Abstract
Enkephalins are involved in neural control of digestive functions such as motility, secretion, and absorption. To better understand their role in pigs, we analyzed the qualitative and quantitative distribution of enkephalin immunoreactivity (ENK-IR) in components of the intestinal wall from the esophagus to the anal sphincter. Immunohistochemical labelings were analyzed using conventional fluorescence and confocal microscopy. ENK-IR was compared with the synaptophysin immunoreactivity (SYN-IR). The results show that maximal ENK-IR levels in the entire digestive tract are reached in the myenteric plexuses and, to a lesser extent, in the external submucous plexus and the circular muscle layer. In the longitudinal muscle layer, ENK-IR was present in the esophagus, stomach, rectum, and anal sphincter, whereas it was absent from the duodenum to the distal colon. In the ENK-IR plexuses and muscle layers, more than 60% of the nerve fibers identified by SYN-IR expressed ENK-IR. No ENK-IR was observed in the internal submucous plexus and the mucosa; the latter was found to contain ENK-IR endocrine cells. These results strongly suggest that, in pigs, enkephalins play a major role in the regulatory mechanisms that underlie the neural control of digestive motility.
Keywords
T
The organization of the enteric nervous system is similar in large mammalian species but differs somewhat from that of small laboratory animals. In addition to the submucosal Meissner's plexus (internal submucous plexus), an external submucous plexus or Schabadasch plexus is apposed to the inner circular muscle layer (Timmermans et al. 1992, 1997). Detailed studies on the internal and external submucous plexus of the pig small intestine and colon have shown that they differ in their topographic, architectural, and structural characteristics (Scheuermann and Stach 1984; Scheuermann et al. 1987, 1989; Brehmer et al. 1994; Timmermans et al. 1997), in the electrophysiological properties of their neurons (Thomsen et al. 1997a, b), and in their neurochemical coding (Scheuermann and Stach 1982; Timmermans et al. 1989, 1990, 1991; Barbiers et al. 1995). All these differences argue in favor of the idea that each of these two submucous plexuses plays a distinct role.
Previous studies on the enkephalinergic innervation of the pig digestive tract have focused mainly on the small intestine (Alumets et al. 1978; Malmfors et al. 1981; Scheuermann and Stach 1985; Scheuermann et al. 1990; Timmermans et al. 1990; De Ridder et al. 1991; Shen et al. 1993; Vento et al. 1998), the esophagus (Uddman et al. 1980), and the antrum (Alumets et al. 1978). In addition, apart from the studies by Timmermans et al. (1990), examination of the distribution of the enkephalinergic innervation was not carried out taking into account the existence of two submucous plexuses in pig digestive tract. It therefore seemed to be worthwhile to perform fairly extensive investigations on the patterns of distribution of the enkephalinergic neural pathways in the entire pig gastrointestinal tract, including the various components of the intestinal wall, especially the ganglionic neural plexuses. These investigations were performed using highly specific methionine- and leucine-enkephalin antibodies that enabled us to compare the respective distribution of Met- and Leu-enkephalin in intramural neural pathways. The use of laser confocal microscopy, combined with a computer analysis of the enkephalin-immunoreactive (ENK-IR) materials, enabled us to develop a more accurate quantitative study of enkephalinergic innervation than those previously performed simply on the basis of visual estimates. To study the distribution of enkephalin innervation with regard to the overall innervation of the digestive tract, ENK-IR was compared with the synaptophysin immunoreactivity (SYN-IR). Synaptophysin is a glycoprotein that is one of the membrane components of the clear synaptic vesicles present in almost all the varicosities of intramural nerve fibers (Navone et al. 1986).
Materials and Methods
Tissue Preparation
Young Pietrain/Hampshire pigs (n = 8; 5-8 weeks old) of both sexes, ranging from 6 to 8 kg, were used in this study. The animals were anesthetized with pentobarbital (10 mg/kg IP) and sacrificed by exsanguination through a carotid incision. Segments of esophagus, cardia, fundus, antrum, pylorus, duodenum, jejunum, ileum, ileocecal junction, proximal and distal colon, rectum, and internal anal sphincter were removed and rinsed in Tyrode's solution. The intestinal segments were immediately fixed by immersing them in 4% paraformaldehyde in 0.1 M PBS, pH 7.4, for 24 hr at 4C and then for 24 hr in PBS containing 20% and 30% sucrose at 4C, and finally frozen in dry ice. The intestinal segments were cut with a cryostat into 20-μm-thick cross-sections.
Immunohistochemistry
The enkephalin distribution was studied on sections rather than on whole mounts with a view to obtaining an overall picture of the ENK-IR throughout the intestinal wall. The sections were first incubated for 1 hr in PBS containing 3% normal goat serum, to prevent any nonspecific binding, and 0.2% Triton X-100. They were then incubated in PBS containing a mixture of Met-enkephalin (ME) or Leu-enkephalin (LE) rabbit polyclonal antiserum and mouse monoclonal antibody anti-synaptophysin (1:15; Boehringer Mannheim, Mannheim, Germany) for 24 hr at 4C. The physicochemical characteristics of the ME and LE antibodies have been exhaustively described elsewhere (Cupo and Jarry 1985; Cupo et al. 1986). They are highly specific for their own antigen; a very weak crossreactive factor was observed between ME antiserum and LE (0.1%) and between LE antiserum and ME (1%). The sections were washed three times for 10 min in PBS and then incubated in a PBS solution containing fluorescein isothiocyanate (FITC)-conjugated anti-rabbit IgG (1:200; Sigma, St Louis, MO) and Texas Red-conjugated goat anti-mouse IgG (1:200; Jackson ImmunoResearch Laboratories, West Grove, PA) for 2 hr at room temperature. They were then washed three times for 10 min in PBS and mounted on microscope slides in an anti-fading medium (Dako Laboratories; Carpinteria, CA). Specificity studies were performed on the ME and LE antibodies by preincubating each specific antiserum with its own antigen at a concentration of 10 μM before incubating it with the tissue sections. This abolished the ENK-IR in all the tissues tested. Negative controls were obtained either by omitting the specific enkephalin and synaptophysin antibodies or by replacing them with nonimmune goat serum to assess the nonspecific adsorption of secondary antibodies.
Confocal Microscopy
Tissue sections were examined under a confocal scanning laser microscope (Leica TCS 4D; Heidelberg, Germany) attached to an Aristoplan microscope (Leica). The source of illumination was an argon/krypton laser with excitation and emission wavelengths of 488 and 522/532 nm, respectively, in the case of FITC and 568 and 605/632 nm in that of Texas Red. Each field of the specimens was scanned sequentially in three dimensions in a series of optical sections with a step size of 1 μm. Five to eight vertical optical sections were scanned at the top of each specimen to ensure that no artifactual decrease would occur in the density of the immunoreactivity, which showed clearly up to a depth of 10 μm. Kalman averages were calculated on each optical section based on four scans. After the recording sessions, the optical sections were displayed in the form of digital images of either 512 × 512 or 1024 × 1024 pixels and processed using the Adobe Photoshop software program (Adobe Systems; San Jose, CA).
Quantification of ENK-IR and SYN-IR was performed in fundus, duodenum, and rectum sections using an appropriate software program (Piclab; Marseille, France). In all experiments, the oil-immersion lens (X63, numerical aperture = 1.4), the laser beam intensity, the pinhole aperture, and the photomultiplier gain were kept constant. Before quantifying the amounts of specific fluorescent signal present in each optical section, removal of background fluorescence was performed. Its threshold level was determined in an area of the optical section devoid of specific fluorescent signal. We found that the background level determined in sections labeled with primary antisera did not differ greatly (±5 of 256 gray levels) from the noise fluorescent signal determined in control sections in which primary antisera were omitted. In each digital image (512 × 512 pixels), the area of the FITC and Texas Red fluorescence labeling was determined and expressed as the number of pixels/μm2. In each region and in each structure studied, the mean area of the FITC and Texas Red fluorescence labeling was calculated using 100 optical sections obtained from four animals, which amounted to a total area of 2.4 mm2. In the Results, the ENK-IR (FITC) present in the various components of the three regions studied is expressed as the ratio between the mean ENK-IR area and the mean SYN-IR area ± SEM as percentages.
The coexistence of the two fluorescence probes in the nerve fibers was also analyzed in the optical sections by use of the Piclab software program. Binary images were generated from digital images, in which all the pixels had gray values of 50 in the case of FITC or 100 in that of Texas Red. FITC and Texas Red binary images based on the same optical section were then summed together to obtain the pixels having both 50 and 100 gray values, and to differentiate between these pixels and the others. Their area was then calculated, using identical processes to those developed for use with the FITC and Texas Red probes. In the Results, coexistence of SYN-IR and ENK-IR in nerve fibers is expressed as the ratio between the mean SYN-IR and ENK-IR co-localized area and the mean SYN-IR area ± SEM as percentages. Statistical tests were performed with StatView (Abacus Concept; Berkeley, CA). Analyses of variances of ENK-IR/ SYN-IR and ENK-IR + SYN-IR area, determined in each ganglionic plexus and in muscle layers of the three regions studied, were performed by multiple comparaison using the ANOVA Bonferroni t-test. Differences between two groups were considered statistically significant at p≤0.05.
Confocal micrographs, except those corresponding to a single optical section, are digital composites of z-series scan of three to five optical sections constructed with Adobe Photoshop (Adobe Systems) software.
Results
Distribution of Synaptophysin and Enkephalin Immunoreactivity
No significant differences were observed between the patterns of distribution of Met- and Leu-ENK-IR. Considering the very weak crossreactivity between Met- and Leu-enkephalin antibodies (see Materials and Methods) the identical pattern of distribution of Met- and Leu-ENK-IR may be due to their genuine co-localization. Therefore, we decided to deal collectively with these two types of immunoreactivity, which will now be referred to as enkephalin immunoreactivity (ENK-IR) (Table 1).
In the myenteric, internal, and external submucous plexuses, SYN-IR nerve fibers were found in all regions studied (Figures 1A-4A). However, ENK-IR nerve fibers were detected only in the myenteric and external submucous plexuses (Figures 1B-4B). In these nervous plexuses, ENK-IR was found in the soma of some neurons (Figures 5B and 6B). In the myenteric and external submucous plexuses, the SYN-IR and ENK-IR nerve fibers were wrapped tightly around the cell bodies of non-ENK-IR neurons and sometimes of ENK-IR neurons (Figures 5A, 5B, 6A, and 6B).
In all regions and the structures studied, synaptophysin-like immunoreactivity was present and is not mentioned in the table.
Enk, presence of enkephalin-like immunoreactivity (neurons and/or fibers); 0, rare or absent enkephalin-like immunoreactivity; LM, longitudinal muscle; CM, circular muscle; MP, myenteric plexus; ESP, external submucous plexus; ISP, internal submucous plexus; MM, muscularis mucosae; M, mucosa; ICJ, ileocecal junction; Prox. colon, proximal colon; IAS, internal anal sphincter.
In the external muscle layers, SYN-IR material was present in the nerve fibers in all regions of the digestive tract studied. These nerve fibers were always particularly abundant in the circular muscle layer whereas in the longitudinal layer, they were more abundant in the proximal (Figure 1A) and distal (Figure 4A) parts of the digestive tract and less so in the small intestine (Figure 2A) and the colon (Figure 3A). The ENK-IR material showed a similar pattern of distribution in the upper part of the digestive tract, extending from the esophagus to the pylorus, to that observed in the lower part running from the rectum to the internal anal sphincter (Figures 1B-4B). However, from the duodenum to the distal colon, the ENK-IR nerve fibers were either rare or completely absent in the longitudinal muscle layer and were mainly found in the circular muscle (Figures 2B and 3B). In most of the regions studied, the SYN-IR and ENK-IR nerve fibers were distributed fairly evenly over these two muscle layers. However, in the small intestine, the SYN-IR and ENK-IR nerve fibers present in the circular muscle layer were distributed according to an increasing gradient, working towards the deep muscular plexus; all these immunoreactive fibers belonged to the outer circular muscle layer (Figures 2 and 3). No SYN-IR and ENK-IR nerve fibers were observed in the inner circular muscle layer.
The mucosa contained mainly SYN-IR nerve fibers and very few ENK-IR nerve fibers. On the other hand, the presence of ENK-IR material was observed in some endocrine cells (Figures 2B and 4B).
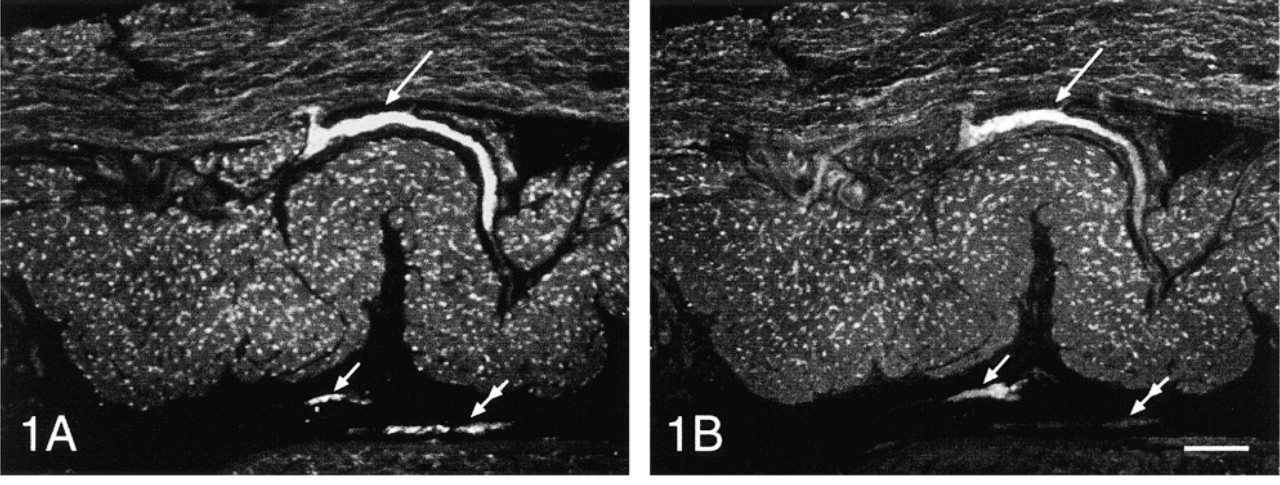
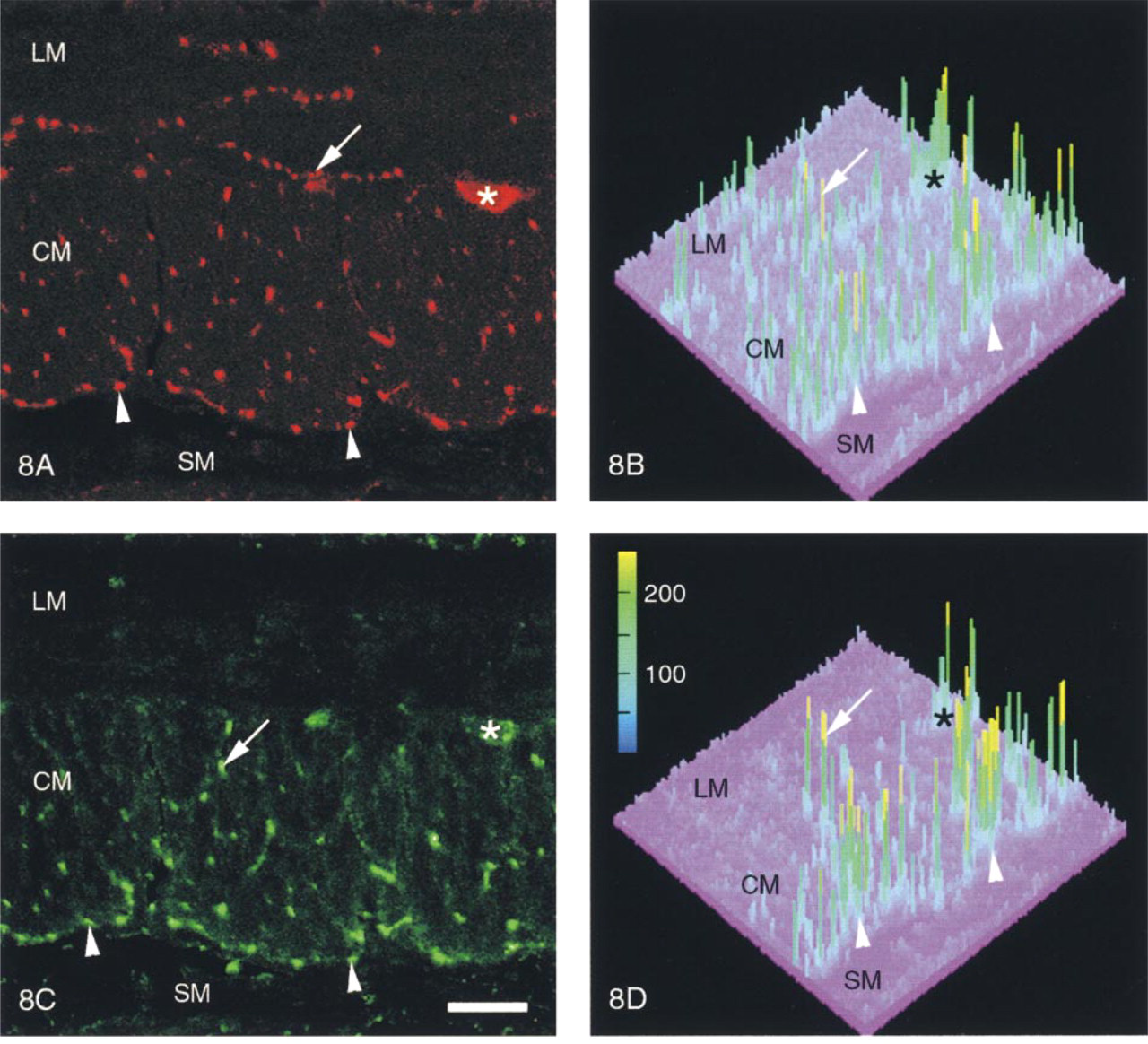
Synaptophysin (SYN-IR, Figures 1A-4A) and enkephalin (ENK-IR, Figures 1B-4B) immunoreactivity in the same transverse section of the fundus (Figure 1), the duodenum (Figure 2), the proximal colon (Figure 3), and the rectum (Figure 4) of the pig. Note the presence of ENK-IR in all the myenteric plexuses (large arrows) and the external submucous plexuses (Figures 1B-4B, small arrows). In the internal submucous plexuses revealed by SYN-IR (Figures 1A-4A, double arrows) ENK-IR was not detected (Figures 1B-4B, double arrows). ENK-IR was contained in endocrine cells (Figures 2B and 4B, arrowheads). Conventional fluorescent microscopy. Bars = 200 μm.
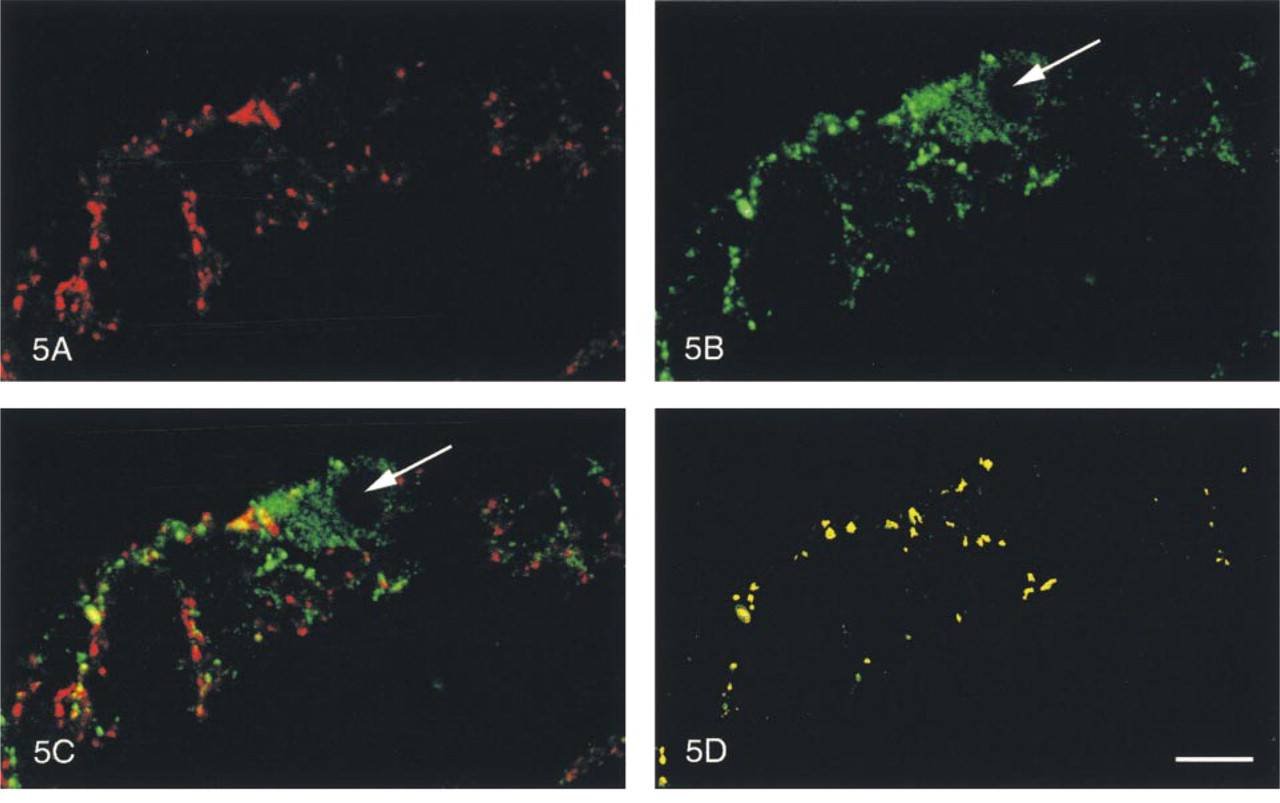
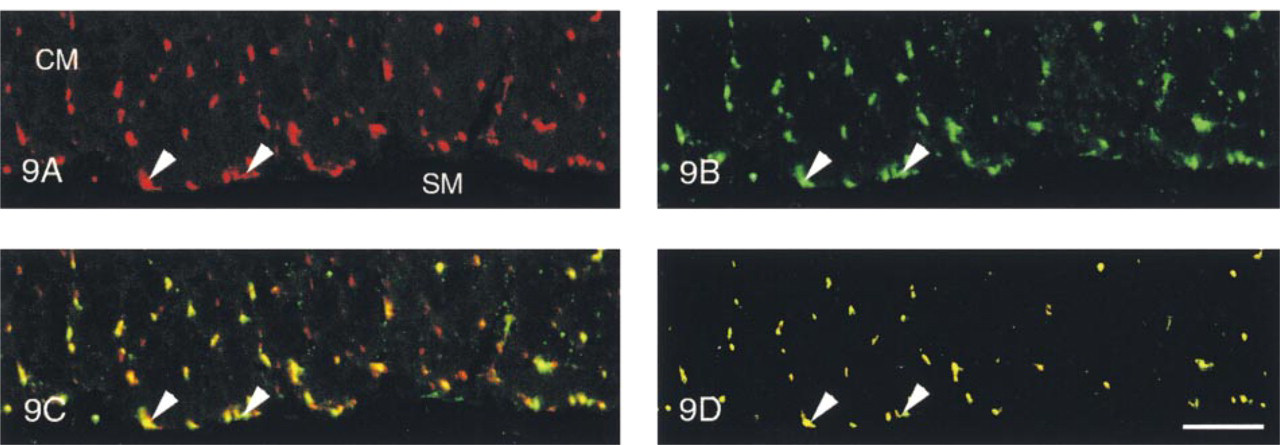
Myenteric plexus (Figure 5) and external submucous plexus (Figure 6) of the pig duodenum. In the same optical section, SYN-IR (Figures 5A and 6A), ENK-IR (Figures 5B and 6B), both SYN-IR and ENK-IR (Figures 5C and 6C), and only co-localized SYN-IR and ENK-IR materials (Figures 5D and 6D) were visualized, respectively. Note that ENK-IR neurons (arrows) were surrounded by co-localized ENK-IR and SYN-IR nerve fibers. Confocal microscopy. Bars:
Quantitative Analysis of SYN-IR and ENK-IR Nerve Fibers
The results of the quantitative analysis are summarized in Figure 7. They are expressed by the ratio of ENK-IR vs SYN-IR mean area ± SEM as percentages, which will be designated percentage of ENK-IR in nerve fibers. Apart from the fact that no ENK-IR was observed in the longitudinal muscle layer of the duodenum, we found that the percentage of ENK-IR was comparable, for a given structure, in the three regions studied. The percentage of ENK-IR was quite high (roughly 90%) in the circular muscle layer and the external submucous plexuses, lower in the longitudinal muscle layer (around 55%), and particularly high (approximately 200%) in the myenteric neural plexuses.
In each region studied, the patterns of distribution of the SYN-IR and ENK-IR nerve fibers were similar (Figures 8A and 8C). Although the distribution of SYN-IR and ENK-IR nerve fibers was relatively homogeneous in the longitudinal and circular muscle layers, the density of the immunoreactive materials revealed in nerve varicosities varied considerably, as demonstrated by the three-dimensional representation of the gray value levels of immunoreactive materials (Figures 8B and 8D). In the entire myenteric and external submucous plexuses, many SYN-IR and ENK-IR nerve fibers showed very high gray value levels. It is worth mentioning that in the duodenum, high gray value levels of SYN-IR and ENK-IR were observed in most of the nerve fibers belonging to the deep muscle plexus (Figures 8B and 8D).
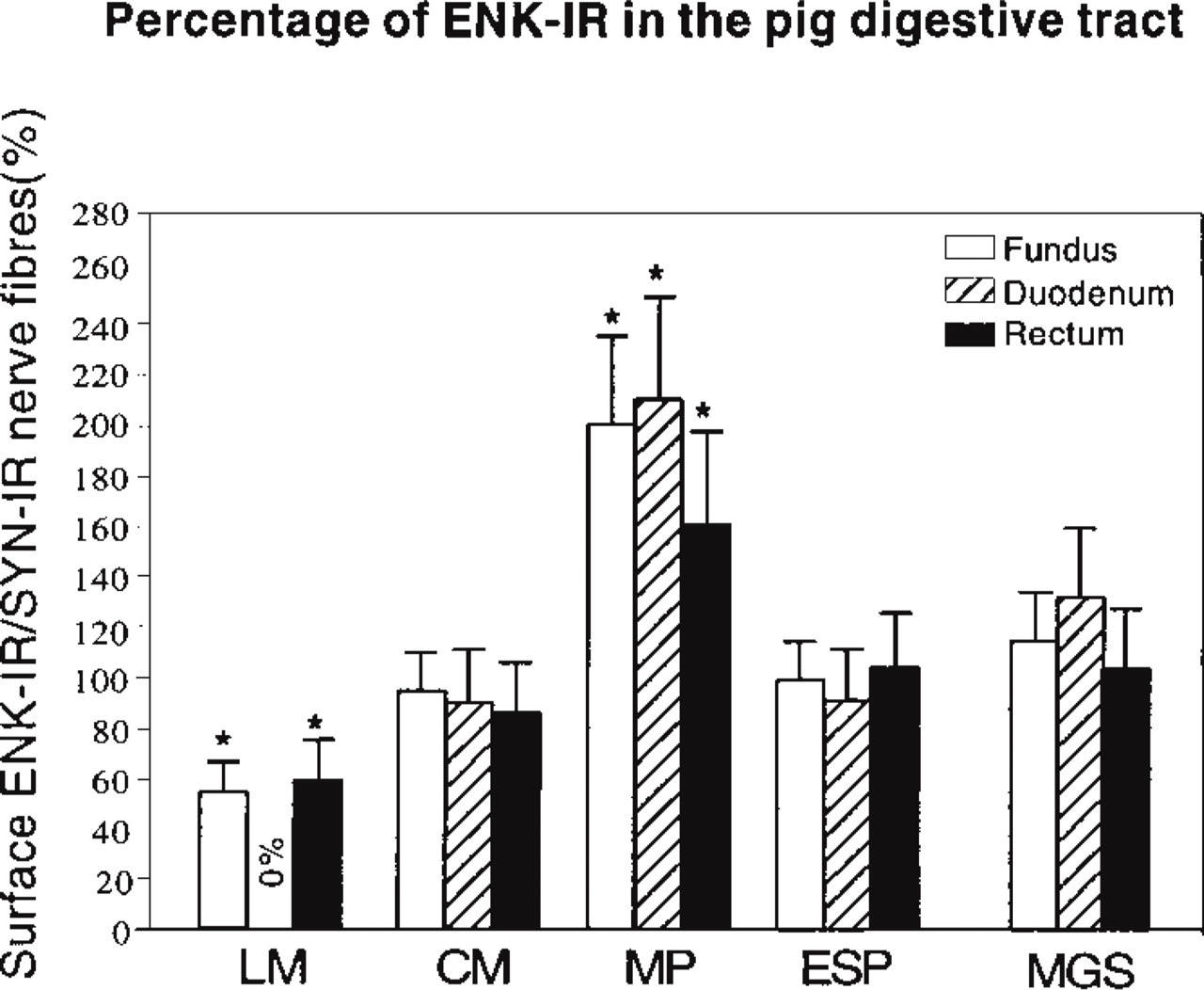
Mean ± SEM of area of ENK-IR vs SYN-IR nerve fibers in the longitudinal muscle (LM), circular muscle (CM), myenteric plexus (MP), external submucous plexus (ESP), and the whole muscle and ganglionic structures (MGS) of the fundus, duodenum and rectum of the pig. The percentage of ENK-IR was lower (∗p<0.05) in the longitudinal muscle layer but higher (∗p<0.05) in the myenteric plexus than in the other structures studied, whatever the region of the digestive tract.
Co-localization of SYN-IR and ENK-IR
Qualitative Aspects. In all three regions of the digestive tract studied here (fundus, duodenum, and rectum), co-localization of SYN-IR and ENK-IR materials was found to occur in many of the nerve fibers located in the myenteric (Figure 5) and external submucous plexuses (Figure 6), and in the external smooth muscle layers (Figure 9). For the neural plexuses, ENK-IR and SYN-IR materials were co-localized along the nerve fibers surrounding both ENK-IR and non-ENK-IR neurons (Figures 5 and 6). In the nerve fibers supplying the smooth muscle layers, SYN-IR and ENK-IR were evenly co-localized (Figure 9). In the duodenum, the SYN-IR and ENK-IR co-localization was particularly evident in the deep muscle plexus (Figure 9).
Quantitative Aspects. The results are summarized in Figure 10, and are expressed as the ratio of SYN-IR and ENK-IR co-localized mean area vs SYN-IR mean area ± SEM as percentages.
The rate of occurrence of nerve fibers in which both SYN-IR and ENK-IR co-localized was higher than 50% in all the smooth muscle layers and the myenteric and external submucous plexuses of the regions studied (Figure 10). In the longitudinal muscle layer, apart from the duodenum, which showed no ENK-IR nerve fibers, comparable percentages of co-localized SYN-IR and ENK-IR were obtained in the fundus and the rectum. In the circular muscle layer and in the myenteric and submucous plexuses, similar percentages of co-localized SYN-IR and ENK-IR were observed.
Discussion
The present study on the distribution of the ENK-IR in piglet digestive tract shows how richly the muscle coat and associated ganglionic plexuses are endowed with enkephalinergic innervation and how rarely (if at all) this innervation occurs in the mucosa. Our results corroborate data previously obtained on the esophagus (Uddman et al. 1980), the stomach, and the small intestine (Alumets et al. 1978; Scheuermann and Stach 1985; Scheuermann et al. 1990; Timmermans et al. 1990; Shen et al. 1993) of pigs, as well as those of other species such as guinea pigs (Schultzberg et al. 1980; Furness et al. 1983; Pfannkuche et al. 1998), rats (Schultzberg et al. 1980), cats (Bagnol et al. 1997), and humans (Polak et al. 1977; Larsson et al. 1987; Crowe et al. 1992).
The finding that ENK-IR is present in the neurons of the external submucous plexuses throughout the digestive tract and that it is absent in the internal submucous plexuses indicates that there may be differences in the neurochemical coding processes at work in these two types of plexuses, as was previously found in the pig small intestine not only for enkephalins but also for other neurotransmitters (Timmermans et al. 1990). Differences have also been observed in the patterns of distribution of various neurotransmitters between the two submucous plexuses in human colon (Crowe et al. 1992). All in all, these data strongly suggest that in both pigs and humans, the submucous plexuses are separate entities that have different functions.
Distribution of SYN-IR (
Circular muscle layer of the pig duodenum. In the same transverse optical section, SYN-IR (
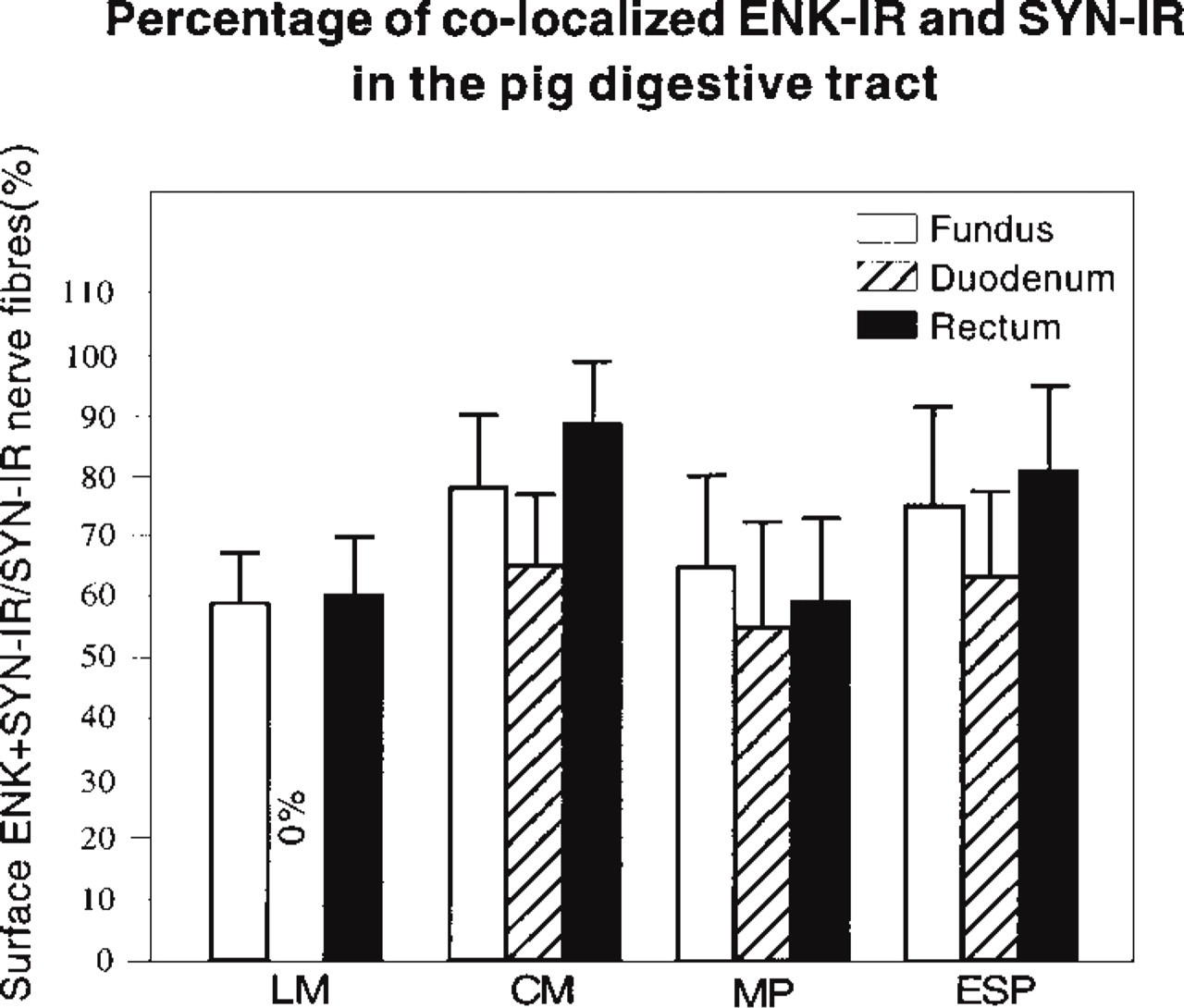
Mean ± SEM of surface of co-localized ENK-IR and SYN-IR in nerve fibers located in the longitudinal muscle (LM), the circular muscle (CM), the myenteric plexus (MP), and the external submucous plexus (ESP) of the fundus, the duodenum, and the rectum of the pig, respectively.
The sparsity or complete absence of ENK-IR observed here in the internal submucous plexuses, the muscularis mucosae, and the mucosa throughout the pig digestive tract is in line with previous findings on the plexuses (Timmermans et al. 1990) and the mucosa of the porcine small intestine (Vento et al. 1998). It therefore appears most likely that, under normal conditions, the enkephalins present in the intramural nervous system may not participate in the absorptive and secretory activity occurring in the intestinal epithelium, whereas the existence of ENK-IR in the enterochromaffin cells (Alumets et al. 1983; Ito et al. 1985) and that of opioid binding sites in the basal parts of villous and crypt cells (Quito et al. 1991) argue in favor of the idea that opiates may play a paracrine role, which would explain the effects they exert on intestinal absorption and secretion in pigs (Ahrens and Zhu 1982). However, given the presence of a few rare enkephalinergic nerve fibers in the internal submucous plexuses and the mucosa, it has been suggested that the neural opioid network may be involved in functional regulation of the mucosa (Timmermans et al. 1997).
We observed no enkephalins in the longitudinal muscle layer throughout the small intestine and colon, although they were present in the longitudinal muscle layer of the upper (esophagus and stomach) and lower (rectum, internal anal sphincter) parts of the digestive tract. A similar pattern of ENK-IR distribution has been observed in cats (Bagnol et al. 1997). Our comparative analysis on the ENK-IR vs SYN-IR patterns of distribution provides evidence that the lack of ENK-IR in the longitudinal muscle layer is not due to a lack of innervation, because many SYN-IR nerve fibers were detected in this layer. Our results therefore indicate that, in the pig small intestine and colon, the enkephalinergic myenteric neurons do not project to the longitudinal muscle layer. Regarding the presence of ENK-IR nerve fibers in the longitudinal muscle layer of the upper and lower regions of the digestive tract, futher investigations will soon be performed to determine their respective origins. In the circular muscle layer, an abundant enkephalinergic innervation was observed here throughout the gastrointestinal tract. These findings are in line with those obtained on other species such as guinea pigs, rats (Schultzberg et al. 1980), and cats (Bagnol et al. 1997). The pattern of enkephalinergic innervation described here in the circular muscle layer was perfectly homogeneous throughout the entire digestive tract except for the small intestine, in which it showed an increasing gradient, working towards the deep muscle plexus. This plexus, which was recently studied at the ultrastructural level in the pig small intestine (Henry et al. 1998), might participate in the control of motility via interstitial cells of Cajal that are interposed between the nerve fibers and the smooth muscle fibers (Hara et al. 1986; Torihashi et al. 1993). In the light of these previous studies and the present data showing the abundance of enkephalinergic innervation in the deep muscle plexus, it appears likely that enkephalins may be involved via the interstitial cells of Cajal in the neuroregulatory mechanisms mediating the motility of the small intestine.
The presence of enkephalinergic neurons in the myenteric plexuses, which has thus far been observed mainly in the pig small intestine (Scheuermann et al. 1990; Timmermans et al. 1990) and esophagus (Uddman et al. 1980), was confirmed here at all levels of the gastrointestinal tract studied. Enkephalinergic neurons are likely to occur in the myenteric plexuses of all the species studied thus far (for reviews see Furness and Costa 1987; Kromer 1988).
The intrinsic origin of the enkephalinergic innervation in the pig digestive tract has been established by performing complete extrinsic denervation by autotransplantation (Malmfors et al. 1981; Shen et al. 1993). However, because enkephalins exist in some of the neurons present in the prevertebral sympathetic ganglia of pigs (Majewski et al. 1991), as well as in those of other species such as cats (Julé et al. 1986; Balayadi et al. 1988; Bagnol et al. 1993) and bovines (Fried et al. 1986), the possibility that the enkephalinergic innervation of the pig digestive tract may be also partly of extrinsic origin cannot be ruled out.
The results of our quantitative study of the patterns of ENK-IR and SYN-IR in the digestive tract provide more detailed information than that previously available about the density of the enkephalinergic innervation, based simply on visual assessments. The present approach to quantifying the intramural enkephalinergic innervation is complementary to the biochemical approach (RIA) with which it is possible to determine the enkephalinergic content of intestinal samples including the external muscle layers and the myenteric plexus (Cupo et al. 1988; Bagnol et al. 1993; Herbrecht et al. 1995). It is noteworthy that, unlike the biochemical approach, this immunohistochemical approach gave a sufficiently detailed picture of the density of the enkephalinergic innervation that we were able to compare the various muscle and ganglionic structures composing the digestive wall at various gastrointestinal levels.
It has been established on the basis of ultrastructural data that synaptophysin labels the small clear vesicles present in nerve fibers (Navone et al. 1986). On the other hand, the ENK-IR fibers in the cat digestive tract are characterized by the presence of both large, dense ENK-IR vesicles and small, clear non-ENK-IR vesicles (Larsson and Stengaard-Pedersen 1982). This set of structural data makes it possible to compare the enkephalinergic innervation with the intramural innervation of the digestive tract as a whole, i.e., that which can be determined by performing synaptophysin labeling. Given the limits of confocal microscopy as a means of detection, it was not possible to determine the density of the enkephalinergic innervation on the basis of the number of nerve varicosities, which is why we focused rather on the area of the ENK-IR and the area of the coexisting ENK-IR and SYN-IR, both referred to the area of the SYN-IR. The present results show that there exist similar areas of ENK-IR in the nerve fibers in the muscle layers and the external submucous plexus, but that these areas were much larger in the myenteric plexuses in all three regions of the digestive tract studied. The particularly high values detected in the myenteric plexuses are probably attributable to the fact that greater numbers of large dense vesicles containing enkephalins may have accumulated at this level and fewer of the small clear vesicles which are immunoreactive to synaptosin. The enkephalinergic contents of the nerve fibers located in the myenteric plexuses therefore appear to be distinctly more abundant than those of the fibers located in the external submucous plexuses and the muscle layers. Our comparative analysis of the patterns of co-localized ENK-IR and SYN-IR shows that comparable areas of the muscle and ganglionic structures were occupied by both types of immunoreactivity in all three regions studied. This strongly suggests that the proportion of the enkephalinergic nerve fibers is very likely comparable in all parts of the intestinal wall as well as in all the regions studied here.
The qualitative and quantitative data obtained in this study on the enkephalinergic innervation of the pig gastrointestinal tract should provide a valuable basis for future studies on the motor deficits associated with various diseases that affect the digestive tract in large mammals, including humans.
Footnotes
Acknowledgments
We wish to express our sincere thanks to Mireille Richelme and Jean Claude Stamegna for their excellent technical assistance.