seen in the lung, accounting for the “lack of expression” previously determined using Northern blot analysis. It should be noted that Mocharla and coworkers (1990) did not analyze CXCL15 expression from many of the tissues investigated in the current study. mRNA was isolated from the tissues reported in the original Northern blot, and in agreement with their findings no expression was seen. Because our demonstration of CXCL15 expression in multiple mucosal and glandular tissues of the C57BL/6J mouse was different than expression reported in the original reports showing limited expression of CXCL15 in the BALB/c murine lung, we tested several different mouse strains (including Balb/cJ and FVB/N) to determine if the expression we observed was strain specific. All strains showed similar patterns of expression, indicating that our findings were not specific for the C57BL/6J strain. Because expression of CXCL15 in mRNA and protein is not a direct 1:1 ratio, it appears that CXCL15 may be regulated at both the RNA and protein levels. Further studies will need to be performed to determine where are the mechanisms of this regulation.
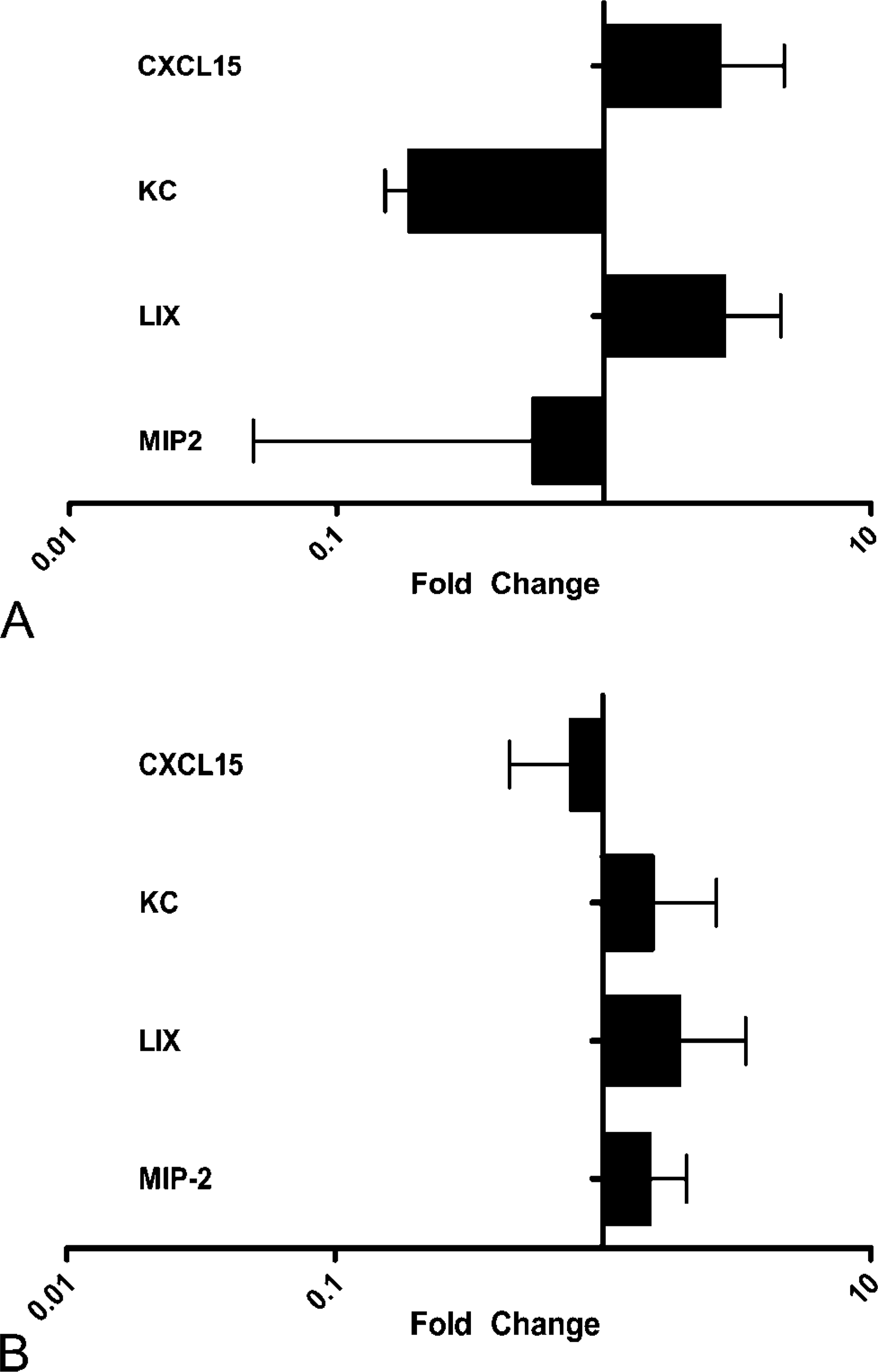
Expression of ELR+ CXC chemokines in select tissues of the gastrointestinal tract. (
Although our results clearly demonstrated a low level of mRNA and protein expression in multiple mucosal and endocrine tissues, they did not indicate what cell type(s) were involved in producing CXCL15. Immunohistochemical experiments on selected tissues clearly demonstrated expression in the epithelial layers of multiple organs including the lung, trachea, colon, uterus, stomach, and small intestine (data not shown). The function of CXCL15 at these epithelial surfaces would be expected to be similar to its identified role in the lung, i.e., to induce neutrophil migration into sites of inflammation and infection. Because there are multiple differentiated epithelial cell types in the stomach, we sought to determine which of these cell types expressed CXCL15. Through the utilization of cell-specific expression markers, the transitional prezymogenic and the mature zymogenic cell were identified as the only cell types that expressed CXCL15 in the stomach. Prezymogenic cells are a group of cells producing secretory granules, which appear intermediate between the granules in mucous neck cells and those of zymogenic cells. These cells eventually differentiate into mature zymogenic cells; however, it is not clear how long cells spend in this transition phase (Karam 1999). They appear to share functions with zymogenic cells and mucus neck cells, i.e., the production of mucus. The prezymogenic cells produce primarily pepsinogen, which is cleaved into active pepsin by hydrochloric acid released by gastric parietal cells. This pepsin then digests ingested food proteins. It is currently unclear what role CXCL15 would play in this transitional cell type.
Because CXCL15 is upregulated in response to multiple pulmonary inflammatory stimuli, we postulated that CXCL15 expression might also be upregulated in gastrointestinal inflammatory diseases. The most common cause of gastric inflammation in humans is H. pylori infection (Kusters et al. 2006); therefore, we utilized a mouse model of Helicobacter-induced gastritis to investigate this hypothesis. As predicted, CXCL15 expression was upregulated during experimental Helico-bacter infection. The function of CXCL15 in this model of inflammation could be to recruit neutrophils because both human and mouse Helicobacter-associated gastritis have a significant neutrophil infiltrate (Dundon et al. 2002 and Supplementary Figure 2). In addition, because this model of gastritis progresses over time to gastric adenocarcinoma, it is intriguing to speculate that CXCL15 could also play a role in this progression toward gastric epithelial cancer (Lynch et al. 1995; Cahill et al. 1996; Nomura et al. 2004). One potential mechanism for this involvement could be the role of CXCL15 as a chemo-attractant for bone marrow progenitor cells because it has been recently reported that Helicobacter-associated gastric cancer originates from bone marrow stem cells (Ohneda et al. 2000; Houghton et al. 2004).
Contrary to what we saw in the H. felis-associated gastritis model, expression of CXCL15 was decreased in two models of murine colitis. Because both models have mild to moderate Gr-1+ neutrophil infiltrate, this implies that these neutrophils are recruited by chemokines other than CXCL15 (Li et al. 1998; Panwala et al. 1998 and Supplementary Figure 2). Our results imply that CXCL15 does not play a significant role in either CD4-mediated infectious or spontaneous murine colitis.
To investigate other potential mechanisms for neutrophil chemoattraction in both the Helicobacter-associated gastritis and the spontaneous FVB.mdr1a −/− colitis models, we decided to investigate the presence of other ELR+ CXC chemokines in the gastrointestinal tract. Analysis of the 8-week infected H. felis gastritis model demonstrated that expression of LIX and CXCL15 chemokines were increased. Intriguingly, the pattern of inflammation-associated expression of ELR+ CXC chemokines was different in the FVB.mdr1a −/− model because expression of KC, LIX, and MIP2 was increased, whereas CXCL15 expression was decreased.
In conclusion, CXCL15 (lungkine) can no longer be considered a lung-specific chemokine. The low-level expression in multiple mucosal tissues implies a much broader role in inflammatory disease than just localized pulmonary inflammation. Localization of CXCL15 to epithelial cells implies that part of its function could be to sense external infection and initiate immune responses. However, the actual functional role of CXCL15 in gastrointestinal disease remains to be elucidated.
Footnotes
Acknowledgements
This work was supported in part by a grant from the American Cancer Society (RPG-99-086-01-MBC), National Institutes of Health (NIH) Grants R01 DK-059911 and P01 DK-071176, and the University of Alabama at Birmingham Digestive Diseases Research Development Center Grant #P30 DK-064400. J.M.S. received salary support from NIH Training Grant T32 AI-07041.
We thank Camalla Kimbrough and Andrea Stanus for expert technical assistance.
