Abstract
Proteins on the membrane of secretory granules (SGs) involved in their biogenesis and exocytosis are poorly characterized compared with those of synaptic vesicle in neurons. Thus the secretory granule membrane was prepared from a mouse pancreatic β-cell line MIN6 by subcellular fractionation, and protein constituents were analyzed by microscale two-dimensional liquid chromatography coupled with electrospray ionization tandem mass spectrometry. Using this proteomics approach, one of the p24 family proteins, p23, was unexpectedly found in the granule fraction, although p24 proteins are generally regarded as functioning in the early secretory pathways between the endoplasmic reticulum and the Golgi apparatus. We further showed that p23 is expressed at high levels in endocrine cells. Furthermore, immunocytochemical analyses of pancreatic β-cells at the light and electron microscopic levels demonstrated that a significant amount of p23 is localized on the insulin granule membrane, although it is most intensely concentrated at the cis-Golgi compartment as previously shown in non-endocrine cells. These findings suggest that a fraction of p23 enters post-Golgi compartments and may function in the biogenesis and/or quality control of SGs.
A
Previously characterized secretory granule membrane (SGM) proteins include vacuolar H+-ATPase, which controls the lumenal environment (Schoonderwoert and Martens 2001), and several molecules involved in the processing of peptide hormones such as prohormone-converting enzymes (Nakayama 1997), carboxypeptidase E (Fricker 1988), and peptide amidating monoxygenase (Eipper and Mains 1988). Although homolog searches of proteins that exist on the membrane of synaptic vesicles have enabled the detection of molecules such as Rab3, synaptotagmins, and synaptobrevin-2 as SGM proteins, these proteins should play roles common to synaptic vesicles and SGs but not those specific to the granules. Monoclonal or polyclonal antibodies raised against SGM preparations have been used to identify granule proteins such as IA-2 and phogrin (Passini et al. 1995; Wasmeier and Hutton 1996). Alternatively, granuphillin and its associated protein Rab27a were discovered by using mRNA differential display and a further examination of various interacting molecules (Wang et al. 1999; Yi et al. 2002). These approaches, however, have not been sufficient to elucidate the whole molecular machinery located on the SGs.
Recently, subcellular proteomics, which combines subcellular fractionation and mass spectrometry, has become a powerful approach to identify the protein complement of cellular compartments. This approach has been applied to biochemically isolated organelles and major suborganellar structures such as mitochondria (Taylor et al. 2003), lysosomes (Journet and Ferro 2004), endoplasmic reticulum (ER)-Golgi intermediate compartment (Breuza et al. 2004), Golgi apparatus (Wu et al. 2000), clathrin-coated vesicles (Blondeau et al. 2004; Ritter et al. 2004), and postsynaptic density (Yoshimura et al. 2004). Each study has identified known proteins not previously understood to associate with the compartment as well as novel proteins described only as predicted open-reading frames from genome-sequencing data.
In the present study we utilized this approach to analyze the protein components of SGM isolated from mouse pancreatic β-cell line MIN6. Our analysis produced the unexpected finding that p23 (also called p24δ, p24c, or Tmp21) is localized on the SGs, although the p24 family proteins including p23 are generally known to be associated with COPI- (Stamnes et al. 1995; Sohn et al. 1996) and/or COPII-coated transport vesicles (Schimmöller et al. 1995; Belden and Barlowe 1996) that cycle between the ER and the Golgi apparatus. We further showed that p23 is expressed at the highest level in endocrine tissues. Furthermore, morphological analyses demonstrated that p23 is actually localized on the membrane of insulin-containing SGs of MIN6 cells and pancreatic β-cells, although most p23 protein is concentrated on the cis-Golgi compartment as previously described in non-endocrine cells (Rojo et al. 1997). Based on the findings, we discuss possible roles of p23 on SGs.
Materials and Methods
Subcellular Fractionation of SGs
MIN6 cells were maintained in DMEM supplemented with 15% fetal bovine serum and 100 μM 2-mercaptoethanol. MIN6 cells in 100-mm plastic plates were detached by a 20-min incubation with PBS containing 10 mM EDTA at 37C. Cells were sedimented by light centrifugation and homogenized in buffer A (250 mM sucrose; 4 mM HEPES, pH 7.4; 1 mM MgCl2; 0.005% DNAase; and a protease inhibitor mixture [1 μg/ml each of aprotinin, leupeptin, and pepstatin A, and 0.5 mM phenylmethylsulfonyl fluoride]). The homogenate was centrifuged at 3000 × g for 2 min, and the resulting supernatant was recentrifuged at 5000 × g for 15 min at 4C. This postnuclear supernatant was centrifuged at 26,000 × g for 15 min. The resultant pellet, the crude organelle fraction, was suspended in 0.8 ml of buffer A with 1.5 mM EDTA and layered onto a sucrose density gradient (20-70%, w/v), which was formed using a tilt tube in a Gradient Master (BioComp Instruments; Fredericton, NB, Canada) as described previously (Hosaka et al. 2004). After centrifugation at 113,000 × g for 18 hr at 4C in a swing rotor, gradients were fractionated by piston displacement from top to bottom using the BioComp Piston Gradient Fractionator. Fractionated samples were concentrated for gel analysis by adding trichloroacetic acid to 7.5% and BSA to 0.01%. Immunoblot analyses of these fractions were performed as described previously (Hosaka et al. 2004).
To prepare the SGM fraction, peak fractions of immunoreactive insulin were diluted and homogenized in a 25-fold volume of buffer B (20 mM HEPES, pH 7.4; 1 mM MgCl2; 1 mM dithiothreitol; and the protease inhibitor mixture) and centrifuged at 100,000 × g for 2 hr at 4C. The pellet was washed twice with buffer B containing 0.15 M NaCl and recentrifuged at 130,000 × g for 2 hr at 4C. The resulting pellet was used for mass spectrometric analysis.
Protein Identification by Automated Two-dimensional Liquid Chromatography (2DLC)-Tandem Mass Spectrometry (MS/MS) and Data Analysis
Purified SGM (50-μg proteins) was dissolved in 1 ml of 7.0 M guanidine-HCl buffered with 0.5 M Tris-HCl, pH 8.0, containing 10 mM EDTA. The preparation was reduced by the addition of 1 mM dithiothreitol and S-carbamoylmethylated with 10 mM iodoacetamide under a nitrogen atmosphere as described previously (Mawuenyega et al. 2003). The S-carbamoylmethylated proteins were subjected to an automated microscale 2DLC-MS/MS system and analyzed as described previously (Isobe et al. 2003; Mawuenyega et al. 2003). The results were extracted using an in-house program ‘stem’ and imported into Microsoft Excel software for further analysis. We first screened the candidate peptides based on a probability-based Mowse score and finally selected the ‘hit’ peptides under the criteria as described previously (Yoshimura et al. 2004).
Molecular Cloning of p23 and Northern Blot Analysis
Mouse p23 was obtained by polymerase chain reaction with oligonucleotide primers containing flanking restriction sites (GAGGAATTCATGTCTGGTTTGTTTGGCCC and CGCGTCGACCTCTATCAACTTCTTGGCC) using MIN6 cDNA as a template.
For Northern blot analysis, total RNAs (10 μg) from mouse tissues and culture cells were electrophoresed on an agarose gel, transferred to a nylon membrane, and hybridized with a 32P-labeled cDNA probe encoding the full-length p23.
Antibodies
Two kinds of rabbit anti-p23 antibodies were generated: one against glutathione S-transferase-fused mouse p23 protein (41-181 amino acids, code #TP01) and the other against a synthetic peptide corresponding to amino acids 43-58 of p23, CLREEIHKDLLVTGAY (code #TP02). These antisera were further purified by affinity chromatography using the corresponding antigens bound to Affi-gels (Bio-Rad; Hercules, CA). The rabbit antibodies against secretogranin III (SgIII) and phogrin were as previously described (Sakai et al. 2003; Hosaka et al. 2004; Torii et al. 2005). The rabbit antisera against β-COP and synaptobrevin-2 (also called VAMP-2) were purchased from BD Biosciences (San Jose, CA) and Wako Pure Chemical (Osaka, Japan), respectively. Mouse monoclonal antibodies against Rab27a, calnexin, cathepsin D, cytochrome C, γ-adaptin, TGN38, and GM130 were from BD Biosciences. Mouse monoclonal antibodies against synaptophysin and β-actin were from Progen Biotechnik (Heidelberg, Germany) and Sigma-Aldrich (St Louis, MO), respectively. Guinea pig anti-insulin antibody for immunoreactive insulin measurement was a gift from H. Kobayashi (Gunma University). To immunocytochemically identify the endocrine cell types in pancreas, guinea pig anti-insulin and rabbit anti-glucagon and anti-somatostatin polyclonal antibodies were obtained from DakoCytomation (Copenhagen, Denmark).
Immunofluorescence Microscopy
Male Wistar rats were used in accordance with the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources, National Research Council, Washington, DC, 1996) under the permission of the experimental animal welfare committee of Asahikawa Medical College (permission #04028). At 10 weeks of age, rats (n=5) were anesthetized with pentobarbital (25 mg/kg; IP) and were perfused with 50 ml of physiological saline followed by 250 ml of 0.1% glutaraldehyde-4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.2. Pancreatic tissues were excised, cut into small pieces, and immersed in the same fixative for 2 hr at 4C. They were then washed thoroughly with the phosphate buffer containing 7.5% sucrose, dehydrated in graded ethanol, and embedded in epoxy resin (Epon 812). Serial sections of 0.5-μm thickness were cut from the tissue blocks with an ultramicrotome and mounted on microscopic slides. After removal of the resin by sodium methoxide (Grube and Kusumoto 1986), semithin sections were incubated with a mixture of a rabbit anti-p23 antibody (#TP01 or #TP02) and a guinea pig anti-insulin serum for 18 hr at 4C. They were then incubated for 1 hr at 20C with a mixture of Alexa Fluor 488-labeled donkey anti-rabbit IgG and Alexa Fluor 594-labeled donkey anti-guinea pig IgG secondary antibodies (Molecular Probes; Eugene, OR). Double staining for p23 and Golgi markers (GM130 or TGN38) was similarly performed with Alexa Fluor 594-labeled donkey anti-mouse IgG. After coverslips were mounted in 90% glycerol (v/v in phosphate-buffered saline) containing 0.1% p-phenylenediamine dihydrochloride (Sigma-Aldrich), the sections were observed with an epifluorescence microscope (Olympus; Tokyo, Japan).
Immunoelectron Microscopy
MIN6 cells grown in 100-mm dishes were fixed with 0.1% glutaraldehyde-4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.2, for 10 min and subsequently with 4% paraformaldehyde in the phosphate buffer containing 3% sucrose for 1 hr at 4C. The cells were gently scraped from dishes and microcentrifuged to pellets, which were dehydrated with 70% ethanol and infiltrated into pure LR White resin (London Resin; Hampshire, UK) for 12 hr at 4C. The pellets were placed in gelatin capsules with fresh LR White resin and polymerized for 24 hr at 60C.
Pancreatic tissues were fixed and excised as described above. They were then washed with 0.1 M phosphate buffer containing 7.5% sucrose and incubated with 0.2% OsO4 in the same buffer for 1 hr at 4C. After postfixation with diluted OsO4, tissue blocks were washed thoroughly with the same buffer, dehydrated three times in 70% ethanol containing 1% phosphotungstic acid for 20 min at 4C, and then embedded in LR white resin as described previously (Sakai et al. 2005).
Immunogold labeling was performed as described previously (Sakai et al. 2003). For removal of osmium, ultrathin sections were treated in 1% sodium metaperiodate (Wako Pure Chemical) for 10 min prior to the immunogold labeling. Then the sections were incubated for 12 hr at 4C with the anti-p23 antibody (#TP01 or #TP02). For double immunostaining, the two-face technique (Bendayan 1982) was applied. Intracellular localizations of p23 and other molecules were distinguished by labeling with different sizes of colloidal gold particles (size of particles: 5 or 15 nm in diameter) conjugated to secondary antibodies (British Biocell International; Cardiff, UK). After the immunolabeling, the sections were contrasted with saturated aqueous solutions of uranyl acetate and lead citrate and examined with an electron microscope (JEM-1010; JEOL, Tokyo, Japan). To evaluate relative labeling densities of immunogold particles on intracellular organelle, rat pancreatic β-cells were randomly photographed at a magnification × 12,000 after immunostaining for p23 or GM130 (20 micrographs per each immunocytochemical staining). The number of immunogold particles localized on the membranes of the cis-Golgi compartment, SGs, rough ER, and mitochondria was separately counted, and the number was then divided by the total profile length of the membrane of the corresponding organelle in the observed fields. The results were statistically analyzed by one-way ANOVA followed by Tukey HSD posthoc test with significance level of p<0.05 (KaleidaGraph software version 3.6; Synergy Software, Reading, PA).
Results
Purification of SGM
To identify proteins on the membrane of insulin-containing granules, subcellular organelles of the mouse β-cell line MIN6 were separated using sucrose density gradient centrifugation. Fractions containing SGs were identified by measuring insulin concentrations (Figure 1A). Granule-associated proteins such as Rab27a, phogrin, and SgIII were concomitantly accumulated with insulin in fractions 10-12 (Figure 1B), although synaptobrevin-2 was only faintly detected in these fractions contrary to a previous finding in the rat β-cell line INS-1 (Regazzi et al. 1995). By contrast, markers for other organelles showed distinct localization patterns: calnexin for ER at fractions 7-9, β-COP for cis-Golgi at fractions 3-9, γ-adaptin for trans-Golgi network at fractions 3-9, and synaptophysin for synaptic-like microvesicles at fractions 2-6. The markers for lysosomes, cathepsin D, and for mitochondria, cytochrome C, were excluded from the starting crude organelle fraction. The buoyant density of fractions 10-12 was 1.176 ± 0.01 g/cm3 (mean of five independent experiments), which was similar to the values previously reported for SG fractions of PC12 and INS-1 cells (Stinchcombe and Huttner 1994; Hosaka et al. 2004). These fractions were lysed in hypotonic solution, washed with high-salt solution to detach nonspecifically associated proteins, and then separated by centrifugation into a membrane pellet and a soluble supernatant. As expected, peripherally granule-associated Rab27a and integral granule-membrane protein phogrin were recovered in the membrane fraction SGM (Figure 1C), whereas the granule cargo insulin was scarcely detected in this fraction (Figure 1D). Thus, the fraction concentrated the proteins localized on the membrane of SGs.
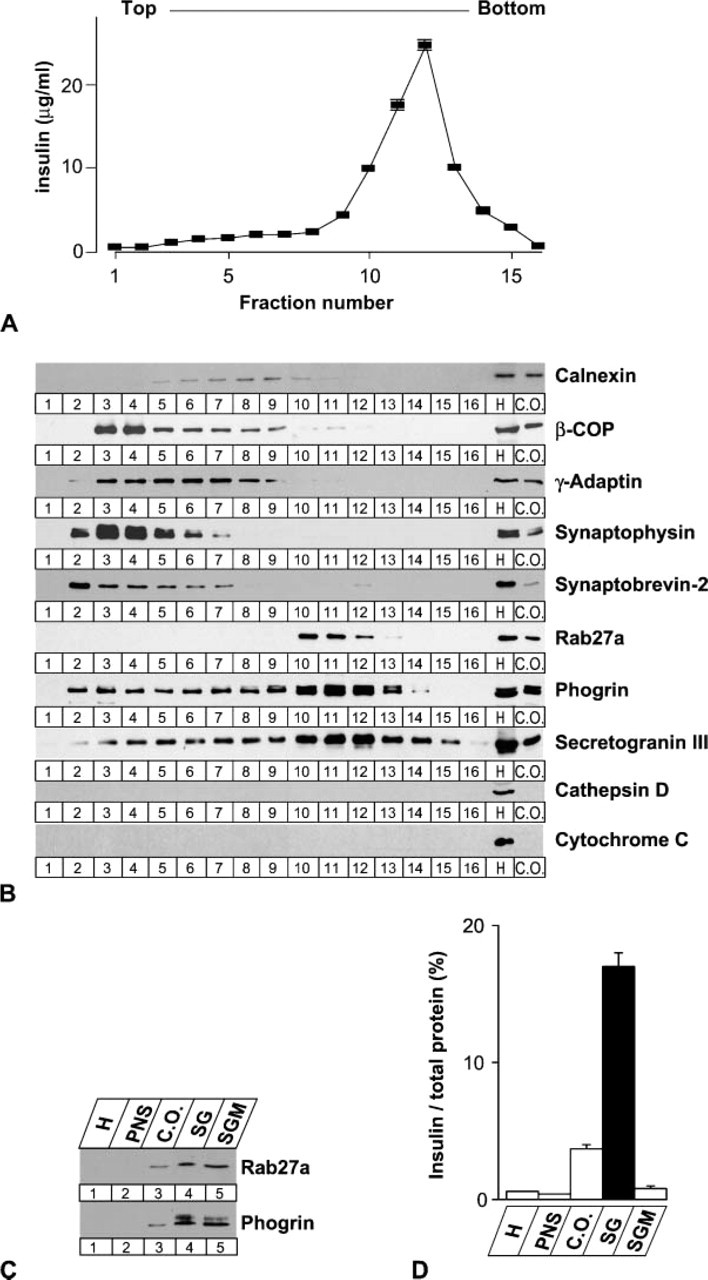
Fractionation of secretory granule (SG) components in MIN6 cells. MIN6 cell homogenate (H), postnuclear supernatant (PNS), and crude organelle fraction (C.O.) were obtained by serial centrifugation as described in Materials and Methods. The crude organelle fraction was further divided into 16 fractions using sucrose density gradient centrifugation. (
SGM fraction was solubilized in 6 M guanidium hydrochloride and digested with trypsin after S-carbamoylmethylation under reducing conditions. The tryptic peptide mixture was then analyzed by an integrated liquid chromatography-based protein identification system, which comprises microscale 2DLC, electrospray ionization MS/MS, and an automated data analysis system (Isobe et al. 2003). As a result, 1994 peptides derived from 550 proteins were assigned, including established granule-resident proteins such as prohormone-converting enzymes. The list would provide a molecular basis for future functional studies on the biogenesis, sorting, and exocytosis of SGs.
Enrichment of p23 in Endocrine Cells
In the present study we focused on one of the p24 family proteins (Emery et al. 1999), p23, which was identified at multiple times in the SGM fraction by our mass spectrometry system. This finding was unexpected because p24 proteins are generally known to associate with COPI (Stamnes et al. 1995; Sohn et al. 1996) or COPII vesicles (Schimmöller et al. 1995; Belden and Barlowe 1996) that cycle between the ER and Golgi apparatus. We first examined the expression of p23 by Northern blot analysis (Figure 2A). mRNA for p23 was highly expressed in adrenal gland, testis, and endocrine-derived cell lines AtT-20 and MIN6. It was moderately expressed in lung and kidney but was barely detectable in heart or liver.
For further analyses at the protein level, we generated two kinds of anti-p23 polyclonal antibodies: one against a glutathione S-transferase-fused mouse p23 protein and the other against a synthetic peptide corresponding to the N-terminal region. Both antibodies recognized a single band of 23-kDa protein in immuno-blots of tissue and cell extracts (Figure 2B). Furthermore, they recognized both endogenous and FLAG-tagged p23 expressed in COS-7 cells (Figure 2B, lower panel at Lane 6; asterisks). These findings indicate that both antibodies specifically recognize p23. The strength of the protein signal was highly correlated with the level of p23 transcripts (Figure 2A). The immunoblot also indicated the high expression level of p23 in pancreatic islets (Figure 2B, upper panel at Lane 9) and in the SGM fraction of MIN6 cells (Figure 2B, lower panel at Lane 7). Enrichment of p23 in endocrine cells suggests its involvement in the secretory processes.
Intracellular Localization of p23
We then examined the intracellular localization of p23 biochemically. Subcellular fractionation of MIN6 cell extracts revealed that p23 protein is broadly distributed in the fraction containing insulin granules (Figure 3), as well as in lighter fractions where the cis-Golgi marker β-COP was localized (see Figure 1B).
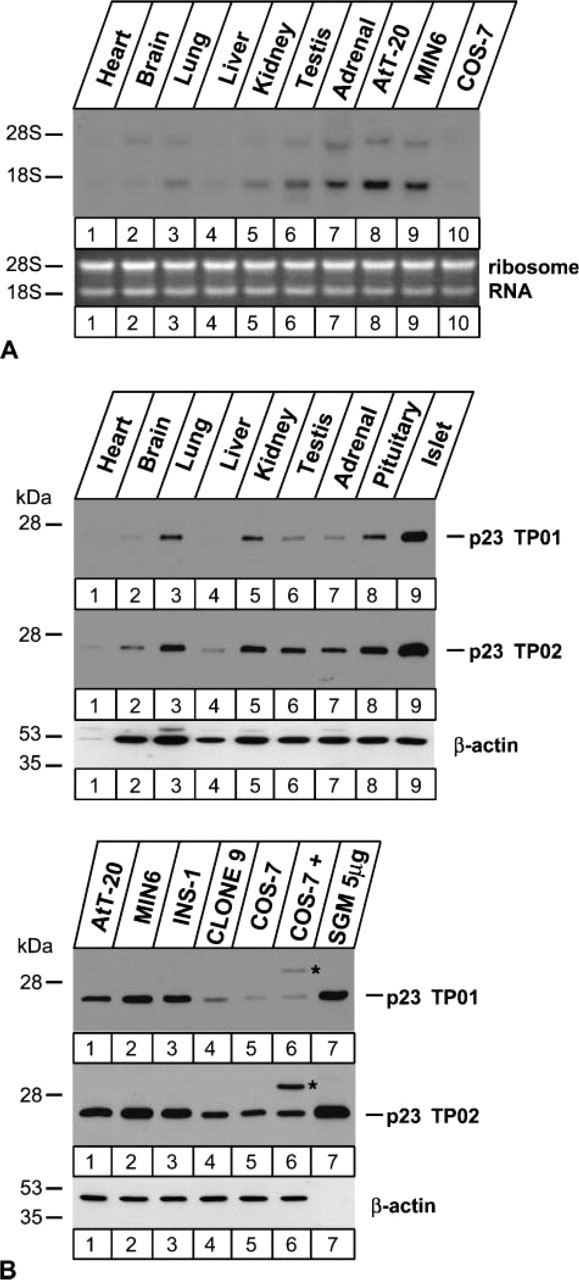
Tissue and cell distributions of p23. Expression of p23 was examined by Northern (
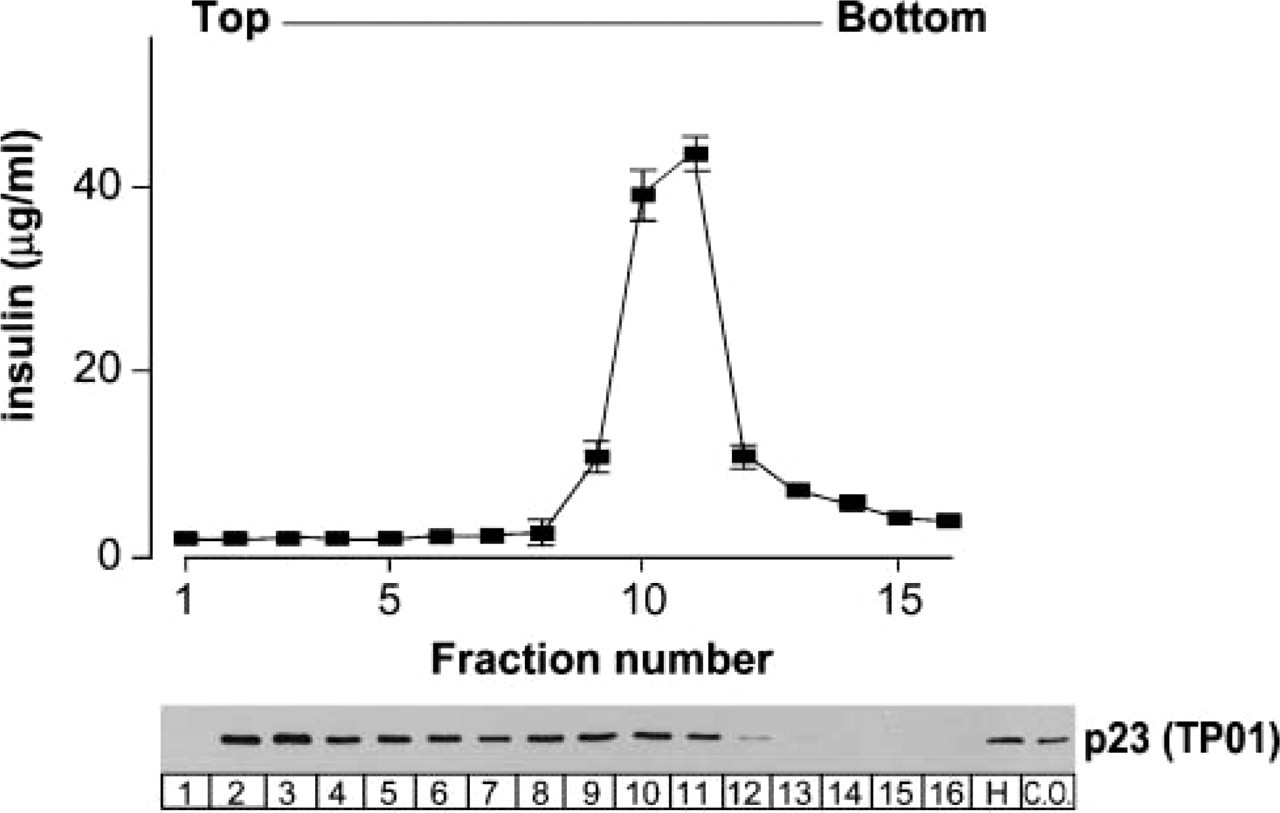
Subcellular fractionation of p23. Crude organelle fraction (C.O.) from MIN6 cell homogenate (H) was fractionated by sucrose density gradient centrifugation as in Figure 1. Insulin levels peaked at fractions 10-11. Distribution of p23 was analyzed by anti-p23 antibody (TP01).
We next performed immunofluorescence microscopic analysis in rat pancreas to examine the intracellular localization of p23 morphologically; p23 protein was intensely distributed in islets, whereas it was only faintly observed in exocrine acini (Figures 4A-4C). In the cytoplasm of the individual endocrine cells, conspicuous immunoreactivity for p23 was colocalized with that for GM130, a marker protein of the cis-Golgi network (Figure 4D), and adjacent to that for TGN38, a marker of the trans-Golgi network (Figure 4E). In addition, faint granular fluorescence immunopositive for p23 was also discerned in the cytoplasm (Figure 4B), suggesting that a relatively small amount of p23 was localized on the SGM. Identical findings were obtained with the two different anti-p23 antibodies (#TP01 and #TP02) and the reactions were specifically inhibited by adsorbing the antibodies with the corresponding antigens prior to the incubation for immunostaining (Figure 5).
To more precisely investigate the localization of p23, we next performed immunoelectron microscopic analyses. Immunogold particles indicative of p23 were significantly localized on the membranes of SGs in MIN6 cells (Figure 6A, arrowheads) and in pancreatic β-cells (Figures 6C and 6D, arrowheads), in addition to their marked accumulation around the Golgi apparatus (Figures 6B and 6D). Immunogold particles indicative of p23 at the Golgi apparatus were colocalized with those of GM130 (Figure 6E) but not with those of TGN38 (Figure 6F), indicating that p23 protein is predominantly accumulated in the cis-Golgi compartment as previously reported in non-endocrine cells (Rojo et al. 1997). Morphometrical analysis revealed that the relative density of p23 on SGs is much lower than that on the cis-Golgi but is significantly higher than that on mitochondria and on rough ER, whereas that for GM130 is restricted to the cis-Golgi compartment (Table 1). These findings indicate that p23 is not merely distributed in the pre-Golgi compartments but is transported to the SGM beyond the Golgi apparatus. In addition to β-cells, immunogold particles indicative of p23 were also observed on the Golgi apparatus of endocrine α- and δ-cells and exocrine acinar cells (Figures 7A-7C). SGM of these cells was barely labeled with immunogold particles indicative of p23. Specificity was confirmed at the electron microscopic level by the fact that no immunogold particles were observed by absorbing the antibodies with the corresponding antigens prior to the incubation for immunostainig (data not shown).
Discussion
In the present study we utilized subcellular proteomics to analyze the protein components of the SGM isolated from MIN6 cells and unexpectedly identified one of the p24 proteins, p23, in addition to the established granule-resident proteins such as prohormone-converting enzymes. We then showed that p23 is highly expressed in endocrine tissues and cells. The following immunocytochemical analyses revealed that p23 is localized not only at the cis-side of the Golgi apparatus as previously reported (Rojo et al. 1997), but also significantly on the membrane of SGs. It may be that the location of p23 on SGs was not previously established because most of the studies were performed in yeast or non-secretory mammalian cells that lack specialized secretory organelles.
Immunoreactivity for p23 by immunofluorescence microscopy was only faint on SGs in pancreatic β-cells, probably because the relative amount of p23 on the granules was much smaller than that on the cis-Golgi. The following immunogold labeling and morphometrical analysis at the electron microscopic level, however, confirmed the significant distribution of p23 on the SGM and revealed higher density on granules than on the rough ER. Although the p24 family proteins are believed to cycle in the early secretory pathway between the ER and the Golgi apparatus (Emery et al. 1999), p23 was absent on the rough ER but significantly labeled on the SG, suggesting that at least a subset of p23 cycles in the post-Golgi compartments. The location of p24 proteins beyond cis-Golgi compartment has also been shown for another p24 family protein; gp27 receives complex oligosaccharides with terminal sialic acid, characteristic of the glycosylating modification at the medial- and trans-Golgi compartments (Fullekrug et al. 1999).
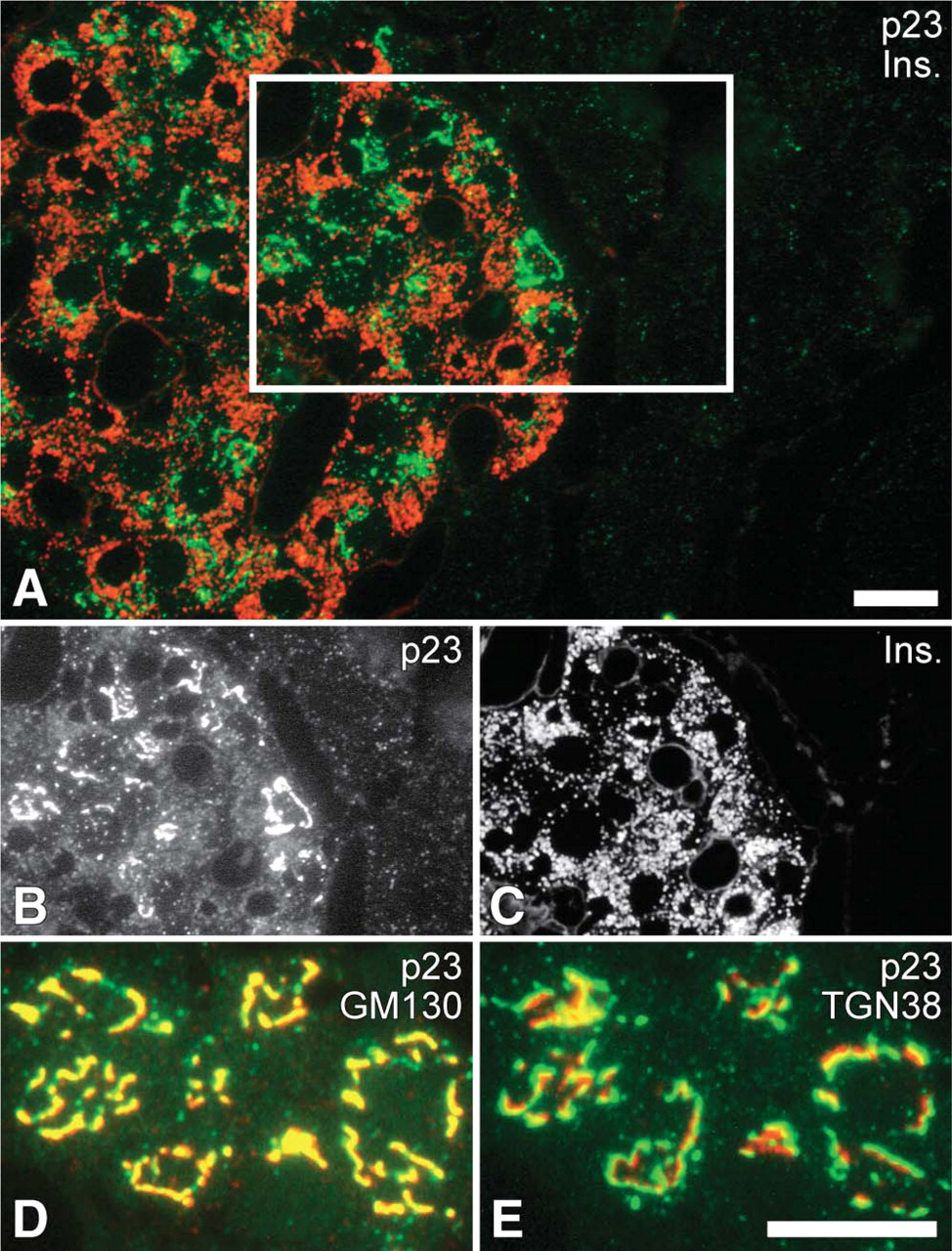
Immunocytochemical distribution of p23 in rat pancreas. (
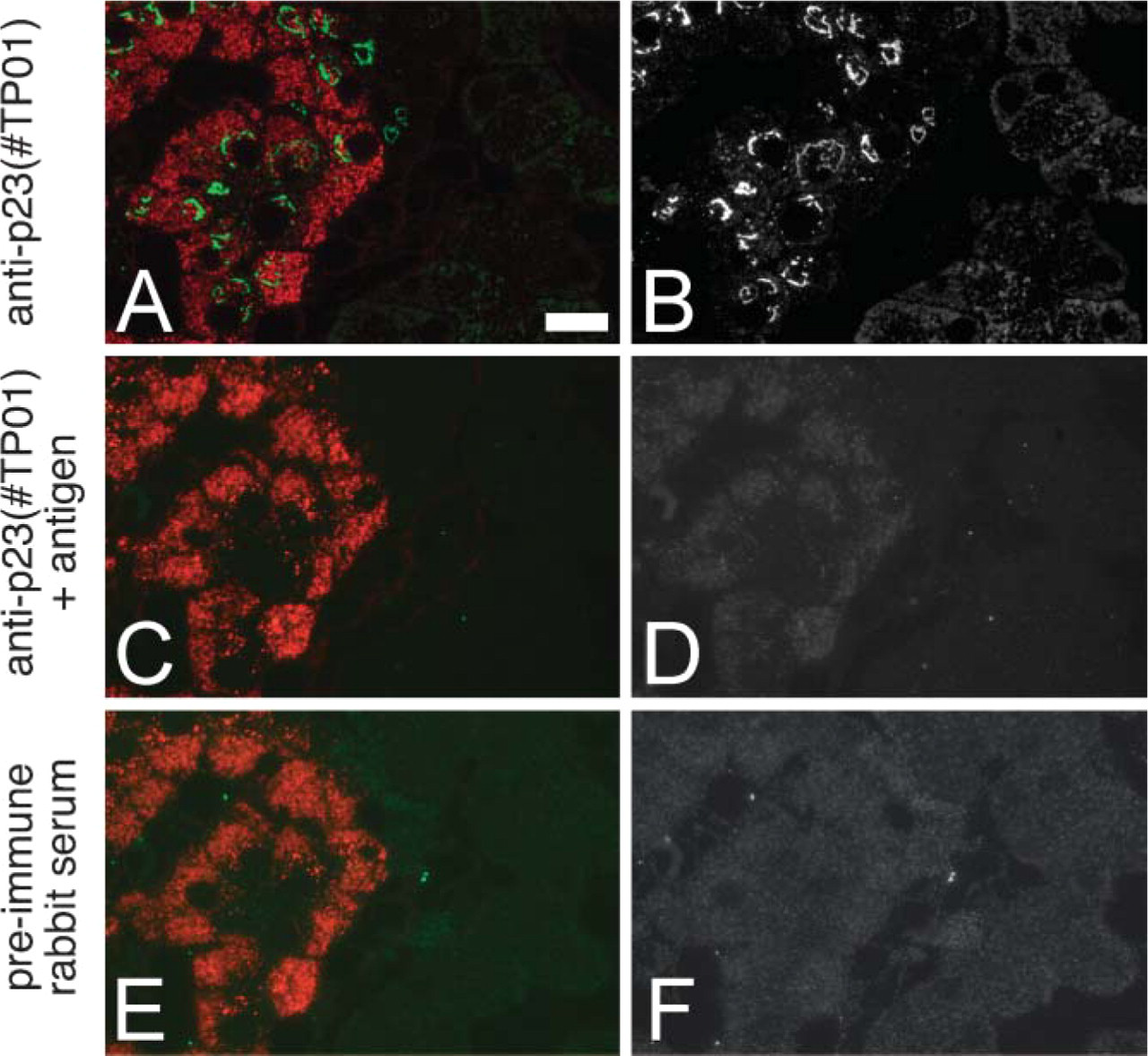
Specificity control of immunostaining. Three serial semithin section (0.5-μm thick) of rat pancreatic tissue (embedded in epoxy resin) were immunostained with the rabbit anti-p23 antibody used in Figure 4 (#TP01;
At present, the function of p24 proteins remains elusive even in the early secretory pathways, although they are generally proposed to function as cargo receptors/adaptors or quality control factors for the COPI and COPII vesicles. In yeast mutants lacking a single p24 molecule, distinct cargo proteins show delayed transport kinetics (Schimmöller et al. 1995; Elrod-Erickson and Kaiser 1996; Marzioch et al. 1999). However, an analysis using a yeast strain in which all eight members of the p24 family are deleted showed that p24 proteins are not essential for the function of either COPI or COPII vesicles (Springer et al. 2000). In contrast to yeast genetics, a disruption of both p23 alleles in mice results in early embryonic lethality (Denzel et al. 2000), which makes detailed analysis difficult. The high expression level of p23 in endocrine cells and its localization on SGs demonstrated in the present study suggest its function in some aspects of the secretory processes. Consistent with this notion, in the intermediate pituitary of Xenopus laevis, expression levels of the p23 homolog, p24δ, and propiomelanocortin concomitantly increased more than 25-fold when frogs were moved from a white background to a black background, a condition known to increase the release of α-melanophorestimulating hormone for color adaptation (Kuiper et al. 2000). The coordinated induction of p24δ with the prohormone suggests its role in granule biogenesis.
Alternatively, p23 on SGs might be involved in the retrograde transport of missorted or unfolded proteins, as the defects of p24 proteins have shown to result in the leakage of ER-resident proteins in yeast (Elrod-Erickson and Kaiser 1996; Marzioch et al. 1999). Such retrievals could also occur at the level of SGs. For example, it has been shown that lysosomal enzymes incorporated in the immature SGs within the pancreatic β-cells are trapped by mannose-6 phosphate receptors and then redestined for the endosomal/lysosomal compartments via AP-1 adaptor/clathrin-coated vesicles (Kuliawat et al. 1997; Klumperman et al. 1998). If the retrieval machinery could properly work even at the post-Golgi compartments, p23 on SGs may be involved in the retrieval of misfolded secretory materials and escaped ER-resident chaperones/folding enzymes. Such demands should be considerably high in pancreatic β-cells because insulin has to be robustly synthesized and correctly folded for continuous regulation of nutrient metabolism. The high expression level of p23 in β-cells may reflect the elaborate quality control processes responsible for the proper posttranslational modification and folding of insulin molecules.
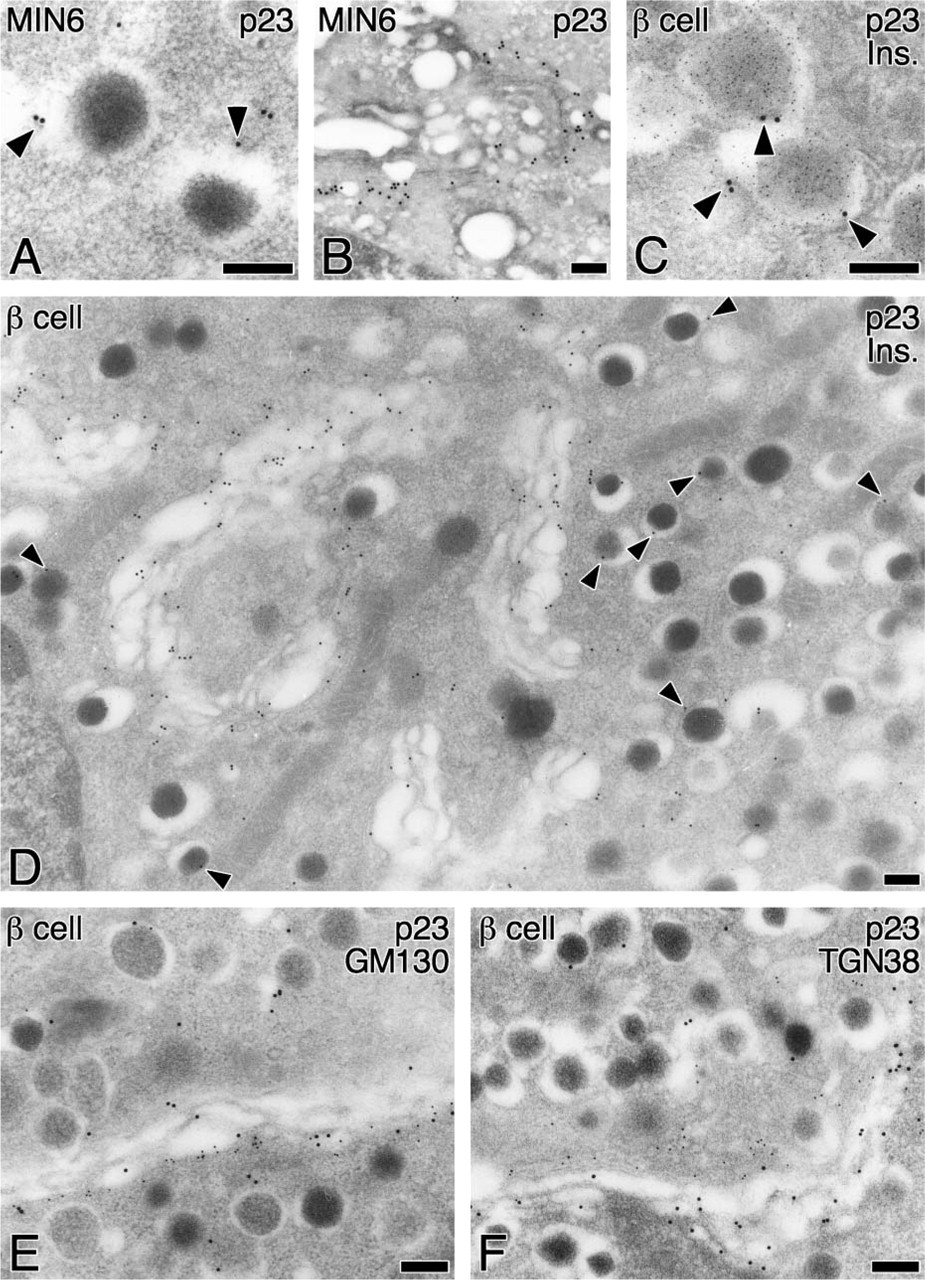
Subcellular localization of p23 in MIN6 cells and pancreatic β-cells. Subcellular localization of p23 was examined by immunoelectron microscopy of urtrathin sections from mouse MIN6 cells (
Relative labeling densities for p23 and GM130 on the intracellular organelles within the rat pancreatic β-cell
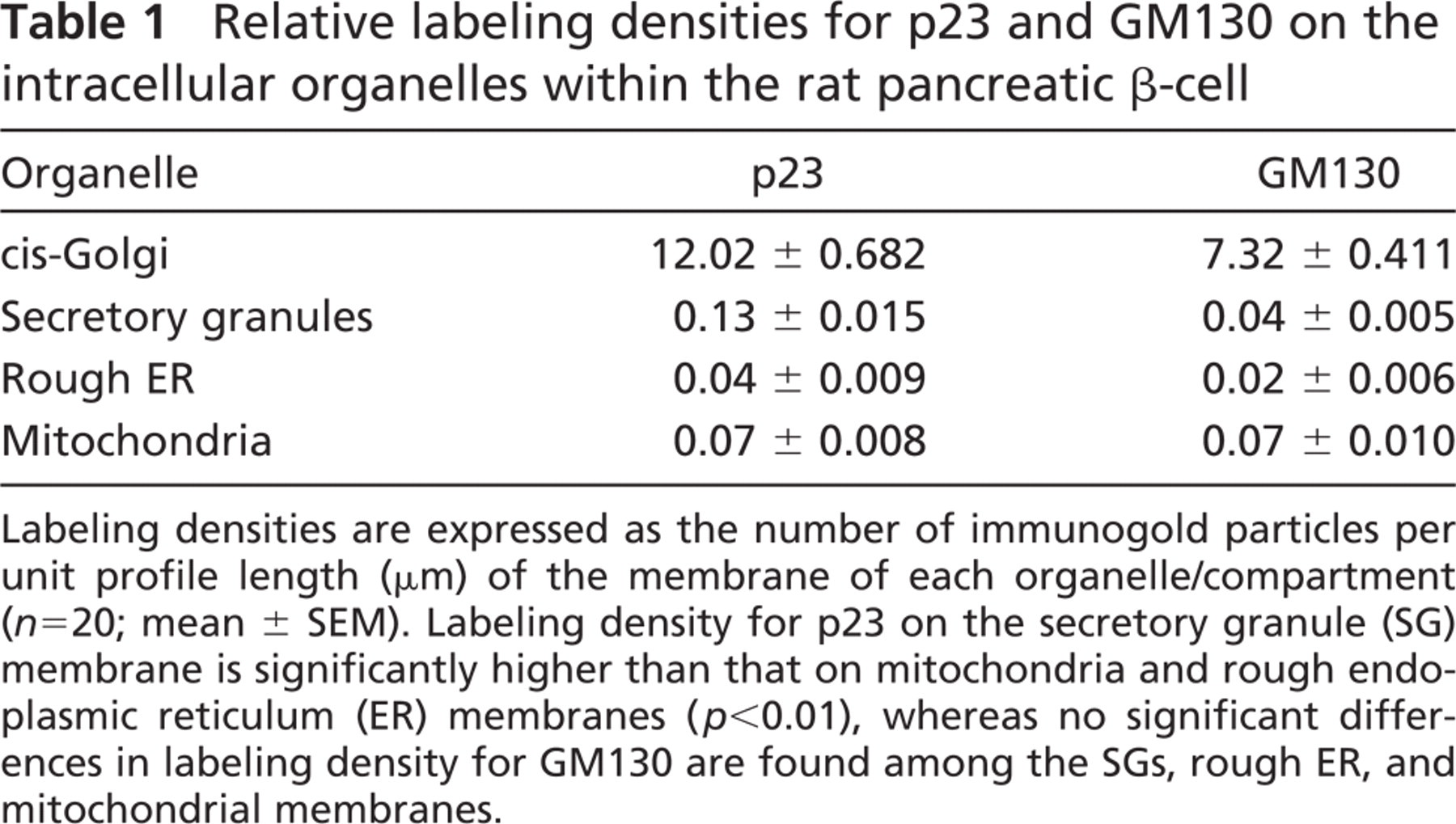
Labeling densities are expressed as the number of immunogold particles per unit profile length (μm) of the membrane of each organelle/compartment (n=20; mean ± SEM). Labeling density for p23 on the secretory granule (SG) membrane is significantly higher than that on mitochondria and rough endoplasmic reticulum (ER) membranes (p<0.01), whereas no significant differences in labeling density for GM130 are found among the SGs, rough ER, and mitochondrial membranes.
In summary, the present study demonstrates the intense expression of p23 in endocrine cells and its significant location on the SGM in pancreatic β-cells. These findings may provide a clue to reconsider the physiological role of p23 not only in the early secretory pathways but also in the post-Golgi compartments, especially in the biogenesis and/or quality control of SGs.
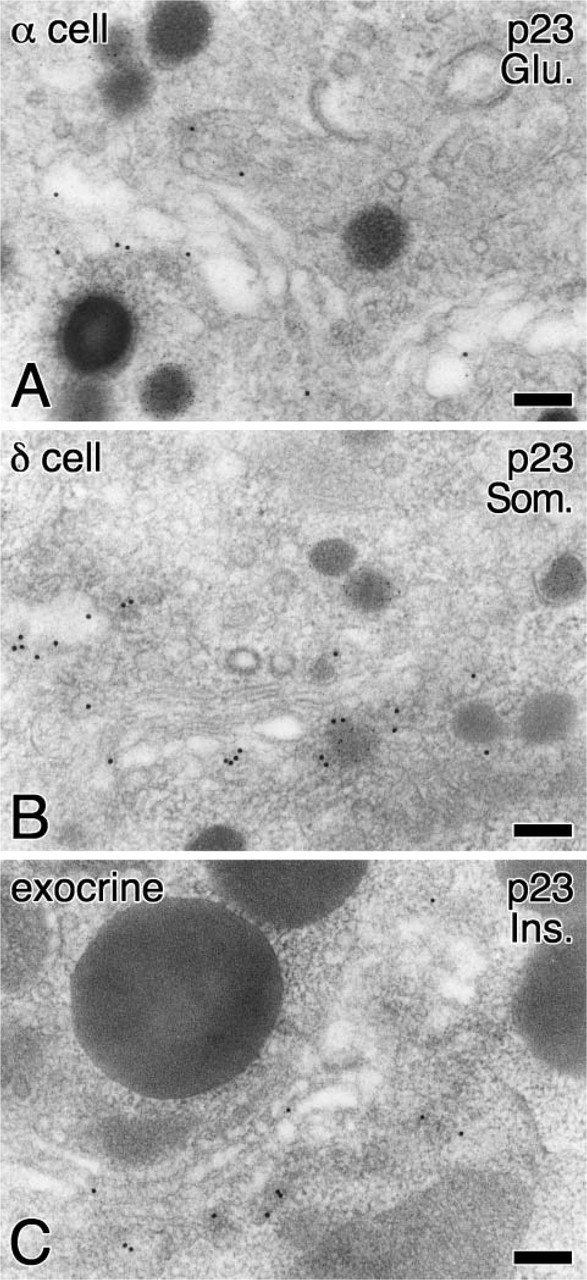
Subcellular localization of p23 in pancreatic α- and δ-cells and exocrine cells. Localization of p23 was visualized with large-sized colloidal gold particles (15-nm in diameter), whereas localization of glucagon (Glu.) (
Footnotes
Acknowledgements
This work was supported by Grants-in-Aid for Scientific Research and the 21st Century Center of Excellent Program from the Ministry of Education, Culture, Sports, Science, and Technology of Japan. The study was also supported in part by grants from the Uehara Memorial Foundation, the Nakajima Foundation, the Yamanouchi Foundation for Research on Metabolic Disorders, and Japan Diabetes Foundation (to MH) and grants from Novo Nordisk Insulin Study Award and Astellas Foundation for Research on Metabolic Disorders (to TI).
We thank M. Hosoi, H. Takemura, and K. Fuse for technical support.
