Abstract
Matrix Gla protein (MGP) is a potent inhibitor of soft tissue calcification, and Mgp gene deletion in mice results in arterial calcification. Our aim was to examine osteopontin (OPN) expression and localization, and posttranslational processing of OPN by the crosslinking enzyme transglutaminase 2 (TG2), in the calcified aorta of Mgp-deficient (Mgp−/−) mice. Using immunohistochemistry and light and electron microscopy, we report that following mineralization occurring in the arterial media of Mgp−/− aortas, OPN is upregulated and accumulates at the surface of the calcified elastic lamellae. Macrophages were observed in direct contact with this OPN-rich layer. Western blot analysis of extracted Mgp−/− aortas revealed that the majority of the OPN was in high molecular mass protein complexes, indicating modification by a crosslinking enzyme. Consistent with this observation, TG2 expression and γ-glutamyl-∊-lysyl crosslink levels were also increased in Mgp−/− aortas. In addition to the mineral-inhibiting actions of OPN, and based on data linking OPN and TG2 with cell adhesion in various cell types including monocytes and macrophages, we propose that TG2 interactions with OPN lead to protein polymerization that facilitates macrophage adhesion to the calcified elastic lamellae to promote clearance of the ectopic mineral deposits.
Keywords
E
Mineral deposits in the vasculature are either associated with lipid-enriched, occlusive atherosclerotic lesions in coronary arteries and other major muscular and elastic arteries or are non-occlusive of the arterial lumen where mineralization occurs mainly in the ECM of the arterial media (medial arterial calcification, elastocalcinosis) (Janzen and Vuong 2001). Vascular calcification of this latter type occurs in human diseases such as Mönckeberg's medial sclerosis, pseudoxanthoma elasticum, idiopathic arterial calcification of infancy, diabetes mellitus, and end-stage renal disease. This pathological mineralization process differs from that found in atherosclerosis and involves increased vessel rigidity and thickness without loss of arterial lumen patency (Proudfoot and Shanahan 2001). Vascular calcification decreases elasticity of the blood vessels and increases their brittleness, leading to increased risk of arterial rupture (Hur et al. 2005).
Although substantial progress has been made in determining the prevalence, extent, and significance of vascular calcification in human populations, much less is known at the tissue level about the cellular and extracellular determinants and mechanisms underlying vascular calcification. In soft tissues that do not normally calcify, pathological (ectopic) mineral deposition may be a passive event that occurs following local removal of potent mineralization inhibitors (Schinke et al. 1999). One such potent protein inhibitor of ectopic calcification in certain soft tissues is matrix Gla protein (MGP), a small, 79-amino acid ECM protein that contains posttranslationally modified γ-carboxyglutamic acid residues resulting from vitamin K-dependent carboxylation of the protein in the endoplasmic reticulum (Price et al. 1983; Price and Williamson 1985). In mice, the Mgp gene is expressed predominantly by chondrocytes and vascular smooth muscle cells (Hale et al. 1988; Shanahan et al. 1993; Luo et al. 1997). Knockout mice deficient in Mgp (Mgp−/−) develop severe vascular calcification that occurs in all elastic and muscular arteries and also ectopically in cartilage (Luo et al. 1997). Vascular calcification leads to arterial rupture and death of the animal normally within the first 8 weeks of life, and ectopic calcification of growth plate cartilage occurring prematurely in the chondrocyte proliferation-zone cartilage (rather than only in the hypertrophic zone) leads to early closure of the growth plates and osteopenia. Vascular smooth muscle cells in arteries of Mgp-deficient mice show a loss of smooth muscle cell markers and, in older Mgp−/− mice, chondrocytes and cartilaginous ECM are observed in the media of large elastic arteries (El-Maadawy et al. 2003). As part of these phenotypic transformations, osteopontin (OPN) is upregulated in Mgp−/− aortas (Steitz et al. 2002).
OPN is a mineral-binding protein abundant in most mineralized tissues and in pathologically calcifying tissues including blood vessels (Steitz et al. 2002; Speer et al. 2005; Giachelli et al. 2005b). OPN is an acidic glycophosphoprotein highly expressed in mineralized connective tissues but is also found in many other non-mineralizing tissues and in most tissue fluids (Sodek et al. 2000). OPN belongs to the SIBLING (Small Integrin-Binding LIgand N-linked Glycoprotein) family of proteins known to interact with cell-surface integrins via their Arg-Gly-Asp (RGD) sequence and to associate with mineral via their phosphate groups and stretches of acidic amino acid residues (Fisher and Fedarko 2003). As part of ECM assembly and its binding to mineral, OPN incorporates into skeletal and dental tissues and shows increased accumulation at mineralized tissue interfaces and surfaces. OPN upregulation in the mineralized aortas of Mgp−/− mice (Steitz et al. 2001) has been proposed to act as a secondary, inducible calcification inhibitor that limits further mineralization of the Mgp−/− arteries. Indeed, mice lacking both Mgp and Opn genes (Mgp−/−; Opn−/− have a more severe vascular calcification phenotype, showing both increased mineral deposition in the aorta and earlier death by aortic rupture relative to that observed in the Mgp−/− mice (Speer et al. 2002). OPN is also a known substrate for transglutaminase 2 (TG2) (also known as tissue transglutaminase) and Factor XIIIA (Prince et al. 1991; Sørensen et al. 1994; Kaartinen et al. 1997, 1999,2002), which are members of the transglutaminase family of protein-crosslinking enzymes that create covalently linked, large protein polymers of its substrate proteins (Lorand and Graham 2003). Transglutaminases are involved not only in matrix formation and stabilization, but also in cell adhesion, spreading, and signaling (Verderio et al. 1998; Gaudry et al. 1999; Lorand and Graham 2003; Akimov and Belkin 2001a,b).
In the present study, our aim was to characterize OPN expression and crosslinking by TG2 in the calcified arteries of Mgp−/− mice by light microscopy immunohistochemistry, by ultrastructural immunogold localization using transmission electron microscopy, and by biochemical methods. We show that OPN expression and localization correlate with the severity of the calcification of the arteries in Mgp−/− mice and that the majority of OPN is in a high molecular mass polymer form likely created by the covalent cross-linking enzymatic activity of TG2, which is also up-regulated in these aortas. We also demonstrate that macrophages make direct contact with the OPN-rich surfaces of the calcified elastic lamellae presumably as part of an attempt to clear this ectopic calcification. Given these recent reports on the role of OPN and TG2 in cell adhesion, our data suggest that in addition to inhibiting ectopic mineralization at the surface of the calcified elastic lamellae in Mgp−/− mice, OPN upregulation might also serve as a signal for removal of the ectopic mineral deposits by macrophages.
Materials and Methods
Animals, Tissue Samples, and Reagents
Generation of Mgp−/− mice was as described previously (Luo et al. 1997), and thoracic aortas were used in the present study. Transgenic founders were generated by pronuclear injection according to standard techniques. All mice were maintained in a pathogen-free standard animal facility, and experiment protocols were approved by the Institutional Animal Care and Use Committee at Baylor College of Medicine. Human coronary artery samples were obtained from Drs. Cecilia M. Giachelli and Charles E. Murry (University of Washington, Seattle, WA) following heart transplantation as a consequence of advanced coronary artery disease, using procedures approved by the Institutional Review Board of the University of Washington. These were fixed in 4% paraformaldehyde and 1% glutaraldehyde in 0.1 M sodium cacodylate buffer. All basic reagents were from Sigma (St Louis, MO) unless otherwise stated. Regions of the human artery not showing any atherosclerotic lesions were selected by light microscopy for further analysis.
Light Microscopy, Immunohistochemistry, and von Kossa Staining
Dissected thoracic aortas from age- and gender-matched Mgp+/+ and Mgp−/− mice were immediately immersed in a 2% paraformaldehyde fixative solution, pH 7.4, containing 75 mM lysine and 10 mM sodium periodate in 0.1 M sodium cacodylate buffer. After overnight fixation at 4C, samples were changed into 0.1 M sodium cacodylate buffer, dehydrated in a graded alcohol series, and embedded in paraffin. Sections were cut on a rotary microtome at a thickness of 7 μm and incubated with either anti-MGP monoclonal antibody (Dr. Leon Schurgers, VitaK; Maastricht, The Netherlands), rabbit anti-human OPN polyclonal antibody (LF-123, Dr. Larry W. Fisher; National Institutes of Health/National Institute of Dental and Craniofacial Research, Bethesda, MD), goat anti-mouse OPN polyclonal antibody (R&D Systems; Minneapolis, MN), anti-guinea pig TG2 monoclonal antibody (Labvision; Fremont, CA), or anti-γ-glutamyl-∊-lysine crosslink-specific monoclonal antibody (Covalab; Lyon, France). Immunoreactions were visualized using the avidin-biotin-peroxidase complex (ABC) technique as described by Hsu et al. (1981). Briefly, dewaxed sections of aorta were incubated overnight with the antibody diluted in 5% normal goat serum containing 0.2% BSA in Tris-buffered saline with Tween-20 (50 mM Tris-HCl, 150 mM NaCl, and 0.01% Tween-20, pH 7.6). Following three buffer washes, sections were incubated for 45 min with the appropriate secondary biotinylated goat anti-rabbit, goat anti-mouse, or swine anti-goat IgG (Caltag Laboratories; Burlingame, CA) and, after additional washing, sections were treated with the Vectastain ABC-AP Kit (Vector Laboratories; Burlingame, CA) according to the manufacturer's instructions. Color development (red) was achieved with a 10-min treatment of Fast Red TR/Naphthol AS-MX phosphate containing 1 mM levamisole as endogenous alkaline phosphatase inhibitor. After rinsing with distilled water, sections were counterstained with methyl green and mounted under coverslips using Kaiser's glycerol jelly. Negative controls consisted of the same treatment except for omission of the primary antibody. Survey-section histology was examined after conventional staining with hematoxylin and eosin. Light micrographs were obtained using a Sony DXC-950 3-CCD camera mounted on a Leica DMRBE light microscope (Leica Microsystems GmbH; Wetzlar, Germany). von Kossa staining for mineral was documented after incubating tissue sections in a 3% silver nitrate solution exposed to bright light while on the sections (for 30 min). This was followed by rinsing with distilled water and light counterstaining for tissue and cell morphology, using methyl green, toluidine blue, or hematoxylin and eosin.
Electron Microscopy and Colloidal-gold Immunolabeling for OPN
Dissected thoracic aortas from age- and gender-matched Mgp+/+ and Mgp−/− mice and the human coronary artery segment were immediately immersed in a 4% paraformaldehyde + 1% glutaraldehyde solution in 0.1 M sodium cacodylate buffer, pH. 7.2, and prepared for electron microscopy and colloidal-gold immunocytochemistry as described previously (McKee et al. 1992). Briefly, after overnight fixation at 4C and rinsing in 0.1 M sodium cacodylate buffer, aortas were diced into smaller pieces, dehydrated in an ascending series of ethanol concentrations to pure ethanol, and infiltrated and embedded in LR White acrylic resin (London Resin Company; Berkshire, UK). Samples were generally left undemineralized prior to embedding, and heat-polymerized (2 days at 55C) LR White resin blocks were sectioned with a diamond knife on a Reichert ultramicrotome (Leica Microsystems GmbH), producing 0.5-μm-thick survey sections for light microscopy. Some samples were demineralized in 4.13% EDTA for 1 week prior to embedding. From these survey sections, selected regions of aorta containing von Kossa-positive aortic segments were trimmed on the block faces and sectioned for transmission electron microscopy. Ultrathin sections (80 nm) were obtained on an ultramicrotome with a diamond knife and transferred to formvar- and carbon-coated nickel electron microscopy grids for colloidalgold immunolabeling as described previously for mineralized tissues (McKee and Nanci 1995a). Briefly, 80-nm-thick grid-mounted tissue sections were processed for immunocytochemistry by incubation with the antibodies described above, and immunolabeling patterns were detected by a second incubation using a protein A–colloidal-gold (15 nm) conjugate (Dr. George Posthuma; University of Utrecht, The Netherlands). Incubated grids were conventionally stained with uranyl acetate and lead citrate and examined in a JEOL 2000FX transmission electron microscope operated at 80 kV (JEOL; Tokyo, Japan).
Protein Extraction
Protein preparations of thoracic aortas from age- and gender-matched Mgp+/+ and Mgp−/− mice were prepared by crushing the tissues under liquid nitrogen into a fine powder with a biopulverizer (Biospec Products; Bartlesville, OK). Tissue powder was extracted for 12 hr with 4 M guanidium (Gu)-HCl/0.5 M EDTA in 50 mM Tris-HCl, pH 7.4, containing protease inhibitors (1 mM phenylmethylsulfonylfuoride, 1 mg/ml benzamidine, and 1 mg/ml leupeptin). Preparations were concentrated with Centricon Plus-20 concentrators (PL-10, molecular mass cutoff 10,000; Millipore, Bedford, MA), and extraction buffer was changed to 8 M urea. Protein concentrations were determined by BCA Protein Assay (Pierce; Rockford, IL). In comparing the calcium-binding properties of monomeric and polymeric OPN, separate extractions were done. Frozen tissue powder was first extracted with 4 M Gu-HCl in 50 mM Tris-HCl, pH 7.4, for 30 min and then subjected to 12-hr extraction with 0.5 M EDTA in 50 mM Tris-HCl, pH 7.4. Preparations were concentrated as described above, and extraction buffers were changed into 8 M urea (for the Gu-HCl extract) and 5 mM NH4HCO3, pH 8 (for the EDTA extract). Mouse bone EDTA extract was used as positive control to demonstrate the presence and electrophoretic pattern of polymeric OPN. Bone extract was prepared as described previously (Kaartinen et al. 2002).
Western Blot Analysis
OPN. Eight μg of total protein of Gu-HCl/EDTA extract (or separate Gu-HCl and EDTA extracts) from Mgp+/+ and Mgp−/− mouse aorta and EDTA extract from mouse bone were subjected to 10% SDS-PAGE followed by electrotransfer onto a PVDF membrane (Bio-Rad Laboratories; Hercules, CA). PVDF membranes were blocked with 5% milk powder in 50 mM TBS/0.1% Tween-20 and incubated for 2 hr at room temperature with anti-OPN (LF-123) antisera diluted in TBS-Tween. Membranes were washed with TBS-Tween and incubated with secondary antibody (anti-rabbit horseradish peroxidase; Caltag Laboratories). After washing, membranes were developed by enhanced chemiluminescence using an ECL-Plus kit according to the manufacturer's instructions (Amersham Pharmacia Biotech; Piscataway, NJ).
TG2 and Isopeptide Crosslinks. Twenty μg of total protein (Gu-HCl/EDTA protein extracts; as above) were separated by native-PAGE (without SDS), followed by electrotransfer onto a PVDF membrane. TG2 was detected using anti-guinea pig TG2 antibody (Labvision) and anti-mouse horseradish peroxidase conjugate (Amersham Pharmacia Biotech). For isopeptide crosslink antibody detection, membranes were stripped with 0.1 M glycine, pH 2.5 (2 × 30 min), and reblocked with 5% non-fat milk for 30 min. Membranes were incubated with isopeptide antibody (Covalab) overnight at 4C, followed by application of anti-mouse horseradish peroxidase (Amersham Pharmacia Biotech). Membranes were developed with the ECL-Plus kit as above.
Results
Immunohistochemistry for Mgp in wild-type mouse aorta revealed intense immunostaining of all elastic lamellae in the arterial media, including the internal elastic lamina (Figure 1A). As expected, sections of Mgp−/− aorta showed complete absence of immunostaining for Mgp (Figure 1B). Whereas wild-type (Mgp+/+) mice had no arterial calcification (Figures 1C and 1D), blood vessel calcification in Mgp−/− mice begins at 1-2 weeks after birth, commencing in large elastic arteries such as the aorta (Luo et al. 1997), shown here in aorta at 4 weeks (Figure 1E) and 6 weeks (Figure 1F) of age. With time, the calcification extends to large muscular arteries and then to all muscular arteries, ultimately leading to death of the mice by arterial rupture and hemorrhaging. Ultimately, the mineral completely fills extended segments of the elastic lamellae (Figure 1E) and, if the mice live long enough, may even extend beyond the elastic lamellae into the adjacent collagen-rich ECM (Figure 1F).
Although it has been shown previously that OPN is upregulated in Mgp−/− mice (Steitz et al. 2001), a detailed characterization by immunohistochemistry at the light microscopic level, ultrastructural analysis and immunolocalization at the electron microscopic level, and protein studies have not been done. Immunohistochemical localization of OPN in paraffin sections of Mgp−/− aorta at 4 weeks (Figure 1G) and 6 weeks (Figure 1H) revealed intense immunostaining at the surface of mineralized elastic lamellae and where calcification had extended into the interlamellar ECM. Immunostaining for OPN was not observed within the central portions of the elastic lamellae, and OPN was not associated with elastic lamellae that were not mineralized. This immunostaining pattern for OPN was similar in both EDTA-decalcified samples (data not shown) and undecalcified samples.
Following LR White resin embedding and sectioning for transmission electron microscopy, calcification (hydroxyapatite) was observed as electron-dense mineral deposits (mineral characterized in Luo et al. 1997) at multiple loci within elastic fibers of Mgp−/− aortas (Figure 2A). With time, these disc-like mineralized structures coalesced into extensive mineral sheets throughout the elastic lamellae (Figures 2B and 2C), with calcification occasionally extending beyond the elastic lamellae and into the surrounding collagenous ECM.
High-resolution, colloidal-gold immunocytochemical localization of OPN showed labeling along elastic lamellae identified as being mineralized by light microscopy (von Kossa staining). Electron microscopy in Mgp−/− aorta revealed abundant gold-particle labeling at the surface of mineralized elastic lamellae (Figures 2D-2G), but not at regions where there was no calcification (Figure 2D). Immunogold labeling for OPN was associated with an electron-dense, organic coating of variable thickness at the surface of the mineralized elastic lamellae that was absent in unmineralized regions. When thicker, this OPN-rich coating sometimes showed a layered appearance (Figure 2E). Where calcification had spread beyond the elastic lamellae and into surrounding collagen, immunolabeling for OPN consistently associated also with this mineralized ECM (Figure 2G).
Using electron microscopy, we also frequently observed macrophages close to the mineralized elastic lamellae in the Mgp−/− aortas (Figures 3A-3C). These macrophages were readily identifiable by their characteristic and abundant numerous cellular processes, which often extended into direct contact with the OPN-rich coating at the surface of the mineralized elastic lamellae (Figures 3B and 3C).
OPN protein expression was also confirmed by Western blotting. Mgp−/− aortas from 4-week-old mice were extracted with EDTA- and Gu-HCl-containing extraction buffer, a combination commonly used to release mineral-binding proteins from bones and teeth (Goldberg et al. 1994). As presented in Figure 4A, abundant OPN was released from the Mgp−/− arteries, but not from the wild-type aortas. Essentially, all OPN expressed in the Mgp−/− aortas was bound to mineral because OPN was extractable only by demineralization with EDTA (Figure 4B); Gu-HCl extraction alone showed an absence of OPN (Figure 4B). As apparent from Western blots, OPN was found in two major forms in the Mgp−/− aortas; one as the 65-kDa monomer form and the other as a high molecular mass polymeric form having a molecular mass of 120 kDa or higher. Of these two forms, the OPN polymers were the predominant form found in the arterial extracts of Mgp−/− mice. We have also previously reported crosslinking of several mineral- and cell-binding SIBLING family proteins, including OPN, by TG2 in both rat and mouse bone and rat teeth (Kaartinen et al. 2002,2005; Nurminskaya and Kaartinen 2006). The observation that OPN is found predominantly in its polymeric form in mineralized tissues such as bones and teeth prompted us to investigate whether TG2 might be upregulated in the Mgp−/− aortas, which are calcified. Immunolocalization of TG2 and the γ-glutamyl-∊-lysine crosslinks created by this enzyme revealed only weak or absent immunostaining of the aortic media in wild-type mice (Figures 5A and 5B), but strong immunostaining in the Mgp−/− aortas (Figures 5C and 5D). Immunostaining for this upregulation of both TG2 and γ-glutamyl-∊-lysine crosslinks in the Mgp−/− aorta was associated mainly with the vascular smooth muscle cells, and an identical staining pattern was observed when EDTA-decalcified samples were used (data not shown). Western blotting of TG2 in aorta extracts after electrophoresis run under native conditions (without SDS) is shown in Figure 6A; TG2 was present only in Mgp−/− aortas and was complexed into high molecular mass polymers. SDS was omitted from the gels because its presence prevented the detection of TG2 and the isopeptide crosslinks. Western blotting using γ-glutamyl-∊-lysine crosslink-specific antibody showed the presence of isopeptide bonds in the high molecular mass region of the blots (Figure 6B).
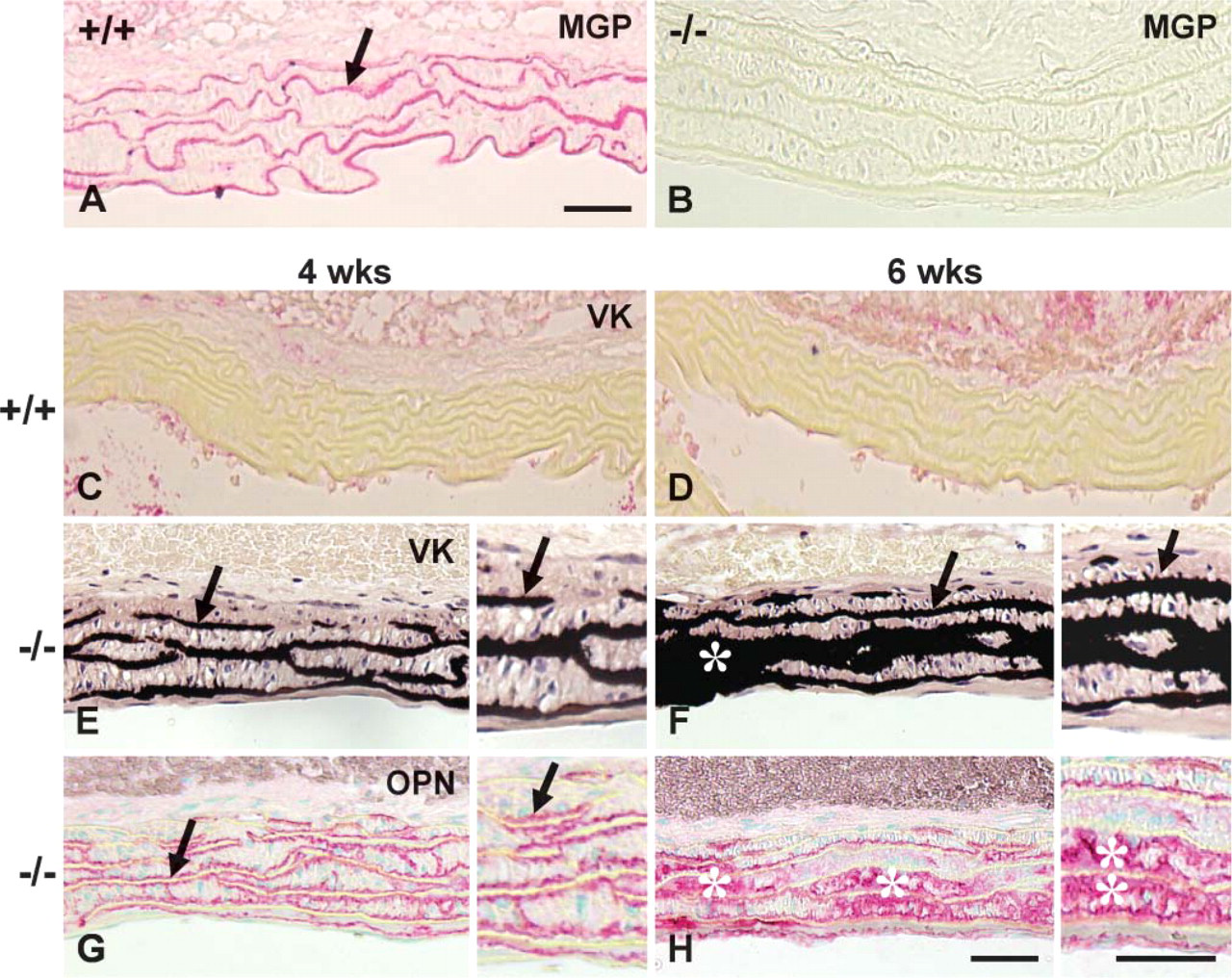
Paraffin section histology, histochemistry, and immunohistochemistry of undemineralized thoracic aortas from wild-type (+/+) and matrix Gla protein (Mgp)-deficient (−/−) mice. (
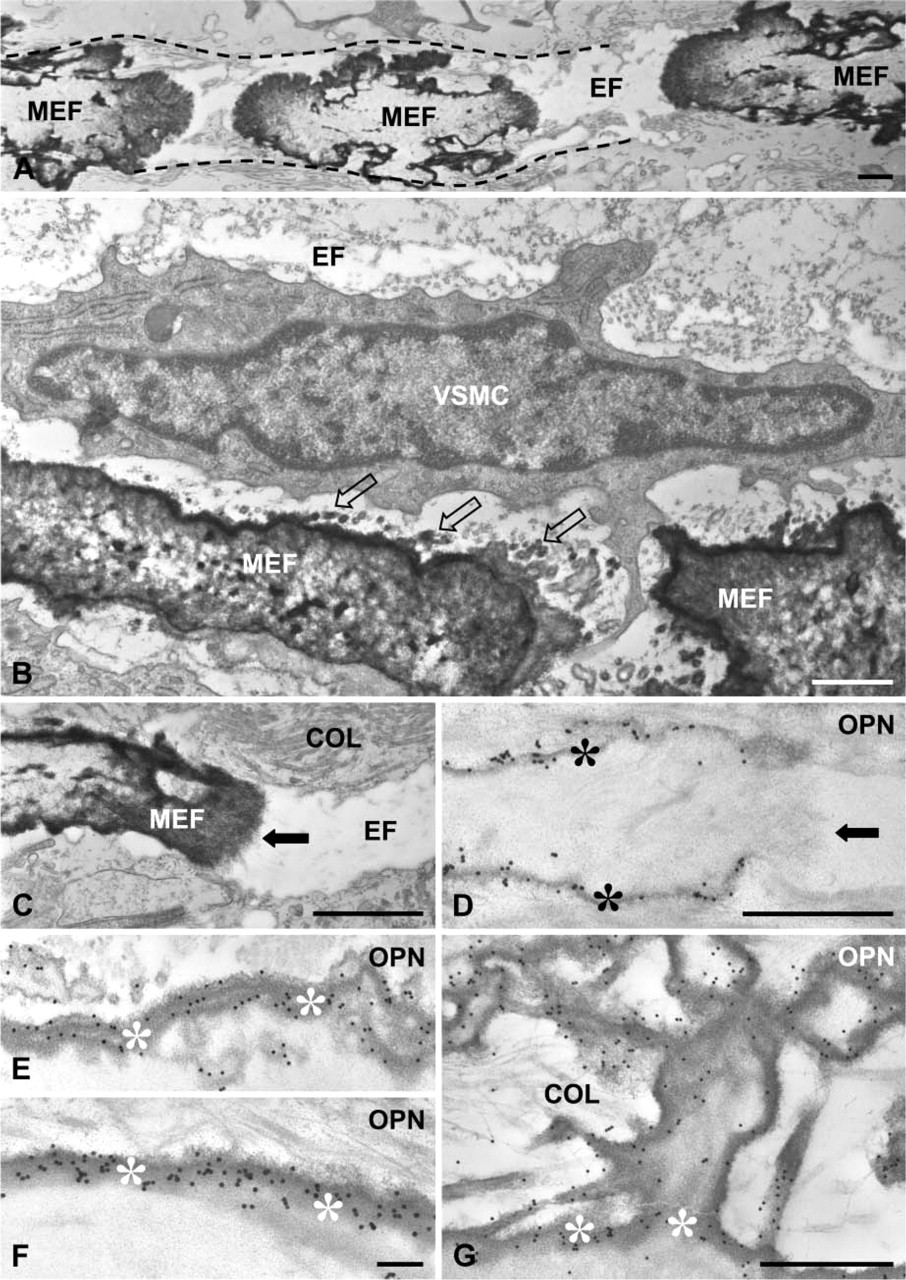
Transmission electron micrographs illustrating early mineralization sites in Mgp−/− aorta. (
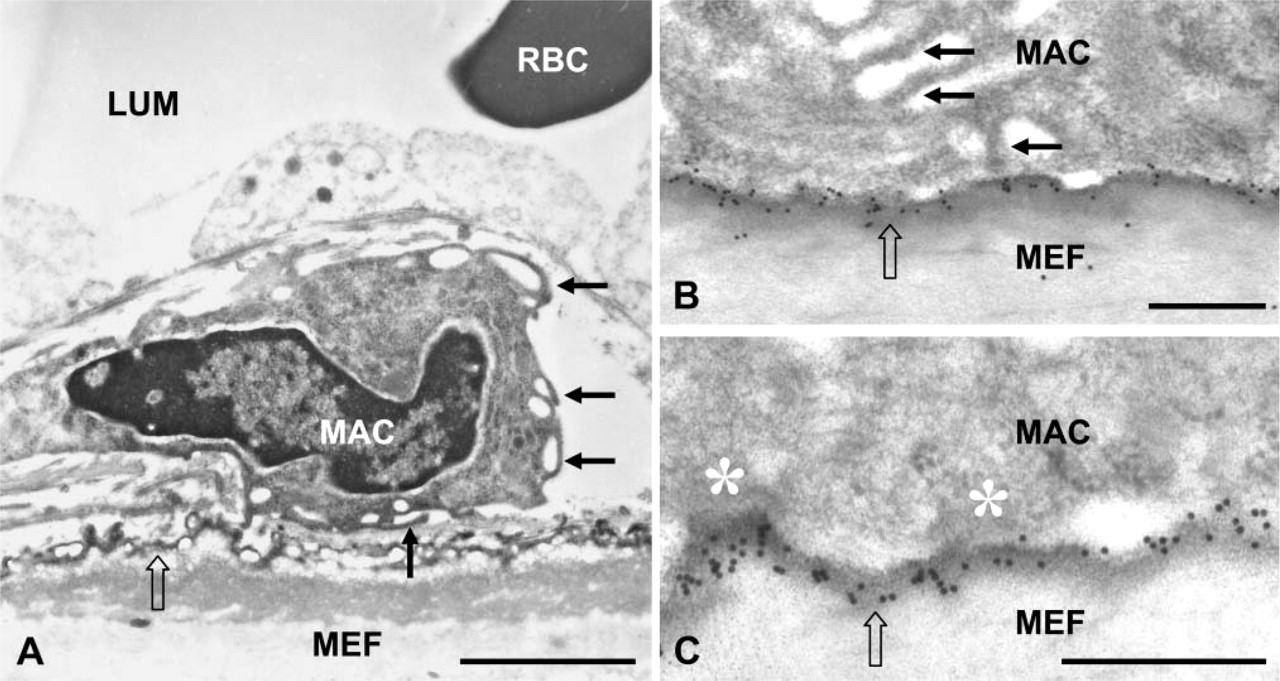
Macrophage interactions with mineralized elastic lamellae and OPN in EDTA-decalcified samples of Mgp−/− aorta. (
As outlined earlier, MGP deficiency in mice is lethal as a result of hemorrhagic rupture of calcified arteries. On the other hand, inactivating mutations of MGP in humans results in Keutel syndrome, where vascular calcification is not observed (but there is ectopic cartilage calcification), and patients have a normal life expectancy (Munroe et al. 1999). This difference in arteries prompted us to investigate whether OPN is expressed in human arteries as a protective mineralization inhibitor. Transmission electron microscopy of thin sections of mouse and human arteries after immunogold labeling for OPN revealed an absence of OPN in normal mouse aorta (Figure 7A), whereas intense labeling for OPN was observed throughout elastic fibers in the human artery (Figure 7B).
Discussion
Although physiological mineralization of the skeleton and dentition serves important supportive, protective, and masticatory functions, ectopic pathological mineralization often interferes with tissue function and can be life threatening, particularly in the case of vascular calcification. Pathological mineralization alters biomechanical properties, modifies the cell-signaling environment, and influences gene expression and cell differentiation. Whereas normally mineralizing tissues have abundant ECM proteins that function to provide the scaffolding and regulatory controls guiding physiological mineralization, “soft” tissues that do not normally mineralize are widely thought to contain potent inhibitors of mineralization. For blood vessels, MGP provides this inhibitory function (Luo et al. 1997); for other soft tissues, different inhibitors likely serve a similar role in preventing their mineralization. In tissues where pathological mineralization has already occurred in the absence of sufficient inhibitory constraints, secondary “damage control” inhibitors such as OPN may be upregulated in an attempt to limit further mineralization (McKee et al. 1995b; Giachelli et al. 2005b). In the present study on mineralized aortas from Mgp−/− mice, we show upregulation of OPN in response to mineral formation, its binding to surfaces of mineralized elastic lamellae, and its polymerization into high molecular mass forms by TG2. Moreover, we provide additional data suggesting that such events involving RGD-containing OPN might serve to promote macrophage adhesion as part of an attempt to clear ectopic deposits of mineral from the vessel wall.
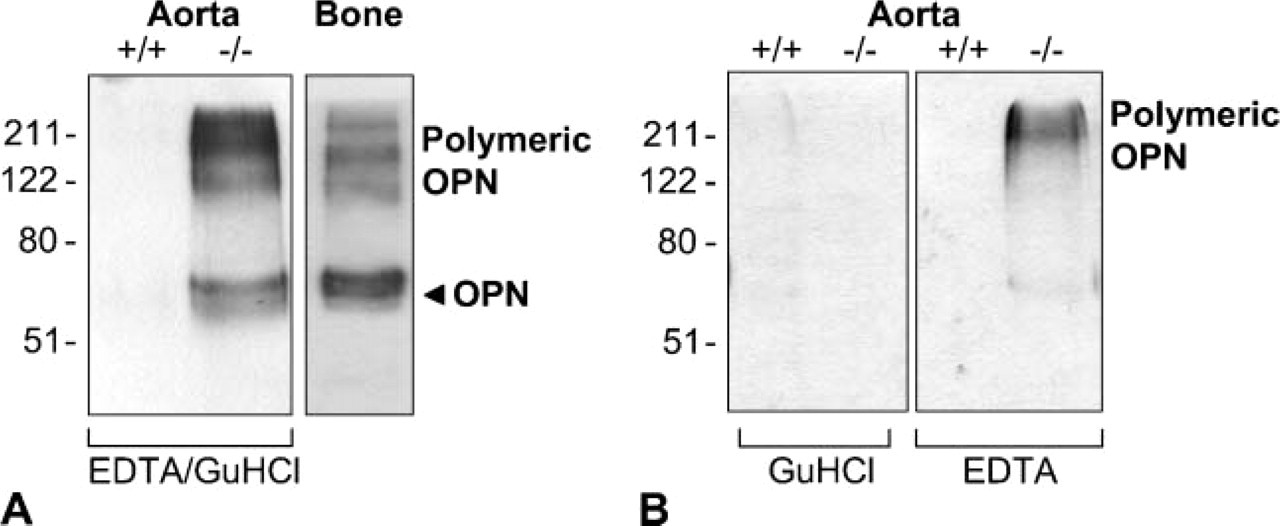
OPN upregulation and polymerization in Mgp−/− aorta as analyzed by Western blotting. (
OPN is abundant in essentially all mineralized tissues (excluding tooth enamel) and also in most pathologically mineralizing tissues (Giachelli 2005a; Giachelli et al. 2005b). Additional roles for this multifunctional protein include important immune system functions (Ashkar et al. 2000) and an influence on the survival and migration of tumor cells (Rittling and Chambers 2004; Sodek et al. 2006). In mineralizing ECMs such as bones and teeth and in pathological mineral deposits in other tissues, OPN is consistently found not only within mineralized areas but is highly concentrated at interfaces and surfaces of mineralized structures. OPN binds directly to hydroxyapatite, calcium oxalate, and calcite crystal surfaces, and this binding potently inhibits crystal growth (McKee et al. 1992; Boskey et al. 1993; Goldberg and Hunter 1995; Asplin et al. 1998). OPN is constitutively expressed by mineralized tissue-resident cells and may be upregulated in these and other cell types, often as a response to local changes in the microenvironment, particularly to shifts in phosphate (Pi) levels (Johnson et al. 2003; Harmey et al. 2004; Giachelli et al. 2005b). Increased extracellular Pi induces both physiological and pathological mineralization both in vitro (Bellows et al. 1992; Steitz et al. 2001) and in vivo in several transgenic mouse models and in human diseases resulting from deficiencies in enzymes regulating extracellular pyrophosphate and Pi levels (Henthorn and Whyte 1992; Ho et al. 2000; Johnson et al. 2003). In mineralized aortas from Mgp−/− mice, OPN is likely expressed by resident vascular smooth muscle cells (VSMCs) as a response to increased local Pi concentrations (Steitz et al. 2001; Giachelli et al. 2005b). Changes in Pi levels have also been shown to influence OPN expression in osteoblasts (Beck and Knecht 2003). Although OPN is also expressed by macrophages (McKee and Nanci 1996; Giachelli et al. 1998) found in the Mgp−/− aortas, their contribution of this protein to the surfaces of the OPN-rich mineralized elastic lamellae is likely minimal, given the known upregulation of OPN by VSMCs and given that macrophages appear in the vessel wall only after mineralization has occurred and only after OPN has accumulated at the surface of the lamellae.
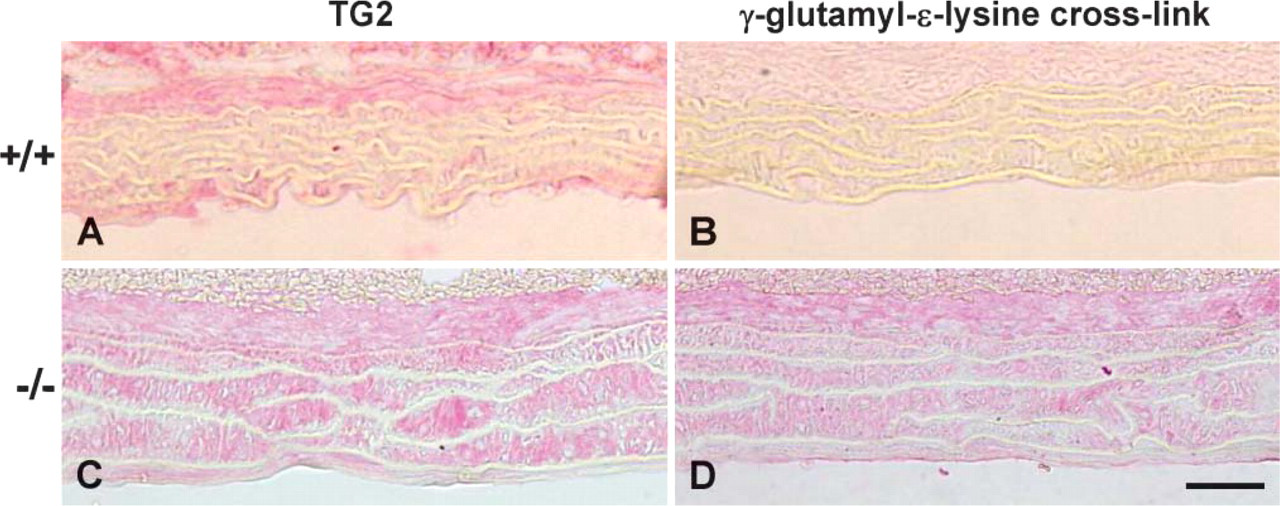
Light micrographs of paraffin sections of wild-type (+/+) and Mgp-deficient (−/−) aortas at 4 weeks of age after immunohistochemistry for transglutaminase 2 (TG2) and γ-glutamyl-∊-lysine crosslinks. Whereas weak or absent immunostaining of the arterial media is observed in wild-type mice (
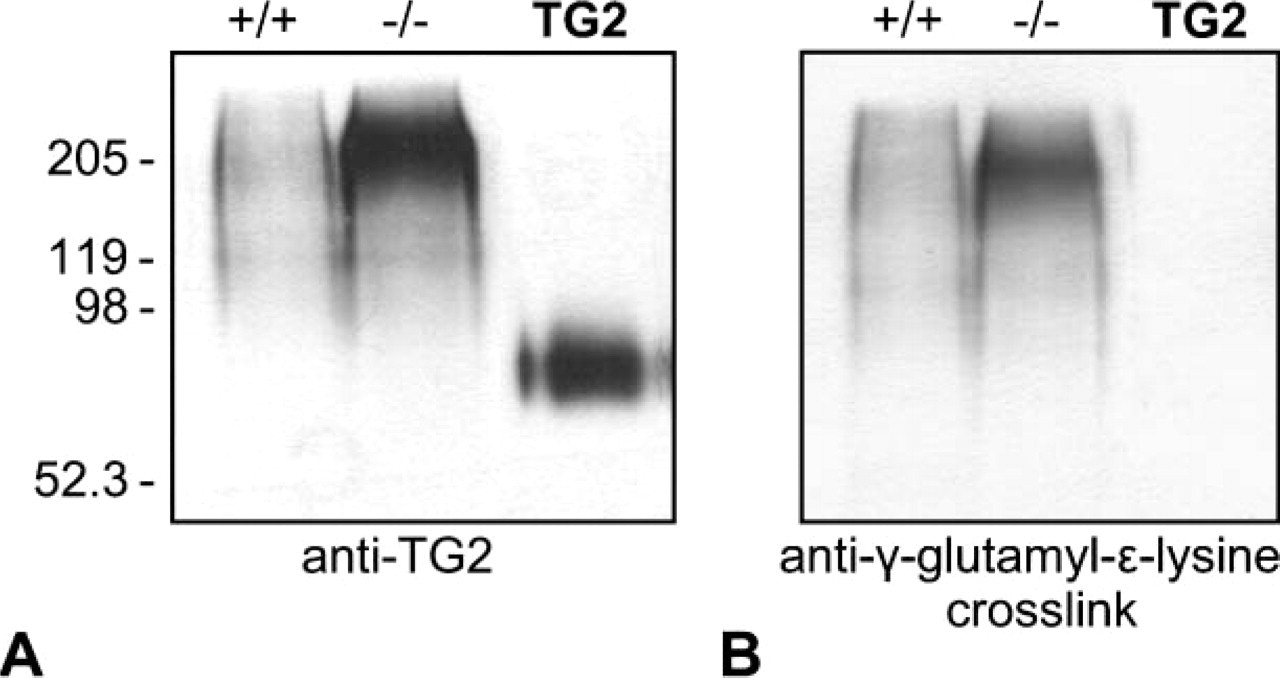
TG2 and isopeptide bond upregulation in Mgp−/− aorta. Mgp−/− aortas were extracted with demineralizing EDTA/Gu-HCl treatment and analyzed by Western blotting. Purified TG2 as a positive control for immunoblotting is shown at the right of each panel. (
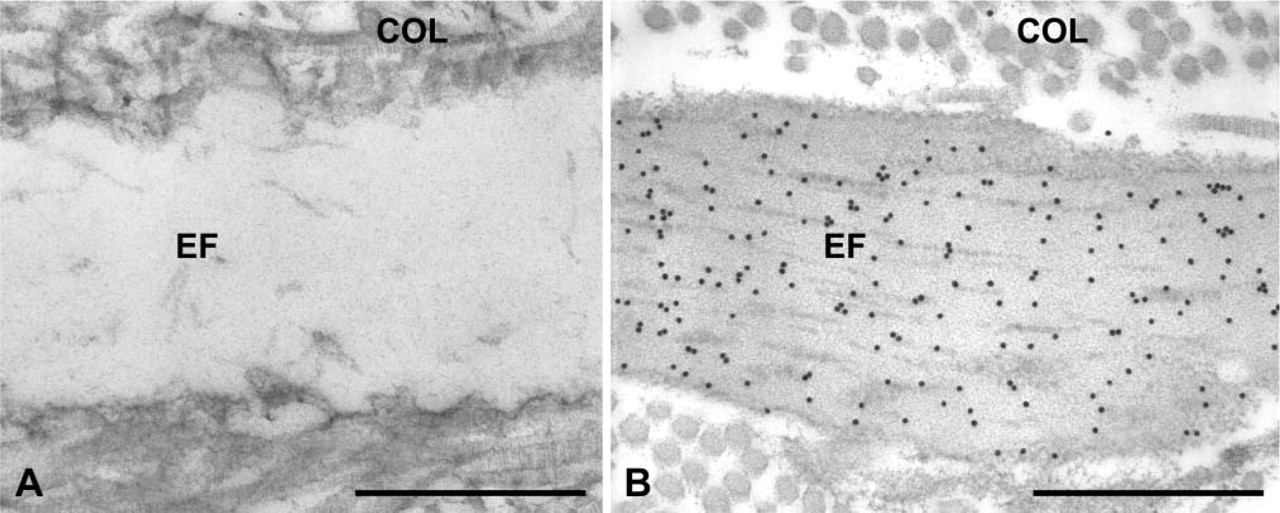
Ultrastructural immunolocalization of OPN in mouse and human artery. (
As detected by Western blotting, OPN in Mgp−/− aortas was found predominantly in its polymeric, high molecular mass form (Prince et al. 1991; Sørensen et al. 1994). This form of OPN arises from the enzymatic, covalent crosslinking activity of either TG2 or Factor XIIIA (Prince et al. 1991; Sørensen et al. 1994; Kaartinen et al. 1997,1999,2002), both members of the transglutaminase family of protein-crosslinking enzymes that create covalently linked, large protein polymers of its substrate proteins (Lorand and Graham 2003). The crosslinked nature of this OPN is supported by the upregulation of TG2 and by the formation of γ-glutamyl-∊-lysine crosslinks in the Mgp−/− aortas at the sites where OPN is found. Although the role of polymeric OPN vs its monomeric form is not fully understood, the crosslinking process is likely linked to cell-matrix interactions because TG2 has been shown to be important for cell adhesion, spreading, and signaling in many cell types (Akimov et al. 2000; Akimov and Belkin 2001a,b). Given the known cellular effects of both OPN and TG2 in diverse tissues, and given that OPN is a substrate for TG2 and that both are upregulated in Mgp−/− aortas to form polymer OPN, it is reasonable to consider that OPN, in addition to restricting mineral growth, could also act as a macrophage adhesion protein at the surface of mineral deposits in the vessel wall; its location there might facilitate clearance of mineral by phagocytosis. In previous work on bone wound healing, we reported that OPN-coated bone particles were phagocytosed by macrophages at the wound site, and OPN opsonization was proposed to facilitate this (McKee and Nanci 1996). Consistent with this proposal is our observation that macrophages make contact with the surface of OPN-coated mineralized elastic lamellae in Mgp−/− aortas. It is possible that TG2 crosslinking of OPN may be involved in this putative opsonization process because TG2 has been implicated in monocyte cell adhesion (Akimov and Belkin 2001b), and TG2-deficient macrophages show defective TGFβ-mediated phagocytosis (Szondy et al. 2003).
MGP has emerged as a major regulator (inhibitor) of soft tissue (blood vessels and cartilage) calcification, and a number of key studies have contributed to our understanding of its function. One view suggests that MGP exerts its effects by acting as an ion scavenger and hence preventing the availability of calcium for mineral deposition (Schinke et al. 1999; Price et al. 2000). A second view describes MGP as an inhibitor of BMP-2 function (Boström et al. 2001). Although the precise mechanisms behind MGP function are poorly understood, it is known that the Gla residues play a critical role in its function because their deletion abolishes its mineral-inhibiting activity (Murshed et al. 2004). Moreover, inhibition of vitamin K production in rats, which is a cofactor required for the γ-carboxylation reaction, results in vascular calcification similar to that observed in Mgp knockout mice (Price et al. 2000). Mutations in the human MGP gene result in a nonfunctional protein causing Keutel syndrome, where patients have abnormal and ectopic cartilage calcification (along with pulmonary artery and tracheobronchial stenosis and neural hearing loss) phenotypically similar to that observed in the cartilaginous growth plates of Mgp−/− mice, but arteries are not mineralized (see below). Also, diseases arising from defective vitamin K metabolism or from exposure to compounds such as warfarin or phenytoin that interfere with γ-carboxylation via vitamin K have manifestations similar to Keutel syndrome (Pettifor and Benson 1975; Pauli et al. 1987; Howe et al. 1995).
The most obvious phenotypic difference between inactivating mutations of MGP in humans vs Mgp gene deficency in mice is that whereas mice die as a result of arterial rupture and hemorrhaging from calcified blood vessels at ∼6-8 weeks of age, humans with MGP mutations lack vascular calcification and have a normal life expectancy (Munroe et al. 1999). Based on our data in the present study showing that OPN is not a constituent of normal, unmineralized elastic lamellae in the mouse, but that in humans OPN can be immunolocalized within the elastic lamellae of human arteries that have not mineralized, we propose that Keutel syndrome patients lacking functional MGP protein have arteries containing mineral-inhibiting OPN that prevents them from calcifying and allows for the extended human survival compared with mice lacking Mgp. The presence of an endogenous, local mineralization inhibitor in human arteries likely decreases their susceptibility for ectopic mineralization. It is also possible that OPN is deposited into the elastic lamellae during development and that the binding is stabilized by transglutaminase activity and isopeptide crosslinks. TG2 appears to be a normal component of healthy human arteries and is expressed by endothelial and vascular smooth muscle cells (Sumi et al. 2002). Also noteworthy is that many of the microfibril components found in the elastic lamellae, such as fibrillin and microfibril-associated glycoprotein, are also transglutaminase substrates (Brown-Augsburger et al. 1994; Qian and Glanville 1997).
In summary, we have shown the upregulation of OPN and its polymerized (covalently crosslinked) form in mineralized aorta from Mgp−/− mice and have demonstrated an OPN-rich coating at the surface of the mineralized elastic lamellae. This OPN likely serves as a secondary response to inhibit further mineral growth into the collagenous ECM of the arterial wall in the absence of Mgp and also serves to initiate removal of the mineral by macrophages. Consistent with previous reports from Giachelli and coworkers (Steitz et al. 2001; Speer et al. 2002), OPN appears to act as an inducible “damage control” mineralization inhibitor at ectopic sites of calcification and, in humans, may be an endogenous protective component preventing arterial calcification.
Footnotes
Acknowledgements
This study was supported by operating grants from the Canadian Institutes of Health Resarch (to MTK and MDM) and from the National Institutes of Health (NIH), Bethesda, MD (to GK).
We thank Dr. Larry W. Fisher (National Institute of Dental and Craniofacial Researh, NIH, Bethesda, MD) and Dr. Leon Schurgers (University of Maastricht, The Netherlands) for generously donating antibodies and proteins for this study. We also thank Ms. Lydia Malynowsky, Dr. Sherif El-Maadawy, and Dr. Neelambar Kaipatur for assistance with the immunohistochemistry, and Dr. Cecilia M. Giachelli and Dr. Charles E. Murry (University of Washington, Seattle, WA) for providing the human tissue.
