Abstract
During development of the nervous system, large numbers of neurons are overproduced and then eliminated by programmed cell death. Puma is a BH3-only protein that is reported to be involved in the initiation of developmental programmed cell death in rodent retinal neurons. The expression and cellular localization of Puma in retinal tissues during development are not, however, well known. Here the authors report the expression pattern of Puma during retinal development in the rat. During the period of programmed cell death in the retina, Puma was expressed in some members of each retinal neuron, including retinal ganglion cells, amacrine cells, bipolar cells, horizontal cells, and photoreceptor cells. Although the developmental programmed cell death of cholinergic amacrine cells is known to be independent of Puma, this protein was expressed in almost all their dendrites and somata of cholinergic amacrine cells at postnatal age 2 to 3 weeks, and it continued to be detected in cholinergic dendrites in the inner plexiform layer for up to 8 weeks after birth. These results suggest that Puma has some significant roles in retinal neurons after eye opening, especially that of cholinergic amacrine cells, in addition to programmed cell death of retinal neurons before eye opening.
The retina originates from the developing brain and belongs to the central nervous system. Because of its highly organized cellular architecture and easy accessibility, the retina has been extensively studied as a model of development in the central nervous system (Stevens et al. 2007; Wakabayashi et al. 2010). During retinal development, excess retinal neurons are born that are later eliminated by programmed cell death (PCD). PCD is a physiological process that is indispensable for normal development of the retina, and impairments in PCD result in abnormal retinal phenotypes (Bähr 2000). Different retinal neurons undergo PCD at specific stages of retinogenesis. In rodents, retinal ganglion cells (RGCs) die predominantly during the first 11 days after birth, most during days 2–5. Amacrine and bipolar cells die mostly between postnatal day (P) 3 and 8 and before P14, respectively (Young 1984; Horsburgh and Sefton 1987). Most retinal neurons die prior to eye opening (around P14), and all PCD in the retina is almost complete by 3 weeks after birth (Péquignot et al. 2003).
During retinal development, Bcl-2 family members have critical roles in the induction of PCD (Bähr 2000). Activation of Bax and Bak, as well as the consequent permeabilization of the mitochondrial outer membrane, is a pivotal step in apoptosis (Chipuk and Green 2008). Antiapoptotic members of the Bcl-2 family (including Bcl-2 and Bcl-xl) prevent activation of Bax and Bak, whereas proapoptotic BH3-only proteins (including Bim, Bid, Puma, Bad, Hrk, and Noxa) activate them (Chipuk and Green 2008). BH3-only protein family members are activated through transcriptional and posttranscriptional mechanisms, depending on the death-inducing stimulus (Wakabayashi et al. 2002; Wakabayashi et al. 2005). A variety of death-inducing stimuli converge on a common downstream pathway through BH3-only proteins (Puthalakath and Strasser 2002). Among BH3-only proteins, Bim, Bid, and Puma are the major initiators of apoptosis because they activate Bax and Bak directly (Kim et al. 2009). The contributions of these molecules to PCD of the developing nervous system have been intensively studied (Putcha et al. 2001; Krajewska et al. 2002). In Bim-null mice, PCD of retinal neurons is delayed, but the effect is transient, and the final number of retinal neurons is not significantly different from that of the wild type (Doonan et al. 2007).
Puma is considered a critical mediator of neuronal apoptosis in normal development as well as in certain pathological conditions (Geng et al. 2007; Steckley et al. 2007; Niizuma et al. 2009). In Puma-deficient mice, the number of RGCs, bipolar cells, and a specific subtype of amacrine cells is increased (Harder and Libby 2011), suggesting that Puma regulates developmental PCD in retinal neurons. However, the expression of Puma in various retinal cell types and at different developmental stages during normal retinogenesis remains to be explored. To elucidate the physiological function of Puma during retinal development, we analyzed the expression pattern and cellular localization of Puma in the rat retina by immunohistochemistry.
Materials and Methods
Animals
Prenatal and postnatal male and female rat pups and male Wistar rats (SLC, Shizuoka, Japan) were used in this study. Animals were raised under 12/12-hour cyclic light/dark conditions. Adult and postnatal animals were sacrificed by intraperitoneal injection of pentobarbital. Prenatal rat pups were decapitated. Treatment of all animals was in strict accordance with institutional and National Institutes of Health guidelines for the care and treatment of laboratory animals. The experimental protocols were approved by the animal ethics committee at Kansai Medical University.
Immunohistochemistry
Immunohistochemical staining was carried out as reported (Wakabayashi et al. 1996a, 1996b; Wakabayashi et al. 2008; Wakabayashi et al. 2010), with minor modifications. Under deep pentobarbital anesthesia, newborn and adult rats were cardiac perfused with 4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4. Eyes were postfixed by immersion in the same fixative overnight. Prenatal rats were decapitated, and their eyes were enucleated and immersion fixed with 4% paraformaldehyde. Flatmount of the retina was prepared as described previously (Wakabayashi et al. 1996a, 1996b; Wakabayashi et al. 2008; Wakabayashi et al. 2010). For cryostat sectioning, 20% sucrose in 0.1 M phosphate buffer, pH 7.4, was used as a cryoprotective agent. Cryogenic sections (20 µm thick) were made through the optic disc in a dorsal to ventral direction. The specimens, both sections and flatmounts, were rinsed with phosphate-buffered saline and incubated with primary antibody, in phosphate- buffered saline containing 0.1% Triton X-100 at 4C overnight. Primary antibodies were detected with a mixture of Alexa Fluor 488- (Life Technologies, Carlsbad, CA) or Cy3-conjugated donkey secondary antibodies (Jackson ImmunoResearch, West Grove, PA). The specimens were counterstained with Hoechst 33258 to visualize the nuclei and observed under a confocal microscope (LSM510 META, Carl Zeiss, Heidelberg, Germany). Alexa Fluor 488 was excited at 488 nm using an argon laser, and the emitted green fluorescence was detected at 505–530 nm. Cy3 was excited at 543 nm using helium neon laser, and the emitted red fluorescence was detected at 561–753 nm. Hoechst 33258 was exited at 405 nm using a laser diode 405, and the emitted blue fluorescence was detected at 420–480 nm.
Primary antibodies used in this study are listed in Table 1. The anti-Puma (StressMarq, Victoria, BC, Canada) is an affinity purified rabbit polyclonal antibody raised against synthetic N-terminal polypeptide of human Puma α isoform (Uniprot number Q9BXH1). The specificity of the anti-Puma antibody was confirmed in this report as described in the Results section. It detects a single 23-kDa band in rat retinal lysate. Immunostaining was prevented by preabsorption with the antigen peptide. Most of the results were obtained using this antibody. We also used another anti-Puma antibody from ProSci Inc (Poway, CA), which was raised against a synthetic polypeptide of the human Puma α N-terminal region. The labeling patterns were consistent between these two anti-Puma antibodies.
Primary Antibodies Used in this Study
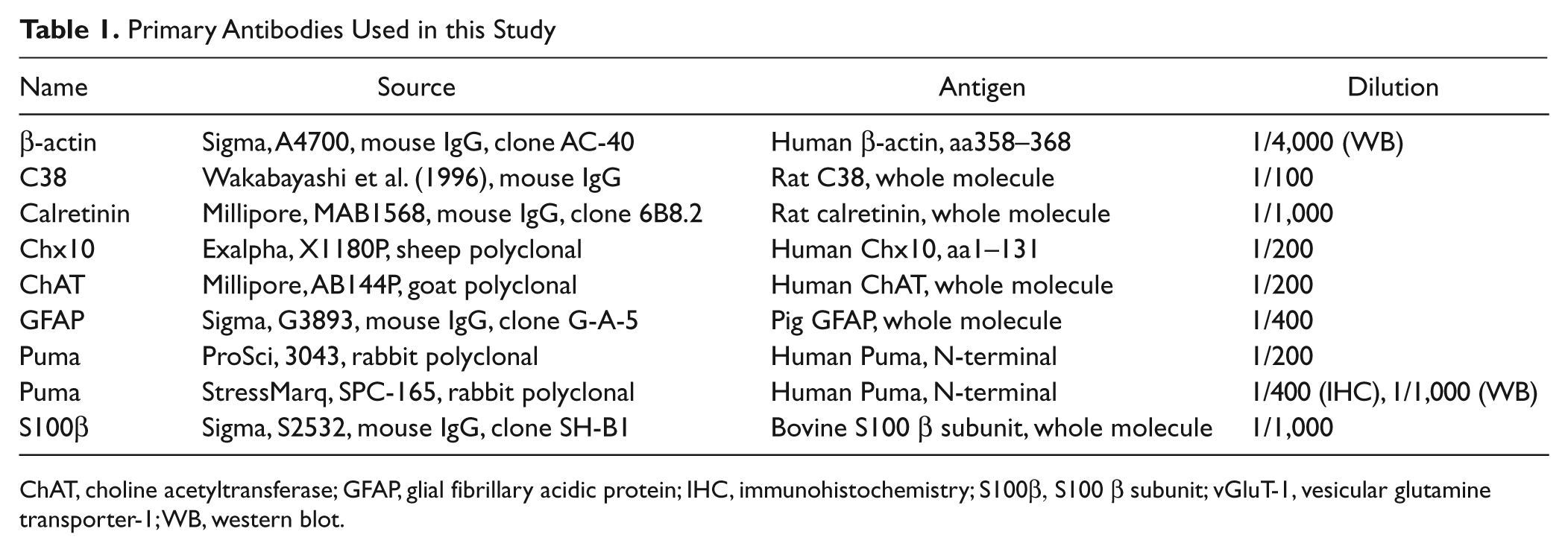
ChAT, choline acetyltransferase; GFAP, glial fibrillary acidic protein; IHC, immunohistochemistry; S100β, S100 β subunit; vGluT-1, vesicular glutamine transporter-1; WB, western blot.
Anti-β-actin (Sigma, St Louis, MO) recognizes a single 42-kDa band on Western blots according to the manufacturer’s datasheet. We detected a single 42-kDa band in rat retinal lysates.
Anti-C38 is a monoclonal antibody raised against rat retinal tissue (Wakabayashi et al. 1996b). It specifically labels RGCs and horizontal cells in rat and mouse retina (Wakabayashi et al. 1996b; Wakabayashi et al. 2010). In Western blots of rat retinal lysate, it recognizes a single 24-kDa band. The immunostaining pattern coincides with the in situ hybridization pattern (Wakabayashi et al. 2010).
Anti-calretinin (Millipore, Bedford, MA) detects a 31-kDa band on mouse brain lysate according to the manufacturer’s datasheet. The labeling pattern that we observed was indistinguishable from that of previous reports (Haverkamp and Wässle 2000; Wakabayashi et al. 2011).
Anti-Chx10 (Exalpha, Shirley, MA) detects a 46-kDa band in rat retinal lysate according to the manufacturer’s datasheet. The labeling pattern that we observed was indistinguishable from that of the previous report (Voinescu et al. 2009).
Anti-choline acetyltransferase (ChAT; Millipore) detects a 68-kDa band (Brunelli et al. 2005), and immunostaining is abrogated following immunodepletion of cholinergic neurons (Yoshida et al. 2001). The labeling pattern that we observed was indistinguishable from that of the previous report (Voinescu et al. 2009).
Anti-glial fibrillary acidic protein (GFAP; Sigma) detects a single 50-kDa band according to the manufacturer’s datasheet. The labeling pattern that we observed was indistinguishable from that of previous reports (Chen et al. 2007; Bosco et al. 2011).
Anti-S100β (Sigma) detects a 20-kDa band in mouse brain lysate (Liao et al. 2008). The labeling pattern that we observed was indistinguishable from that of previous reports (Rambotti et al. 1999; Liao et al. 2008).
Peptide Neutralization
The specificity of the anti-Puma antibody was confirmed by competition with an excess amount of antigen peptide (Wakabayashi et al. 2011). Anti-Puma antibody was raised against the N terminus of the human Puma α isoform. A peptide corresponding to the N-terminal 30 amino acids of the Puma α isoform was chemically synthesized (ABIMED AMS-422, Abimed, Langenfield, Germany). This sequence (MARARQEGSSPEPVEGLARDSPRPFPLGRL) was found to be unique and to have no significant similarities to other antigens in a search against all GenBank sequences. Anti-Puma antibody was preincubated overnight at 4C with a 100-fold excess of either the antigen peptide or a nonspecific peptide in phosphate-buffered saline containing 1% bovine serum albumin. Immunohistochemistry was performed as described above using these preincubated solutions.
Western Blotting
Western blots were performed as described (Wakabayashi et al. 1996b; Wakabayashi et al. 2011) with minor modifications. Briefly, tissue lysates (30 µg total protein) were resolved by 12.5% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto Immobilon-P membranes (Millipore, Billerica, MA). The blots were incubated with primary antibody followed by horseradish peroxidase-conjugated secondary antibody (Jackson ImmunoResearch). Signals were detected by chemiluminescence (ECL Prime Western Blotting Detection Reagent, GE Healthcare Japan, Tokyo, Japan).
Results
Developmental Expression of Puma in the Retina
Puma localization in the developing rat retina was evaluated by immunohistochemistry. During retinal development, PCD is first observed around embryonic day (E) 16. PCD of RGCs occurs predominantly in the first postnatal week and that of other retinal neurons peaks at the second postnatal week and is almost complete within the first 3 weeks after birth (Young 1984; Péquignot et al. 2003). Thus, we first examined retinal expression of Puma from E16 to P21. Puma immunoreactivity was detected in the ganglion cell layer (GCL) at E16 (Fig. 1). At prenatal and early postnatal stages, Puma expression was relatively restricted to the GCL, but scattered labeling was also observed throughout the neuroblastic layer (Fig. 1, E16–P2). Puma immunoreactivity in the neuroblastic layer was predominantly restricted to the inner half of the layer by P7, when the neuroblastic layer is divided into the inner and outer nuclear layers (INL and ONL) by the formation of the outer plexiform layer (OPL) (Wakabayashi et al. 2010). Thus, Puma appears to have been restricted to the INL (Fig. 1, P7; Fig. 2, P7). At P7, Puma was also detected in the segment layer of photoreceptors.
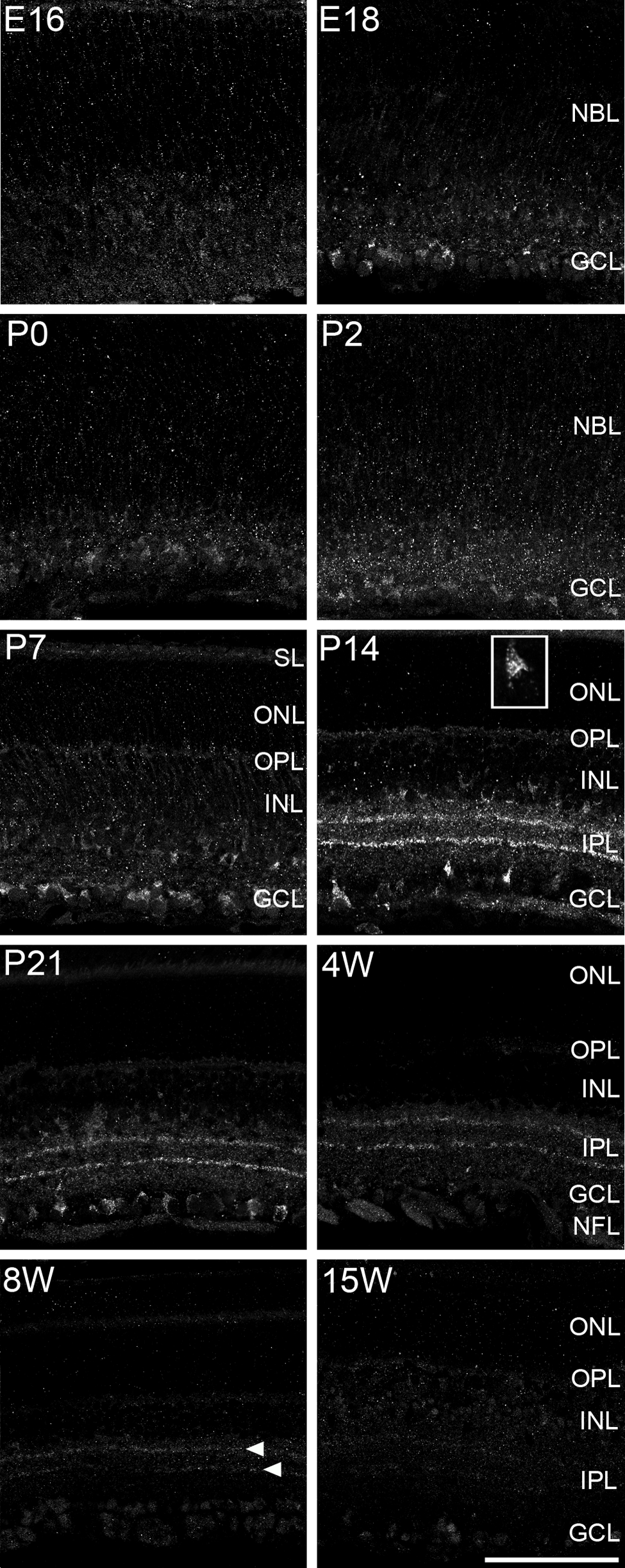
Expression of Puma antigen during development of the rat retina. Specimens were labeled with anti-Puma. Puma was detected in the GCL as early as E16. At P14 and P21, Puma was expressed from the GCL to the OPL but predominantly in the GCL and the inner INL. Immunoreactivity was selectively localized in the cytoplasm (see inset in P14: high magnification of Puma-positive cell in the GCL). Segment layer of photoreceptors was also labeled as obvious in P7. In addition, two bands in the IPL were detected from P14 up to postnatal 8 weeks (8W; arrowheads). E, embryonic day; GCL, ganglion cell layer; INL, inner nuclear layer; IPL, inner plexiform layer; NBL, neuroblastic layer; NFL, nerve fiber layer; ONL, outer nuclear layer; OPL, outer plexiform layer; P, postnatal day; SL, segment layer. Bar = 100 µm.
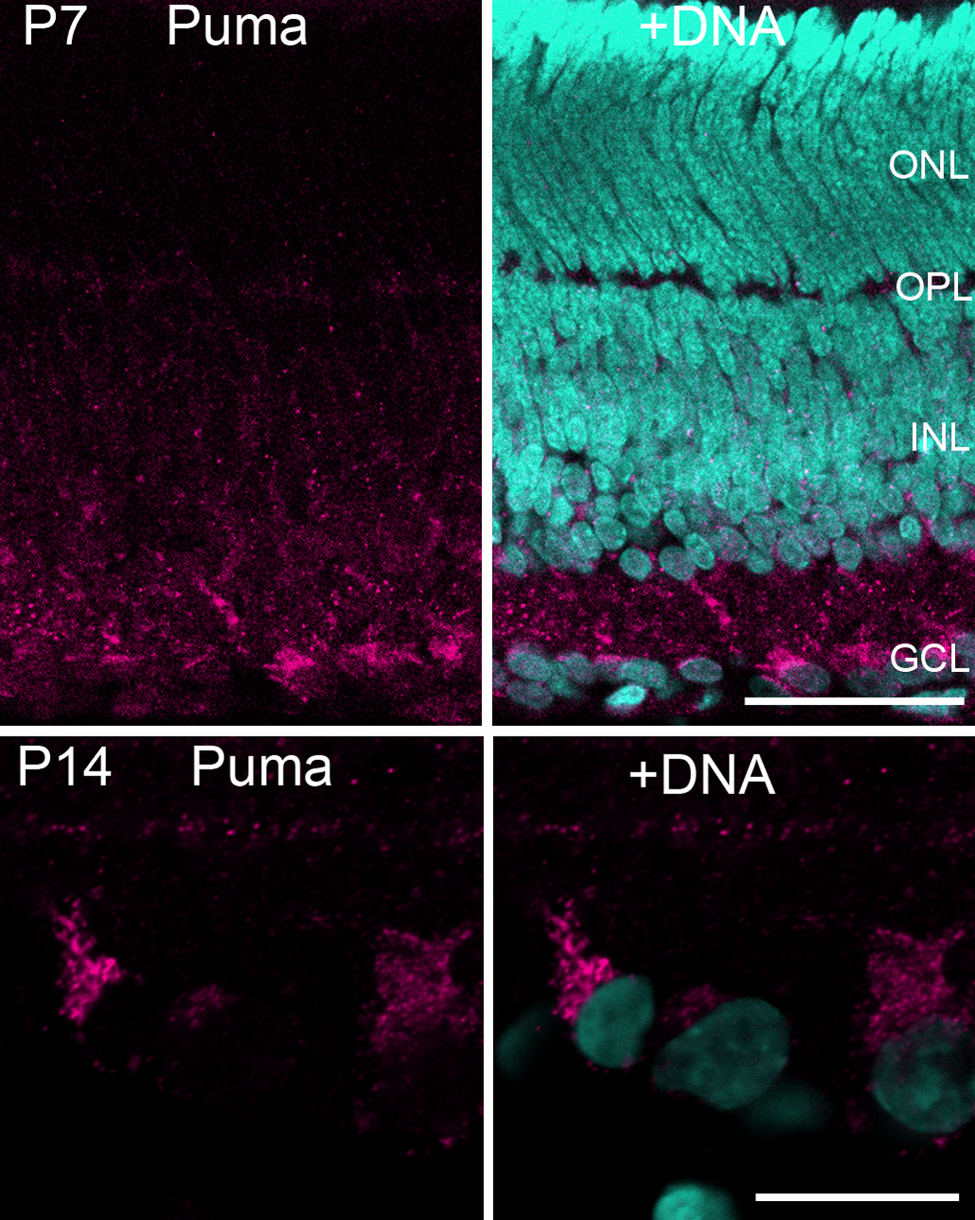
Enlarged figures of the P7 and P14 retina labeled by the anti-Puma antibody. (P7) At this stage, the neuroblastic layer is divided into the INL and the ONL by the OPL. Puma immunoreactivity was predominantly restricted to the INL rather than the ONL. Bar = 50 µm. (P14) Puma-labeled cells in the GCL. Puma immunoreactivity is mostly detected in the cytoplasm rather than the nuclei. Bar = 20 µm. The specimens were counterstained with Hoechst 33258 to visualize nuclei (+DNA). GCL, ganglion cell layer; INL, inner nuclear layer; ONL, outer nuclear layer; OPL, outer plexiform layer; P, postnatal day.
Intense Puma immunoreactivity was detected at P14 and P21. At P14, immunoreactivity was clearly localized in the cytoplasm rather than the nucleus (Fig. 1, P14 inset; Fig. 2, P14). The antibody labeled the cell somata in the GCL and the innermost part of the INL more strongly as compared with other layers. Labeling was faint in the ONL, the OPL, and the middle of the INL. In addition, two linear dense bands were readily detected in the inner plexiform layer (IPL).
Because Puma antigen was readily detected at P21, we continued to investigate expression in the older animals. After P21, immunoreactivity declined with age, especially in the cell somata in the INL and GCL. However, two bands in the IPL were still detected even at 8 weeks (Fig. 1, arrowheads). No significant Puma immunoreactivity was detected in the retina at 15 weeks. Thus, Puma expression was predominantly detected in the cell somata of the rat retina until P21, and faint but significant labeling in two bands of the IPL continued for at least 8 weeks after birth.
To confirm the specificity of the antibody, we performed peptide neutralization experiments. The antibody was raised against the N terminus of the Puma α isoform, which does not share homology to the Puma β isoform (Nakano and Vousden 2001). When the Puma antibody was preincubated with a synthetic peptide corresponding to the N-terminal 30 amino acids of Puma α, no significant staining was detected in the P14 rat retina (Fig. 3b). In contrast, in the absence of the peptide (Fig. 3a) or in the presence of a nonspecific peptide (Fig. 3c), Puma immunoreactivity was readily detected. Furthermore, we labeled the retinal specimens with another anti-Puma antibody (Fig. 4) and found the staining patterns were consistent between these two anti-Puma antibodies (Figs. 1, 4).

Peptide neutralization experiments indicate that anti-Puma specifically recognizes rat Puma antigenicity in rat retinal tissue. Anti-Puma preincubated with the antigen peptide (b) showed no significant labeling in P14 retinal tissue. In the absence of antigen-specific peptide (a) and in the presence of nonspecific peptide (c), Puma immunoreactivity was readily detected. Bars = 100 µm. GCL, ganglion cell layer; INL, inner nuclear layer; IPL, inner plexiform layer; ONL, outer nuclear layer; OPL, outer plexiform layer; P, postnatal day.
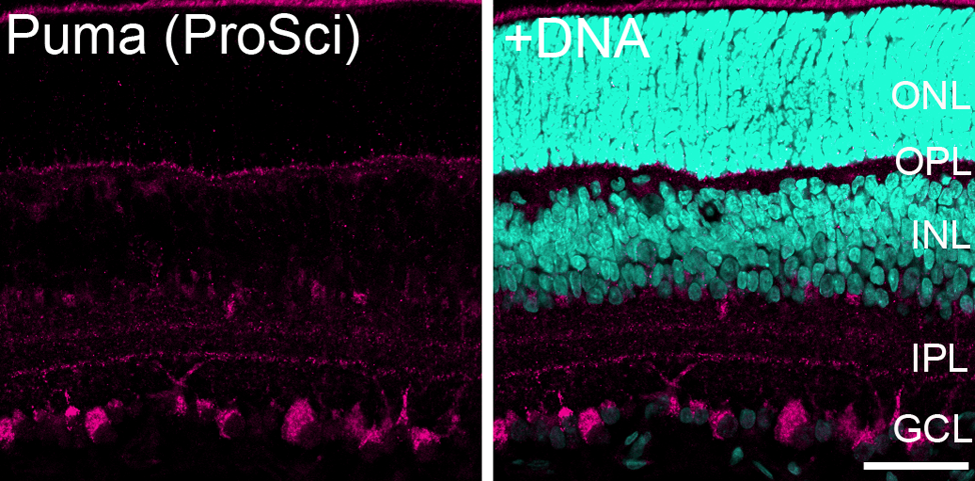
Immunofluorescence images of the P14 rat retina labeled with another anti-Puma antibody. The labeling pattern demonstrated by this anti-Puma antibody (ProSci, #3043) was similar to that observed in Fig. 1. Bar = 50 µm. The specimens were counterstained with Hoechst 33258 to visualize nuclei (+DNA). GCL, ganglion cell layer; INL, inner nuclear layer; IPL, inner plexiform layer; ONL, outer nuclear layer; OPL, outer plexiform layer; P, postnatal day.
We also evaluated the expression of Puma protein by western blotting. Anti-Puma specifically labeled a 23-kDa protein in rat retina, which corresponds to the known size of the α isoform of Puma protein (Fig. 5). This result supports the specificity of the Puma antibody. Puma expression was detected from birth and was highly expressed until P14. After postnatal 4 weeks, the expression levels were low but still detectable.
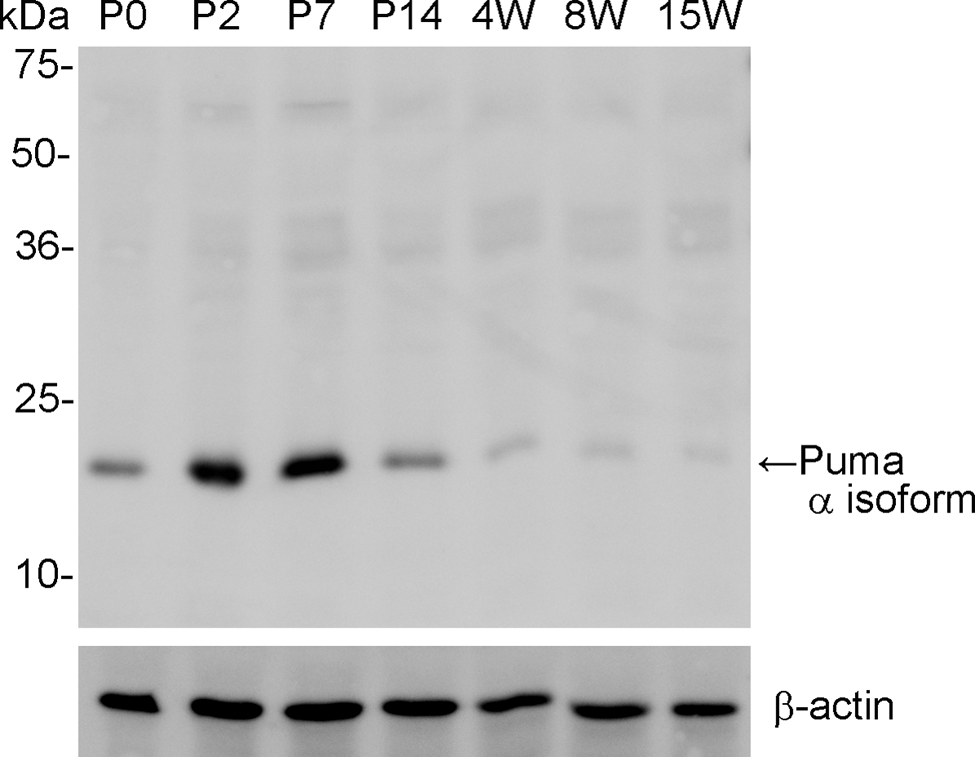
Specificity of Puma antibody by Western blotting against retinal samples from rats of different ages. Anti-Puma specifically labeled a 23-kDa band corresponding to the α isoform of Puma protein in retinal samples. Puma protein was highly expressed in the retina until P14. After that period, expression was faint but significant even at 8 weeks. β-actin was used as an internal control. P, postnatal day; W, postnatal weeks.
Retinal Cell Types that Express Puma Antigen
To identify which cell types express Puma, double labeling was performed for Puma and several cell type–specific markers. Because Puma antibody labeling was intense in the P14 retina, we performed most double-immunolabeling experiments at this stage.
The β subunit of S100 (S100β) is specifically expressed in Müller cells in the INL (Terenghi et al. 1983). In double-labeling experiments, S100β-positive cells were not co-labeled with anti-Puma (Fig. 6a−d). The somata of the S100β-positive cells were localized in the middle layer of the INL (Fig. 6a, arrowhead), whereas Puma-positive cells were predominantly localized in the inner layer of the INL (Fig. 6b, arrow). Colocalization of Puma and S100β was also not observed in the retina at P21 (data not shown).
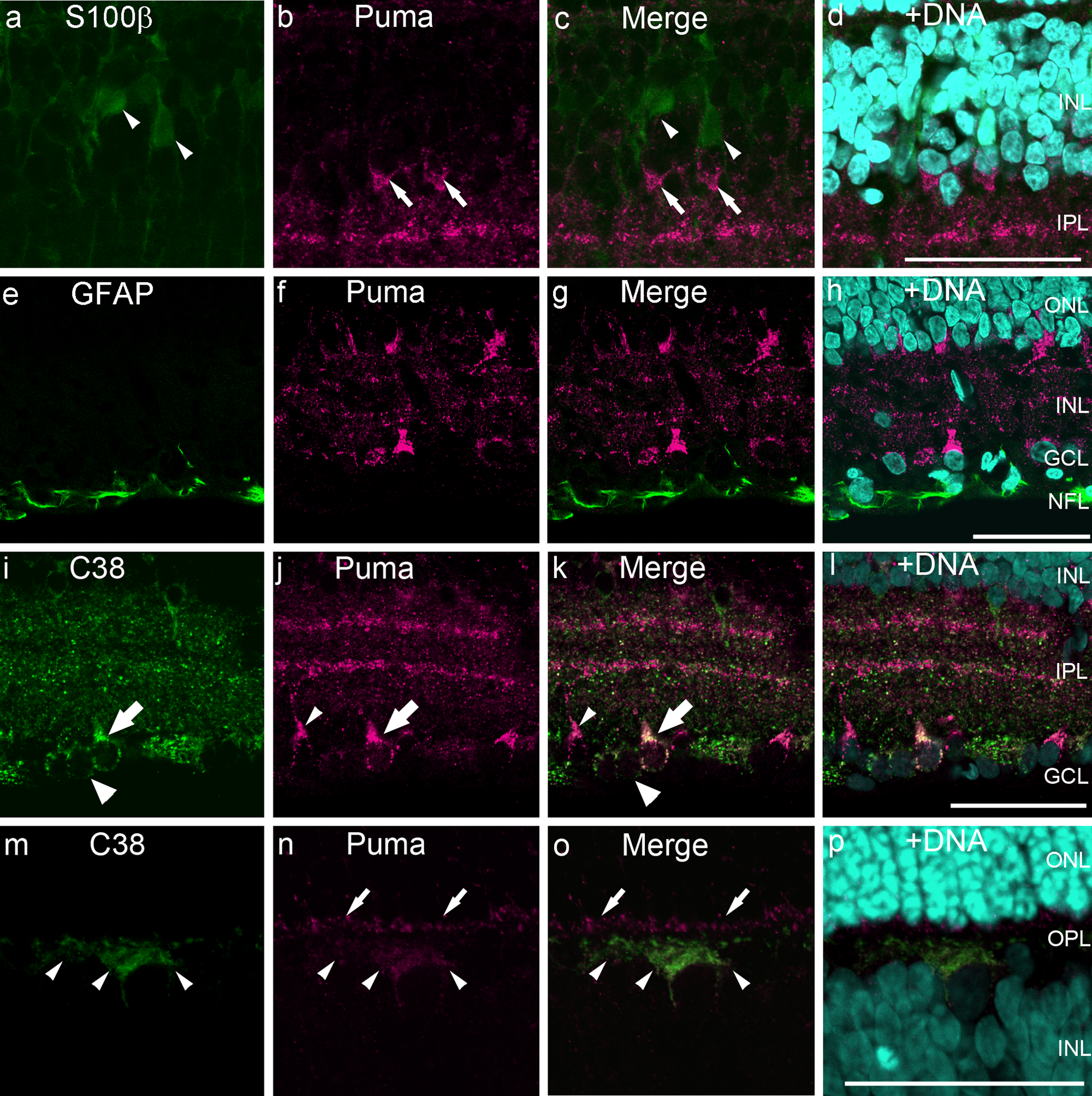
Double-labeling of P14 rat retinas for Puma and cell-specific markers S100β, GFAP, and C38. (a–h) Puma was not expressed in S100β-positive Müller cells in the INL (a–d) or GFAP-positive astrocytes in the NFL (e–h). Arrows and arrowheads indicate Puma- and S100β-positive cells, respectively. (i–p) Colabeling of Puma and C38, focused on the GCL to the inner INL (i–1) and on the OPL (m–p). Some cells in the GCL were positive for both C38 and Puma (large arrows), but others were labeled only with C38 (large arrowheads) or Puma (small arrowheads). In the inner part of the OPL, C38-positive horizontal cell processes were labeled with Puma (m–o, small arrowheads). In the outer OPL, Puma-positive but C38-negative staining was observed (small arrows). The specimens were counterstained with Hoechst 33258 to visualize nuclei (+DNA; d, h, 1, p). Bars = 50 µm. GCL, ganglion cell layer; INL, inner nuclear layer; IPL, inner plexiform layer; NFL, nerve fiber layer; ONL, outer nuclear layer; OPL, outer plexiform layer; P, postnatal day.
GFAP is an astrocyte-specific marker in the retina (Chen et al. 2007; Bosco et al. 2011). GFAP-positive astrocytes in the nerve fiber layer were not labeled by the Puma antibody (Fig. 6e–h).
The monoclonal anti-C38 labels RGCs and horizontal cells in the rat retina (Wakabayashi et al. 1996a, 1996b; Wakabayashi et al. 2010). The Puma antibody labeled subpopulations of C38-positive cells in the GCL (Fig. 6i−1, large arrows). Approximately, 70% of C38-positive cells were also positive for Puma on the flatmount preparation (Fig. 7). In addition, some cells were labeled by anti-Puma alone (Fig. 6i−1, small arrowheads). Weak but significant Puma immunoreactivity was also detected in some C38-positive cells in the inner half of the OPL (Fig. 6m−p, small arrowheads). This inner OPL colabeling was found in processes and occasionally in the somata of horizontal cells (Fig. 6m−p). In addition, Puma immunoreactivity was detected in the outer half of the OPL, which was not labeled by C38 (Fig. 6n, o, small arrows).
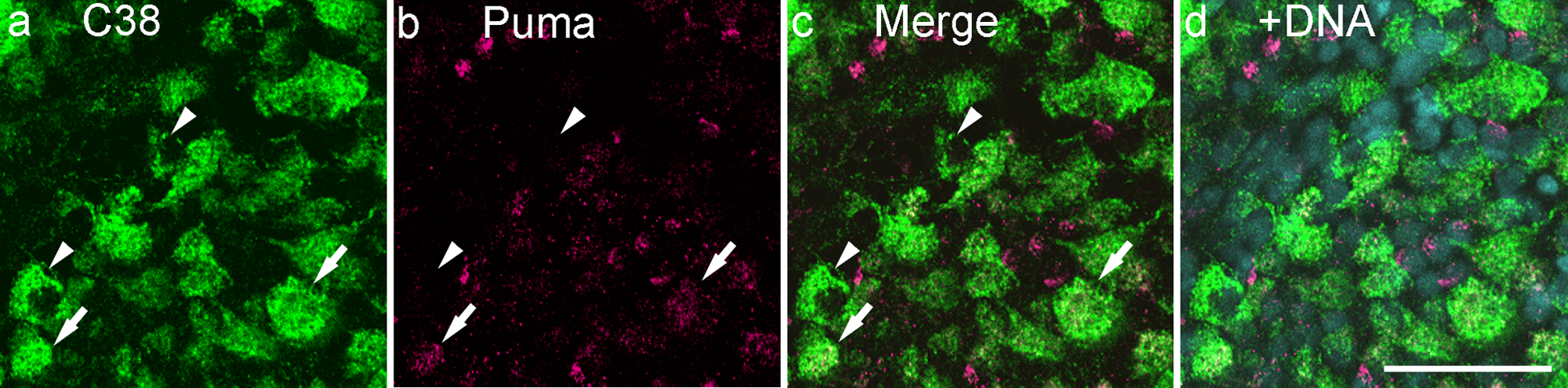
Double-labeling for Puma and C38 on the flatmount of the P14 (postnatal day 14) retina. Among C38-positive retinal ganglion cells, both Puma-positive (arrows) and -negative (arrowheads) cells were observed. Bar = 50 µm. The specimens were counterstained with Hoechst 33258 to visualize nuclei (+DNA).
The ChAT antibody labeled the cell bodies of cholinergic amacrine cells in the inner INL and the GCL (Fig. 8a, e, arrows) as well as their dendrites extending into two narrow bands in the IPL (Fig. 8a, e, arrowheads) (Haverkamp and Wässle 2000). Almost all the ChAT-positive cells were also positive for Puma in both the somata and dendrites (Fig. 8a−h; Fig. 9).
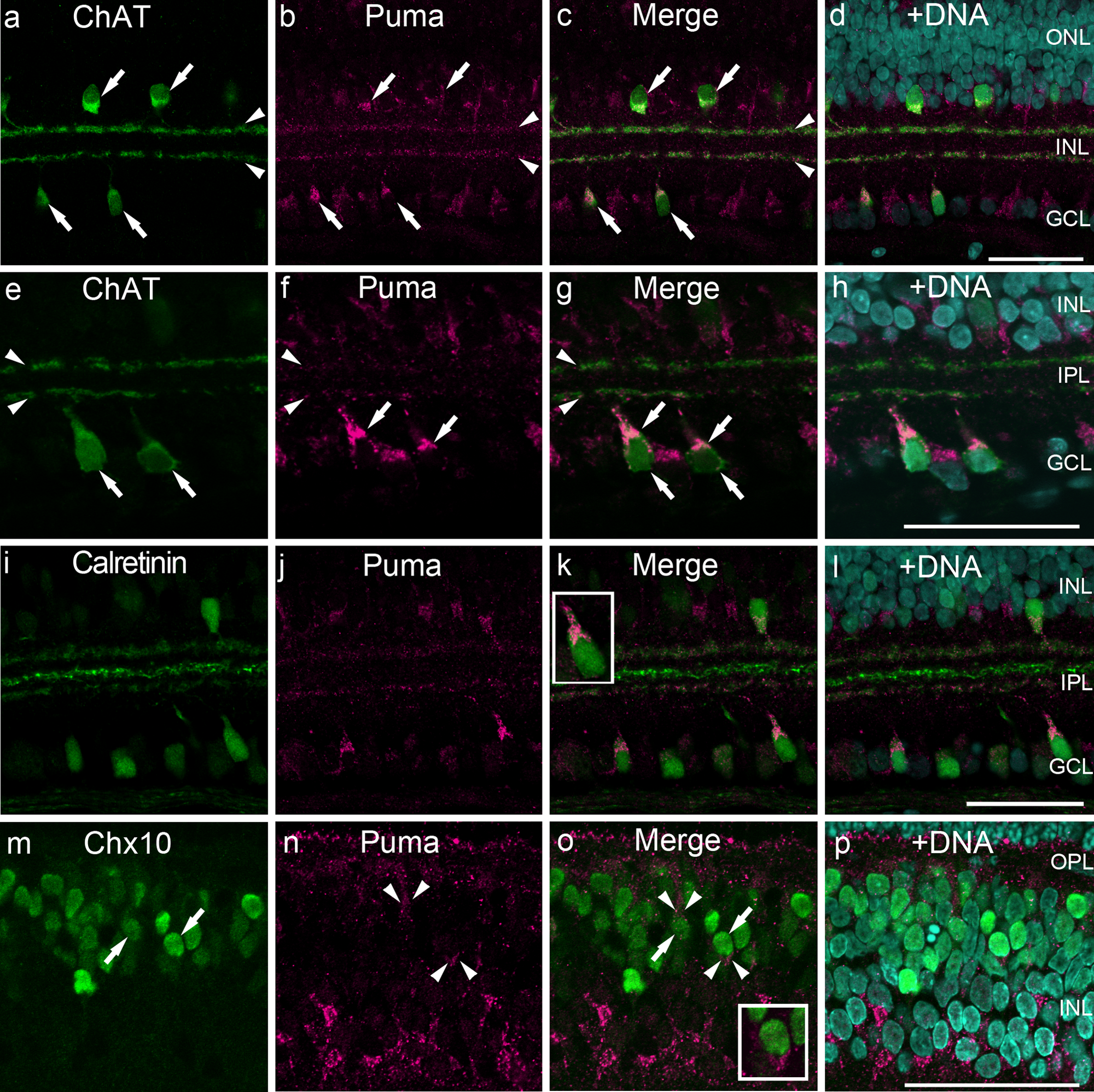
Double labeling of P14 rat retinas for Puma and cell-specific markers ChAT, calretinin, and Chx10. (a–h) Colabeling of Puma and ChAT. Low magnification (a–d), focused on the GCL to the inner INL (e–h). Almost all ChAT-positive somata were positive for Puma (arrows), as were the ChAT-positive cholinergic bands in the IPL (arrowheads). (i–1) Subpopulations of calretinin-positive somata were labeled with anti-Puma. In the INL, anticalretinin labeled three distinct bands, and the outer and inner bands were also positive for Puma. (inset in k) High magnification of Puma and calretinin double-positive cell. (m–p) Subpopulations of Chx10-positive cells (arrows) were also positive for Puma (arrowheads). (inset in o) High magnification of Puma and Chx10 double-positive cell. The specimens were counterstained with Hoechst 33258 to visualize nuclei (+DNA; d, h, 1, p). Bars = 50 µm. GCL, ganglion cell layer; INL, inner nuclear layer; IPL, inner plexiform layer; ONL, outer nuclear layer; OPL, outer plexiform layer; P, postnatal day.
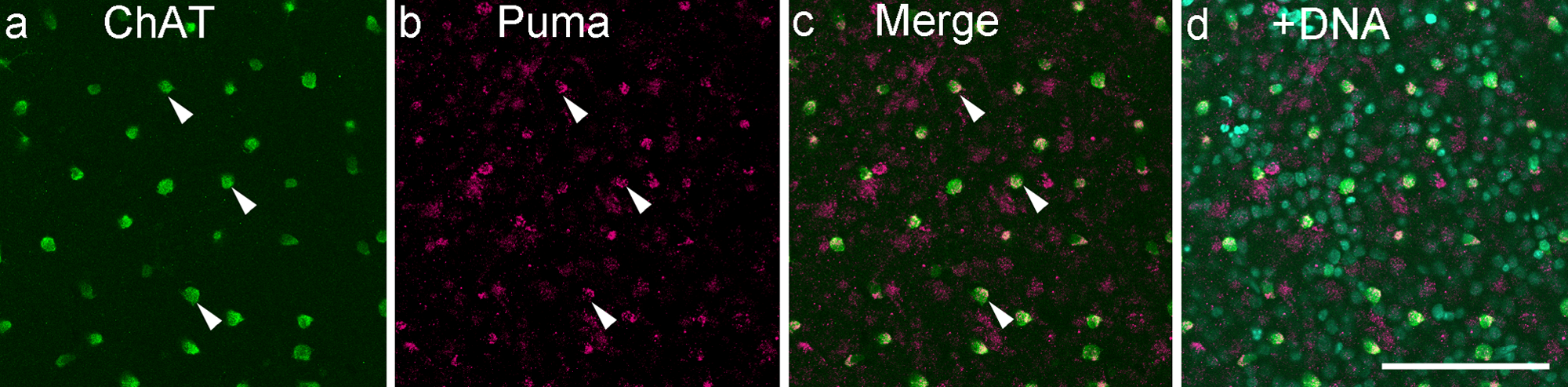
Double labeling for Puma and ChAT on the flatmount of the P14 (postnatal day 14) retina. Almost all ChAT-positive cells were also positive for Puma (arrowheads). Bar = 100 µm.
Calretinin labels three distinct bands in the IPL (Fig. 8i). The outer and inner bands represent cholinergic bands, whereas the middle of the IPL represents the stratification of nitric oxide synthase–positive amacrine cells (Haverkamp and Wässle 2000). Calretinin also labels the somata of amacrine cells (Fig. 8i, k). The Puma antibody labeled subpopulations of the calretinin-positive somata (Fig 8k, inset) and the outer and inner cholinergic bands but did not label the middle band (Fig. 8i−1).
Chx10 is a pan-bipolar cell marker that is localized in the nuclei (Elshatory et al. 2007). Chx10 positive cells (Fig. 8m, o, arrows), surrounded by weak but significant Puma labeling, were occasionally observed (Fig. 8n, o, arrowheads, inset), particular in the outer INL.
Continuous Expression of Puma in Cholinergic Bands
In 4- to 8-week-old rats, Puma immunoreactivity was detected in the retina but restricted predominantly to two bands in the IPL, with little detectable expression in the cell somata (Fig. 1). Because in P14 retina the Puma immunoreactivity in the INL was colocalized with ChAT, it is most likely that Puma localized in the cholinergic bands even in the elderly rats. Synapses of RGCs, cholinergic amacrine cells, and cone bipolar cells are formed within these cholinergic bands (Mariani and Hersh 1988). To confirm this, double labeling was performed with Puma and ChAT in P21 to 8-week-old rat retina. As expected, Puma immunoreactivity in the INL was colocalized with ChAT labeling up to 8-week-old (Fig. 10, arrowheads).
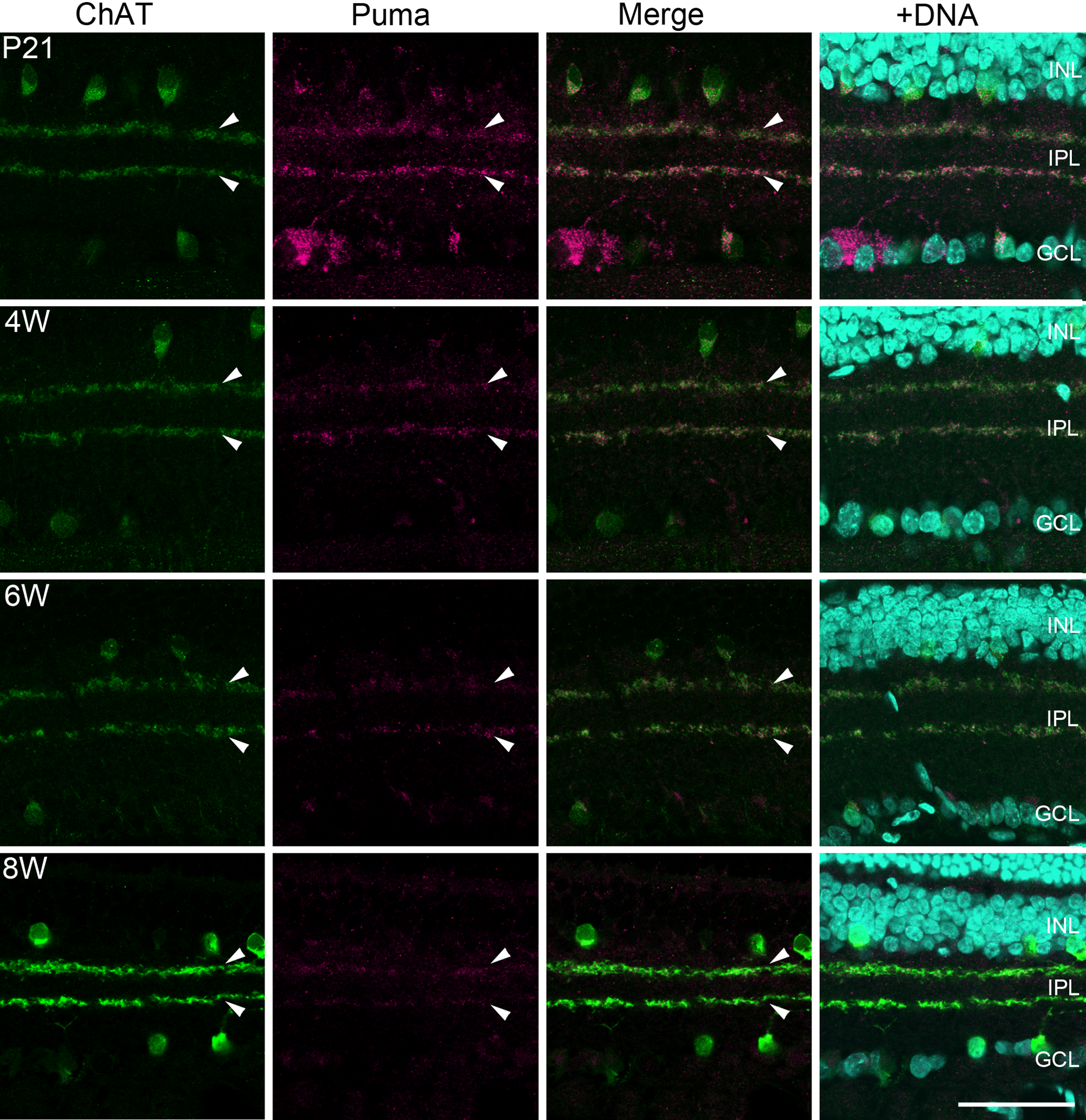
Double labeling of rat retinas from P21 to 8W for Puma and ChAT. The labeling of Puma was gradually restricted to the IPL with age but still colocalized with ChAT-positive bands (arrowheads) up to postnatal 8 weeks. Bar = 50 µm. GCL, ganglion cell layer; INL, inner nuclear layer; IPL, inner plexiform layer; ONL, outer nuclear layer; OPL, outer plexiform layer; P, postnatal day; W, postnatal weeks.
Discussion
Cell Types that Express Puma During Retinal Development
Most of the retinal neurons express Puma during postnatal development, which includes RGCs, amacrine cells, bipolar cells, horizontal cells, and photoreceptor cells. In contrast, Puma is most likely not expressed in glial cells, because the Puma-positive cells were not immunoreactive for the Müller cell marker S100β (Terenghi et al. 1983) or the astrocyte marker GFAP. In the GCL, some Puma-positive cells were also positive for C38, which labels RGCs. Approximately 70% of RGCs were positive for Puma; thus, subpopulations of RGCs were negative for Puma. There are more than 20 subtypes of RGCs even in rodents, and we did not identify which subtypes were negative for Puma. It is possible that certain RGC subtypes do not express Puma during the period of development. A subset of cells in the GCL was positive for Puma but negative for C38. These were displaced amacrine cells, because approximately half the neurons in the rat GCL are displaced amacrine cells (Perry 1981), which are not labeled by C38 (Wakabayashi et al. 1996b). Some of the Puma-positive cells in the GCL were also positive for ChAT or calretinin, which labels subpopulations of amacrine cells (Keeley et al. 2007). But calretinin-positive cells negative for Puma were also observed. This further supports the possibility that Puma is expressed in subpopulations of displaced amacrine cells. The Puma antibody also labeled the somata of amacrine cells in the INL.
Puma immunoreactivity was faint in the outer half of the INL as compared with the GCL and the inner half of the INL. Nevertheless, Chx10-positive bipolar cells that expressed Puma were occasionally observed, and C38-positive horizontal cells in this layer were weakly labeled with Puma. Furthermore, Puma labeled the segment layer of photoreceptors. Taken together, these observations show that Puma is developmentally expressed at least in some members of each of the retinal neurons, including RGCs, amacrine cells, bipolar cells, horizontal cells, and photoreceptor cells.
Contribution of Puma to Developmental Cell Death in Retinal Neurons
In Puma-deficient mice, RGCs, bipolar cells, and a subpopulation of amacrine cells were increased in number in the adult retina (Harder and Libby 2011). These results, in part, are comparable to our observations that Puma was expressed in RGCs and bipolar cells, both of which require Puma for developmental PCD. In these cells, Puma is predominantly localized in the cytoplasm rather than the nuclei. Puma exhibits its proapoptotic activity by activating proapoptotic or inhibiting antiapoptotic proteins interacting in the cytoplasm (Vousden 2005). In addition, Bcl-2 family proteins are known to regulate dendritic development and synapse formation by regulating mitochondrial morphology and functions (Liu and Shio 2008). Thus, Puma is mostly localized to the cytoplasm. In contrast, although the number of ChAT-positive amacrine cells and the number of horizontal cells were not significantly different between wild-type and Puma-knockout mice, we found that Puma was expressed in these cell types. Cholinergic amacrine cells are overproduced and subsequently reduced in number by PCD during postnatal development (Strettoi and Volpini 2002), and we clearly showed that Puma expression was detected in almost all cholinergic amacrine cells (Fig. 8a−h; Fig. 9). Although Puma was highly expressed in cholinergic amacrine cells during development, there is no apparent increase in the number of cholinergic amacrine cells in Puma-deficient mice (Harder and Libby 2011). The reasons for this discrepancy are not currently clear. It is possible that other BH3-only proteins such as Bim or Bid are responsible for developmental PCD in cholinergic amacrine cells. However, in Bim- or Bid-deficient mice, PCD during the development of retinal neurons is delayed, but in the adult there are no apparent changes in retinal cell numbers as compared with the wild type (Doonan et al. 2007; Harder and Libby 2011). The antiapoptotic Bcl-2 family proteins can antagonize the proapoptotic effect of Puma in cholinergic amacrine cells, but Bcl-2 is predominantly expressed in Müller cells rather than in retinal neurons after P10 (Chen et al. 1994). Almost all cholinergic amacrine cells were labeled by anti-Puma, in contrast to the subpopulations of Puma-positive cells in other retinal neuron types. We suggest that at least in cholinergic amacrine cells, Puma may have some role(s) other than as an inducer of apoptosis, as discussed below. In the case of horizontal cells, they are not overproduced in large numbers during retinal development, and their developmental PCD is independent of Bcl-2 family proteins (Strettoi and Volpini 2002). Thus, it may be possible that even if Puma is expressed in horizontal cells, apoptosis is not induced in these cells.
Functional Significance of Puma Expression in Retinal Neurons After Eye Opening
During retinal development in the rat, PCD begins to taper off before the eyes open and is complete by P21 (Young 1984; Péquignot et al. 2003). We also confirmed that there were no apoptotic cells in the retinas after 4 weeks (data not shown). Puma was highly expressed in retinal neurons until postnatal 2−3 weeks. This suggests that Puma has a significant role in developmental PCD in at least some cell types. However, even after eye opening, Puma was expressed in the inner retina. Furthermore, for up to 8 weeks postnatally, Puma was expressed in two bands in the IPL that corresponded to cholinergic amacrine cell dendrites. Puma was expressed in almost all cholinergic amacrine cell bodies, in which PCD is independent of Puma activity (Harder and Libby 2011). These results strongly suggest that Puma has additional roles in cholinergic amacrine cells other than inducing PCD. Increasing reports indicate that Bcl-2 family of proteins regulate morphological and functional maturation of dendrites and synapses by modifying mitochondrial function (Hansen et al. 2007; Li et al. 2008; Tsubouchi et al. 2009; Jiao and Li 2011). The Bcl-2 family of proteins regulate synaptic function without inducing cell death (Jonas et al. 2003; Jonas et al. 2005). More recently, caspase 3, a key mediator of apoptosis that is activated by Puma, has been shown to regulate synaptic activity (D’Amelio et al. 2010). Thus, it is possible that Puma protein expressed in cholinergic amacrine cells contributes to the regulation of dendrite and synapse development. Continuous expression of Puma predominantly in the dendrites rather than in the somata suggests this possibility. Synaptic activity reduces Puma-induced apoptosis (Léveillé et al. 2010), and during retinal development, cholinergic amacrine cells receive rhythmic synaptic inputs and undergo spontaneous activity (Zhou 1998). Thus, it is possible that synaptic activity suppresses the proapoptotic effects of Puma in cholinergic amacrine cells. The retina is a powerful tool for analyzing the contribution of Bcl-2 family proteins to synaptic activity in an in vivo model of the central nervous system, because it is relatively easy to manipulate synaptic activity in this system (i.e., through light deprivation). Further investigation is necessary to resolve the reasons for continuous Puma expression in the retina.
Conclusion
During retinal development, Puma was expressed in most retinal neuron types. Puma expression was predominantly found in neurons in the inner retina, although faint but significant expression was also seen in the outer retina. Puma immunoreactivity continued for up to 8 weeks after birth, mostly in the dendritic processes of cholinergic amacrine cells. Our results suggest that Puma may have significant functions in the retina other than in initiating PCD.
Footnotes
Acknowledgements
We thank Mr. S. Tamaru, Central Research Laboratory, Okayama University Medical School, for his excellent technical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grants-in-Aid for Scientific Research (C) from the Japan Society for the Promotion of Science to TW and JK, “Academic Frontier” Project for Private Universities: matching fund subsidy from the Ministry of Education, Culture, Sports, Science and Technology of Japan to HY and Research Grant D from Kansai Medical University, Japan to TW.
