Abstract
β1–6 branching of
G
Galectin-3 is expressed in a variety of cells including epithelial and endothelial cells (Glinsky et al. 2001; Lin et al. 2002; Mengwasser et al. 2002; Khaldoyanidi et al. 2003), activated macrophages (Dong and Hughes 1997; Kim et al. 2003), and dendritic cells (Swarte et al. 1998; Vray et al. 2004). A potential role for galectin-3 in inflammation has been suggested by experiments using galectin-3 null mice (Colnot et al. 1998; Hsu et al. 2000; Bernardes et al. 2006). In fact, recent evidence suggests that galectin-3 is a pro-inflammatory protein (Colnot et al. 1998; Hsu et al. 2000). It downregulates interleukin (IL)-5 gene expression in human eosinophils (Cortegano et al. 1998), activates NADPH oxidase (Almkvist et al. 2004), stimulates superoxide production from neutrophils (Yamaoka et al. 1995), and promotes monocyte chemotaxis (Sano et al. 2000). In the extracellular space, galectin-3 may act as a de-adhesion molecule, as shown in the interaction of thymocytes and thymic microenvironmental cells (Villa-Verde et al. 2002) and stimulate cell migration (Silva-Monteiro et al. 2007) and cell death (Stillman et al. 2006; Silva-Monteiro et al. 2007). These diverse galectin-3 functions seem related to the ligands engaged in different cell types and tissues.
As a lectin, galectin-3 recognizes preferentially polylactosamines found in tri- or tetra-antennary
Materials and Methods
Materials
All reagents were purchased from Sigma (St Louis, MO) unless stated otherwise and were of analytical grade or higher. Either biotin- or digoxigenin-conjugated lectins used were L-PHA,
Cells and Antibodies
Cell lines used were the murine melanoma B16-F10 and two sarcoma cell lines (CCR2 and Σ12). Murine melanomas were cultured in RPMI 1640, supplemented with 10% fetal calf serum (Gibco; Grand Island, NY) in a humidified incubator equilibrated with 5% CO2 at 37C. CCR2 and Σ12 cells were cultured in DMEM supplemented with 10% fetal calf serum under the same conditions. Cells were harvested at subconfluence with trypsin and EDTA. To inhibit the processing of
The hybridoma-secreting rat anti-galectin-3 monoclonal antibody (TIB166, M3/38) was purchased from the American Tissue Culture Collection (ATCC; Manassas, VA) and was maintained in DMEM supplemented with 10% fetal calf serum under culture conditions. Supernatants were harvested, pooled, and tested for activity and specificity using a panel of cell extracts and the purified recombinant protein galectin-3. Rabbit anti-bacterial AP was purchased from Chemicon (Temecula, CA), anti-rat IgG horseradish peroxidase (HRP) conjugates were from KPL, Kirkegaard and Perry Laboratories (Gaithersburg, MD), and anti-rabbit IgG HRP conjugates were from Sigma.
In Vivo Experiments
Female Balb-c and C57bl/6 mice (6–8 weeks old) were used in the experiments. Animals were housed under controlled environmental conditions in 12-hr light cycles. Samples from normal murine tissues (e.g., thymus) were collected after approval by the University of São Paulo Medical School Ethics Committee for Research (both on animals and humans). When indicated, animals were inoculated with either the melanoma cell line (B16F10, isogenic to C57bl/6 mice) or the CCR2 sarcoma cell line (isogenic to Balb-c mice). Animals were sacrificed when tumors reached 0.5 cm3. Tumors were then excised and processed for histopathological analysis.
Construction of the Hybrid Galectin-3 Molecule
The human galectin-3 (gal3) gene was cloned in the pLIP6-GN vector, which encodes the AP gene,
The gal3 gene was PCR amplified from the pKK322-Gal3 clone using primer gal3NTSfi (5′-attagggcccagccggccgcagacaatttttcg-3′) and primer gal3CTNot (5′-gagaggcggccgctatcatggtatatga-3′), which added a
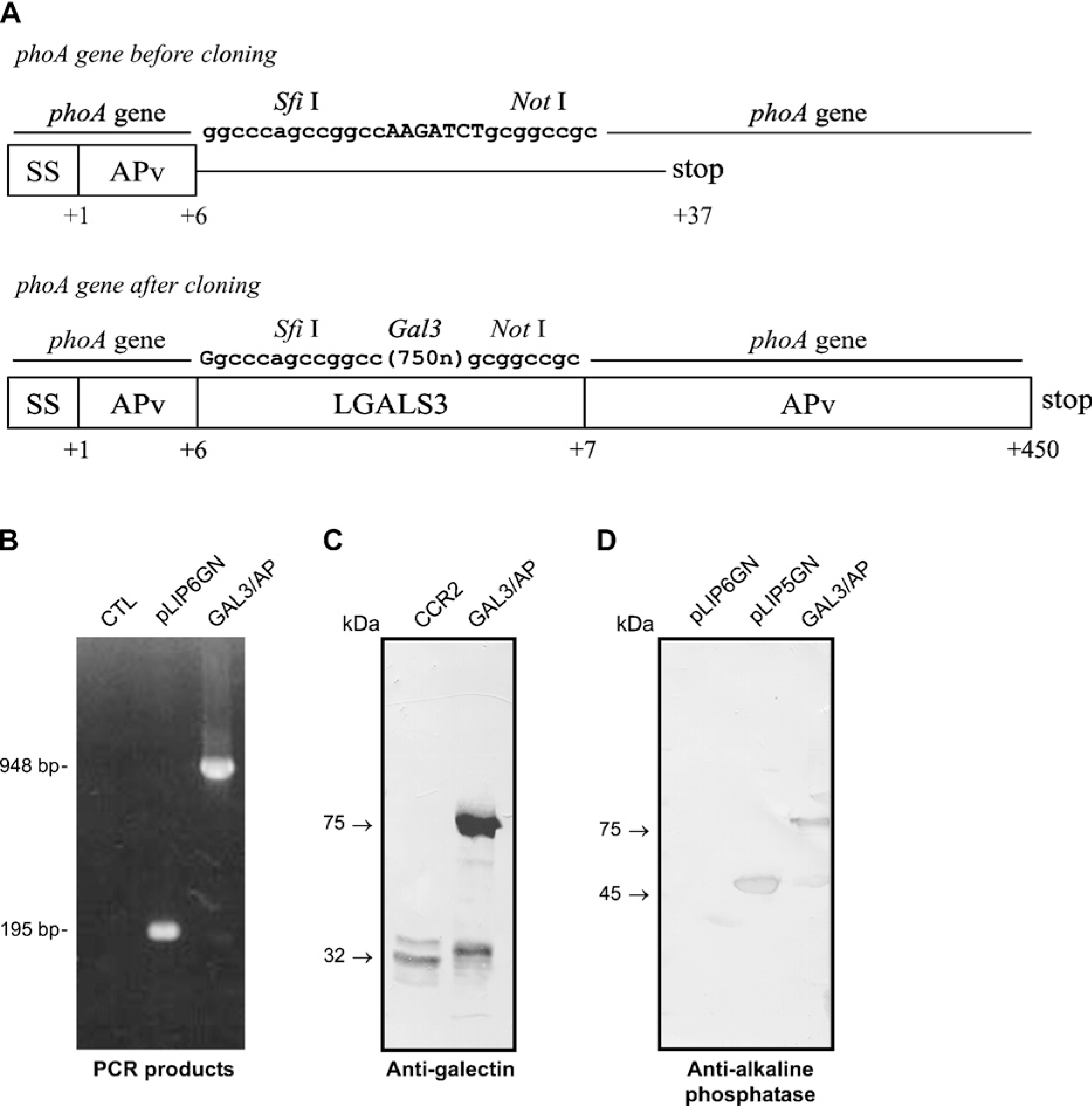
Construction and expression of a chimeric probe containing both galectin-3 and alkaline phosphatase (AP) activities. (
Protein Expression and Purification
A single clone was used to transform
Western and Lectin Blot Analysis
The presence of the recombinant fusion protein was detected by SDS-PAGE after staining with Coomassie blue or by Western blotting, using antibodies that recognize galectin-3 and AP or alternatively using the AP substrate, 5-bromo-4-chloro-3-indolyl phosphate in the presence of nitroblue tetrazolium solution (NBT/BCIP; Boehringer Mannheim, Indianapolis, IN). After periplasmic extraction, the samples were clarified by a 20-min centrifugation at 4C, and the protein concentration was determined using the BCA protein assay reagent (Pierce Biotechnology; Rockford, IL). Equal amounts of periplasmic extracts were boiled in SDS sample buffer under reducing conditions (240 mM Tris–HCl, pH 6.8, SDS 0.8%, glycerol 40%, β-mercaptoethanol 200 mM, and bromophenol blue 0.02%) for 5 min. Samples were separated by electrophoresis on 12.5% SDS–polyacrylamide gels under reducing conditions and transferred to a polyvinylidene difluoride membrane (Millipore; Bedford, MA). After protein transfer, the membranes were incubated with 5% non-fat dry milk dissolved in PBS (10 mM sodium phosphate, 150 mM NaCl, pH 7.2) and incubated with rat monoclonal anti-galectin-3 M3/38 or with 2 μg/ml rabbit polyclonal anti-bacterial AP overnight at 4C. After three washes with 5% non-fat milk in PBS, the membranes were incubated for 1 hr with 0.5 μg/ml HRP-conjugated rabbit anti-rat IgG or HRP-conjugated anti-rabbit IgG. Following three washes, the reaction was developed using 3.75 mg DAB in Tris-buffered saline (50 mM Tris–HCl, pH 7.5, containing 150 mM NaCl) in the presence of H2O2 (1 μl/ml; Fluka, Seelze, Germany).
For lectin blots, protein extracts were transferred to PVDF membranes, which were blocked with PBS-1% BSA and incubated with biotin-conjugated L-PHA (
Enzyme-linked Lectin Assay (ELLA) and Statistical Analysis
ELLA was performed to evaluate the lectin and AP activity of gal3/AP. To assay the lectin activity, wells from a flat-bottom microtiter plate from Corning (Corning, NY) were coated with 10 μg/ml of laminin-1 for 1 hr at 37C and washed with PBS. Nonspecific sites were blocked with 1% BSA for 30 min at 37C and washed with PBS. Increasing concentrations of gal3/AP periplasmic extract (in the range of 0–20 μg/ml) were added to the wells either in the absence or presence of 100 mM of lactose (Sigma) and incubated overnight at 4C. After washing, because affinity constants of galectin-3 binding are in the micromolar range, the bound lectin was then cross-linked with 1% paraformaldehyde for 15 min. The wells were then washed three times with blocking solution and incubated with rat monoclonal anti-galectin-3 M3/38 for 1 hr at 37C. After three washes, 0.5 μg/ml rabbit anti-rat IgG HRP conjugate was added and incubated for 1 hr at 37C. Following washes, the freshly prepared substrate (0.4 mg/ml
Direct Binding Assay
Direct binding assay was performed to evaluate the fine specificity of gal3/AP. Diverse glycans (Glycan Array version v2.3, NIGMS Consortium for functional glycomics) were added in a 384-well HBC NeutrAvidin Black plate (ligand coating, 30 pmol/well). For the complete list of glycans used in this assay, please consult the site http://www.functionalglycomics.org/static/consortium/resources/resourcecoreh2.shtml. The plate was then washed with PBS with 0.05% Tween 20 (wash buffer) and incubated with 15 μg/ml gal3/AP in wash buffer and 1% BSA. The bound lectin was detected with anti-AP antibodies followed by incubation with Alexa 488-conjugated secondary antibodies, using a plate fluorimeter.
Tissue Staining
Sequential tissue microarray slides containing panels of human tumors of diverse origins and their normal counterparts were obtained from the Hospital A. C. Camargo, São Paulo, Brazil after approval from the local Ethics Committee and tumor bank director. Sections of buffered paraformaldehyde-fixed, paraffin-embedded thymuses or experimental murine sarcomas or melanomas were also used. Tissue sections were routinely processed, and antigen retrieval was done by heating the specimens in citrate–phosphate buffer, pH 6.0, in the microwave for 10 min. After blocking with PBS-1% BSA, the sections were incubated with 30 μg/ml gal3/AP in the blocking solution overnight at 4C, either in the absence or presence of 100 mM lactose. After washing, the reaction was developed using Fast Red in Naftol phosphate Tris–HCl buffer (1 mg/ml; Dako, Carpinteria, CA). Specimens were also stained using anti-galectin-3, anti-CD34 antibodies, and L-PHA and then developed with peroxidase-conjugated secondary reagents.
Results
Production of the Gal3/AP Hybrid Protein
Galectin-3 cDNA was successfully subcloned into the pLIP6-GN vector, giving rise to the construct shown in Figure 1A. A blue colony was screened by PCR, which confirmed the presence of a 948-base pair band corresponding to the galectin-3 gene (Figure 1B). This clone was further sequenced confirming the correct in-frame cloning of the galectin-3 cDNA. The pLIP6/gal3 clone was subsequentially used to transform
Gal3/AP Has Both Lectin and AP Activities
Periplasmic extracts were assayed for both AP and lectin activities. Galectin-3 interacts with polylactosamines containing glycoconjugates including laminin. To examine the lectin activity of gal3/AP, ELISA plates were coated with laminin-1, and increasing concentrations of gal3/AP were incubated in the presence or absence of 100 mM lactose. Figure 2A shows that gal3/AP bound to laminin-1 in a dose-dependent manner and that this interaction was inhibited by lactose. To investigate the AP activity of the chimeric protein,
Gal3/AP Has the Same Specificity of Parental Galectin-3
Direct binding of gal3/AP to a panel of >190 glycans (Consortium for Functional Glycomics, Glycan plate arrays, versions 1.2 and 2.7) allowed us to determine the specificity of the chimeric lectin. Table 1 indicates the eight distinct glycans recognized by gal3/AP with higher affinity. These are essentially the same glycans recognized by the parental molecule, using the same version of the glycan plate array (galectin-3, data from 2004, available at the site of the Consortium for Functional Glycomics, http://www.functionalglycomics.org, under CFG data, Core H-glycan screening). Signal-to-noise ratio is an indirect measurement of the apparent affinity. Sialylation did not interfere with gal3/AP binding (Table 1). This was further confirmed in lectin blots using sialidase-treated protein extracts (data not shown). Note that more recently the Consortium for Functional Glycomics extended the platform using a printed array, which now includes >250 different glycans. Among them, galectin-3 bound strongly to two other glycans that were not present in the plate array used herein, namely, Galβ1–4GalNAcaα1–3(Fucα1–2)Galβ1–4GlcNAcβ and GlcNAcβ1–3Galβ1–4GlcNAcβ1–3Galβ1–4GlcNAcβ which, however, share similar determinants to blood group A (type 2) and dilactosamine (LN)2, respectively.
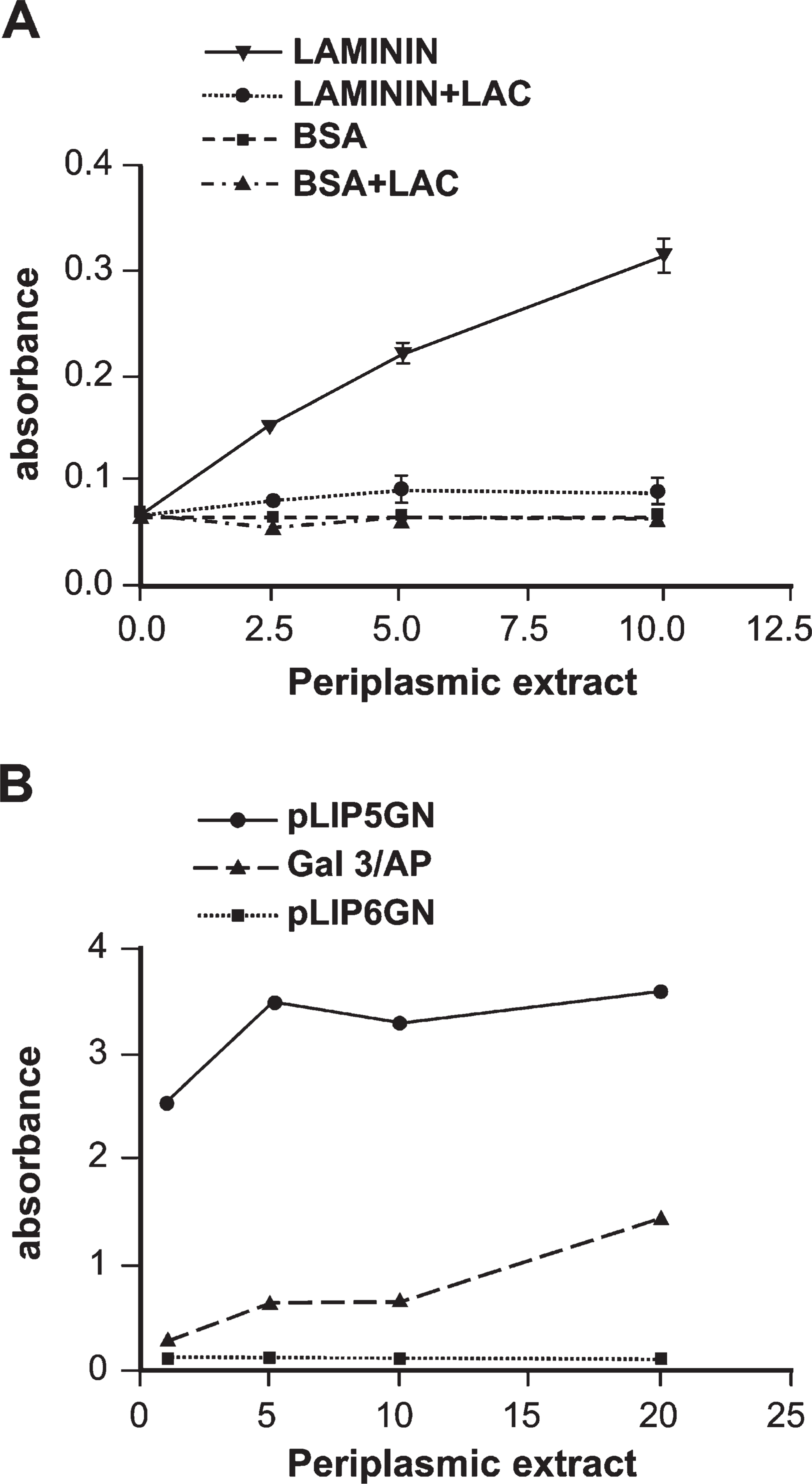
Gal3/AP shows both lectin and AP activities. (
Gal3/AP binding specificity
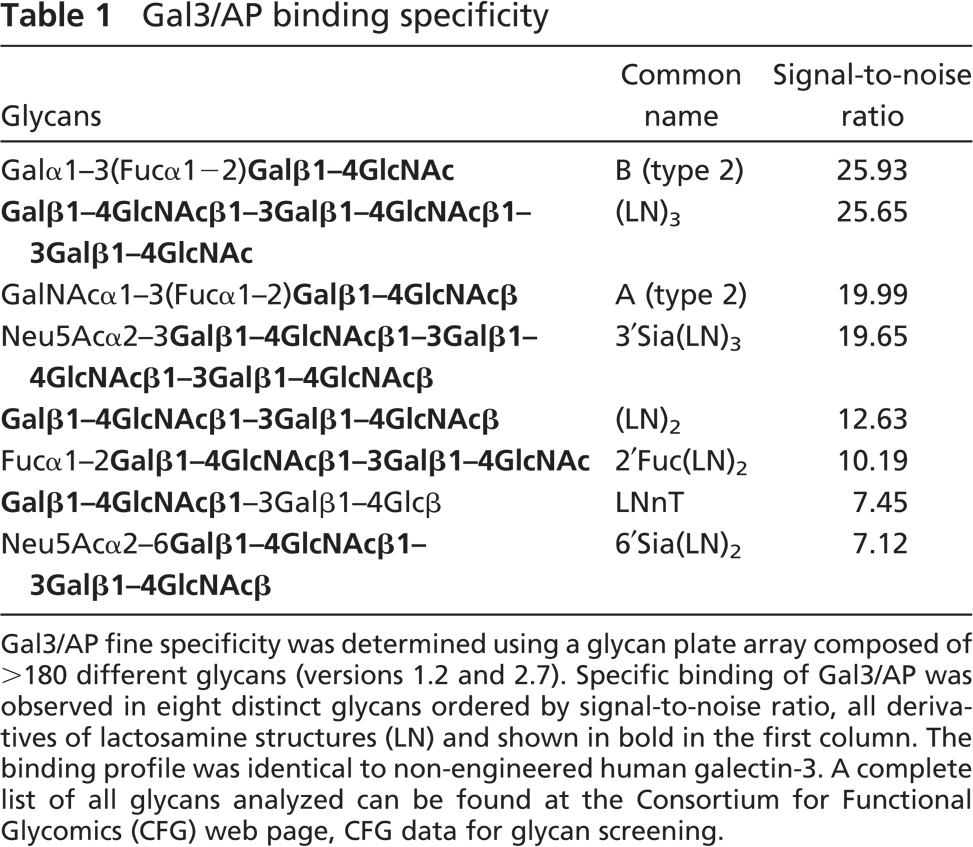
Gal3/AP fine specificity was determined using a glycan plate array composed of >180 different glycans (versions 1.2 and 2.7). Specific binding of Gal3/AP was observed in eight distinct glycans ordered by signal-to-noise ratio, all derivatives of lactosamine structures (LN) and shown in bold in the first column. The binding profile was identical to non-engineered human galectin-3. A complete list of all glycans analyzed can be found at the Consortium for Functional Glycomics (CFG) web page, CFG data for glycan screening.
Gal3/AP Recognition Depended at Least in Part on the N -linked Oligosaccharides of Different Glycoproteins
Gal3/AP was useful in ligand blottings, as shown in Figure 3. The pattern of gal3/AP binding was compared with L-PHA binding in protein extracts derived from established cell lines. To evaluate the binding of gal3/AP to
In a second set of experiments, B16F10 and Σ12 cells were treated with 1 μg/ml of swainsonine (SW), an inhibitor of mannosidase II, for 48 hr. After that, protein extracts from B16F10 and Σ12 cells were subjected to lectin blot analyses, using L-PHA and gal3/AP (Figure 3B). Several bands were recognized by L-PHA in B16F10 extracts. Major bands included a polydisperse band of 120–140 kDa and an 80-kDa band. The 120-to 140-kDa polydisperse band was also found in Σ12 cell extracts. This band was also recognized by Gal3/AP. Upon SW treatment, a significant decrease in the binding of both L-PHA and Gal3/AP was found. The 80-kDa band was no longer identified after SW treatment in B16F10 cells. On the other hand, two smaller bands of 50 and 60 kDa apparent molecular mass were observed upon SW treatment. It is clear from both approaches that L-PHA and gal3/AP binding may be similar but not identical and are at least in part dependent on
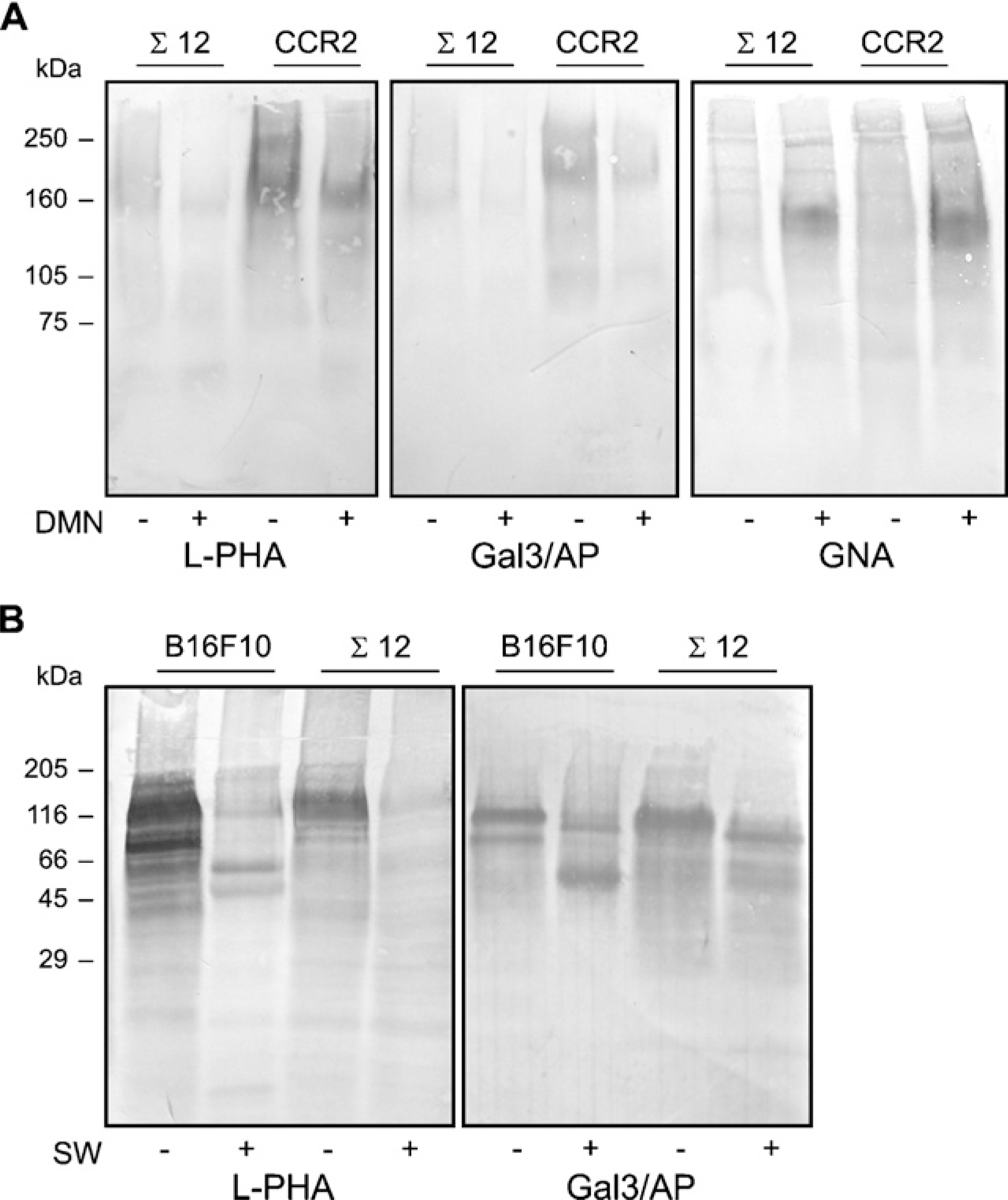
L-PHA and gal3/AP recognition patterns are similar but not identical. We compared the binding profiles of both leukoagglutinin from
Expression of Galectin-3 Ligands in Different Types of Tumors by Tissue Assay
Gal3/AP could be used to identify galectin-3 ligands in situ. Paraffin-embedded murine thymuses (see example in Figures 4A–4D) and sarcomas (derived from CCR2 murine cell line, Figures 4E–4H) were used to compare the binding pattern of the plant lectin L-PHA (Figures 4B and 4F) and gal3/AP (Figure 4C, 4D, 4G, and 4H). Gal3/AP binding was abolished by coincubation of the lectin with 100 mM lactose (data not shown and supplemental figures, SF1–SF9), as observed in direct binding assays using laminin as a galectin-3 ligand (Figure 2A). Presence of galectin-3 is also shown in Figure 4 (A, E).
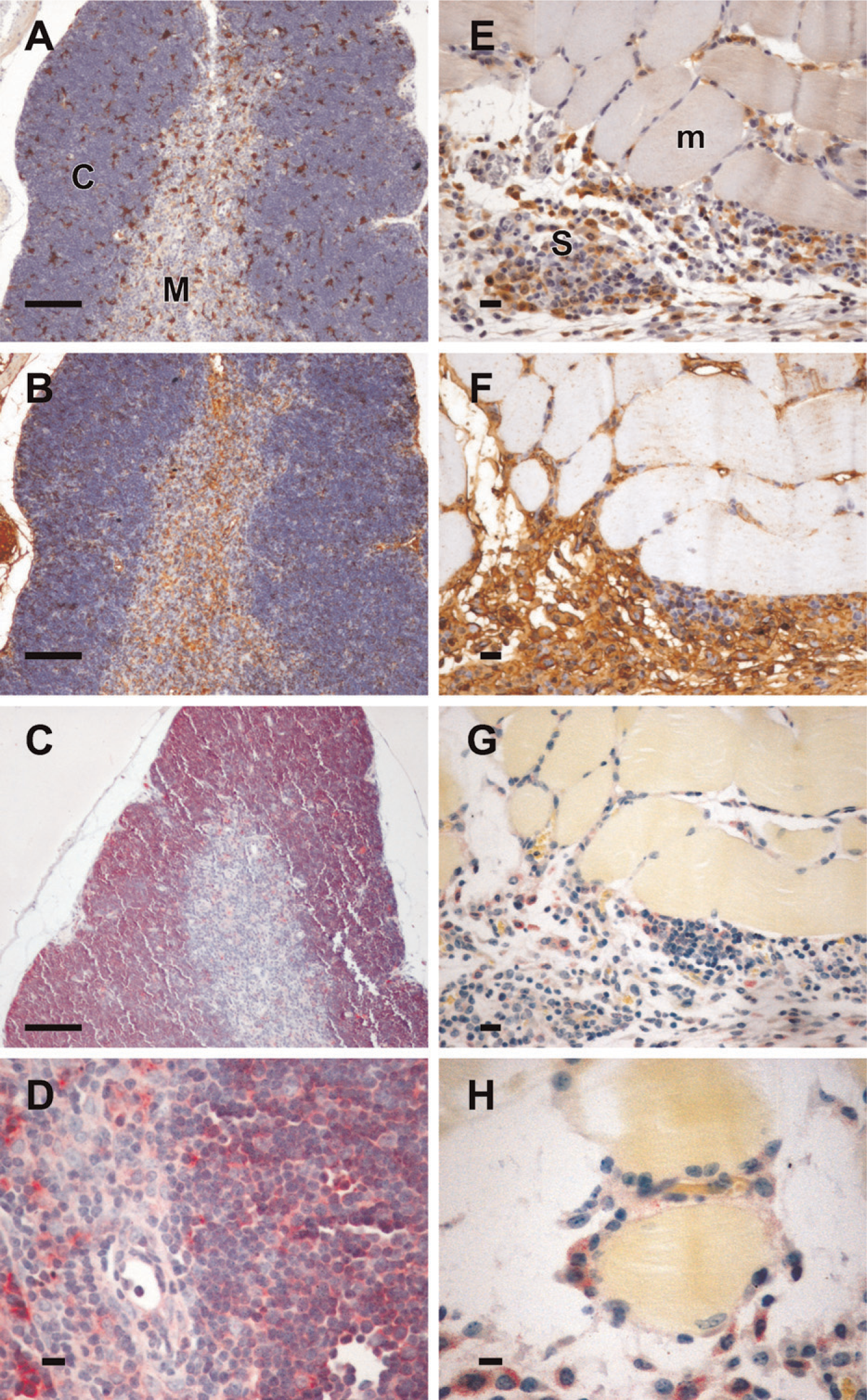
Lectin histochemistry using Gal3/AP indicates that not all L-PHA-positive cells bear galectin-3 ligands. Murine thymus (
In the murine thymus, cells expressing galectin-3 are scattered in cortex and medulla. Whereas L-PHA structures are found in both thymic compartments, especially in the vascular structures in the medulla, gal3/AP bound mainly cortical cells (Figure 4C) and a few cells in the medulla (Figure 4D). De novo expression of sialyltransferases is a common hallmark of thymocyte differentiation (Wu et al. 1997; Starr et al. 2003). As mentioned above, loss of galectin-3 binding was not due to masking of galectin-3 ligands by the addition of sialic acid residues because treatment of thymocyte extracts with sialidase did not expose novel gal3/AP binding sites (data not shown). Experimental sarcomas (Figures 4E–4H) express galectin-3 (Figure 4E) and migrate onto basement membranes of muscle fibers, as discussed elsewhere (Jasiulionis et al. 1996) and indicated in Figure 4H (note that Gal3/AP-stained sarcoma cells are adhered to muscle fibers). In this example, gal3/AP binding was not as evident as L-PHA binding, although as shown in Figures 4F–4H, infiltrating sarcoma cells stained with both plant and animal lectins.
Specimens from different human tumors were also analyzed using a tissue microarray. Examples of tissue staining by Gal-3/AP that were more representative are shown in Figure 5, which depicts examples of staining from breast carcinomas (Figures 5A and 5B), prostate carcinomas (Figures 5C and 5D), a glioblastoma (Figure 5E), and a melanoma (Figure 5F). Strong immunoreactivity was detected in the apical portion of well-differentiated breast carcinomas, including in the lumen secretion (Figure 5A). A trend to lose gal3/AP binding was observed in poorly differentiated tumors (Figure 5B). A similar trend was observed in prostate cancer (Figure 5C illustrates a well-differentiated tumor; Figure 5D illustrates a poorly differentiated tumor). Stromal reactivity was intense and found either in vascular elements (Figure 5D) or reactive glial cells (as indicated in the glioblastoma, arrow in Figure 5E). Heterogeneity of gal3/AP binding was observed in human melanomas, as illustrated in Figure 5F (arrow indicates an example of a strongly reactive cell in an infiltrated microenvironment). Supplemental figures illustrate the broad pattern of galectin-3 ligand distribution in a selected variety of human tumors, including squamous cell carcinoma, breast, prostate, pancreas carcinomas, melanomas, neuroendocrine tumors, neuroblastomas, and glioblastomas.
In murine melanomas (Figure 6), gal3/AP bound both vascular structures and infiltrating stromal cells. Gal3/AP binding (Figures 6B and 6D) was comparable to anti-CD34 antibodies, as illustrated in Figures 6A and 6C. In this regard, the probe we have developed could be used to determine microvascular density within experimental tumors. Moreover, a subset of dendritic-like stromal cells (Figure 6D) also stained intensely with gal3/AP.
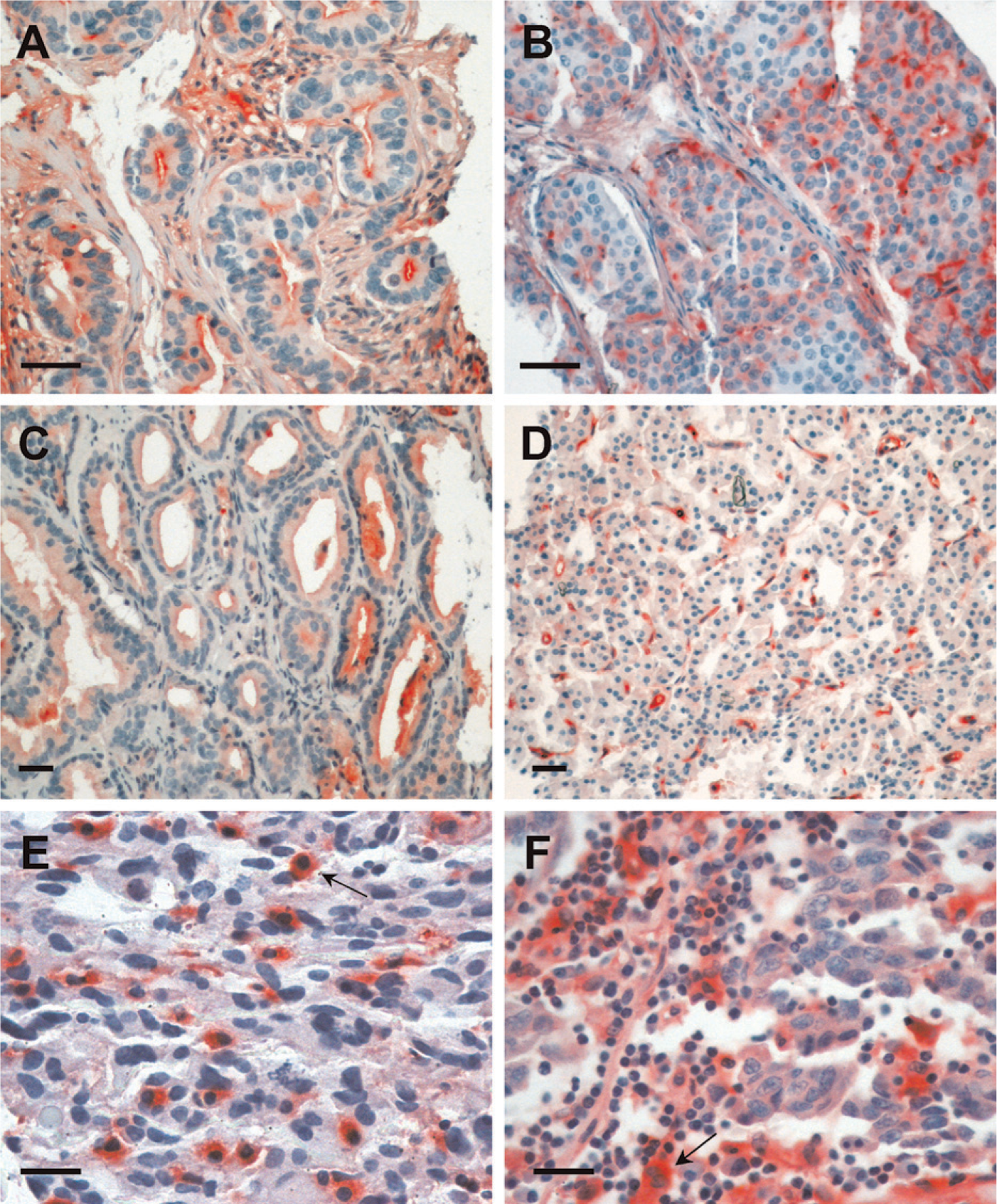
Human tumors are recognized by gal3/AP. Examples of the heterogeneous reactivity of gal3/AP in different human cancers. Breast (
Discussion
We have described here the production, expression, and some applications of gal3/AP, a hybrid molecule displaying both the carbohydrate binding properties of galectin-3 and enzymatic activity of AP. The binding profile of this chimeric molecule was compared with the binding of the plant lectin L-PHA. Although the binding specificities of L-PHA and galectin-3 are similar at the oligosaccharide level (Cummings and Kornfeld 1982; Sato and Hughes 1992; Demetriou et al. 2001), we showed here that neither glycoprotein profiling nor in situ binding of each lectin was identical. Therefore, it seems incorrect to use L-PHA and galectin-3 staining data interchangeably. As the probe presented here is derived from galectin-3 itself, it is a more adequate tool for pathophysiological studies on the roles of galectin-3 than the plant lectin. Gal3/AP will be useful to identify the binding patterns of galectin-3 and will be useful for the in situ determination of galectin-3 ligand distribution. This tool is an alternative to biotinylated lectins, which had been widely used to describe the distribution pattern of glycoconjugates in normalcy and malignancy. Indeed, biotinylated galectin-3 had been successfully used by different groups, following the pioneering work of Gabius and colleagues (Choufani et al. 1999; Plzak et al. 2001). Gal3/AP is an effective alternative to biotinylated galectin-3; the former, however, can be produced in a single step. As no chemical modifications are necessary for the generation of the chimeric probe, it is anticipated that Gal3/AP-specific activity would be rather constant from batch to batch, in addition to allowing a direct determination of galectin-3 ligands in situ, thus overcoming the potential problem of loss of specificity due to the use of secondary reagents for development of the reaction. Furthermore, Gal3/AP represents a suitable alternative for studying the distribution of galectin-3 ligands in tissues where endogenous biotin levels are high.
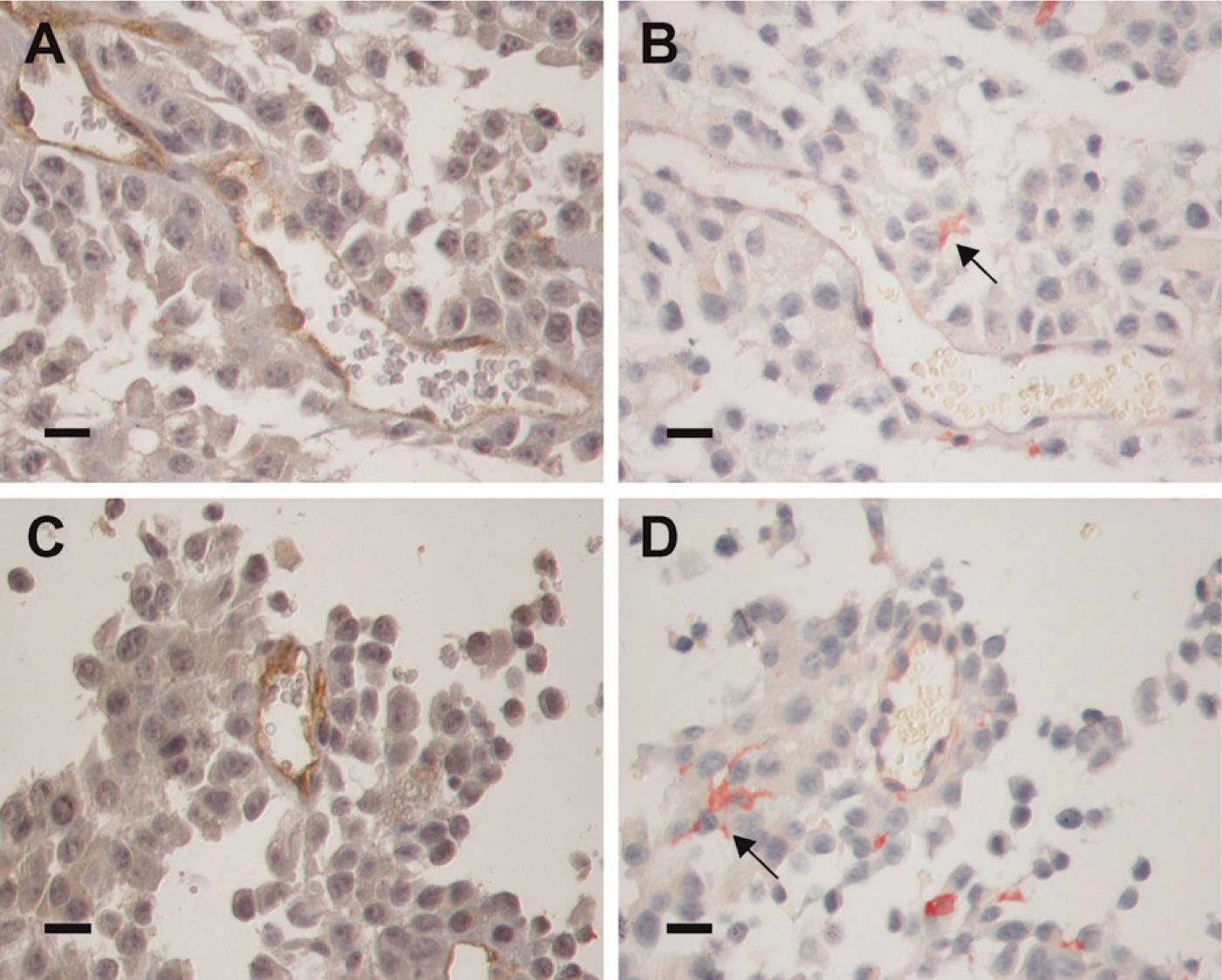
Gal3/AP is a useful probe for analysis of tumor microenvironmental cells. (
Immunopurified molecules can be probed directly with the chimeric galectin-3. Binding of the chimeric probe to immobilized glycoproteins in lectin blots has been used recently to identify different glycoforms of integrins as ligands for galectin-3 (Chammas and Bellis, unpublished data). The lectin blot and ligand overlay assays do not favor conformation-dependent protein–protein interactions, although there are a few examples of peptide–protein receptor interactions in ligand blottings in the literature (Chammas et al. 1994b). It seems likely that most of the binding partners of galectin-3 revealed in lectin blots are glycoproteins, and that the oligosaccharide residues present in the blotted glycoproteins are the actual determinants of galectin-3 binding. Indeed, lactose inhibited gal3/AP binding to blotted proteins (data not shown), as it did in ELISA assays (Figure 2A). Due to galectin-3 specificity, most proteins revealed in lectin blots are likely to be either cell surface or extracellular matrix glycoproteins. The finding that the mannosidase inhibitors, dMN and SW, led to a decreased binding of gal3/AP (Figure 3) corroborates the notion that
There is increasing interest in determining the apparently non-glycosylated intracellular partners of galectin-3. Two hybrid systems led to the identification of molecules such as gemin 1 and 4 and β-catenin as galectin-3 binding molecules, among others (Park et al. 2001; Shimura et al. 2004). Optimal conditions for binding of gal3/AP to intracellular proteins may be determined in enzyme-linked lectin assays, a useful application of this chimeric molecule. These assays are likely to be done under reducing conditions, as those found in the cytoplasm.
The most direct application of the molecule described here is its use in direct lectin histochemistry. The involvement of galectin-3 in inflammation, development of both innate and adaptive immune responses, and tumor progression is anticipated by several lines of evidence and has been reinforced by studies using galectin-3 null mice (Colnot et al. 1998; Hsu et al. 2000; Bernardes et al. 2006; Silva-Monteiro et al. 2007). Although some of these roles may be exclusively due to the functions of intracellular galectin-3, some will involve its lectin function. The chimeric probe will aid in the identification and a more precise description of tissue microenvironments where galectin-3 may play a role.
In the murine thymus, for example, most of the ligands for galectin-3 are confined to the cortical thymocytes, different from what was observed to L-PHA staining. We have shown that extracellular galectin-3 acts as a de-adhesion molecule disrupting the interactions between thymocytes and thymic microenvironmental cells, such as the cortical thymic epithelial cells found in the thymic nurse complexes (Villa-Verde et al. 2002). Data shown in Figure 4D suggest that most of the galectin-3 ligands are in immature thymocytes found in the thymic cortex. This notion was recently confirmed by independent groups of experiments. On one hand, we have shown that galectin-3 mainly affects migration of CD4+CD8+ cells (Silva-Monteiro et al. 2007), whereas Baum's group demonstrated that galectin-3 induces CD4−CD8− cell death (Stillman et al. 2006). In experimental sarcomas, the probe was useful to illustrate the pattern of dysfunctional migration of sarcoma cells on laminin-rich muscle basement membranes (Figures 4E–4H).
However, the higher impact of the use of this probe will be on studies on the clinical significance of a specific pattern of aberrant glycosylation, namely, β1–6 branching of
The survey in different tumors clearly indicated that not only tumor cells, but also microenvironmental cells as infiltrating leukocytes, express galectin-3 ligands (Figure 5 and Figure 6). Positive identification of these ligands is warranted, and gal3/AP will be a useful tool for this objective. Moreover, gal-3/AP stained vascular structures in tumor sections as well as the commonly used antibodies anti-CD31 and anti-CD34 (Figure 6). Accordingly, recent findings indicate that galectin-3 participates in tumor angiogenesis (Nangia-Makker et al. 2000; Fukushi et al. 2004; Shekhar et al. 2004) This finding will be useful to determine the microvascular density of tumors that do not express CD34 or in tumors that aberrantly express CD34, as is the case for experimental renal carcinomas (data not shown).
Finally, as Gal3/AP is a recombinant chimeric probe, its production in
Footnotes
Acknowledgements
This work was funded by FAPESP (Center for Cell-based Therapy Research, 1998/14247-6) and CNPq grants. The glycan-array analysis was conducted by the Protein–Carbohydrate Interaction Core (H) of The Consortium for Functional Glycomics funded by the National Institute of General Medical Sciences (Grant GM62116).
We are grateful to Dr. Frederic Ducancel, CEA-Saclay, Gif-sur-Yvette, France, for providing the vectors used in this work and to Dr. Paulo Lee Ho, Centro de Biotecnologia do Instituto Butantan, for sequencing the constructs. We thank the Core H staff, Ms. Angela Lee, and Mrs. Carole Davis for their help in conducting the assays.
