Abstract
Nestin, which was initially identified as a marker of neural stem cells, has been reported in regenerating pancreas as well as in early embryonic stem (ES) cell derivatives. However, little is known about its specific roles in stem cells as a functional regulator. We investigated the source of the action of nestin in ES and adult pancreatic ductal stem (PDS) cells in regard to the neogenesis of insulin-secreting β-cells. In ES cells, suppression of nestin by gene silencing led to an increased expression of the pluripotency-associated genes, including
P
Nestin is an intermediate filament protein transiently expressed during early development in neuronal cells as well as in non-neuronal embryonic and adult cells (i.e., cardiac, skeletal, hepatic, and pancreatic cells) and during tissue regeneration (Lendahl et al. 1990; Dahlstrand et al. 1992; Morshead et al. 1994; Matsuoka et al. 2002; Shibuya et al. 2002; Wiese et al. 2004). In pancreas, nestin expression is considered a marker for pancreatic stem cells and for islet progenitor cells (Kim et al. 2004). Previous studies have shown that insulin-secreting cells are generated from mouse and human (Soria et al. 2000; Assady et al. 2001) ES cells by the selection of progenitor cells expressing nestin (Lumelsky et al. 2001; Blyszczuk et al. 2003). In adult rat pancreas, nestin-positive cells were identified in the islet as a non-endocrine subpopulation that can differentiate ex vivo into cells having pancreatic endocrine, exocrine, and hepatic phenotypes (Zulewski et al. 2001). In addition, we previously observed that nestin is highly expressed at the earlier stages of ductule morphogenesis. Culture of these nestin-positive duct stem cells led to their full differentiation into endocrine cells, mostly β-cells, having the phenotype of glucose-stimulated insulin secretion (Kim et al. 2004). However, it remains unclear whether nestin in ES and adult stem cells supports intermediate regulators for the self-renewal of ES cells and for the differentiation into β-cells. To address this, we have tested two different cells, ES cells and pancreatic ductal stem (PDS) cells in which nestin was depleted using the gene silencing approach.
Materials and Methods
Culture of ES Cells
Mouse D3 ES cells obtained from the American Type Culture Collection (ATCC; Rockville, MD) were maintained undifferentiated on mitomycin C–treated STO cells as feeder layers in DMEM-high glucose (GIBCO/BRL; Grand Island, NY) supplemented with 15% defined fetal bovine serum (FBS; Hyclone, Perbio Science, Company, Logan, UT), 107 U/ml ESGRO (Chemicon; Chandlers Ford, Hampshire, UK), 1% non-essential amino acids (GIBCO/BRL), 2 mM
In Vitro Differentiation of ES Cells
According to a previously described protocol (Kahan et al. 2003), with some modifications, we promoted the differentiation of mouse ES cells into insulin-secreting β-cells. After transfection, to establish embryoid bodies (EBs), these cells were plated onto bacteriological culture dishes in DMEM containing 15% FBS in suspension for 4 days (early EB). After 4 days of suspension culture, intact EBs were plated onto gelatin-coated surfaces in 24-well or 60 × 15–mm culture plates (BD Biosciences; Franklin Lakes, NJ). EBs were allowed to differentiate further for 10 days (mid-EB) and 24 days (late EB) with the same medium. Each stage of EBs was characterized by their developmental maturity as follows: early EB as the initial stage at which the EB starts to form, mid-EB as the stage at which differentiation into ecto-, meso-, and endodermal lineages and outgrowth occurs, and late EB as the terminal stage of differentiation that commits progenitors.
Isolation and Culture of PDS Cells
Subtotal pancreatectomy (Px) was performed on 5-week-old male Sprague-Dawley rats (100–120 g; Daehan Experimental Animal, Seoul, Korea) to induce pancreatic regeneration. All animals were treated according to the guidelines of the Laboratory Animal Care Committee of the School of Medicine at Inha University. We isolated the neogenic ductules from the pancreatic remnants 3 days after Px to harvest PDS cells according to protocols described previously (Kim et al. 2004). The isolated ductules were plated onto coverslips in 12-well plates. The tissue fragments were cultured with RPMI medium (GIBCO/BRL) supplemented with 10% heat-inactivated FBS (Hyclone), 100 U penicillin/100 μg streptomycin/ml (GIBCO/BRL), and 2.5 μg/ml of fungizone (GIBCO/BRL) for 4 days until the cell explants developed from the primary ductal tissues. No growth factor or glucose was added to the culture medium, to exclude their effects on cell proliferation and differentiation during the culture (Soria 2001).
Synthesis of Small Interfering RNAs (siRNAs)
The siRNAs corresponding to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (as a control template in a standard silencer siRNA cocktail kit reaction) and nestin gene were synthesized using the Silencer siRNA cocktail kit (Ambion; Austin, TX). To generate a transcriptional template for both strands, single-stranded RNA (ssRNA) and long ssRNA were synthesized from the gene-specific PCR products amplified with T7 promoter containing PCR primers (sense and antisense) using separate PCRs. The GAPDH control template is a linear GAPDH double-stranded DNA (dsDNA) fragment with opposing T7 promoters that yields a 440-bp double-stranded RNA (dsRNA) product. Nestin template was used in separate PCR procedures amplified with the following sequences. Producing nestin siRNAs requires four primers containing the T7 promoter: sense 5'-GCGTAATACGACTCACTATAGGGCCTGTACTATCTGAG-3’ and antisense 5'-GAGAATTCCCTGAGTCCTG-3'; sense 5'-GGGGTACCTGTACTATCTG-3’ and antisense 5'-GCGTAATACGACTCACTATAGGGGAGACTTCCCTGAGTCC-3’ (primers giving a 1.1-kb product). After the transcription reaction, both strands of RNA were annealed to form nestin dsRNA. Then 15 μg of dsRNA for each gene was digested with RNase III (Ambion) and the siRNAs were purified according to the manufacturer's specifications. Here, RNase III–generated siRNA cocktails are a good alternative to standard methods of siRNA production. The siRNA population generated by this method does not exhibit high toxicity or nor does it have nonspecific effects on either nestin or GAPDH gene expression.
Transfection of siRNAs
To determine whether inhibition of nestin affects stemness of ES cells at the early stage, transfection into feeder-free ES cells was carried out with nestin siRNAs at a starting confluence between 40% and 60% at day 1 of culture using the siPORT lipid transfection agent (Ambion). Transfection with reagent alone served as a negative control. Briefly, 100 nM siRNA was incubated with 3 μl siPORT Lipid in 100 μl OPTI-MEM (GIBCO/BRL) for 35 min at room temperature. The mixture was added to the cells in a final volume of 0.5 ml. PDS cells were also transfected at day 3 of culture as above. The cells were provided with fresh medium 4 hr after transfection and were cultured for 24 hr. The cells were harvested by trypsinization to assess modification of mRNA transcripts of genes and proteins. In addition, the pellets of harvested cells were fixed with Bouin's solution or 4% paraformaldehyde for analysis of protein expression by immunocytochemistry.
Quantitative PCR (qPCR) Analysis
RNA was isolated with TRIzol (Invitrogen; Carlsbad, CA). Using the ImProm-II Reverse Transcriptase (Promega; Madison, WI), 1 μg of total RNA was then reverse transcribed into cDNA. qPCR analysis was performed on an iCycler iQ multicolor detection system (Bio-Rad, Foster, CA; 94C for 30 sec, 56C or 58C for 30 sec, 72C for 30 sec) with several PCR primer sets, as listed in Tables 1 and 2. All transcript levels were normalized against GAPDH. The cycle number for the nestin siRNA–transfected cells was calculated relative to mock control. For each evaluated marker, the amplification threshold was defined as a fold value of 1.
Western Blot
ES and PDS cells were harvested 24 hr after transfection. Cells were extracted with 1% NP-40, 0.5 mM EDTA, 150 mM NaCl, 50 mM Tris (pH 7.4), and 1 mM PMSF. Cellular debris was removed from the lysate by centrifugation at 13,000 rpm for 15 min at 4C. Supernatants were collected, and proteins were separated by electrophoresis through 10% Tris-HCl polyacrylamide gels. Proteins were transferred to polyvinylidene fluoride membranes (Bio-Rad) and incubated with mouse anti-nestin [clone rat 401, 1:500; Developmental Studies Hybridoma Bank (DSHB), Iowa City, IA] and rabbit anti-Oct 4 (1:2000; Santa Cruz Biotechnology, Santa Cruz, CA) overnight at 4C. After washing, membranes were incubated with horseradish peroxidase–conjugated secondary antibodies for 1 hr at room temperature. Finally, membranes were revealed using the Enhanced Chemiluminescence Kit (Roche; Mannheim, Germany).
The primers used for qPCR

GAPDH, glyceraldehyde-3-phosphate dehydrogenase; qPCR, quantitative PCR.
The primers used for conventional and qPCR

Immunocytochemistry
For ES cells in culture, coverslips were rinsed three times with PBS and fixed with 4% paraformaldehyde for 15 min at room temperature. Cells were permeabilized with 0.25% Triton X-100 (Sigma) in PBS for 2–4 hr at room temperature and processed for antibody staining. PDS cells were fixed with Bouin's solution for 3 min at room temperature and treated with 3% H2O2 in MetOH for 5 min at −20C. Cells were permeabilized with 0.1% Triton X-100 (Sigma) in 10 mM sodium citrate (pH 6.0) for 3 min at −20C and blocked. Immunostaining was carried out by the avidin-biotin-peroxidase complex method (ABC; Vector Labs, Burlingame, CA) (Hsu et al. 1981) or by immunofluorescence. Double immunostaining for nestin and insulin was performed to trace transitional cells undergoing differentiation from nestin-expressing cells into insulin-secreting cells. Double immunoperoxidase labeling was carried out as previously described (Kim et al. 2001). Primary antibodies were as follows: mouse anti-nestin (clone rat 401, 1:250; DSHB), rabbit anti-nestin (1:250; Abcam, Cambridge, UK), mouse anti-insulin monoclonal antibody (1:1000; Biogenex, San Ramon, CA), rabbit anti-insulin (1:500; Santa Cruz Biotechnology), rabbit anti-
Measurement of Insulin Secretion
Culture media were collected for measurement of insulin secreted by ES and PDS cells after transfection with mock or nestin siRNAs. Immunoassay for insulin concentrations was performed using an ultrasensitive insulin ELISA kit displaying standard ranges between 0.02 and 5.5 ng/ml (Alpco Diagnostics; Windham, NH).
Morphometry
For morphometric analysis, we counted insulin-positive cells as well as non-immunoreactive duct epithelial cells in explants immunocytochemically stained on coverslips (18 × 18 mm). Data on differentiation are presented as number of insulin-positive cells per 1000 cells of the explants at 24 hr after mock or nestin siRNA transfection.
Statistical Analyses
All values are expressed as means ± SEM. Comparisons were performed by two-tailed unpaired Student's
Results
Nestin Expression in ES and PDS Cells
We previously reported that neogenic ductules isolated from the regenerating pancreas following partial Px show eccentric expansion with an increase in nestin-positive cells (Kim et al. 2004). Consistent with those results, nestin expression in PDS cells as found in the present study increased and peaked at day 3, but it subsequently decreased to levels lower than those of day 1 (Figure 1A). For ES cells, nestin mRNA expression was significantly increased in EBs at early stages compared with ES cells (Figure 1B), but decreased afterward in mid- and late EBs. Nestin expression in mid- and late EBs was significantly less than that in ES cells.
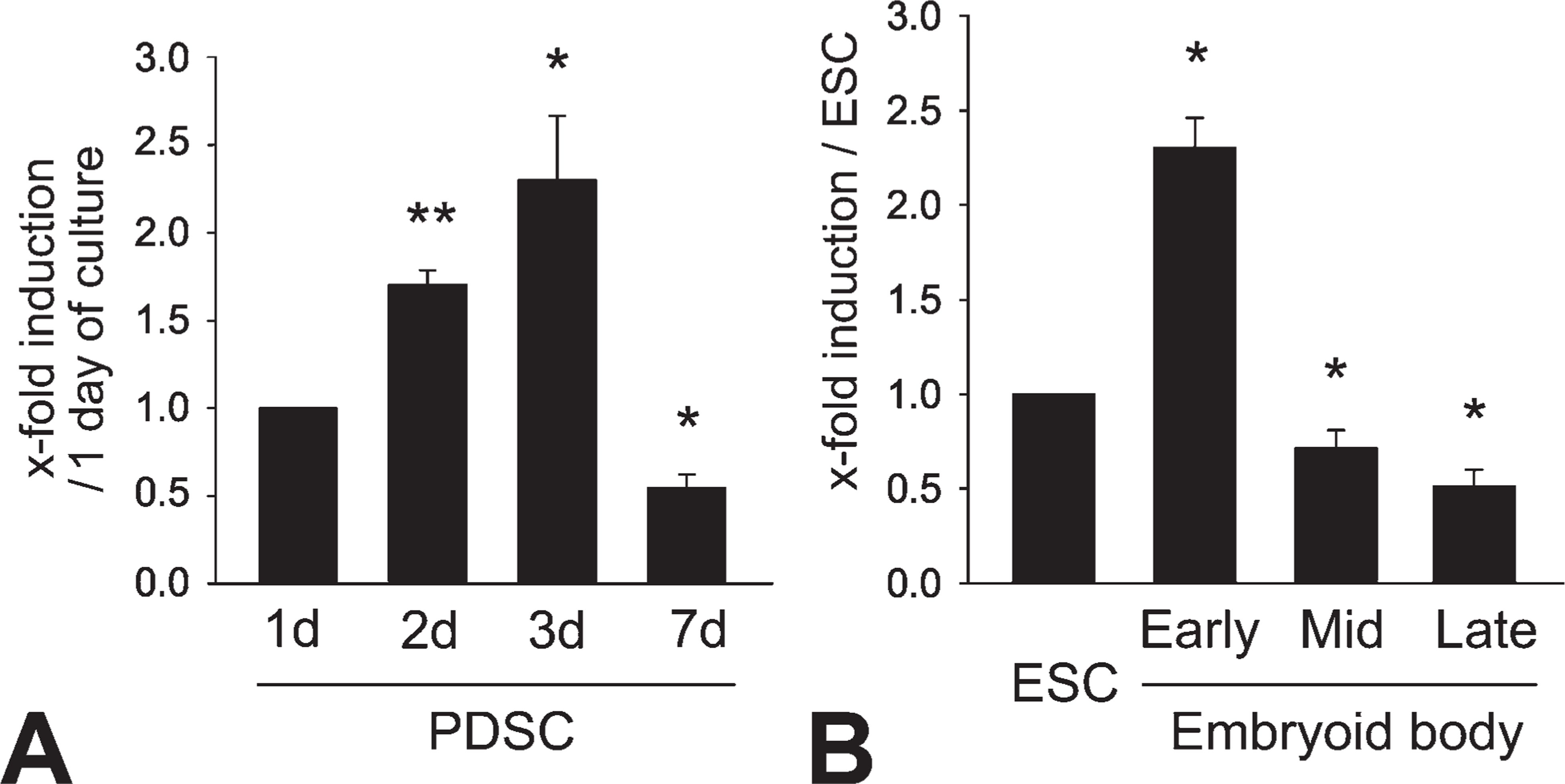
Nestin expression in embryonic stem (ES) cells and pancreatic ductal stem (PDS) cells. Changes of nestin mRNA transcripts were determined by quantitative PCR (qPCR) in PDS cells
siRNA-mediated Inhibition of Nestin Expression in ES and PDS Cells
We suppressed nestin expression in stem cells by siRNA to reveal some of the roles that nestin plays in differentiating stem cells. Almost complete absence of nestin expression was reached in both embryonic and adult pancreatic stem cells at the mRNA level as well as protein upon transfection of the cells with nestin siRNA (Figure 2).
Modifications of Stage-specific Genes in ES Cells by Nestin Suppression
We examined the changes of stage-specific genes of stem cells, including
Nestin siRNA–mediated Suppression of Transcription and Differentiation Factors
To highlight some of the roles of nestin during insulin cell differentiation, we assessed the expression of specialized transcription factors, islet hormones, and insulin cell–specific genes in nestin-suppressed cells (Figures 4 and 5). qPCR analyses were performed in mid- and late-EB and PDS cells. Most of the factors determined by qPCR analysis showed a decreased mRNA expression after transfection with nestin siRNAs (Figures 4A, 4B, and 5). Marked decrease was registered for endodermal, pancreatic transcriptional factors, and β-cell–related genes in mid-EB (Figure 4A) as well as in late EB (Figure 4B). In PDS cells, similar reduction of these factors was seen after downregulation of nestin expression. More-detailed evidence of inhibition of pancreatic transcription factors, insulin, and glucagon (1.8–3.7-fold reduction) was determined by qPCR analysis (Figure 5). These results imply that nestin expression in stem cells is closely related to differentiation and that its suppression retains the cells in a stem cell stage.
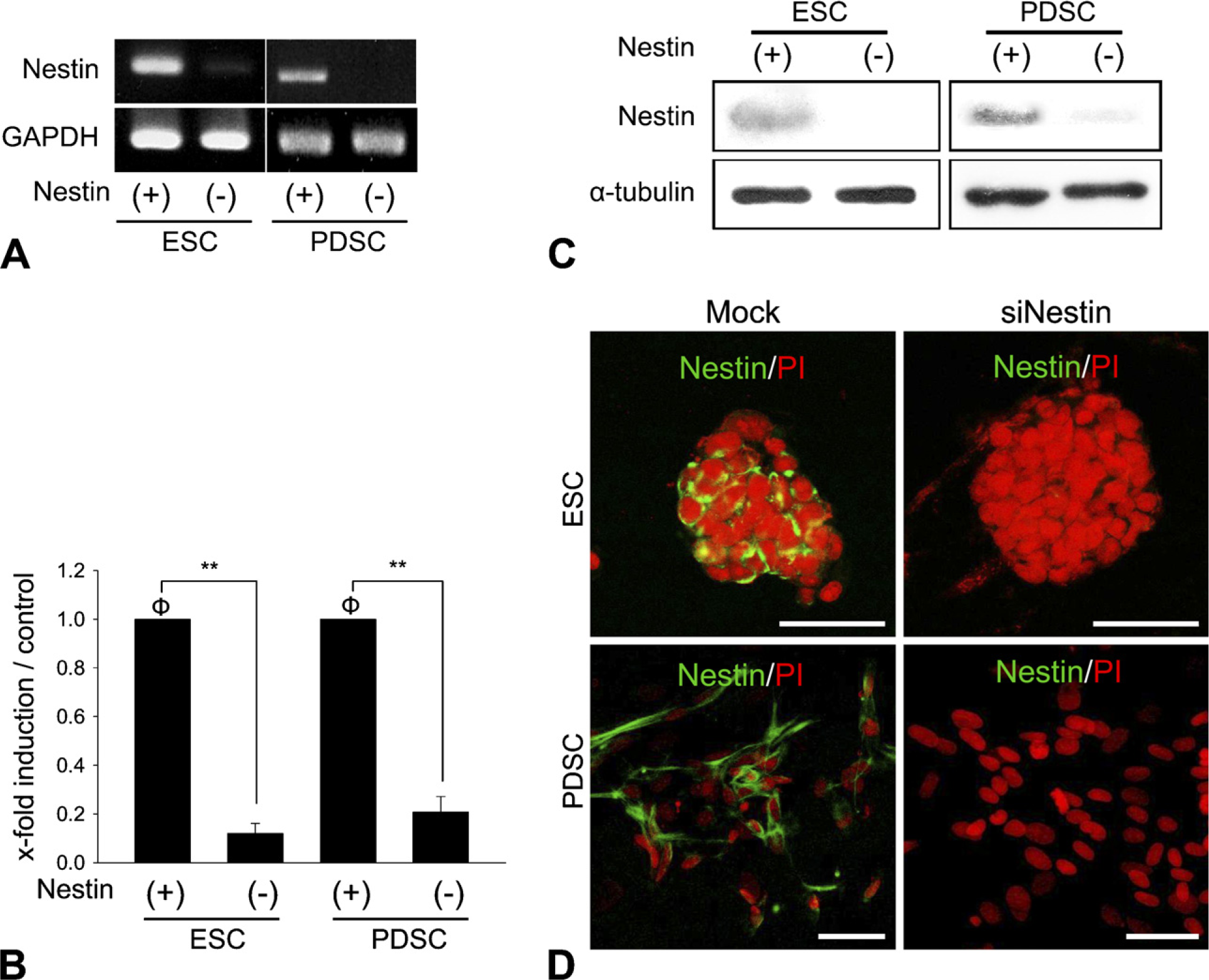
Inhibition of nestin expression by RNA interference in ES and PDS cells. Conventional
Insulin Cell Differentiation in ES and PDS Cells
Expression of C-peptide and insulin was assessed by immunocytochemistry in differentiated ES and PDS cells after nestin siRNA transfection and mock-control transfection (Figure 6). In late EB, C-peptide–positive cells were numerous in the control, whereas only a few of these cells were observed among the nestin siRNA–transfected cells (Figure 6A). We also found a remarkable decrease of insulin immunoreactivity among the PDS cells after transfection of nestin siRNA (Figure 6A). The number of differentiated cells among the PDS cells was estimated by the number of insulin-positive cells (1 × 103). The insulin-positive cells among nestin siRNA–transfected cells decreased by 2.1-fold (4.5 ± 1.14 cells/103 duct cells) 24 hr after transfection compared with the control (9.6 ± 1.46 cells/103 duct cells) (Figure 6B). To detect transitional cells undergoing transformation from nestin-positive cells to insulin-positive cells, we performed double labeling at day 7 of the culture. We found some cells that simultaneously expressed both insulin and nestin. Figure 7 illustrates a particular cell that displays nestin and insulin in two different poles, which indicates cellular transformation from nestin-positive stem stage into differentiated insulin cells.
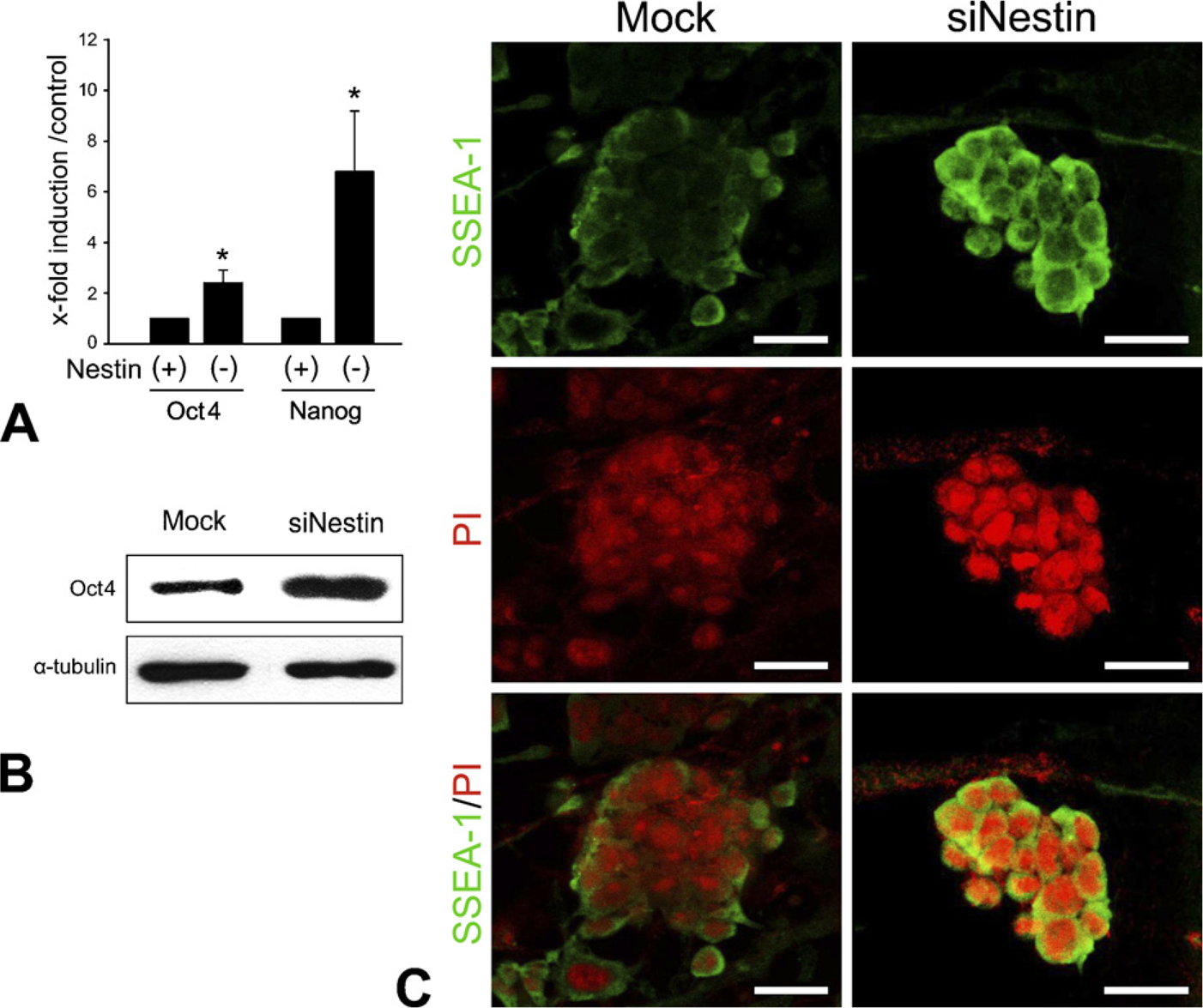
Activation of the pluripotent cell state–associated genes by suppression of nestin in ES cells.
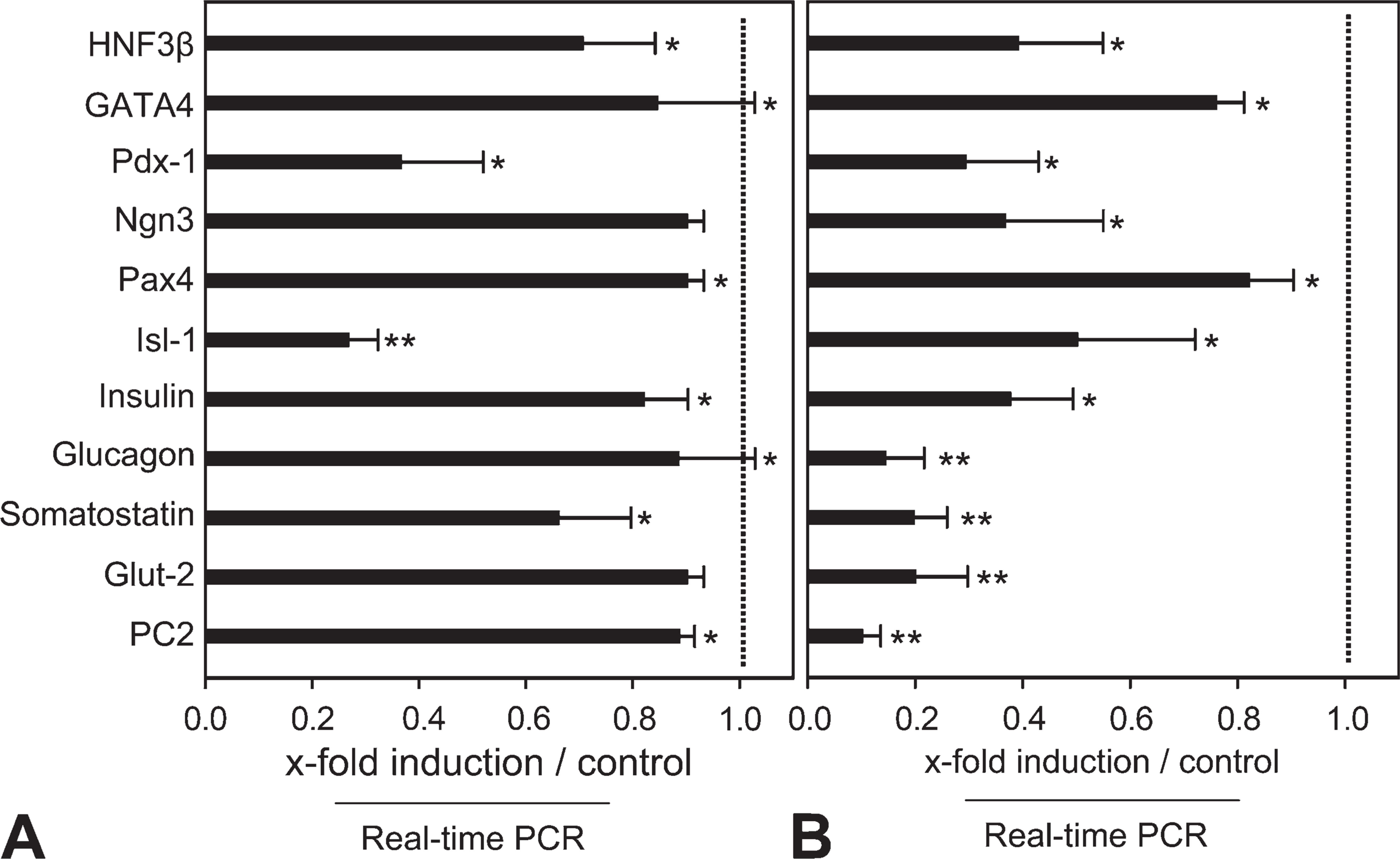
Changes in differentiation factors in EBs.
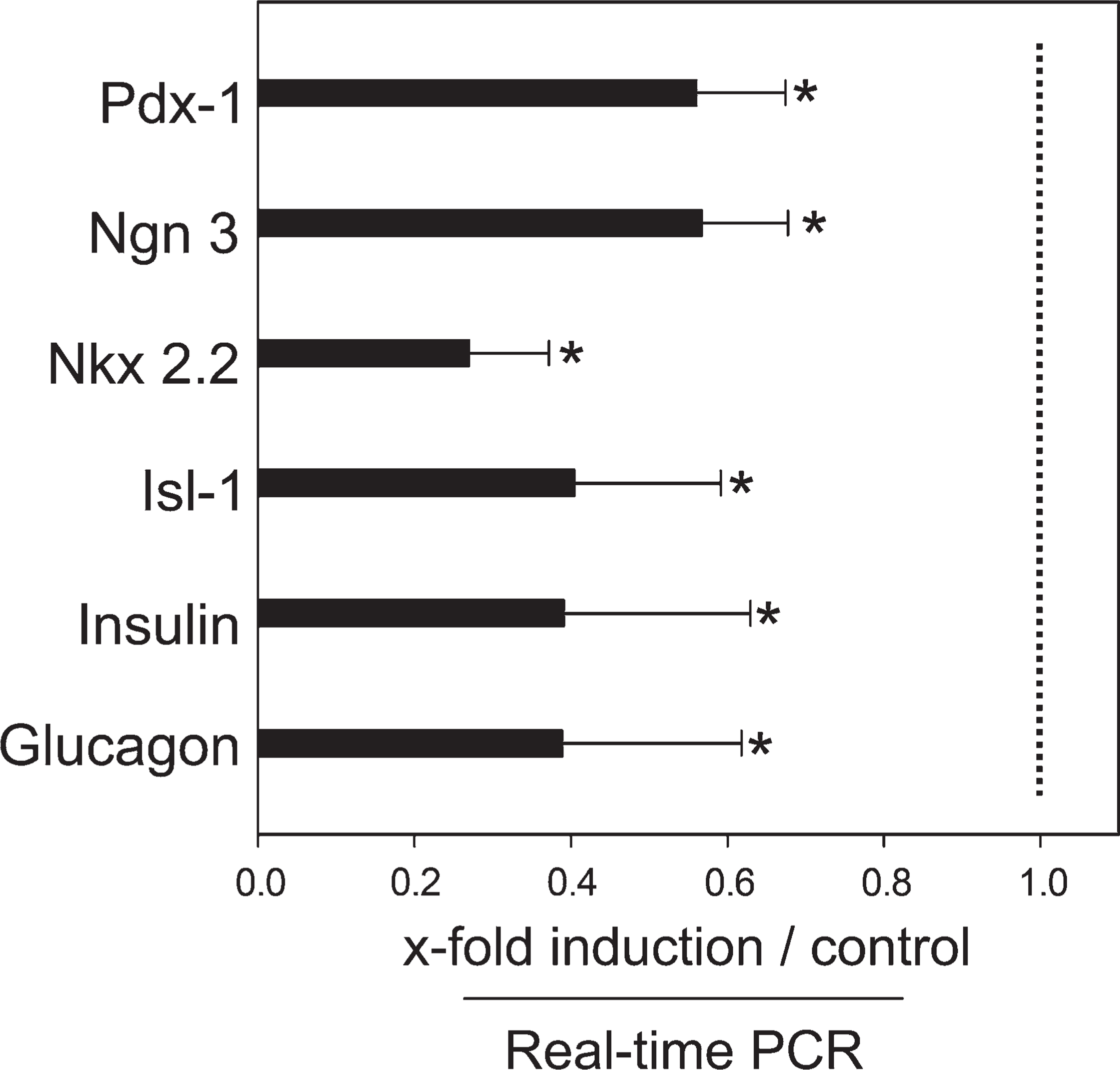
Changes in expression of pancreatic genes in PDS cells. Nestin siRNA mediates decreases of transcriptional factors and pancreatic endocrine genes in PDS cells as compared with controls. Mean values ± SEM. ∗
Insulin Secretion
Insulin concentration in the medium was found to be significantly lower in nestin siRNA–transfected ES cells at late EB (4.19 ± 0.11 pmol/l,
Discussion
Stem cells can undergo either proliferation or multi-lineage differentiation, with the outcomes of either self-renewal or differentiation. Differentiation occurs under the influence of both intrinsic factors and extrinsic signals (Fujikura et al. 2002; Chambers et al. 2003; Boyer et al. 2005). Nestin is a class VI intermediate filament protein that is expressed transiently in early developmental stages and during tissue regeneration in response to injury in a large variety of cell types (Lendahl et al. 1990; Lin et al. 1995; Vaittinen et al. 2001; Shibuya et al. 2002). Recently, in vitro, intestinal epithelial cells derived from nestin-positive cells showed a high but definite proliferative capacity and were able to differentiate into cells expressing neural, pancreatic, or hepatic transcripts and proteins (Wiese et al. 2006). In addition, a distinct subpopulation of nestin-positive mesenchymal stem cells primed toward neural differentiation was described (Lamoury et al. 2006), indicating that nestin expression is a required step for the transformation of stem cells into differentiated functional cells. In pancreatic cells, nestin expression has been suggested as an essential process for stem cell differentiation into insulin-secreting cells (Lumelsky et al. 2001; Zulewski et al. 2001; Kim et al. 2004; Maria-Engler et al. 2004). Although it is still unclear whether nestin expression is required to trigger the differentiation of stem cells into functional cells, there is increasing evidence that intermediate filament proteins are involved in major biological processes, including regulation of cell differentiation, growth potential, and cytoprotection (Franke et al. 1982; Tolstonog et al. 2001,2002). Self-renewal and differentiation are inherent and programmed processes of stem cells, but are distinctive cellular events that are regulated individually by different intrinsic factors (Niwa et al. 1998; Takeda et al. 2006). In our previous study on pancreatic cells, we suggested that nestin expression is an intermediate cellular event in the process of differentiation (Kim et al. 2004). In the present study, we attempted to further elucidate the roles of nestin as an intermediate factor in the balance between self-renewal of stem cells and differentiation. We observed that spontaneous expression of nestin is markedly enhanced in early EB and at day 3 in PDS cell cultures. This implies that maximal expression of nestin is concurrent with a shifting stage in stem cells, from cell proliferation dominance to cell differentiation dominance. Several transcription factors used as stem cell markers are known to instigate self-renewal, suppressing differentiation of ES cells. Expression of Oct 4, a class V transcription factor of POU (Pit-Oct-Unc) factors, is crucial for the maintenance of ES cell self-renewal (Niwa et al. 2000).
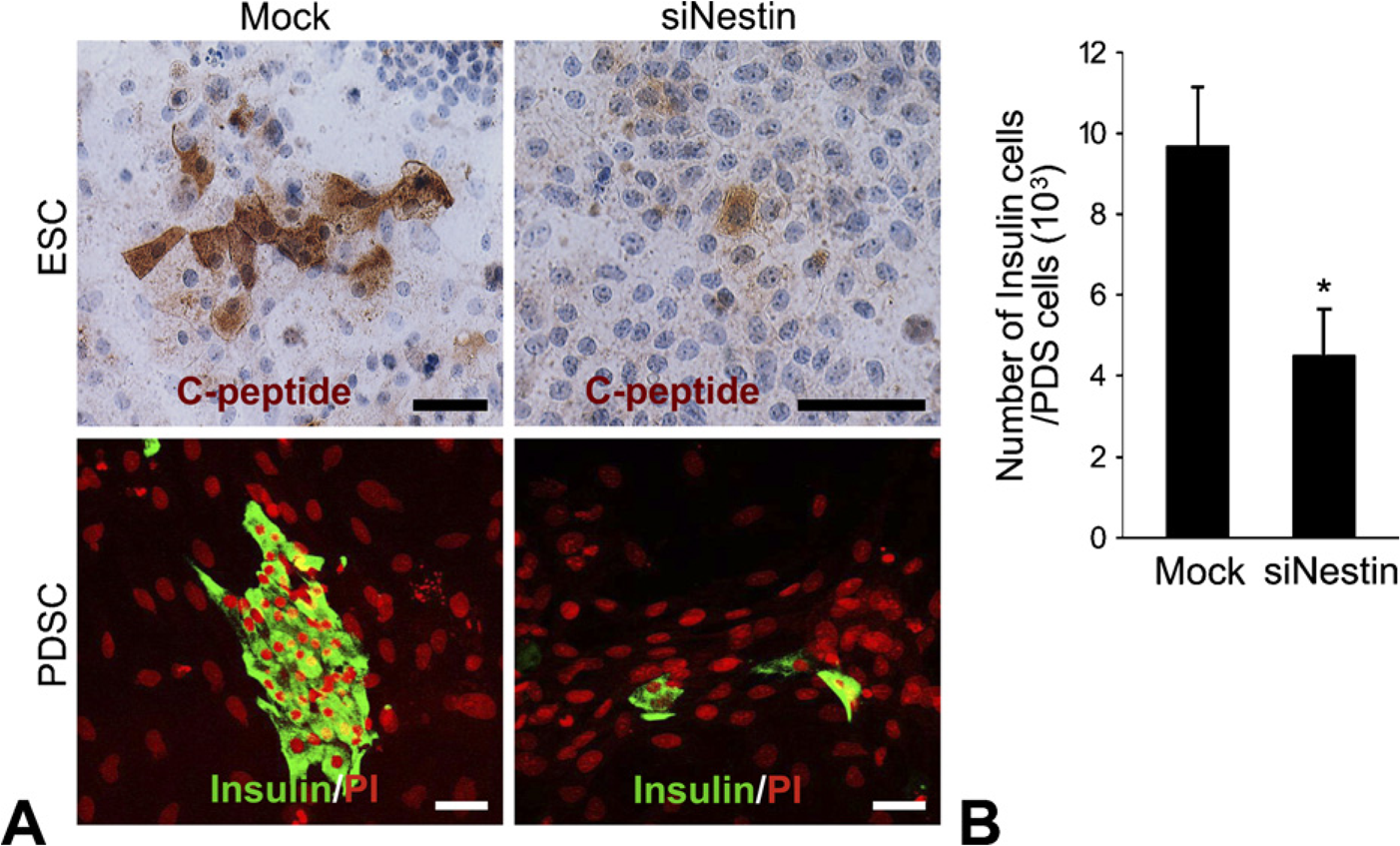
Determination of insulin cell differentiation in
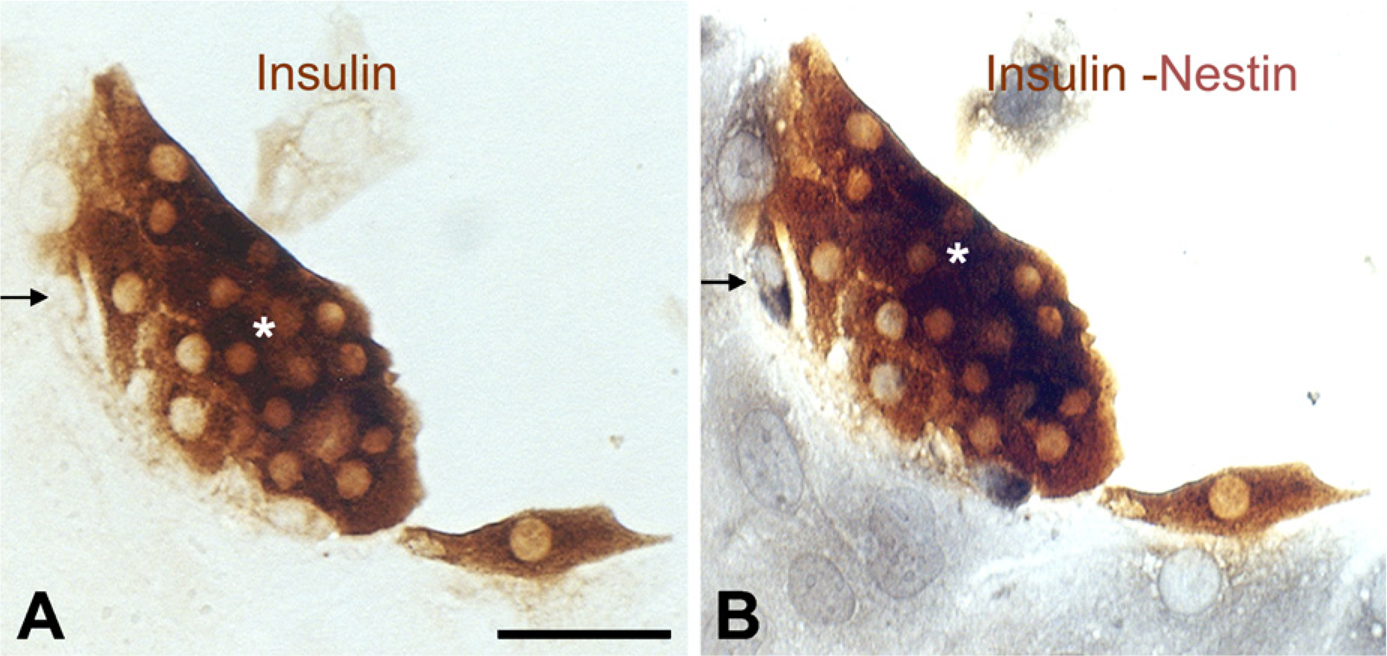
Detection of transitional cells undergoing transformation from nestin-positive cells to insulin-positive cells.
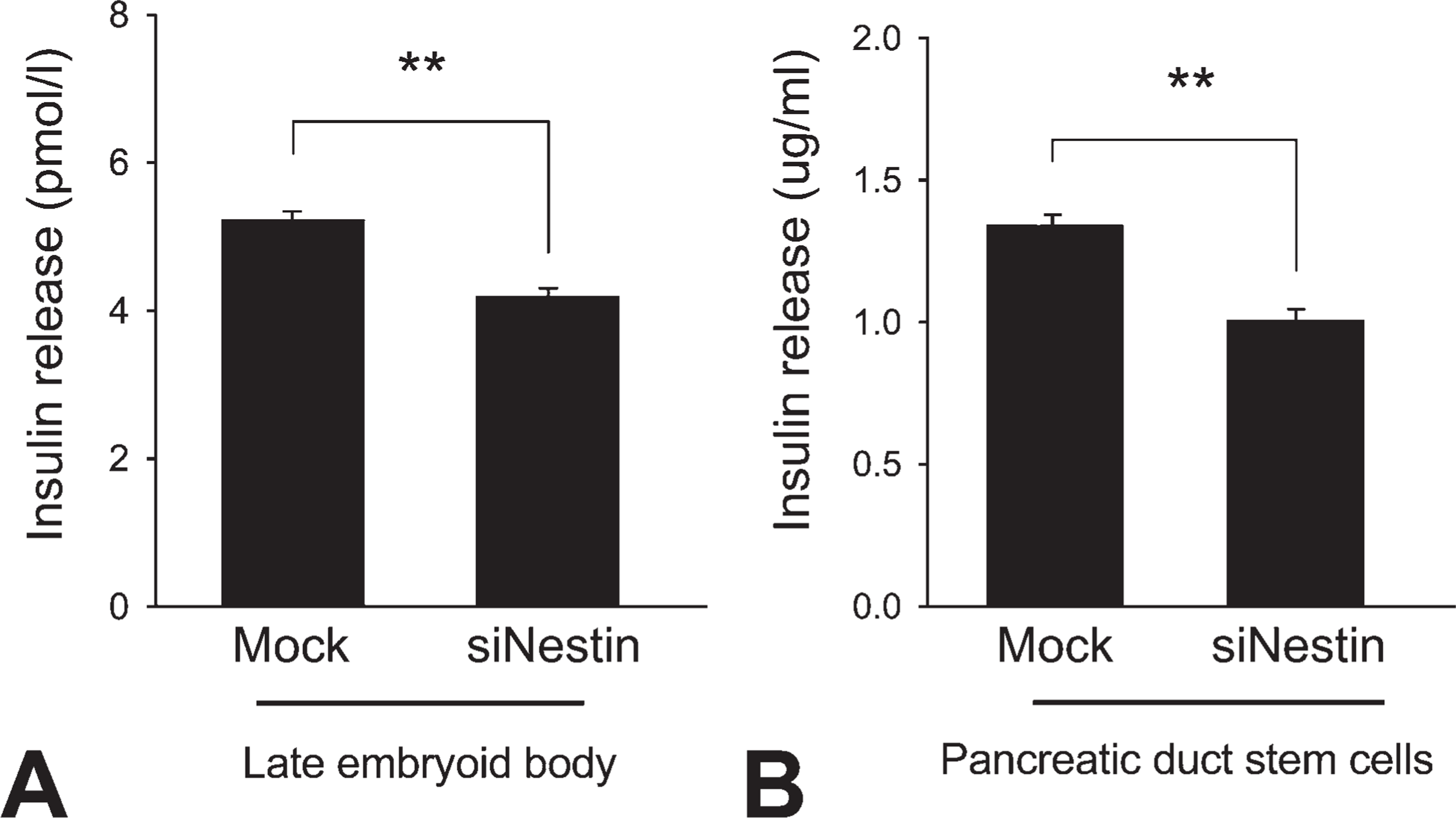
Quantification of insulin secretion by differentiated cells. Insulin release from the cells was measured by ELISA in late-EB cells
Inhibition of nestin expression not only downregulated the genes associated with insulin cell development, but also reduced the differentiation of insulin cells from stem cells. As shown in Figures 4 and 5, there was significant downregulation of
Footnotes
Acknowledgements
This work was supported by the Korea Science and Engineering Foundation (2006-2005402 and 2009-0091914).
