Abstract
Cholinergic agents affect the light responses of many ganglion cells (GCs) in the mammalian retina by activating nicotinic acetylcholine receptors (nAChRs). Whereas retinal neurons that express β2 subunit-containing nAChRs have been characterized in the rabbit retina, expression patterns of other nAChR subtypes remain unclear. Therefore, we evaluated the expression of α7 nAChRs in retinal neurons by means of single-, double-, and triple-label immunohistochemistry. Our data demonstrate that, in the rabbit retina, several types of bipolar cells, amacrine cells, and cells in the GC layer express α7 nAChRs. At least three different populations of cone bipolar cells exhibited α7 labeling, whereas glycine-immunoreactive amacrine cells comprised the majority of α7-positive amacrine cells. Some GABAergic amacrine cells also displayed α7 immunoreactivity; α7 labeling was never detected in rod bipolar cells or rod amacrine cells (All amacrine cells). Our data suggest that activation of α7 nAChRs by acetylcholine (ACh) or choline may affect glutamate release from several types of cone bipolar cells, modulating GC responses. ACh-induced excitation of inhibitory amacrine cells might cause either inhibition or disinhibition of other amacrine and GC circuits. Finally, ACh may act on α7 nAChRs expressed by GCs themselves.
Keywords
T
Functional studies in cat (Ikeda and Sheardown 1982; Schmidt et al. 1987) and rabbit retinas (Masland and Ames 1976; Ariel and Daw 1982a,b; Masland et al. 1984) revealed that application of ACh or acetylcholinesterase inhibitors influenced responses in many GC subsets, the visually driven responses of ON- and OFF-center GCs, and the responses of all GCs that are sensitive to motion. In vitro experiments with cat retina showed that intra-arterially applied ACh increased both the rod- and cone-driven b-wave amplitude of the electroretinogram, indicating increased depolarization of bipolar cells. ACh also induced a decrease in the optic nerve response, which reflects the summed activity of GCs (Jurklies et al. 1996). Electrophysiological studies revealed that the actions of ACh are mediated in part via nicotinic ACh receptors (nAChRs) (Masland and Ames 1976).
nAChRs are members of the family of ligand-gated ion channels. They are pentameric receptors comprised of subunits α2-α6 and β2-β4 in α/β combinations or of subunits α7-α9 in homomeric forms (McGehee 1999; Lindstrom 2000; Dani 2001). The subunit composition of nAChRs has been shown to determine the pharmacological and functional properties including agonist/antagonist affinity, open time, and desensitization rate. For instance, α7 nAChRs are characterized by their sensitivity to blockade by α-bungarotoxin (αBgt, a protein from the venom of the banded krait), rapid desensitization, and relatively low sensitivity to activation by ACh. In contrast, heteromeric nAChR subtypes such as αβ2 are αBgt-insensitive. Furthermore, αβ2 nAChRs desensitize more slowly than the α7 nAChRs and are potently activated by ACh (Nelson and Lindstrom 1999). Optical recordings of intracellular free calcium changes in rabbit retinal GCs suggested that the actions of ACh on these cells are mediated solely by αBgt-insensitive nAChRs and are not attributable to indirect effects mediated by glutamatergic bipolar cells (Baldridge 1996). However, data from subsequent extracellular recordings, single-cell RT-PCR of rabbit retinal GCs (Reed et al. 2002; Strang et al. 2003,2005a), and the results of molecular and pharmacological studies of rat retina (Moretti et al. 2004) suggest that both αBgt-sensitive and -insensitive nAChR subtypes are involved in generating the light responses of some GCs. Both receptor subtypes are expressed directly by GCs, and nAChRs are expressed by upstream cells as well. Indeed, immunohistochemical (IHC) studies have demonstrated that in rabbit retina many GCs as well as several types of amacrine cells express αBgt-insensitive nAChRs containing β2 subunits, some of which are in combination with α3 and possibly other subunits (Keyser et al. 2000; Dmitrieva et al. 2001,2003).
However, identification of cholinoceptive neurons expressing αBgt-sensitive nAChRs in mammalian retina has been more problematic. Pourcho (1979) described the distribution of αBgt-binding sites in mouse retina as shown by application of a conjugate of αBgt and horseradish peroxidase. In this study, labeled synaptic contacts were observed between amacrine cells and bipolar cells and at amacrine cell to amacrine cell synapses. No labeled contacts were described at amacrine to GC synapses, yet physiological data suggests that retinal GCs express either αBgt-sensitive or -insensitive nAChRs or both (Strang et al. 2005a). Thus, the identity of the retinal neurons that express solely αBgt-sensitive or both αBgt-sensitive and -insensitive nAChRs in mammalian retina and the subcellular localization of αBgt-sensitive nAChRs within the neurons are not clear. Furthermore, it remains to be determined whether these cholinoceptive neurons are the part of rod- and/or cone-driven retinal circuits. To address these questions, we examined the distribution of α7 nAChRs by means of single-, double-, and triple-label IHC using α7 subunit-specific antibodies together with antibodies against α3β2 nAChRs and with specific markers for specific types of bipolar, amacrine cells and for synaptic sites in rabbit retina. As a test of the specificity of the α7 nAChR antibody used, we also performed studies using fluorescently labeled αBgt together with the α7 antibody. Our observations indicate that α7 nAChRs are implicated in information processing in multiple retinal circuits.
Materials and Methods
Tissue Preparation
Eyes were obtained from adult Dutch-belted rabbits. Animal treatment was in accordance with NIH guidelines and the policies of the Animal Resources Program of the University of Alabama at Birmingham. Rabbits were sacrificed with an overdose of sodium pentobarbital (120 mg/kg). Eyes were removed, the anterior pole of each eye was cut away, and the vitreous was removed completely. For IHC, the retinas from five individual animals were used. Eyes were hemisected, and the posterior eyecups were fixed by immersion in 1% paraformaldehyde with 0.34%
For gel electrophoresis, retinas from four individual animals were removed from pigment epithelium. Protein from these retinas was analyzed by Western blots as described below.
Antibodies
Goat polyclonal antibody sc-1447 against the nAChR α7 subunit was purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Antibody sc-1447 was raised in goat against a peptide with the sequence WKFAACVVDRLCLMAFSVFT, mapping at the cytoplasmic loop between the third transmembrane domain (TM) and TM4 and partially into TM4 of the human α7 subunit (NCBI GenBank accession number P36544). Specificity of this antibody in rabbit retina was tested in competition studies with blocking peptide (Santa Cruz Biotechnology) and in Western blot analysis.
To evaluate expression of α7 nAChRs by different types of neurons, we incubated rabbit retina with antibody sc-1447 in conjunction with other commercially available antibodies against different neurochemical markers. These antibodies have been previously used in rabbit retina to identify specific subpopulations of retinal neurons. Rabbit anti-NR1 subunit of NMDA receptor (Chemicon; Temecula, CA) and mouse anti PSD-95 (Affinity Bioreagents; Golden, CO) have been used to localize postsynaptic density protein PSD-95 and NMDA receptors in rat and monkey retinas. Results for each of the markers used in this study were consistent with those previously reported. The reference for each of the markers, types of retinal neurons they have been reported to label, and the working dilutions of antibodies are listed in Table 1. All secondary antibodies were raised in donkey and specifically prepared for multiple-labeling studies (Jackson Immunoresearch Laboratories; West Grove, PA).
List of the antisera used to identify retinal cell populations in the present study
Peptide Neutralization Studies
Specificity of goat polyclonal antibody sc-1447 was tested by competing with an excess of antigen (blocking peptide, sc-1447 P; Santa Cruz Biotechnology) followed by indirect avidin-biotin IHC as previously described (Dmitrieva et al. 2001). According to the manufacturer's protocol, we determined that the highest antibody dilution at which a consistently positive staining of rabbit retina achieved was 1:1000. Antibody sc-1447 was diluted 1:1000 in 0.1 M saline PB containing 0.3% Triton X-100 (PBS/Triton) combined with peptide diluted 1:100 in the same buffer and rotated overnight at 4C (preabsorbed antibody solution). Cryostat sections of rabbit retina were washed in three changes of PBS and treated with 0.8% 2-mercaptoethanol (Bio-Rad; Hercules, CA) in PBS/Triton for 1 hr at room temperature (the rationale for this treatment is discussed in the IHC section). Tissue was washed in four changes of PBS and then preincubated for 1 hr at room temperature with 10% donkey normal serum in PBS/Triton. After preincubation, retinas were treated with avidin-biotin blocking kit (Vector Laboratories; Burlingame, CA) according to manufacturer's instructions and were then exposed to preabsorbed antibody solution overnight at 4C. Simultaneously, some of the sections were exposed to antibody sc-1447 diluted 1:1000 in PBS/Triton overnight. Tissue was next rinsed in three changes of PBS and incubated in biotinylated secondary donkey anti-goat IgG in PBS/Triton for 1 hr. Following incubation in secondary antibody, sections were washed three times in PBS and incubated in Vectastain Elite ABC solution (Vector Laboratories) in PBS/Triton for 1 hr. After rinsing, tissue was incubated in Metal Enhanced DAB Substrate working solution (Pierce Biotechnology; Rockford, IL) until easily visible staining was achieved. Sections were washed three times in PBS and coverslips were applied. Sections were examined with conventional light microscope equipped with differential interference contrast optics, and images were collected with Spot Camera (Diagnostic Instruments; Sterling Heights, MI).
SDS-PAGE and Western Blotting
In four experiments, isolated retinas from two eyes were homogenized in five volumes of ice-cold lysis buffer (PBS), pH 7.4, containing 1% NP-40, 0.5% sodium deoxycholate, 0.1% sodium dodecyl sulfate (SDS), and protease inhibitor cocktail (1:30; Sigma) and incubated at 4C for 30 min (Jones et al. 2001). Tissue homogenate was then centrifuged (15,000 x g for 20 min), the supernatant was retained and mixed with an equal amount of sample buffer and heated for 5 min at 95C. For gel electrophoresis, 15 μl of sample was loaded onto 10% SDS polyacrylamide gels and run at 100 V for 2 hr, after which the proteins were transferred to nitrocellulose membranes (Pierce Biotechnology). Nitrocellulose membranes were blocked with 7% non-fat milk (Bio-Rad) in PBS/0.1% Tween-20 (PBST) for 2 hr at room temperature, followed by incubation in goat antibody sc-1447 (1 μg/ml) in PBST /blocking solution overnight at 4C. After rinsing in PBS (3 × 10 min), the membrane was incubated in mouse anti-goat IgG (Jackson ImmunoResearch Laboratories) containing horseradish peroxidase diluted in PBST for 2 hr and washed three times. Immunoreactive bands were detected with a colorimetric detection Opti-4CN kit (Bio-Rad). Controls were performed by omitting the primary antibody from the procedure.
IHC
Indirect fluorescence was used to perform single, double, and triple labeling with different antibodies (see Table 1). Cryostat sections of rabbit retina were washed three times in PBS and then pretreated with 0.8% of 2-mercaptoethanol (Bio-Rad) in PBS/Triton solution for 1 hr at room temperature. We incorporated the mercaptoethanol treatment step to improve α7 recognition by sc-1447 antibody. 2-Mercaptoethanol reduces disulfide bonds in nAChRs (Watty et al. 1997), resulting in increasing the exposure of the antigenic epitope by either denaturation or dissociation of interacting proteins (Wilson and Bianchi 1999). It should be noted that mercaptoethanol retrieval of α7 antigenicity did not influence labeling patterns revealed by the other antibodies used in these studies. After the treatment, the tissue was rinsed at least four times in PBS, blocked in 10% donkey normal serum/PBS/Triton for 1 hr, and in avidin-biotin blocking solutions to eliminate nonspecific background binding. Sections were then incubated in primary antibody, sc-1447, diluted 1:50 in PBS/Triton for 24-48 hr at 4C. The tissue was next washed in three changes of PBS followed by incubation in secondary biotinylated donkey anti-goat IgG for 1 h. After incubation in the secondary antibodies, retinas were rinsed and incubated in streptavidin conjugated to Oregon Green 488 (green fluorescence; Molecular Probes, Eugene, OR) for 1 hr, rinsed, mounted in PermaFluor Aqueous mounting medium (Thermo Shandon; Pittsburgh, PA), and coverslipped.
In double- or triple-labeling IHC experiments, the tissue was exposed to a mixture of primary antibodies followed by a mixture of species-appropriate secondary antibodies made in donkey and tagged to Texas Red (red fluorescence) and to aminomethylcoumarin (AMCA, blue fluorescence). In doublelabeling experiments with goat sc-1447 and rat MAb210, the sections initially were stained for MAb210 with Texas Red and then for sc-1447 with Oregon Green 488. For goat sc-1447 and mouse anti-kinesin, the tissue was labeled first for α7 by goat sc-1447 (Oregon Green 488) followed by labeling mouse antibody against kinesin with Texas Red. Control procedures involved systematically omitting each of the primary and secondary antibodies.
As an independent test of the specificity of the α7 nAChR antibody used, we used Alexa-Fluor 594-conjugated αBgt in conjunction with sc-1447. This allowed us to compare the pattern of α-Bgt binding in the IPL with that of the anti-α7 antibody. The αBgt-binding protocol was modified from that of Pourcho (1979). Live dark-adapted retinas were incubated in oxygenated Ames medium at 4C containing 6.7 μM Alexa-Fluor 594 conjugated to αBgt (Invitrogen/Life Technologies; Gaithersburg, MD) for 2 hr. To prevent competitive binding by ACh released by the population of OFF starburst amacrine cells in dark conditions, synaptic transmission was blocked by the addition of 2 mM cobalt chloride to the medium during incubations. After incubation, the retina was washed for 1/2 hr in 4C oxygenated Ames medium followed by fixation in 1% PLP for 2 hr at room temperature. Following fixation, 200 μM vertical sections were cut and incubated in 0.8% 2-mercaptoethanol (Bio-Rad) in 0.1 M PBS/0.3% Triton for 4 hr at room temperature. Sections were blocked overnight at 4C with 10% donkey normal serum in 0.1 M PBS/0.3% Triton containing avidin blocking solution. Tissue was then incubated free floating with sc-1447 (1:200) in 0.1 M PBS/0.3% Triton containing 3% sodium azide and biotin blocking solution for 10 days at 4C. Incubation with the primary antibody was followed by four 30-min washes, incubation with biotinylated donkey anti-goat (1:200) for 2 days at 4C, four 30-min washes, and incubation with streptavidin-conjugated Alexa 488 (1:200) for 2 days at 4C. Some retinas were preincubated in 6.7 μM unlabeled αBgt in 4C oxygenated Ames medium for 20 min prior to incubation with the labeled αBgt, as described in the protocol above. This pretreatment abolished nearly all of the labeling, with only occasional flecks of fluorescence remaining (data not shown).
Sections were examined with a Leica TCS NT confocal laser scanning microscope (Leica Microsystems; Mannheim, Germany) using argon and krypton lasers. Each channel was scanned separately, and the images were saved as digital graphics files. Other sections were viewed with a Zeiss Axioplan 2 Imaging microscope (Göttingen, Germany) and images were collected with a Zeiss AxioCam MRm camera.
Cytofluorochrome masking as described in Strang et al. (2003) was used in some cases to provide an objective assessment of colocalization. Briefly, using the Leica software, a frequency distribution of the pixel colors and intensities, each 2-channels, was generated for optical sections. Pixels that represented simultaneous excitation of both channels (yellow) fell in the center of the frequency distribution. The simultaneous fluorescence of two fluorophores within one pixel was defined as indicative of colocalization and masked back onto the image as white pixels.
Brightness and contrast adjustments and figure preparation were completed with Adobe Photoshop (Adobe Systems; San Jose, CA).
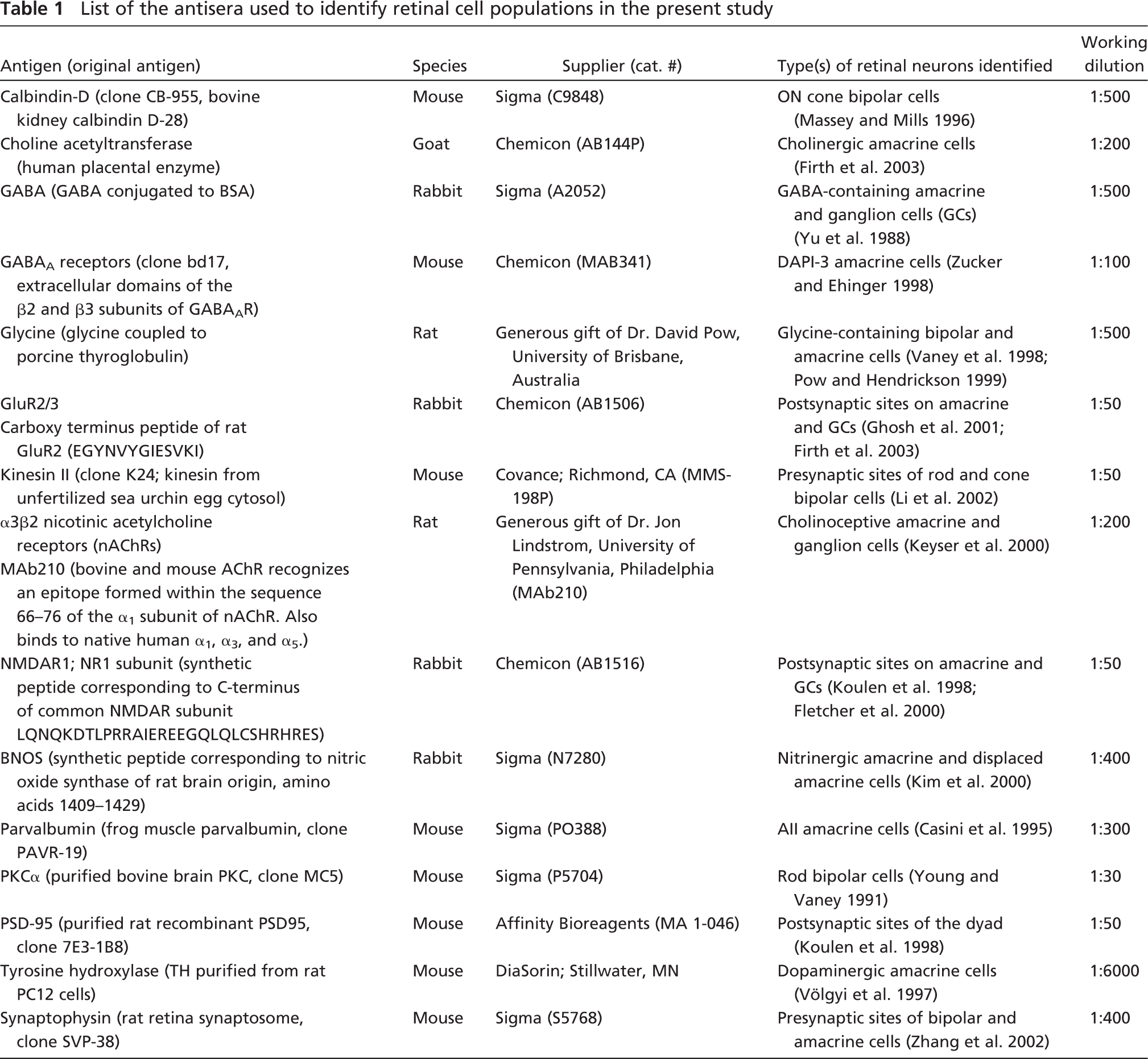
Results
Specificity of the Antibody
Goat polyclonal antibody sc-1447 used in this study was raised against a 20-amino acid peptide from the extracellular carboxy terminal domain of human nAChR α7 subunit. This region is identical to the corresponding rat sequence. Anti-α7 polyclonal antibody (sc-1447) has been used previously for immunostaining α7 nAChRs in transfected cells (line QT6) (Kassner et al. 1998), in rat hippocampal neurons (Kawai et al. 2002), and in chick spinal cord neurons (Roth and Berg 2003). In this latter study, specificity of sc-1447 was confirmed by colabeling with a previously characterized anti-α7 antibody, MAb306 (Roth and Berg 2003). In Western blot analysis of proteins extracted from the hippocampal formation, MAb306 recognizes a band of ∼56 kDa (Fabian-Fine et al. 2001). A band of the same size was recognized by sc-1447 on Western blotting of rabbit retina, further demonstrating that sc-1447 and MAb306 bind to the same protein (Figure 1). The labeled band was eliminated when the primary antibody was omitted from the protocol. In our hands, IHC using the anti-α7 antibody yielded a specific and repeatable pattern of labeling in the different layers of rabbit retina (Figure 2). Incubation of retinal tissue with sc-1447 preabsorbed with the corresponding 20-amino acid synthetic peptide used for immunization abolished staining in rabbit retinal neurons (not shown).
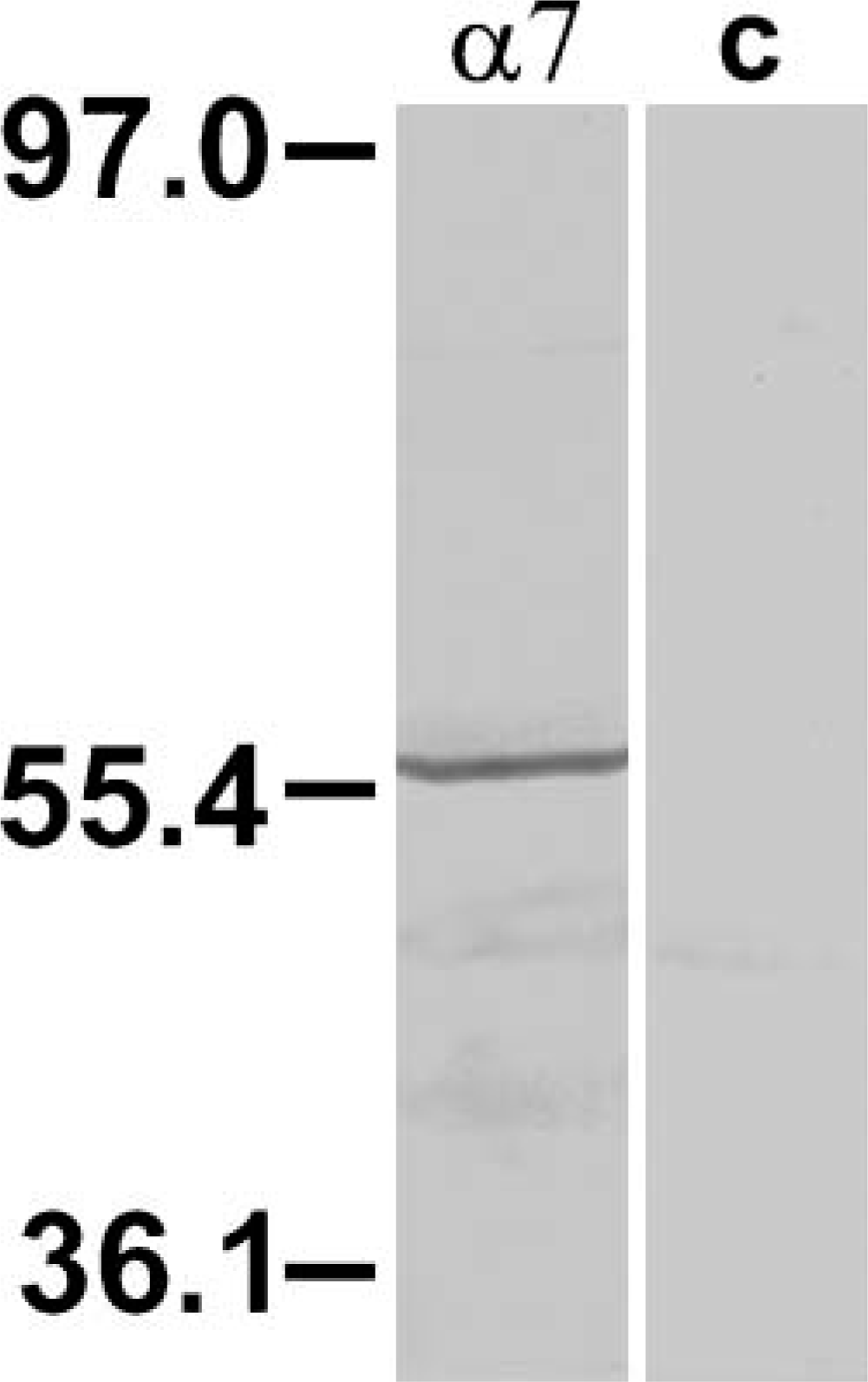
Western blot analysis of rabbit retina probed with sc-1447 antisera. The sc-1447 antisera labeled a 56-kDa band corresponding to the α7 nicotinic acetylcholine receptor (nAChR) subunit.
Distribution of α7 nAChRs in Rabbit Retinal Neurons and Their Processes as Revealed by IHC and αBgt Binding
Immunoreactivity to the α7 antibody sc-1447 was observed in four layers of rabbit retina. Immunoreactivity in the somata and processes of neurons located in the INL and in the GCL contributed to sparse staining in the OPL and to more intense diffuse staining in the IPL (Figure 2). In the INL, ∼49% of the immunoreactive cells exhibited the characteristic appearance of bipolar cells. Specifically, the somata were adjacent to the OPL, the dendrites could be followed into the OPL, and the axons descended directly into the IPL (Figure 2).
Somata of other labeled cells (∼32%), presumably amacrine cells, were found in the inner and in the middle or upper middle part of the INL. The presumptive amacrine cell dendrites could be traced into the IPL until they joined diffuse bands of labeled processes that ramified throughout the IPL (Figures 22-B2). There appeared to be at least two different types of amacrine cells labeled by sc-1447: those that had a single primary dendrite that ramified in two or more levels of the IPL after branching (Figures 2B and 2C) and presumptive “flag cells” (MacNeil and Masland 1998) that sent a single oblique process with thin branching dendrites into the middle of the IPL (Figures 2D and 2E).
There were fewer labeled cells in the GCL. Immunoreactive GCs comprised 12.5% of the total number of immunoreactive cells. In some instances, dendrites of intensely labeled neurons in the GCL could be traced to the inner margin of the IPL (Figure 2). However, because of diffuse labeling throughout the IPL, we were not able to systematically discern the arborization patterns of individual cells in the GCL. Occasionally, labeled cells in the GCL could be seen to give rise to labeled axons and were therefore identifiable as GCs.
It is worth noting that the intensity of α7 labeling was very low and almost undetectable in the retinal neurons in the area of optic nerve head. This pattern was consistent across retinas and reflects regional differences in the expression of α7 nAChRs in rabbit retina similar to the expression patterns of β2-containing nAChRs (Keyser et al. 2000). Alternatively, the reduced labeling may be an artifact arising from poor penetration of the fixative or other reagents into this part of the retina. However, distribution of other cell-specific markers did not appear to be comparably affected.
As an independent test of the specificity of the sc-1447 labeling in the IPL, we carried out studies using Alexa-Fluor 594-tagged αBgt. This method yielded consistent punctate labeling on dendrites within the IPL (Figures 3A and 3B). Incubation of retinas with unlabeled αBgt prior to exposure to the Alexa-tagged αBgt abolished nearly all of the labeling, with only occasional flecks of fluorescence remaining (not shown). Doublelabeling studies with the α7 antibody (Figures 3C and 3D) and the labeled αBgt revealed an excellent, but not 1:1, correspondence between puncta labeled by the two methods (Figures 33-E3). There was consistently more antibody labeling than αBgt binding. The imperfect correspondence between the αBgt binding and sc-1447 labeling is addressed in Discussion.
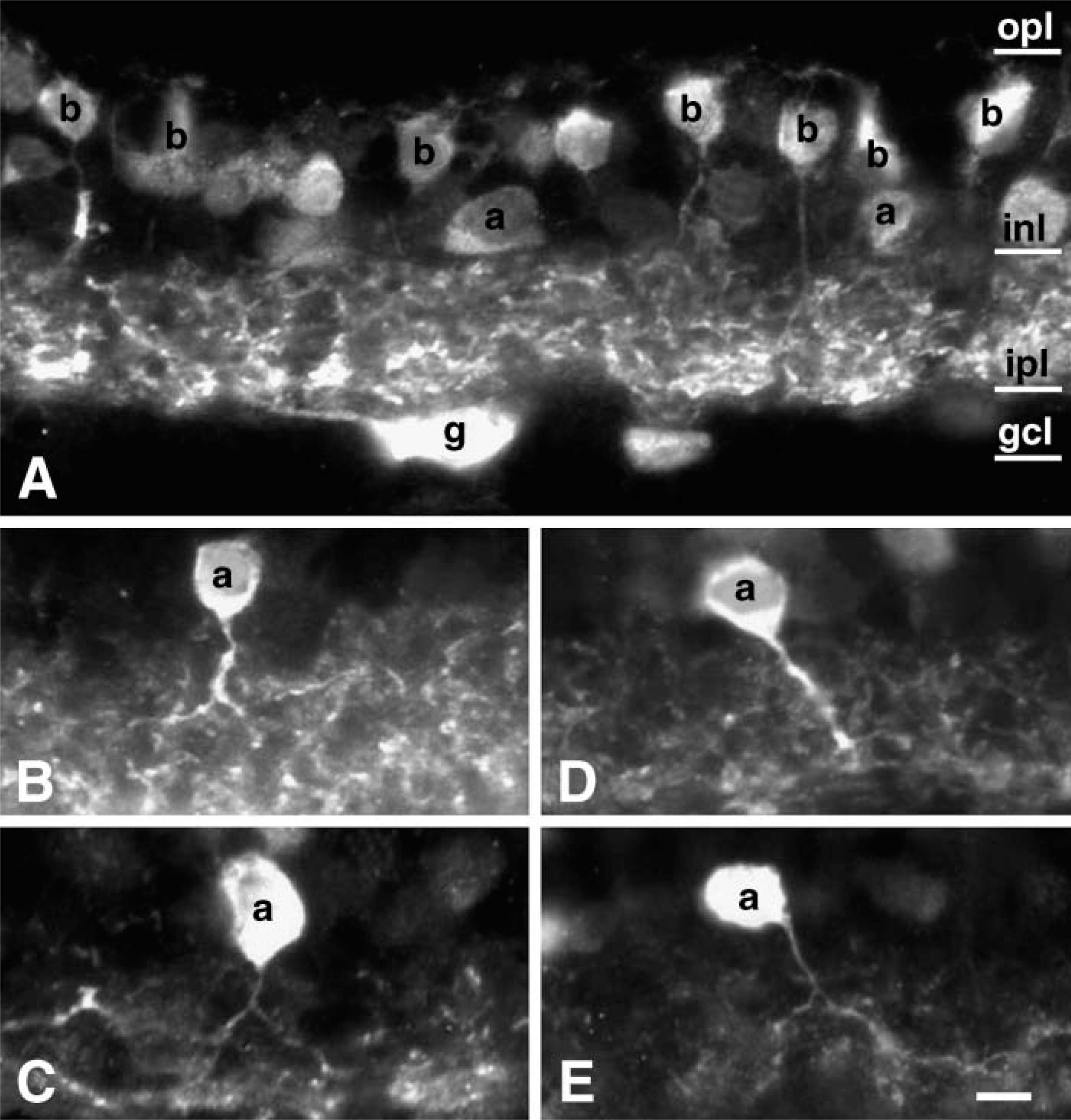
Microscopic images of vertical sections of rabbit retina labeled with goat antibody against α7 nAChRs. Staining was observed in the somata of bipolar cells (b), amacrine cells (a), and ganglion (g) cells and in their dendrites in the OPL and in the IPL (
Expression of α7 nAChRs by Different Subpopulations of Bipolar Cells and Amacrine Cells
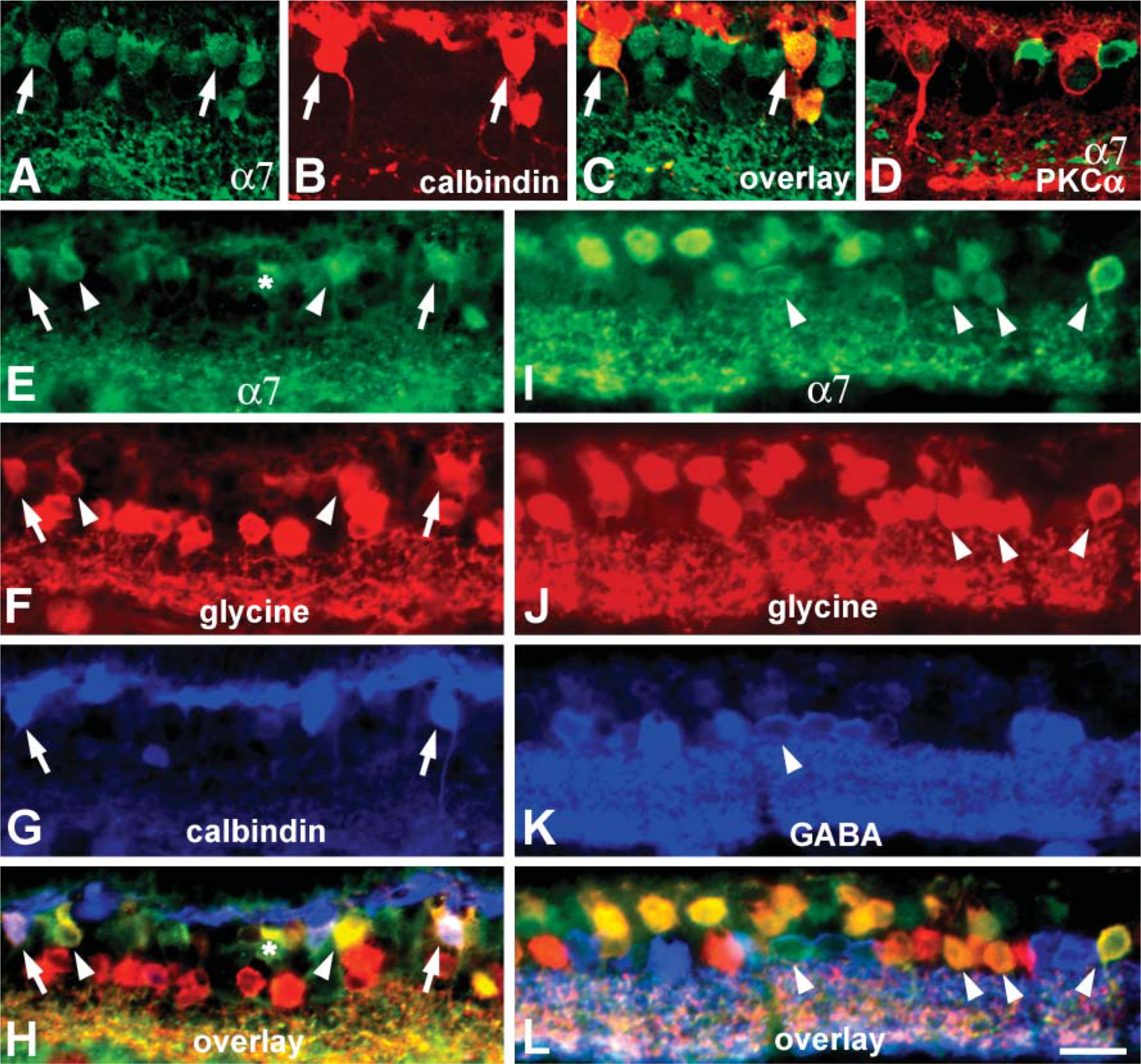
To investigate expression of α7 nAChRs by neurons in different information processing pathways, we carried out double- and triple-labeling studies with sc-1447 and antibodies that label specific populations of bipolar and amacrine cells in rabbit retina. In mammalian retina, the vertical pathways are subdivided into rod and cone pathways used for vision in dark- and light-adapted conditions, respectively. Rods and cones provide input to one type of rod bipolar cell and several subtypes of cone bipolar cells, respectively (Boycott and Wässle 1999; McGillem and Dacheux 2001). Antibodies against calbindin, a member of the family of calcium-binding proteins, prominently label ON-type cone bipolar cells in rabbit retina (Massey and Mills 1996). Simultaneous visualization of sc-1447 and calbindin immunoreactivity revealed that all calbindinpositive bipolar cells expressed α7 nAChRs, but not every α7 immunoreactive bipolar cell contained calbindin (Figures 44-A4).
Approximately half of the cone bipolar cells in rabbit retina contain elevated levels of the inhibitory transmitter glycine due to the coupling of bipolar cells through gap junctions with glycinergic amacrine cells (Vaney et al. 1998). Triple-labeling experiments with antibodies against glycine, calbindin, and α7 resulted in further differentiation of the sc-1447-positive bipolar cells. Some of the α7-expressing bipolar cells were labeled for calbindin and glycine, others exhibited only glycine immunoreactivity, and the third subset of α7-containing bipolar cells was negative for calbindin and glycine staining (Figures 44-E4).
Rod bipolar cells in rabbit retina are intensely immunoreactive for PKCα (Young and Vaney 1991). Somata of rod bipolar cells are located in the outmost portion of the INL, give rise to several primary dendrites that branch in the OPL, and their axons descend unbranched through the whole depth of the IPL and arborize as multiple lobes in the innermost part of the IPL (Figure 4D). Double labeling of vertical sections with antibodies against PKCα and α7 nAChRs showed no colocalization of the two markers, indicating that α7 nAChRs are not likely to be involved in the modulation of the rod pathway at the level of the first synapse. Thus, only cone but not rod bipolar cells were found to be cholinoceptive, and there were at least three different subsets of α7-expressing cone bipolar cells.
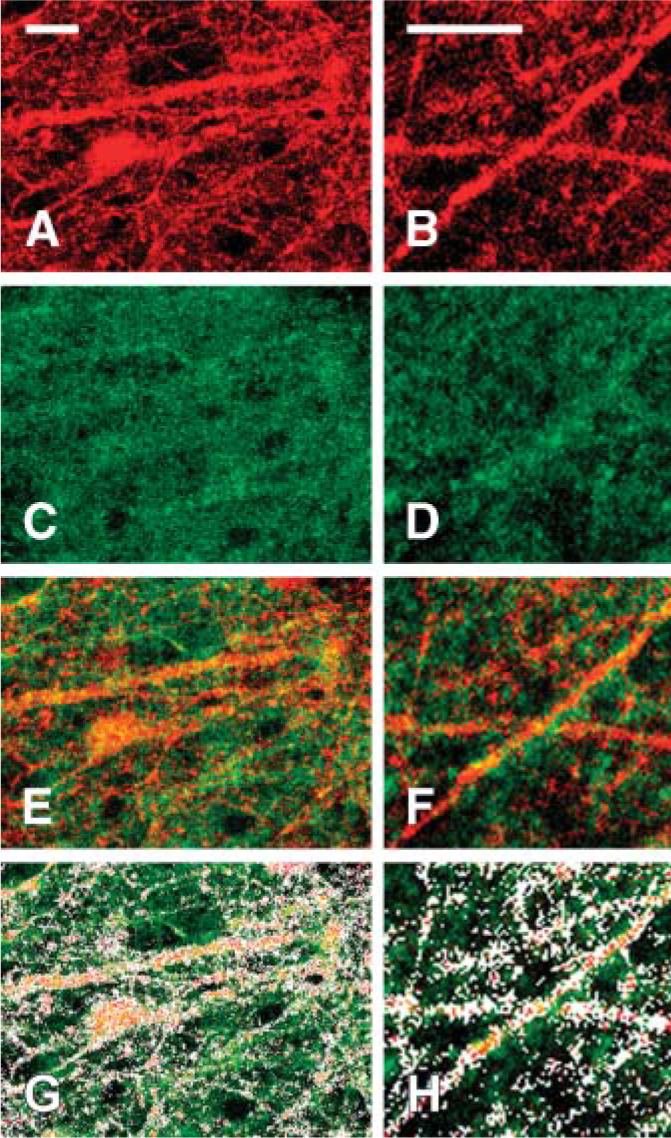
Single optical confocal sections showing Alexa-Fluor 594-conjugated α-bungarotoxin (αBgt) binding (
In mammalian retina including rabbit retina, ∼50% of cholinoceptive amacrine cells are glycinergic (Dmitrieva et al. 2003), whereas >35% contain GABA (Dmitrieva et al. 2001). To test whether or not α7-positive amacrine cells contain glycine or GABA, we performed triple-label IHC. Our results demonstrated that the majority of α7-expressing cholinoceptive amacrine cells displayed glycine immunoreactivity. Only a small population of amacrine cells were double labeled for α7 and GABA (Figures 44-I4).
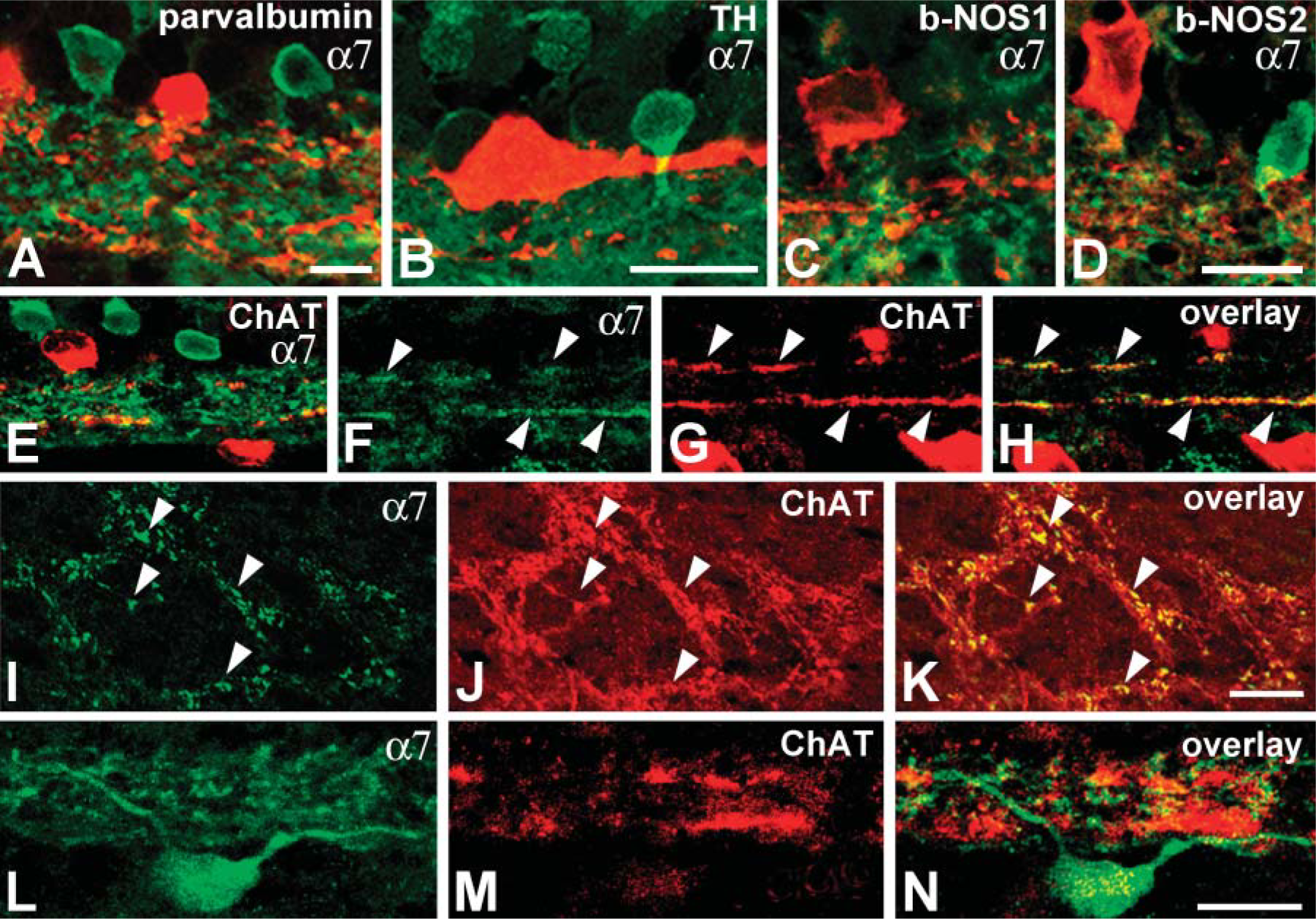
Among the 26 distinct morphological types of amacrine cells in rabbit retina, several types of glycine- and GABA-containing cells can be selectively labeled with specific antibodies (Table 1). Glycinergic AII amacrine cells that serve as a conduit for transferring the rod signal through the IPL are immunoreactive for parvalbumin, a member of a family of calcium-binding proteins (Casini et al. 1995; Bloomfield and Dacheux 2001). Dopaminergic amacrine (interplexiform) cells modulate AII-AII cell coupling under different light adaptation conditions (Bloomfield and Dacheux 2001) and can be labeled with antibodies against tyrosine hydroxylase (TH) (Brecha et al. 1984; Völgyi et al. 1997). GABA-containing cholinergic and nitrinergic amacrine cells and the glycinergic DAPI-3-positive amacrine cells in rabbit retina collect information from cone bipolar cells and other amacrine cells. These amacrine cells arborize with the dendrites of different types of amacrine and GCs (Brecha et al. 1988; Famiglietti 1991; Wright et al. 1997; Kim et al. 2000) and can also be selectively labeled. Cholinergic amacrine cells have been identified by immunostaining for choline acetyltransferase (ChAT) (Brandon 1987); nitrinergic cells have been consistently labeled with antisera against neuronal nitric oxide synthase (NOS) (Kim et al. 2000), and antibodies against the β2/3 subunit of GABAA receptor prominently label DAPI-3 cells (Zucker and Ehinger 1998).
The α7 receptor labeling was never detected in somata and processes that exhibited parvalbumin, TH, or NOS immunoreactivity (Figures 55-A5), further indicating that α7 nAChRs have the potential to modulate information transfer along cone, but not rod, pathways. Somata of ChAT-positive cells were also sc-1447 negative, but the processes of cholinergic cells in the IPL appeared to be labeled with anti-α7 antibody (Figure 5E). Colocalization of ChAT and α7 in the processes could easily be seen in the IPL above optic nerve head where reduced α7 labeling of processes and somata was observed (Figures 55-I5).
In addition, double-label experiments with ChAT and α7 revealed that the dendrites of some α7-labeled GCs arborized immediately adjacent to the ON and OFF ChAT-immunoreactive bands in the IPL, stratification consistent with that of ON-OFF bistratified GCs (Figures 55-L5).
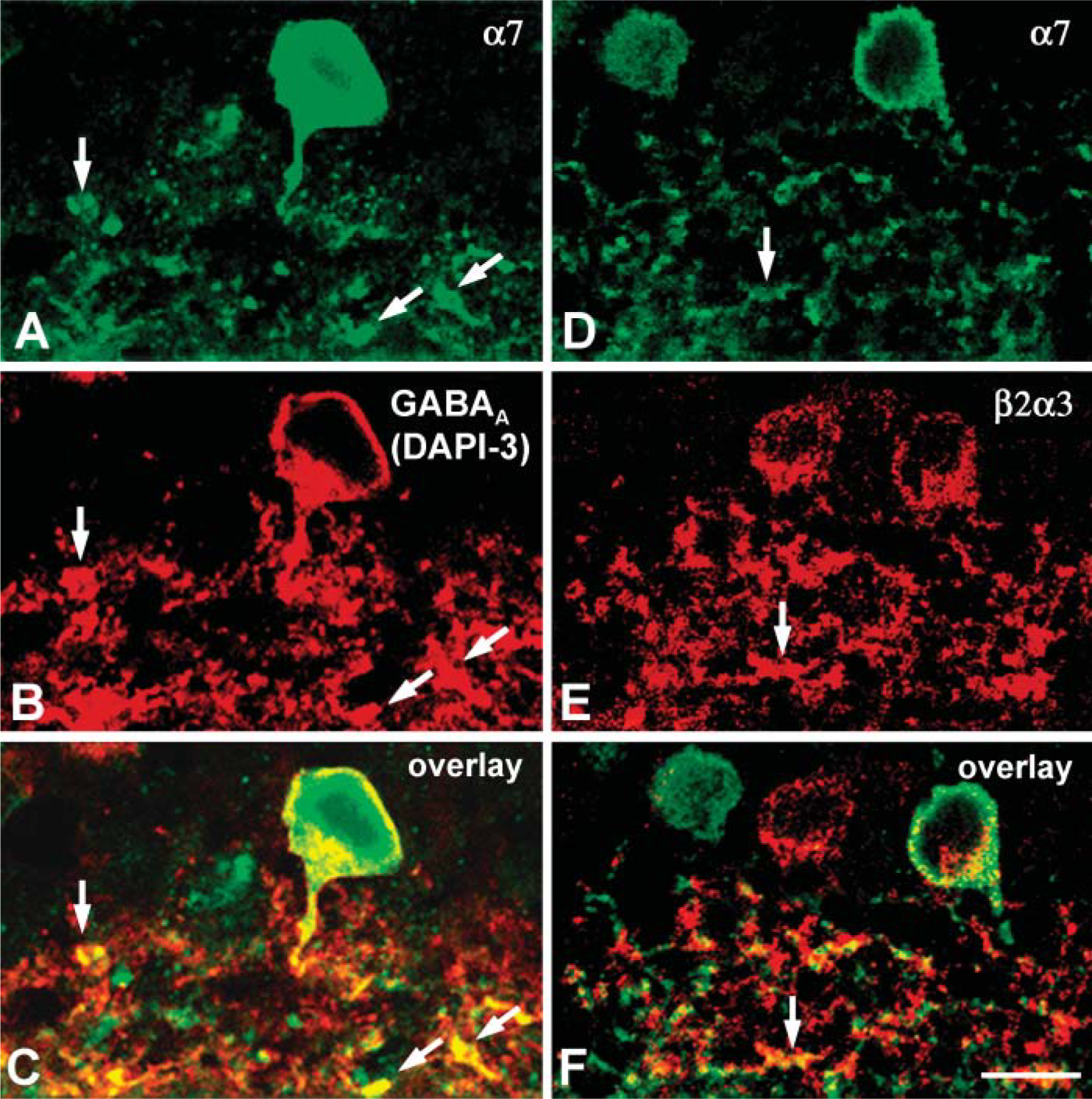
Somata of glycinergic DAPI-3 cells in the INL, as well as many sites on their dendritic arborizations in the IPL, showed colocalization of GABAA and α7 immunoreactivity (Figures 66-A6). This might reflect the clustering of α7 nAChRs and GABAA receptors on the same or adjacent membrane domains of DAPI-3 cells.
Colocalization of α7 and α3β2 nAChRs in Rabbit Retinal Neurons
Expression of α7 nAChRs by amacrine cells and some cells in the GCL of rabbit retina raised the question whether they colocalize with β2-containing nAChRs present in subsets of amacrine and GCs as well (Keyser et al. 2000; Dmitrieva et al. 2001,2003). Simultaneous visualization α3β2 nAChRs labeled by MAb210, and α7 nAChRs revealed that although some cell bodies in the INL and the GCL were positively labeled by both antibodies, dendritic varicosities in the IPL showed no apparent colocalization of two markers, with only a few exceptions (Figures 66-D6). Thus, different subsets of retinal amacrine cells and GCs can express homomeric α7 or heteromeric α3β2 nAChRs or both. In cases where both receptor subtypes were expressed in the same retinal neurons, they were often in juxtaposition with each other but did not appear to occupy the same spot on the membranes of dendrites.
Images of double- and triple-labeled vertical sections of rabbit retina using sc-1447 antiserum (green) and four different markers: anti-calbindin (red in
Subcellular Distribution of α7 nAChRs
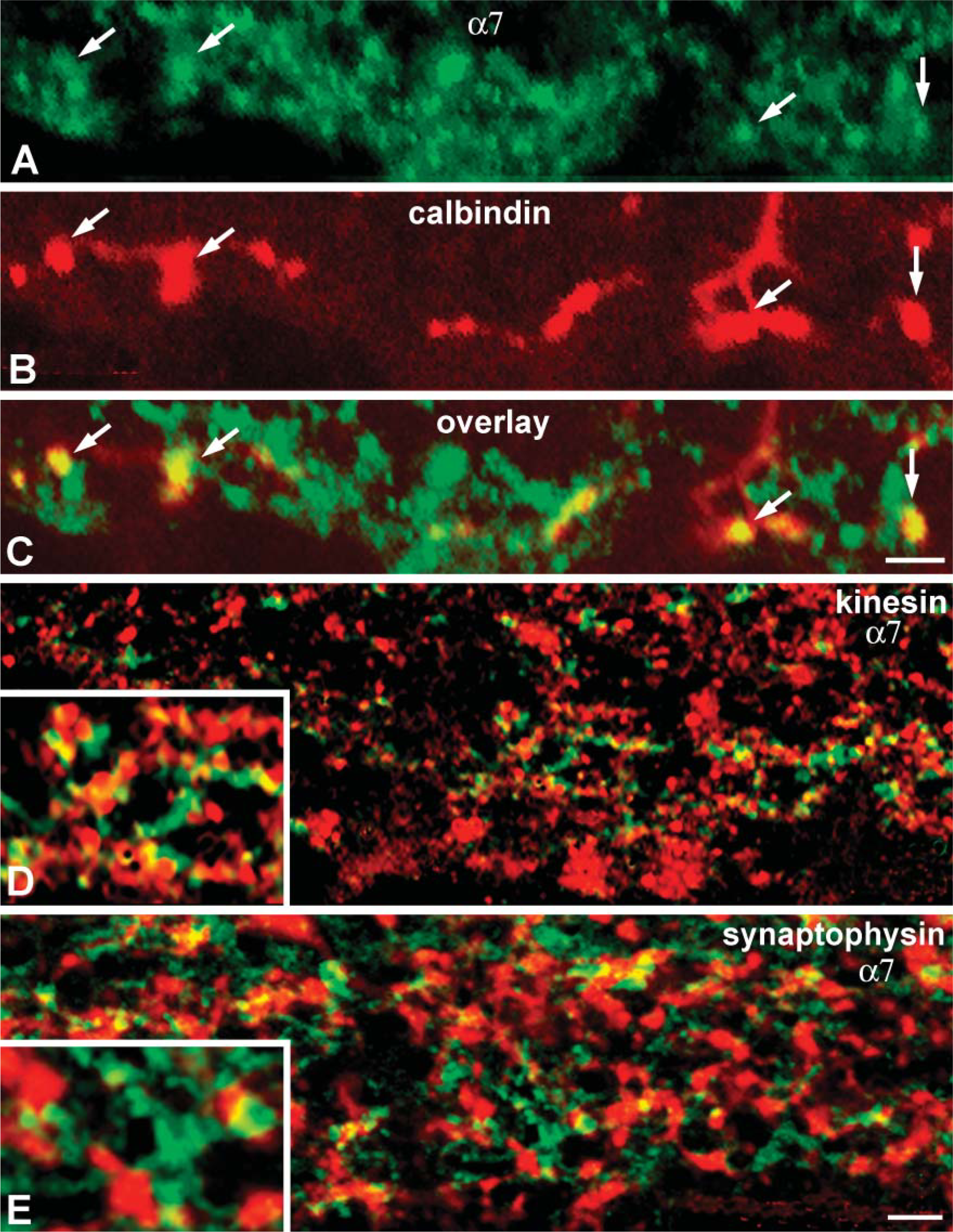
The functions of nAChRs rely on their channel properties and cellular and subcellular distribution (Sargent 1993; Le Novére et al. 2002; Lindstrom 2002). Previous studies have shown that in brain, α7-containing receptors can be found in diverse locations both at synaptic and non-synaptic sites (e.g., Berg and Conroy 2002). In rabbit retina, expression of α7 receptors clearly coincided with the axonal arborizations of calbindinpositive bipolar terminals at strata 4 (Figures 77-A7). These terminals make ribbon synapses to dyads of postsynaptic dendrites that belong to either ganglion or amacrine cells (Strettoi et al. 1994). Thus, our data imply that α7 nAChRs are expressed by presynaptic elements at ribbon synapses of calbindin-positive bipolar cells.
Double labeling of selective types of amacrine (red) and ganglion cells (GCs) with sc-1447 antibody (green) in rabbit retina. α7 Immunoreactivity was not detectable in the somata and dendrites of AII amacrine cells (
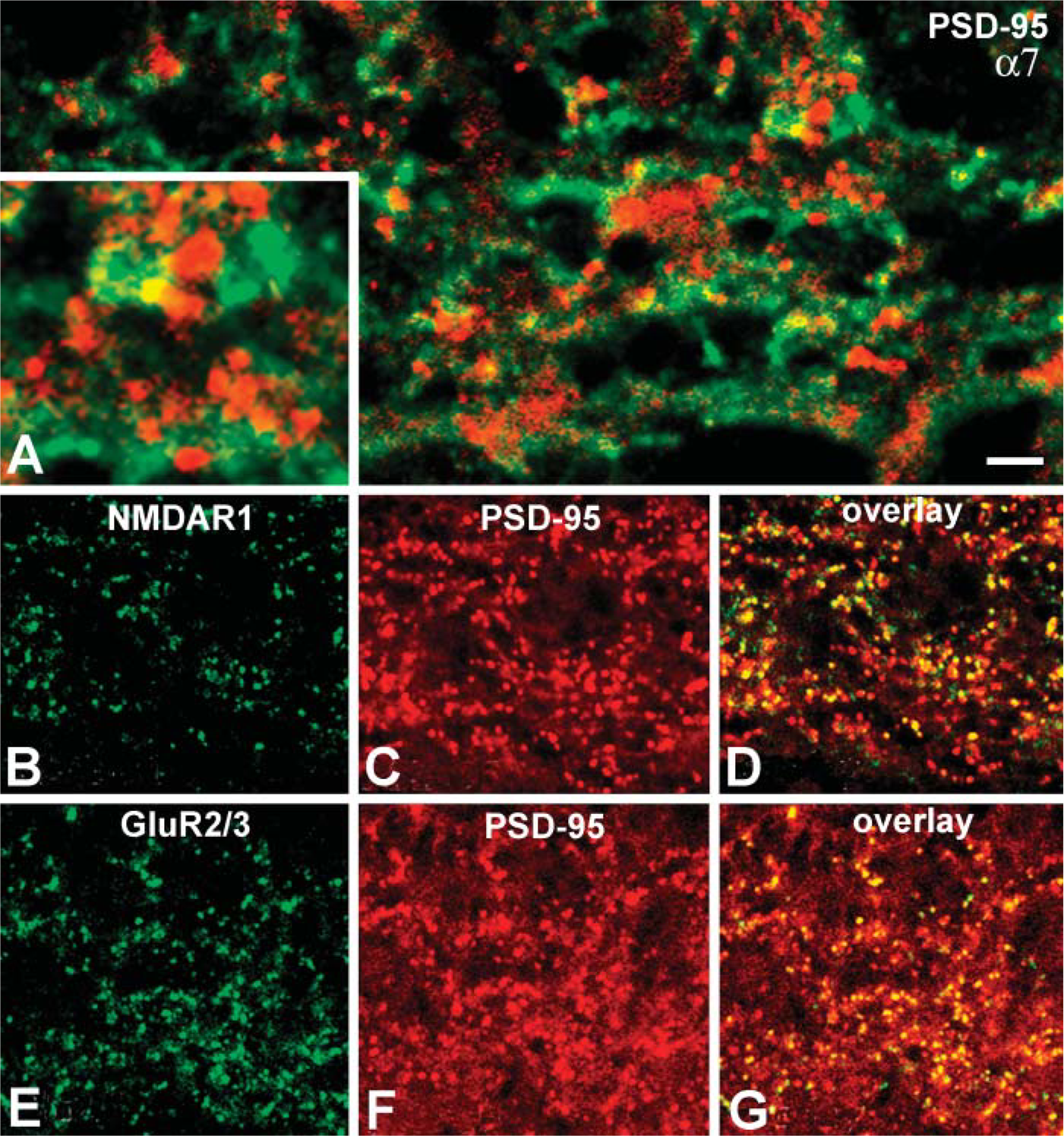
As previously noted, at least three subpopulations of bipolar cells and different types of amacrine cells were labeled by sc-1447. To determine whether α7 nAChR clustering on the processes of the other cone bipolar cell types, amacrine cells, and/or GCs is associated with presynaptic or/and postsynaptic elements in the IPL, we performed double-labeling experiments with sc-1447 and antibodies against kinesin II, synaptophysin, and PSD-95. In rabbit retina, anti-kinesin II has been shown to bind selectively to presynaptic ribbons in photoreceptors and bipolar cells (Li et al. 2002), and antisynaptophysin labels presynaptic sites of bipolar and amacrine cells in the IPL (Zhang et al. 2002). The third protein, anti-PSD-95, is a member of a family of glutamate receptor (GluR)-clustering molecules, presumably associated with postsynaptic elements of glutamatergic synapses in rat and monkey retina. The majority of NMDA receptors and all AMPA, GluR2/3 receptor subunits have been reported to be colocalized with PSD-95 (Fletcher et al. 2000; Kennedy 1997; Ghosh et al. 2001).
Consistent with the localization of α7 nAChRs on calbindin-positive bipolar cells, many yellow-orange patches, indicating simultaneous fluorescence captured by the red and green channels, were visible in different strata of the IPL in vertical sections of rabbit retina processed either for α7 and kinesin (Figure 7D) or for α7and synaptophysin (Figure 7E). Higher magnification areas (inset panels) revealed colocalization and/or close apposition of labeled sites. This pattern of association could mean that many α7 nAChRs are expressed at presynaptic elements of different bipolar cell (kinesin positive) synapses, as well as at presynaptic (synaptophysin positive) elements of amacrine cell synapses.
In rabbit retina, PSD-95 immunoreactive clusters coincided with punctate labeling of NMDR1 receptors and with all Glu2/3 receptor puncta in the IPL (Figures 8B-8D, 8E-8G). These results are identical to patterns of association shown to represent the postsynaptic aggregations of GluRs at glutamatergic synapses on the dendrites of amacrine and GCs in rat and monkey retina (Fletcher et al. 2000; Ghosh et al. 2001). Therefore, we considered the anti-PSD-95 stained clusters in the IPL as a reliable marker for postsynaptic densities in rabbit retina. In contrast to the results with markers for presynaptic elements, double-labeling studies with sc-1447 and PSD-95 detected little apparent colocalization between α7 nAChRs and postsynaptic densities. Rather, PSD-95 immunoreactivity appeared to be adjacent to α7-contining profiles (Figure 8). In total, the results with pre- and postsynaptic markers indicate that α7 nAChRs are likely to be expressed at the presynaptic terminals of bipolar and amacrine cells but are less likely to be expressed by the postsynaptic members at bipolar cell synapses.
Confocal microscopic images of two vertical sections of rabbit retina. One section was exposed to both sc-1447 (green) and antisera against GABAA receptors (red). Somata of DAPI-3 amacrine cells (red) in the INL and some sites on their dendritic arborizations in the IPL were labeled with anti-α7 antisera (arrows in
Discussion
Specificity of the sc-1447 antibody was confirmed in several ways. First, Western blotting revealed a band of ∼56 kDa, the expected molecular mass for mammalian α7 subunits. Second, sc-1447 labeling was abolished by preincubation with the blocking peptide supplied by the vendor. Third, we demonstrated with double-label experiments using Alexa-Fluor 594-conjugated αBgt in conjunction with sc-1447 that there is good, but not 1:1 correspondence between labeled Alexa-Fluor 594-αBgt binding and sc-1447 labeling in the IPL. The difference is likely due to differences in the probes themselves. Reliable αBgt binding requires incubation with live retinas, which should yield binding only to surface receptors. For the sc-1447 studies, the tissue must be fixed and permeabilized with detergent because the epitope spans part of one of the transmembrane domains and continues into a portion of the intracellular sequence. Thus, in contrast to the surface αBgt binding, the sc-1447 labeling includes newly synthesized receptors that are in transit to membrane insertion sites, as well as receptors that are cycling out of the membrane. Consistent with this possibility, sc-1447 labeling was more extensive and less punctate than αBgt labeling.
α7 nAChR Involvement in the Rod and Cone Pathways and in On-Off Circuitry
These results demonstrate, for the first time, that α7 nAChRs are expressed by classes of bipolar and amacrine cells in the INL, in addition to GCs in the mammalian retina. Expression of α7 nAChRs by bipolar and amacrine cells indicates that the activation of this subtype of nAChRs in rabbit retina might significantly influence the processing of visual information by upstream neurons, as well as affecting processing by GCs.
Confocal images of three vertical sections of rabbit retina at the level of the IPL. Section shown in
Confocal images of transverse sections of rabbit retina at the level of the IPL. (
The α7-containing bipolar and GCs transmit signals from photoreceptors forward to the brain. The α7-expressing amacrine cells may modify forward transmission by releasing glycine or GABA in response to stimulation by ACh or choline. Our imunohistochemical data are consistent with results from extracellular recordings in rabbit retina that suggest the expression of functional α7 nAChRs by GCs and upstream cells as well (Reed et al. 2002; Strang et al. 2005a). Figure 9 provides a summary of the different cell types that were immunoreactive to antibodies against α7 nAChRs.
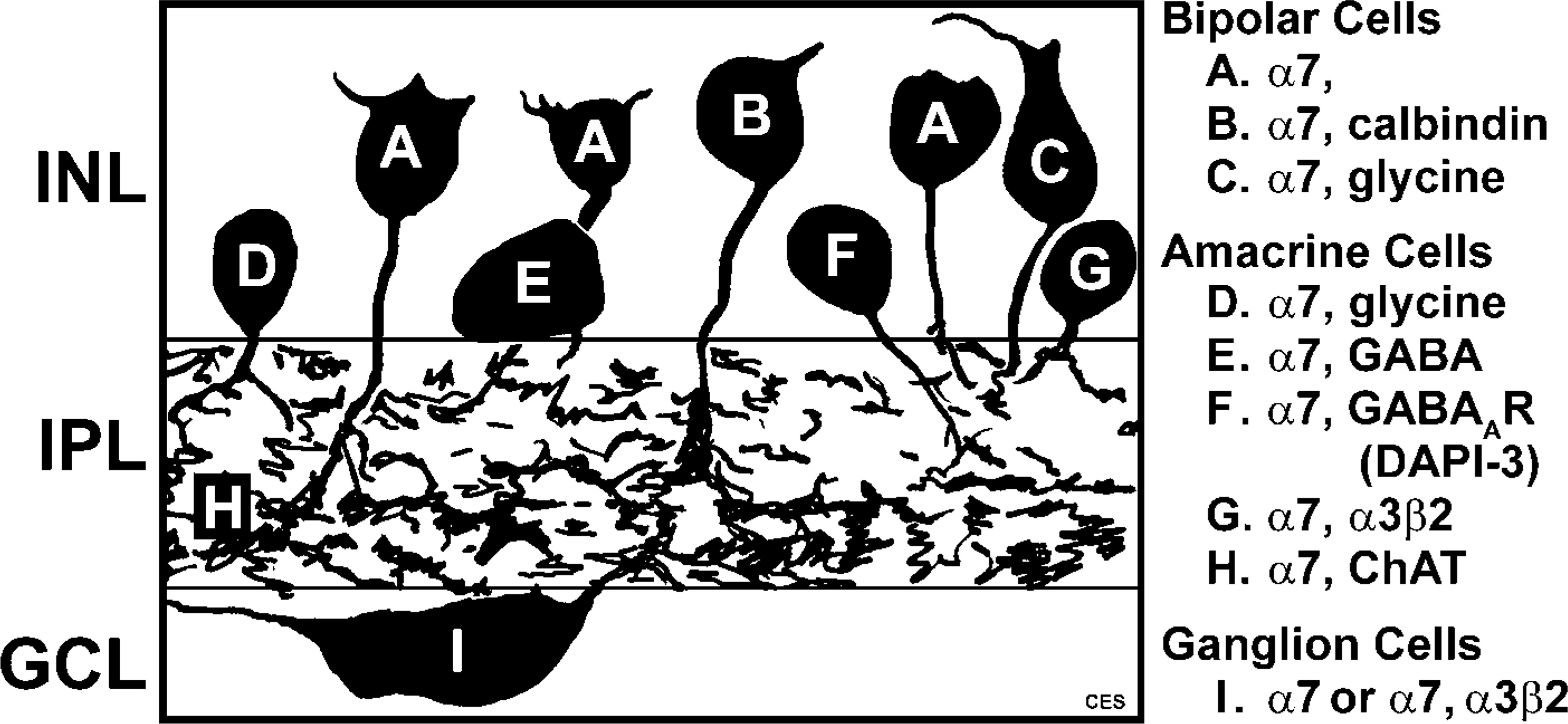
Diagram summarizing the α7-expressing cell types in the rabbit retina. Letters on the drawing correspond to the description to the right of the image.
The retina includes separate circuits that serve rod scotopic (low- and medium-light levels) and cone photopic (high-light levels) vision. Rods and cones synapse onto one type of rod bipolar cell and many types of cone bipolar cells, respectively, and different types of cone bipolar cells make synaptic contacts with selected subtypes of GCs. In contrast, rod bipolar cells connect to GCs only indirectly via connections from AII amacrine cells that synapse onto cone bipolar cells. In addition, the rod signal can be transferred to cones through gap junctions and reach GCs through cone bipolar cells (for review, see Bloomfield and Dacheux 2001; Kolb 2003).
Our data suggest that the dedicated rod pathway neurons, rod bipolar cells, and AII amacrine cells do not express α7 nAChRs, nor do the dopaminergic and nitrinergic amacrine cells that modulate the activity of the AII amacrine cells. In contrast, many cone bipolar cells exhibited α7 immunoreactivity, including calbindinpositive cone bipolar cells. Interestingly, the calbindinpositive cone bipolar cells can receive input from AII amacrine cells and transmit both rod- and cone-mediated signals to ON-type GCs (Strettoi et al. 1994; Massey and Mills 1996). These data suggest that the release of ACh affects cone signals directly but may also affect rod signals downstream of the dedicated rod pathway.
α7 nAChR Involvement in ON and OFF Circuitry
To perceive contrast between light and shadow under photopic conditions, parallel sets of visual channels that process ON and OFF qualities are present in mammalian retina (reviewed by Kolb 2003). In the rabbit retina, there are six distinct types of ON cone bipolar cells that connect to ON GCs in sublamina b of the IPL and six distinct types of OFF cone bipolar cells that connect to OFF GCs in sublamina a of the IPL (McGillem and Dacheux 2001). In our study, numerous α7-positive cone bipolar cells were also labeled with anti-glycine antibodies, and they may comprise four or five common types of ON of cone bipolar cells (Vaney et al. 1998). At least two types of ON cone bipolar cells—calbindin/glycine positive and calbindin negative/glycine positive—exhibited α7 immunoreactivity. The third type of α7 immunoreactive cone bipolar cells, which displayed neither calbindin nor glycine immunoreactivity, remains to be identified. These results indicate that primarily the ON cone bipolar pathway is influenced by α7 nAChRs.
Amacrine cells comprise the second major class of retinal neurons in rabbit retina that displayed α7 immunoreactivity. Our finding that most of the α7-positive amacrine cells contained glycine and that a smaller subset of α7-positive amacrine cells were GABAergic strikingly resembled the expression patterns of α3β2 subtype of nAChRs. Specifically, α3β2-expressing glycinergic amacrine cells were found to make up the majority of cholinoceptive amacrine cells in rabbit retina (Dmitrieva et al. 2003). However, the patterns of α7 and α3β2 immunoreactivity were not identical; some amacrine cells expressed both types of nAChRs (DAPI-3 glycinergic cells), whereas others, such as the putative “flag” B glycinergic cells, were positive for α7 alone.
Glycinergic amacrine cells are usually “small field” cells and make more synapses onto amacrine and ganglion than onto bipolar cells. Their processes can span several strata within the IPL, but they are more compact in the plane parallel to the vitreal surface (Pourcho and Goebel 1985; MacNeil and Masland 1998; Menger et al. 1998; Kolb 2003). Different types of ON and OFF GCs in rabbit retina have been shown to express functional glycine receptors that mediate direct inhibitory input onto the GCs (Rotolo and Dacheux 2003a,b). One might predict that the release of ACh and its activation of αBgt-insensitive and -sensitive nAChRs on glycinergic cells preferentially control the activity of lateral circuits that feedback to other amacrine cells rather than information transfer to bipolar or GCs. However, organization of these local circuits is not well enough understood to speculate further.
α7-Immunoreactive cells in the GCL constituted the numerically smallest population of cholinoceptive neurons in rabbit retina. α7-Immunoreactive neurons in the GCL represented two distinct types: some GCL cells expressed α7 and α3β2 nAChRs and others exhibited only α7 immunoreactivity. The apparent heterogeneity of nAChR expression by neurons in the GCL as judged by IHC studies is consistent with the observed pharmacological diversity of GCs in rabbit retina. Many physiologically identified GC types, including subsets of sustained OFF and sustained ON, transient OFF (Strang et al. 2005a), and some directionally selective ON-OFF cells may express β2-containing and α7 nAChRs (Reed et al. 2002; Strang et al. 2005b). Responses to cholinergic agonists of other subsets of GCs were mediated solely by α7 nAChRs (Strang et al. 2005a). Our data, together with the physiological studies cited above, suggest that ACh can directly affect the responses of GCs through the activation of α7 nAChRs and/or β2-containing nAChRs on the GCs themselves, as well as on the upstream cells.
Putative Role of α7 nAChRs in Visual Processing
In the central nervous system, nAChRs mediate fast synaptic transmission only on rare occasions. More commonly, they modulate the release of neurotransmitters such as glutamate and GABA (Radcliffe et al. 1999). Light and electron microscopic studies have shown a “mismatch” between the locations of ACh release sites and the location of corresponding high-affinity receptors, providing anatomic evidence for a modulatory role (Descarries et al. 1997; Herkenham 1999). We observed a similar “mismatch” in the rabbit retina. There are reports that cholinergic cells in rabbit retina make synaptic outputs only onto GCs and other cholinergic cells (Brandon 1987; Famiglietti 1991). However, other studies of αBgt labeling (Pourcho 1979; Yamada et al. 2003) showed that starburst amacrine cells make presynaptic contacts onto bipolar cell terminals. Our data show that in addition to GCs and cholinergic cells, numerous bipolar cells and amacrine cells exhibited α7 immunoreactivity. In light of the studies by Brandon (1987) and Famiglietti (1991), our data are consistent with the probability that the α7 nAChRs expressed by GCs reflect direct cholinergic transmission from starburst cells, whereas activation of α7 nAChRs on amacrine and bipolar cells may exert modulatory effects on GC responses.
In our studies, α7 immunoreactivity often coincided with “hot spots”, varicosities on the dendrites of cholinergic amacrine cells. Ultrastructural studies suggest that the majority of these varicosities are postsynaptic to bipolar and amacrine cells or presynaptic to GC dendrites (Famiglietti 1991). Therefore, α7-immunoreactive varicosities on cholinergic cells may represent pre-or/and postsynaptic sites at contacts with GCs or other cholinergic cells. These ultrastructural studies are consistent with our double-labeling studies with the presynaptic markers kinesin II and synaptophysin, suggesting a presynaptic role for α7 nAChRs on starburst cells.
At glutamatergic synapses, α7 nicotinic receptors were strictly confined to presynaptic structures in the IPL. Localization of α7 nAChRs, which have high Ca2+ permeability, on presynaptic terminals of bipolar cells raises two possibilities. First, ACh might increase the spontaneous release of glutamate from the bipolar terminal through activation of presynaptic α7 receptors, thus affecting the responses of specific postsynaptic GCs or amacrine cells. Second, the activation of α7 receptors coincident with transmission of bipolar signals might enhance glutamate-driven responses of target ganglion and amacrine cells. Consistent with our data, the application of MLA, a partial antagonist for α7 nAChRs, decreased the maintained firing and light-evoked responses in some identified GCs in rabbit retina (Reed et al. 2002; Strang et al. 2005a), and these effects may be at least partially attributable to a modulation of upstream glutamate release.
Our finding of α7 nAChRs in close association with synaptophysin on the dendrites of glycinergic and some GABAergic cells close to synapses indicates that the release of glycine and GABA could be enhanced via the activation of presynaptic α7 receptors and cause either inhibition or disinhibition of other amacrine and GCs involved in the same local circuits. However, our findings do not rule out a postsynaptic localization for α7 nicotinic receptors. In the CA1 region of adult hippocampus, double labeling for αBgt and synaptophysin revealed abundant expression of both epitopes, often in close apposition, that represented labeling of α7 at both pre- and postsynaptic elements at the electron microscopic level (Fabian-Fine et al. 2001).
The lack of reliable postsynaptic markers for inhibitory synapses in the retina that are compatible with the sc-1447 protocol did not allow us to test for the postsynaptic localization of α7. However, some of the α7 nAChRs in our studies coincided with GABAA receptors on the dendrites of DAPI-3 glycinergic cells. GABAA receptors are frequently found at postsynaptic sites at synapses with other amacrine cells in the IPL of rabbit retina (Greferath et al. 1994). This suggests that clustered α7 nAChRs and GABAA receptors might be present at postsynaptic elements as well.
Post- or perisynaptic α7 nAChRs may mediate an increase in [Ca2+]i through influx directly through the α7 nAChR, the activation of voltage-sensitive Ca2+ channels, or by release from intracellular stores. This increase could be sufficient to influence the activity of calcium-dependent kinases and phosphatases, thus regulating the function of GABAA receptors at GABAergic synapses (Berg and Conroy 2002).
Physiological experiments have demonstrated that selected GCs in rabbit retina (Strang et al. 2005a) including directionally selective GCs (Strang et al. 2005b) express both α7 and non-α7 nAChRs. Furthermore, our results indicate that, in addition to GCs, some amacrine cells express at least two types of nAChRs. They localize close to each other on the dendrites but apparently occupy different spots on the plasma membrane. In brain, expression of two functional subtypes of nAChRs, α7 and α4β2, on the same neuron has been demonstrated by CA1 GABAergic interneurons in rat hippocampal slices (Alkondon et al. 1999). The mediation of fast (phasic) excitation of interneurons through the α7 subtype and slow (tonic) excitation of interneurons through α4β2 results in two types of inhibition on target neurons. Similar to these findings, expression of both α7 and α3β2 nAChRs on some inhibitory amacrine cells in rabbit retina raises the possibility of dual modulation of the release of glycine and/or GABA.
Thus, the apparent expression of α7 nAChRs by different retinal neurons at diverse subcellular locations suggests that they can serve many functions. Release of ACh may boost the responses of ON and ON-OFF types of GCs through the activation of postsynaptic α7 receptors on GCs or by enhancing ON bipolar cell signals via activation of presynaptic α7 nicotinic receptors. α7 nAChR activation on inhibitory amacrine cells that synapse onto other types of inhibitory amacrine cells likely would cause inhibition or disinhibition of multiple local circuits resulting in modulation of the responsiveness of selected GCs. Finally, activation of postsynaptic α7 nicotinic receptors at inhibitory synapses could modulate the activity of GABAA receptors by triggering a cascade of calcium-dependent processes in cholinoceptive amacrine cells.
The possibility that cholinergic modulation of GCs via pathways involving bipolar cells has been previously suggested. Specifically, observations in chick (Vogel et al. 1977), mouse (Pourcho 1979), goldfish (Zucker and Yazulla 1982), and turtle (James and Klein 1985) all demonstrated αBgt binding associated with bipolar cells or amacrine-bipolar synapses. In addition, previous work from this laboratory (Keyser et al. 1993) showed αBgt-sensitive nAChR (α8 sub-units) antibody labeling of bipolar cells in the avian retina. Finally, data from non-human primate retina revealed that cholinergic cells synapse directly onto cone bipolar cell terminals, in addition to synapsing on amacrine and GCs (Yamada et al. 2003). In this respect, the observations we report here concerning bipolar cells are consistent in the context of a body of work that spans nearly 30 years. Details of the organization of cholinoceptive local circuits, the expression of additional nAChR subtypes in the retina, and their subcellular distribution remain issues for future investigation.
Footnotes
Acknowledgements
This work was supported by NEI grants P30 EY-03039 and EY-07845.
