Abstract
Current scientific literature lacks data on the prognostic value of the expression of RAD51 and BRCA2 in gastric adenocarcinoma. Therefore, we aimed to evaluate those and other homologous recombination-related proteins (ATM, ATR, BRCA1, CHK2, γH2AX, p53) in gastric cancer, assessing their correlation with clinical prognosis. Paraffin-embedded samples were obtained from surgical specimens collected in total or subtotal gastrectomy procedures. Between 2008 and 2017, 121 patients with advanced gastric adenocarcinoma underwent surgical resection and were included in this study. Negativity for nuclear RAD51 correlated with vascular invasion, lymph node metastasis, larger tumor size, and lower overall survival and disease-free survival in univariate analysis. However, nuclear RAD51-negative cases presented better response rates to adjuvant therapy than the positive ones. Nuclear ATR negativity correlated with larger tumor size and a higher histological grade. Positivity for ATM was associated with more prolonged disease-free survival. Positivity for nuclear BRCA2 correlated with lower overall survival and diffuse histological type, whereas its high expression was associated with vascular invasion. Nevertheless, tumors positive for nuclear BRCA2 were more frequently low grade in the intestinal histological type. Our findings indicate that RAD51 and BRCA2 are valuable immunohistochemical prognostic markers in gastric adenocarcinoma.
Introduction
Gastric adenocarcinoma is the fifth malignant neoplasm in incidence and the third in mortality worldwide; it represents a group of biologically and genetically heterogeneous tumors with a multifactorial etiology that involves the interaction between environmental conditions and genetic/epigenetic alterations. 1
Homologous recombination is the most accurate pathway for repairing DNA double-strand breaks. Defects in DNA repair favor tumor development by allowing the accumulation of mutations and chromosomal aberrations. In many cases, the low expression of homologous recombination proteins is characteristic of more aggressive tumors; such neoplasms, however, tend to be more sensitive to therapeutic agents such as cytotoxic drugs, radiotherapy, and inhibitors of poly-ADP ribose polymerase.2–5
Among the various proteins that participate in the homologous recombination mechanism, the following stand out: the MRE11-RAD50-NBS1 complex, which is a DNA break sensor; ataxia telangiectasia mutated (ATM), a central element at the beginning of the signaling cascade that phosphorylates and activates multiple proteins; intermediate pathway elements such as checkpoint kinase 2 (CHK2), breast cancer susceptibility protein type 1 (BRCA1), BRCA2, ATM and RAD3-related protein (ATR), and the phosphorylated histone γH2AX; and RAD51, which is the great effector protein that searches for the homologous DNA sequence that will serve as a template for the repair.6,7 In parallel with homologous recombination, the activation of p53 stops the cell cycle or promotes apoptosis. 8
To date, few studies have assessed the expression of RAD51, ATR, and BRCA2 in gastric cancer. Identifying potential prognostic or treatment response markers is key to determining the best therapeutic strategy. This study evaluated the immunohistochemical expression of RAD51, BRCA2, ATR, ATM, p53, CHK2, BRCA1, and γH2AX in samples of gastric adenocarcinoma, correlating it with clinicopathological characteristics.
Materials and Methods
This was a retrospective cohort study conducted at the University Hospital of the Ribeirão Preto Medical School, at the University of São Paulo (HCFMRP-USP). Patients undergoing total or subtotal gastrectomy due to gastric adenocarcinoma infiltrating at least the muscularis propria layer were included. Patients who died less than 30 days after the surgery were excluded. The study was approved by the HCFMRP-USP Research Ethics Committee (process no. 12349/17).
Histological Examination
All cases were histologically reviewed and classified into Laurén’s diffuse, intestinal, or mixed types. 1 Intestinal tumors were classified as well-differentiated (grade 1), moderately differentiated (grade 2), and poorly differentiated (grade 3). Diffuse and mixed neoplasms were always considered grade 3. 1 Pathological staging; the presence of vascular (lymphatic or blood vessel) or perineural invasion; and the presence of gastritis, intestinal metaplasia, and Helicobacter pylori in non-neoplastic mucosa were also assessed.
Tissue Microarrays (TMAs)
TMAs were performed using two representative neoplastic samples and one non-neoplastic mucosal sample from each case, extracted from paraffin blocks with 2-mm gauge needles.
Immunohistochemistry
Immunohistochemistry was performed using 4-µm histological sections obtained from TMAs and placed on silanized slides. For deparaffinization, successive xylol and alcohol baths were used. Antigen retrieval was done with citrate in pH 6.0 for p53, tris-EDTA in pH 9.0 for CHK2, and with Diva Decloaker 10X (Biocare Medical, Pacheco, CA 94553, USA) for the other markers. The slides were incubated with antibodies to p53 (Novocastra, Buffalo Grove, IL 60089 USA, NCL-p53-DO7, 1:50 dilution), CHK2 (Novocastra, DCS270.1, 1:100), ATM (St John’s Laboratory, London, E16 2RD, United Kingdom, STJ97797, 1:400), ATR (Abcam, ab178407, 1:100), BRCA1 (St John’s Laboratory, STJ113833, 1:300), RAD51 (St John’s Laboratory, STJ95330, 1:100), γH2AX (Abcam, ab26350, 1:100), for 12 hr at –4C, and BRCA2 (St John’s Laboratory, STJ91885, 1:100), for 1.5 hr at –4C. Detection was performed using the Reveal biotin-free detection kit (Biogen, MA 02142, USA). Sections were incubated with a solution of diaminobenzidine (DAB) and hydrogen peroxide, and were counterstained with Harris’s hematoxylin.
Immunohistochemical Evaluation Criteria
Staining intensity (weak, moderate, or strong) was evaluated and the percentage of stained cells was estimated. Then, the histochemical score (H-score) for each case was calculated by multiplying the percentage of marked cells by 1, 2, or 3 in case of weak, moderate, or strong marking, respectively. Therefore, results could range from 0 to 300. Nuclear and cytoplasmic staining were evaluated separately for RAD51, ATR, BRCA1, and BRCA2, whereas p53, CHK2, ATM, and γH2AX had only nuclear staining considered.
Cases with 10% or less marking of neoplastic cells were considered negative; those above 10% were considered positive.9–11 Positive cases were divided into low expression (H-score less than or equal to the median of positive cases) and high expression (H-score greater than the median). 12 Not all samples were evaluable due to the absence of tumor cells or tissue in some histological sections.
Statistical Analysis
Student’s t-test was used for comparing means between two groups. The chi-square or Fisher’s exact tests verified the homogeneity between proportions. Spearman’s correlation coefficient was used for assessing correlations between markers based on individual H-score values. The analysis of overall survival and disease-free survival was performed using the Kaplan–Meier survival curve with a log-rank test. Cox uni- and multivariate regression models were used for calculating hazard ratios (HRs). The level of significance considered for the tests was 5%.
Results
Characteristics of the Study Population
The clinicopathological characteristics of the 121 patients included in the study, operated between April 2008 and June 2017, are summarized in Table 1.
Clinicopathological Characteristics of the Patients.

Abbreviation: ECX, epirubicin, cisplatin, and capecitabine.
Immunohistochemistry Results
In tumor samples (Fig. 1), p53 was positive in 55.4% (67/121) of the cases, CHK2 in 99.2% (120/121), ATM in 94.1% (112/119), nuclear ATR in 40.8% (49/120), cytoplasmic ATR in 37.5% (45/120), nuclear BRCA1 in 85% (102/120), cytoplasmic BRCA1 in 93.4% (113/121), nuclear BRCA2 in 36.4% (44/121), cytoplasmic BRCA2 in 38.8% (47/121), nuclear RAD51 in 81.5% (97/119), cytoplasmic RAD51 in 84.0% (100/119), and γH2AX in 52.9% (64/121). Table 2 shows correlations between expressions of nuclear RAD51, nuclear ATR, ATM, and nuclear BRCA2 and clinicopathological parameters.
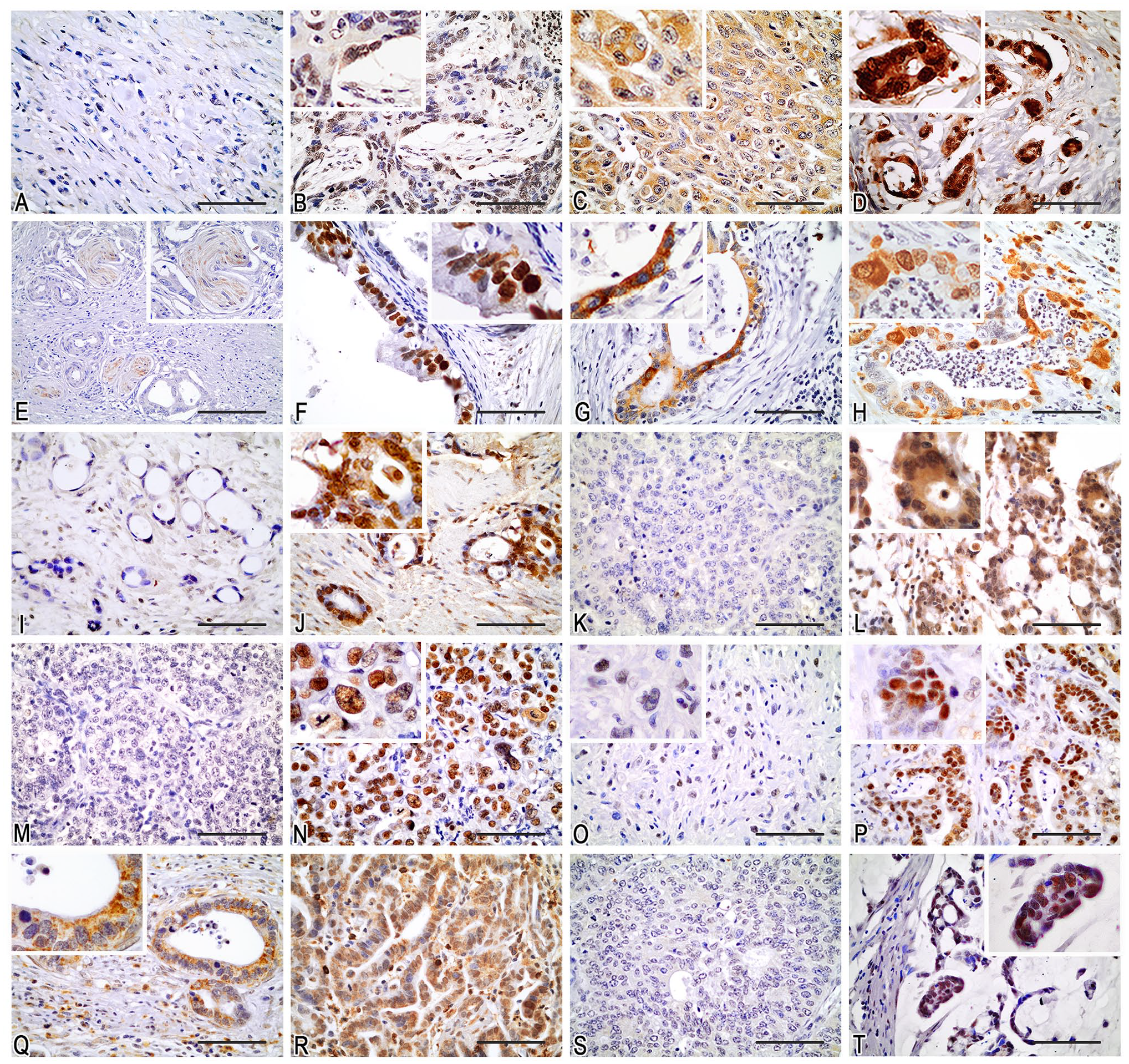
Immunohistochemical staining of tumor samples. RAD51: (A) negative; (B) mainly nuclear; (C) cytoplasmic; (D) nuclear and cytoplasmic. ATR: (E) negative (perineural invasion with stained nerve fibers); (F) nuclear; (G) cytoplasmic; (H) nuclear and cytoplasmic. ATM: (I) negative; (J) positive (nuclear staining was considered). BRCA2: (K) negative; (L) nuclear and cytoplasmic. p53: (M) negative; (N) positive (nuclear). CHK2: (O) weak (nuclear); (P) strong (nuclear). BRCA1: (Q) cytoplasmic; (R) nuclear and cytoplasmic. γH2AX: (S) negative; (T) positive (nuclear). Scale bars = 100 μm.
Correlations Between the Expressions of Nuclear RAD51, Nuclear ATR, ATM, and Nuclear BRCA2 and Clinicopathological Parameters in Gastric Adenocarcinoma.
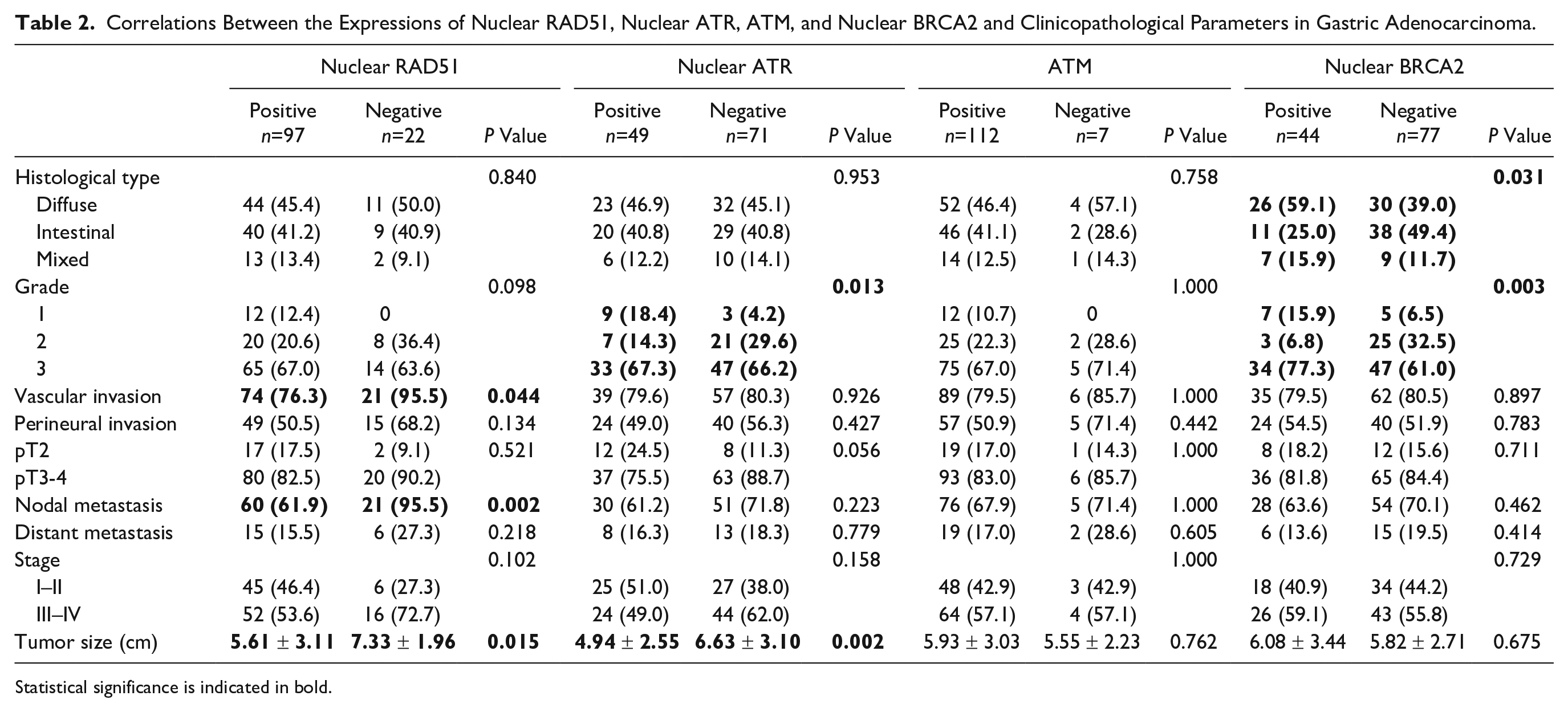
Statistical significance is indicated in bold.
In non-tumor mucosa, no immunostaining for p53 was observed. CHK2 showed weak to moderate diffuse nuclear staining in the lower portion of foveolar epithelium (proliferative compartment), and only in sparse cells in the upper portion of foveolar epithelium and glands. Other markers showed weak and focal marking, mainly nuclear, and probable nonspecific cytoplasmic staining was also observed, especially in parietal cells.
Correlation Between Immunohistochemical Markers
We observed a weak positive correlation (r = 0.180, p=0.048) between the nuclear and cytoplasmic expression of RAD51. No correlation was observed between the respective nuclear and cytoplasmic expressions of BRCA1, BRCA2, and ATR. Table 3 shows the correlations of the nuclear expression of the markers. The strongest correlations, all positive, were between ATM and nuclear BRCA1 (r = 0.428, p<0.001), and between γH2AX and p53 (r = 0.407, p<0.001), nuclear ATR (r = 0.552, p<0.001), and nuclear RAD51 (r = 0.491, p<0.001).
Spearman’s Correlation r Coefficients Between Nuclear Markers and Their Respective P Values.
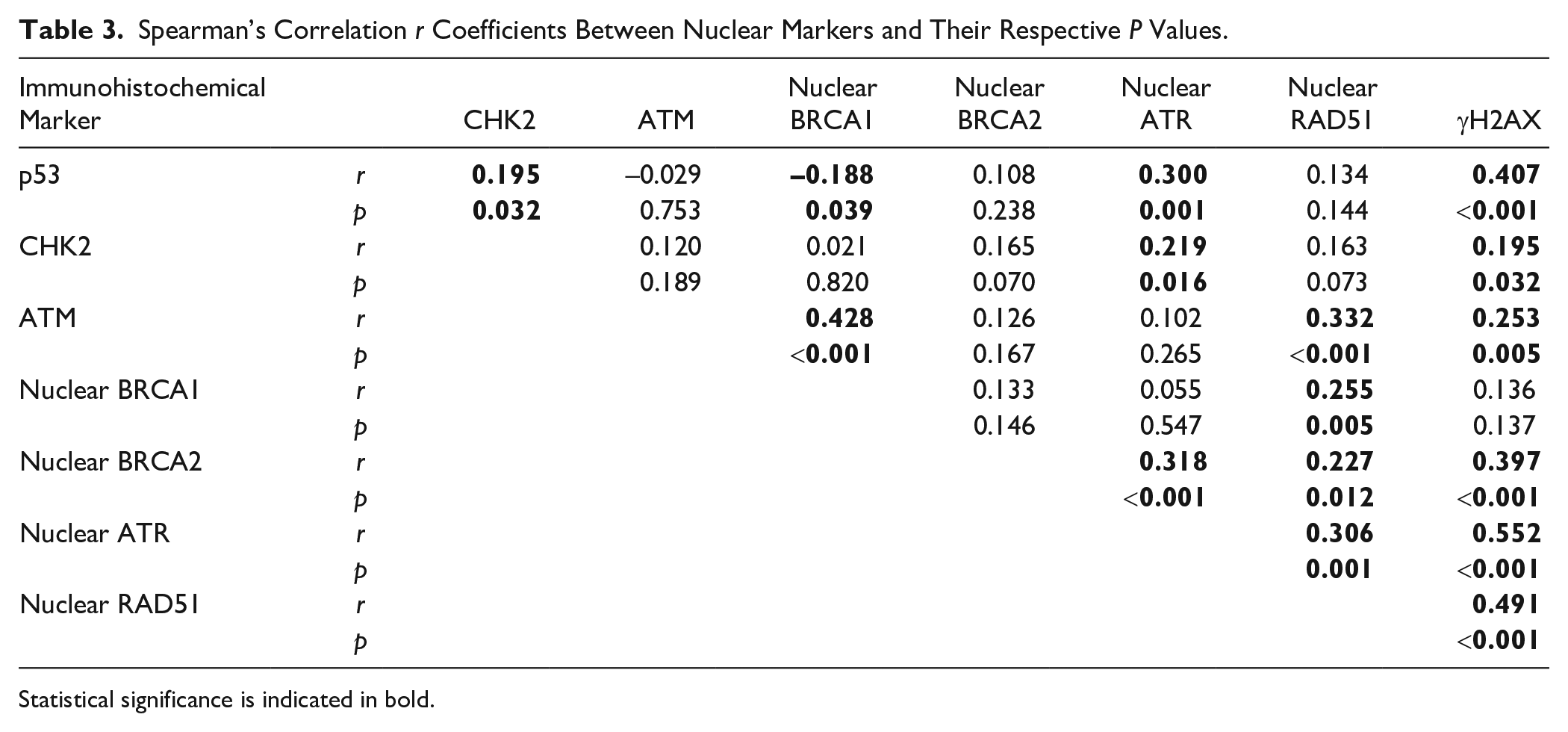
Statistical significance is indicated in bold.
RAD51
A higher proportion (p=0.037) of nuclear RAD51-positive tumors was observed in cases with prior neoadjuvant therapy—93.75% (30/32)—than in those without neoadjuvant therapy—77.01% (67/87).
Mean tumor size was larger in nuclear RAD51-negative tumors than in positive ones (7.33 cm vs 5.61 cm, p=0.015). This finding was independent (p=0.043) from the correlation between this marker and neoadjuvant therapy, which reduced tumor size.
Tumors that were negative for nuclear RAD51 showed a higher frequency of lymphatic or blood vessel invasion than the positive ones [95.5% (21/22) vs 76.3% (74/97) in the total population, p=0.044; 95% (19/20) vs 80.6% (54/67) in cases without neoadjuvant therapy, p=0.124]. Nuclear RAD51-negative tumors also had a greater frequency of metastasis in regional lymph nodes [95.5% (21/22) vs 61.9% (60/97) in the total population, p=0.002; 95% (19/20) vs 64.2% (43/67) in cases without neoadjuvant therapy, p=0.008].
Nuclear RAD51-negative adenocarcinomas showed a correlation with lower overall survival and disease-free survival (Tables 4 and 5). In the analysis of overall survival, positive cases had an HR of 0.576 (p=0.048) compared with negative cases; mean global survival was 1073 and 684 days, respectively. In the group of patients without adjuvant or neoadjuvant treatment, HR was 0.345 (p=0.044); in the group with adjuvant or neoadjuvant treatment, it was 0.718 (p=0.327): 0.745 (p=0.416) in the adjuvant group and 1.269 (p=0.817) in the neoadjuvant group (only two negative cases were observed in this group). Therefore, there was no significant association between nuclear RAD51 and survival in these chemo- or radiotherapy submitted groups (Table 4 and Fig. 2). In the analysis of disease-free survival (Table 5), in the total population, HR of positive to negative nuclear RAD51 cases was 0.531 (p=0.022).
Uni- and Multivariate Analysis of Overall Survival.
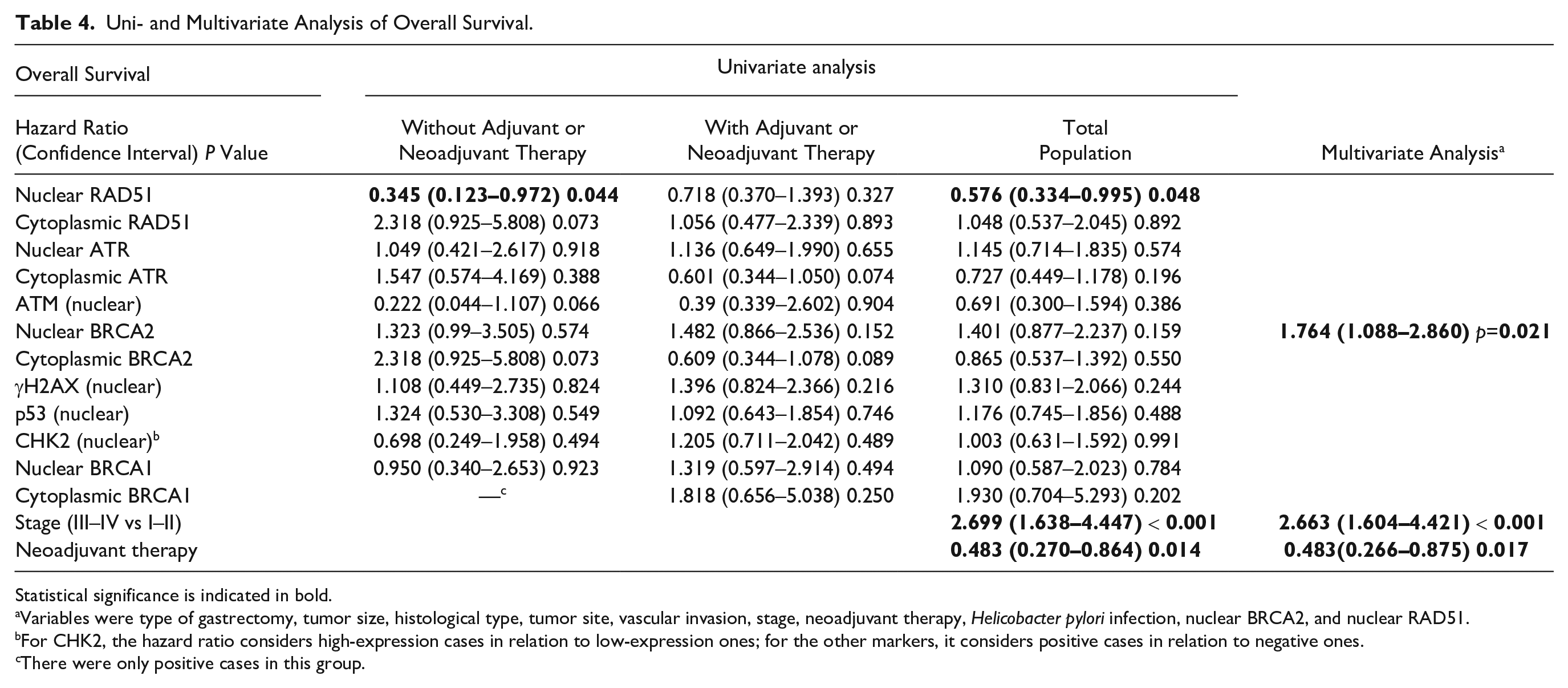
Statistical significance is indicated in bold.
Variables were type of gastrectomy, tumor size, histological type, tumor site, vascular invasion, stage, neoadjuvant therapy, Helicobacter pylori infection, nuclear BRCA2, and nuclear RAD51.
For CHK2, the hazard ratio considers high-expression cases in relation to low-expression ones; for the other markers, it considers positive cases in relation to negative ones.
There were only positive cases in this group.
Uni- and Multivariate Analysis of Disease-free Survival.
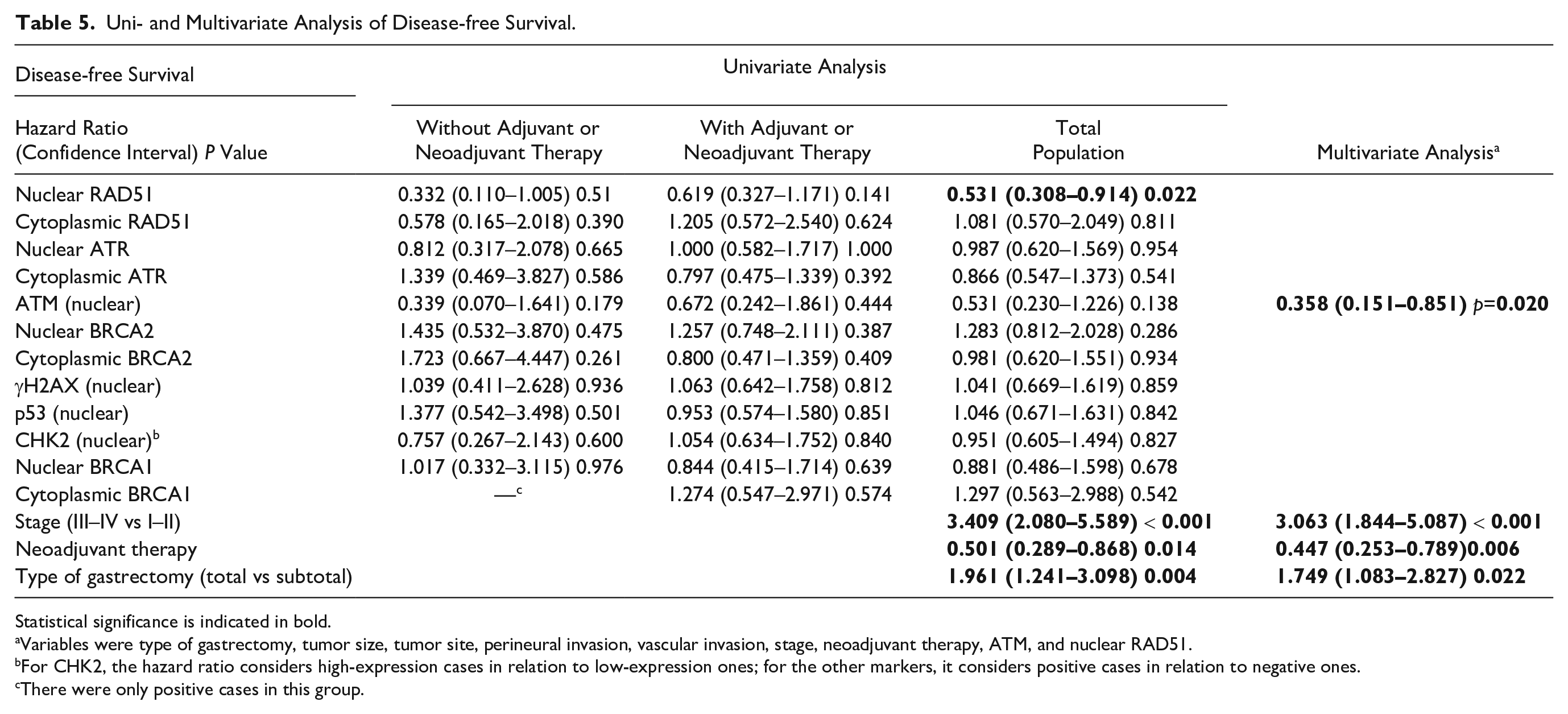
Statistical significance is indicated in bold.
Variables were type of gastrectomy, tumor size, tumor site, perineural invasion, vascular invasion, stage, neoadjuvant therapy, ATM, and nuclear RAD51.
For CHK2, the hazard ratio considers high-expression cases in relation to low-expression ones; for the other markers, it considers positive cases in relation to negative ones.
There were only positive cases in this group.
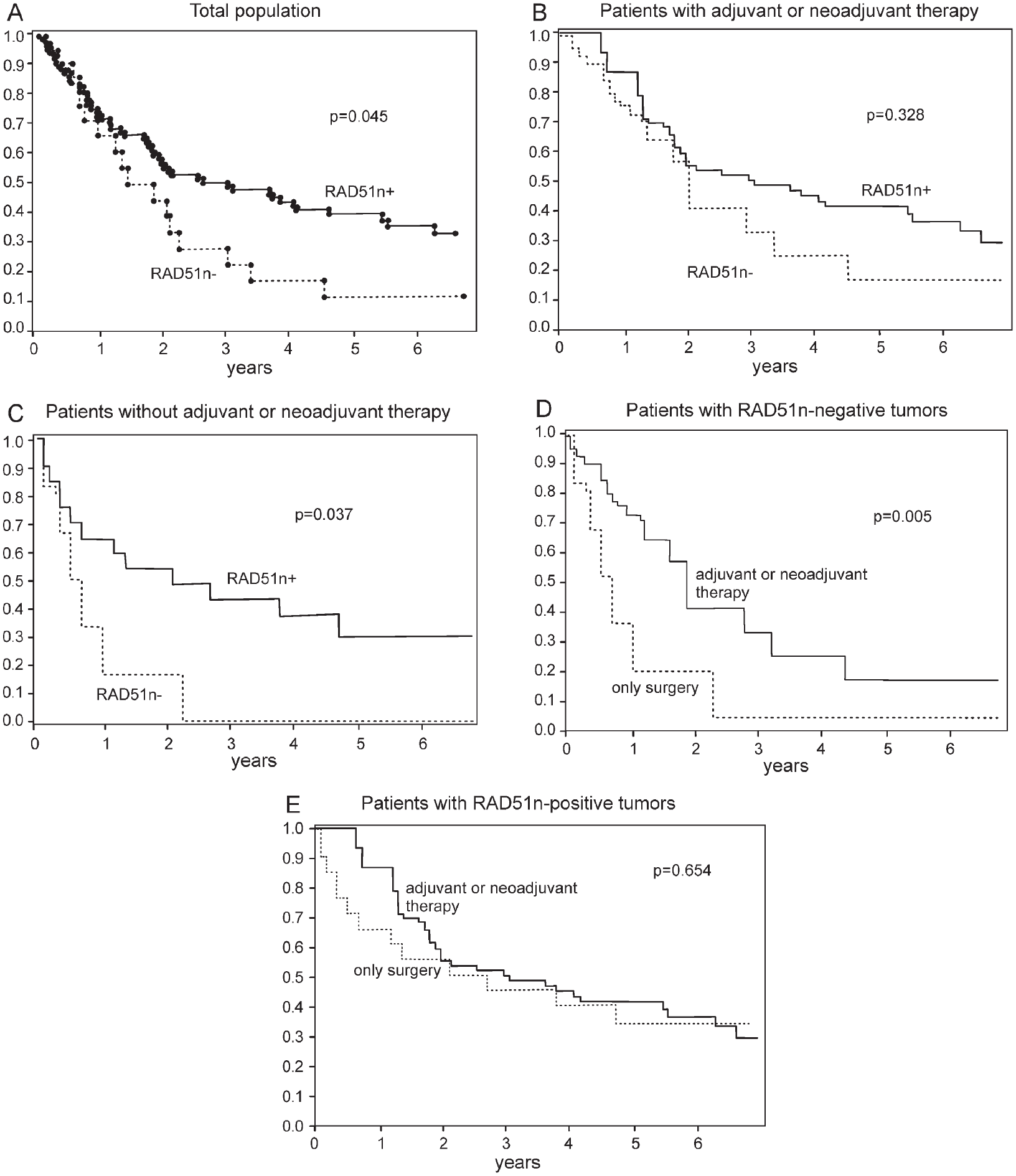
Kaplan–Meier curves of overall survival comparing patients with positive and negative gastric adenocarcinomas for nuclear RAD51 in the total population (A), in the group of patients with adjuvant or neoadjuvant treatment (B), and in the group of patients treated only with surgery (C); curves comparing surgically treated patients with those who received adjuvant/neoadjuvant therapy, grouped in nuclear RAD51-negative (D) and RAD51-positive (E) groups. The p values were calculated using the log-rank test.
Considering patients whose tumors were negative for nuclear RAD51 (Fig. 2), adjuvant or neoadjuvant treatment evaluated in conjunction (HR: 0.260, p=0.004, n=16) or adjuvant treatment (HR: 0.280, p=0.008, n=14) was associated with greater overall survival in comparison with exclusively surgical treatment (n=6); this was not the case of nuclear RAD51-positive tumors (adjuvant or neoadjuvant: HR: 0.739, p=0.322, n=76; adjuvant: HR: 0.900, p=0.742, n=46; in comparison with only surgery group: n=21). Neoadjuvant treatment results were not statistically significant (nuclear RAD51-negative: HR: 0.231, p=0.138, n=2; nuclear RAD51-positive: HR: 0.567, p=0.142, n=30).
ATR
Mean tumor size was larger in nuclear ATR-negative tumors than in positive ones (6.63 cm vs 4.94 cm, p=0.002). Nuclear ATR was also correlated with histological grade. The proportion of grade 3 cases was similar in positive [67.3% (33/49)] and negative [66.2% (47/71)] tumors, but there was a higher proportion (p=0.013) of grade 2 cases among negative tumors [29.6% (21/71) vs 14.3% (7/49)] and of grade 1 cases among positive tumors [18.4% (9/49) vs 4.2% (3/71)].
ATM
ATM negativity correlated with lower disease-free survival in the multivariate analysis (Table 5), with HR of 0.358 for positive cases compared with negative ones (p=0.020). In this analysis, the following variables were considered: type of gastrectomy, tumor size, tumor location, perineural invasion, vascular invasion, stage, therapy, ATM, and nuclear RAD51.
BRCA2
Positivity for nuclear BRCA2 was correlated with diffuse histological type [59.1% (26/44) among positive cases; 39% (30/77) among the negative ones; p=0.031]. It also correlated with a higher proportion of grade 3 and grade 1 tumors (Table 2). Association with grade 3 is due to greater proportion of diffuse type. Grade 1 tumors, on the contrary, are of intestinal type by definition. Vascular invasion was more frequent in adenocarcinomas with high nuclear BRCA2 expression [95.45% (21/22)] than in those with negative or low expression [76.77% (76/99), p=0.047].
In the multivariate analysis (Table 4), positivity for nuclear BRCA2 in gastric adenocarcinoma was correlated with a lower overall survival, with an HR of 1.764 when comparing positive cases with negative ones (p=0.021). In this analysis, the following variables were considered: type of gastrectomy, tumor size, histological type, tumor location, vascular invasion, stage, treatment, H. pylori infection, nuclear BRCA2, and nuclear RAD51.
Considering the intestinal type alone, positive cases for nuclear BRCA2 had a death HR of 1.691 compared with negative ones in univariate analysis (p=0.204). The cases with high expression of nuclear BRCA2 had 100% (5/5) of presence of vascular invasion, and the others, 65.91% (29/44; p=0.117).
Discussion
To date, few studies have assessed the clinical importance of RAD51, BRCA2, and ATR in gastric cancer. We evaluated and correlated them with more extensively studied homologous recombination-related proteins to broaden the understanding on such matter.
Our results revealed an overall trend of positive correlation between the nuclear expressions of most evaluated repair proteins. The phosphorylated histone γH2AX correlated positively with nuclear ATR, nuclear RAD51, CHK2, ATM, nuclear BRCA2, and p53. A hypothetical explanation for this finding is the reactive expression of repair proteins in a situation of increased DNA damage, because γH2AX presence indicates the occurrence of DNA double-strand breaks. 13 A possible cause of DNA damage accumulation is p53 mutation itself. The immunohistochemical detection of p53 commonly reflects a dysfunctional mutant protein with a lower degradation rate. 10 Other possible explanations to p53 and γH2AX correlation include the fact that DNA double-strand breaks reduce p53 ubiquitination by MDM2 through several mechanisms, 8 and the caspase-3-mediated H2AX phosphorylation during apoptosis triggered by p53.14,15 The correlation between γH2AX and ATM has also been reported in thyroid carcinomas. 16 This correlation could be attributed to the fact that ATM phosphorylates H2AX. However, DNA-PKcs and ATR also redundantly perform such phosphorylation. 17
We also detected a weak positive correlation of p53 with CHK2 and nuclear ATR. Both proteins stabilize p53—CHK2 by direct phosphorylation and ATR by MDM2 phosphorylation. 8 The association between CHK2 and p53 has already been demonstrated in gastric cancer. 18 On the contrary, the correlation we observed between nuclear BRCA1 and p53 was negative, albeit weak. One possible explanation is that positivity for p53 may reflect in some cases the accumulation of normal protein, as one of the usual roles of p53 is to transport BRCA1 from the nucleus to the cytoplasm. 19
Typically, the absence of a DNA repair protein favors tumor progression due to a propensity to mutations and chromosomal aberrations. In this sense, our findings suggest that negative nuclear RAD51, nuclear ATR, and ATM correlate with more aggressive gastric cancers.
Unlike these markers, nuclear BRCA2 proved to be characteristic of more aggressive neoplasms. Its positivity correlated with a lower overall survival in the multivariate analysis and with a diffuse histological type, whereas high expression was associated with vascular invasion. These findings are aligned with those by Zhang et al., 20 who correlated the nuclear expression of BRCA2 with a higher frequency of lymph node metastasis in gastric adenocarcinoma.
However, nuclear BRCA2 was associated with well-differentiated tumors (grade 1), in the intestinal histological type. This could suggest a different role of BRCA2 in the pathogenesis of intestinal type tumors. Nonetheless, a trend toward the association of nuclear BRCA2 with vascular invasion and lower survival, similar to that in the whole population, was also noted in the intestinal type, although it was not statistically significant.
Nuclear ATR negativity was associated with larger tumors, a higher proportion of histological grade 2, and a lower proportion of grade 1 tumors. These findings contrast with those by Zhang et al., 20 who did not detect a correlation between the ATR expression in gastric cancer and clinicopathological characteristics. Abdel-Fatah et al. 21 also observed different results in breast cancer, with an association between ATR expression and greater tumor aggressiveness.
ATM negativity was associated with lower disease-free survival in the multivariate analysis. The correlation between low ATM expression and a worse prognosis in gastric cancer was also noted by Abdel-Fatah et al., 22 Lee et al., 9 and Kim et al. 23
Neoadjuvant therapy was correlated with a higher proportion of gastric adenocarcinomas that were positive for nuclear RAD51, which suggests a possible defense reaction of the neoplasm to the DNA damage caused by the treatment.
Negative nuclear RAD51 results were associated with larger tumors, vascular invasion, metastasis to regional lymph nodes, and lower overall and disease-free survival in the univariate analysis. An association between low RAD51 expression and a worse prognosis has already been described in glioblastoma, 24 breast cancer,25,26 and non–small cell lung carcinoma. 27 However, for several other tumors, studies have shown a correlation between high RAD51 expression and unfavorable clinicopathological characteristics.11,28–34 This could be caused by the stimulation of EBPβ expression by RAD51. EBPβ is a transcription factor related to the epithelial–mesenchymal transition, metalloproteinase synthesis, and metastatic potential. 31
We observed that adjuvant or neoadjuvant treatment did not alter the survival of patients with nuclear RAD51-positive tumor, indicating resistance to chemoradiotherapy. It is in accordance with previous demonstrations that RAD51 promotes resistance to DNA-damaging treatment in cell cultures of various cancer types, 2 including gastric cancer. 35
Conversely, we found an increased overall survival in patients undergoing adjuvant treatment or in adjuvant plus neoadjuvant groups compared with exclusively surgical cases in patients with nuclear RAD51-negative tumors. The survival of nuclear RAD51-negative cases treated with chemotherapy or radiotherapy was similar to those with RAD51-positive neoplasms. Nonetheless, nuclear RAD51 negativity was significantly associated with lower overall survival among patients treated with surgery alone.
Tan et al. 35 also evaluated RAD51 expression in gastric cancer. They studied 52 patients and detected no prognostic correlation in the total population. However, they observed lower survival of the RAD51-positive cases limited to the group of patients treated with 5-fluorouracil. Those results are different from ours, but they may also suggest RAD51-associated chemoresistance. The results reported by Gachechiladze et al. 27 in non–small cell lung carcinoma are similar to ours; they observed an association between the absence of RAD51 expression and a worse prognosis only in patients treated exclusively with surgery.
Our results suggest that RAD51 immunohistochemistry could be used to determine the need for adjuvant treatment or to indicate specific types of therapy, depending on future research results. One of the limitations of this study is that we included different therapeutic schemes, and the small sample hampered individual analyses on each line of treatment.
In summary, the immunohistochemical analysis of nuclear RAD51, nuclear BRCA2, and ATM in gastric adenocarcinoma seems to be important in prognostic evaluation. Nuclear RAD51 is also probably related to the prediction of chemotherapy or radiotherapy response.
Footnotes
Acknowledgements
We acknowledge and thank Alfredo Ribeiro-Silva (in memoriam) for his contribution toward the conception of this study, Laura Midori Kawasse Ferreira and Márcia Aparecida Ferreira for performing immunohistochemistry, Guilherme de Paula Lemos and Flávio Henrique Leite for TMA construction, and Creusa Maria Roveri Dal Bó for statistics.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MOB designed the study. JDBP and CFAM collected clinical data. JDBP and MOB performed histological and immunohistochemical analysis. JDBP, MOB, and AKS contributed to statistical analysis. ATF, AKS, JSJ, and DPCT provided experimental support and intellectual input. JDBP and MOB wrote the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the São Paulo Research Foundation (FAPESP-N. 2017/24081-0).
