Abstract
A collagen column is a structure of the extracellular matrix that helps to maintain the flatness and width of gill lamella. Collagen columns are unique in that they are enfolded by plasma membrane of pillar cells that form two-dimensional vascular networks between parallel sheets of respiratory epithelia. Despite their unique structure and fundamental importance in the physiology of aquatic animals, little is known about their properties and molecular components, owing to the lack of detection methods. In this study we demonstrated that collagen columns can be visualized by staining with fluorescencelabeled concanavalin A (ConA), a lectin that specifically recognizes the trimannoside core of N-glycosylated proteins and histidine-tagged green fluorescent protein (His6-Xpress-GFP), a fluorescent substrate for transglutaminase. We constructed a three-dimensional image of a pillar cell and visualized the spatial relationship between collagen columns and contractile apparatuses within the pillar cell body. This manuscript contains online supplemental material at
Keywords
T
To analyze the nature and makeup of the pillar cells and collagen columns, a simple staining protocol is required that allows visualization of the collagen columns by fluorescence. Our previous report demonstrated that pillar cells have numerous actin fibers whose bundles line the outer side of the collagen columns and that these actin fibers are easily visualized by staining with fluorescence-labeled phalloidin (Mistry et al. 2004). This makes it possible to visualize collagen columns as negatively stained holes lining the inner side of actin fibers. However, no method for the direct staining of collagen columns has been reported to date.
In this study we report two protocols for the staining of collagen columns. First, we found that fluorescencelabeled ConA strongly stains collagen columns of gill sections. ConA is a lectin from the legume Canavalia ensiformis, which specifically recognizes the trimannoside core of the N-glycosylated proteins abundant in ECMs (Reeke et al. 1975). Second, when frozen sections of unfixed gills were thawed, dried, and then incubated with His6-Xpress-GFP, the collagen columns were specifically labeled. His6-Xpress-GFP is a fluorescent substrate for transglutaminase developed in our laboratory and covalently anchored to ECMs where transglutaminase activities are concentrated (Furutani et al. 2001). Transglutaminases are a family of calciumdependent enzymes that catalyze the covalent cross-linking of proteins by forming isopeptide bonds between peptide-bound glutamine and lysine residues (Ichinose et al. 1990; Greenberg et al. 1991; Aeschlimann and Paulsson 1994; Lorand and Graham 2003) and have an important role in the maintenance of the integrity of ECMs. These results have provided useful tools enabling us to visualize collagen columns for the first time, as well as suggesting that ECM proteins in collagen columns are covalently cross-linked and mechanically reinforced by transglutaminase.
Materials and Methods
Animals and Tissue Preparation
Japanese eels (Anguilla japonica) were purchased from a local dealer and reared in a 150-liter tank containing fresh water. Eels were anesthetized by immersion in 0.1% ethyl m-amino-benzoate methanesulfonate (MS-222 or tricaine; Nakarai, Kyoto, Japan) before being killed. Animal protocols and procedures were approved by the Institutional Animal Care and Use Committee of Tokyo Institute of Technology.
Fluorescence Histochemistry
Eels were perfused with ice-cold PBS for 2 min by intraarterial infusion to remove blood and were subsequently fixed by perfusion with 2% paraformaldehyde fixative (2% w/v paraformaldehyde in PBS) for 5 min. Gills were excised, immersed in the same fixative for 2 hr at 4C, rinsed in PBS containing 10% (w/v) sucrose, immersed in PBS containing 20% sucrose for 16 hr at 4C, and frozen in optimum cutting temperature compound (OCT, Tissue-Tek; Torrance, CA) in a cryomold (Tissue-Tek). Frozen sections (7 μm) were cut on a cryostat at −20C and mounted on glass slides (MAS-coated Superfrost; Matsunami, Osaka, Japan) and air dried at room temperature for 30 min. After being washed with PBS, sections were incubated for 1 hr at room temperature with PBS containing Alexa 488-labeled ConA (50 μg/ml; Molecular Probes, Eugene, OR), TRITC-labeled phalloidin (0.15 μM; Sigma-Aldrich, St Louis, MO), and Hoechst 33342 (0.1 μg/ml; Molecular Probes) in a moist chamber, washed three times in excess PBS, and mounted on antifade glycerol (90% glycerol, 10% 10X PBS, and 0.1% 1,4-phenylenediamine, pH 7.4). Samples were analyzed using a fluorescence microscope equipped with an ApoTome (Carl Zeiss; Oberkochen, Germany). Images were obtained with a high-resolution digital charge-coupled device camera (AxioCam HRm, Carl Zeiss) and processed using the AxioVision 4.1 imaging software (Carl Zeiss).
Activity Staining of Transglutaminase in Tissue Sections With His6-Xpress-GFP
Eels were perfused with ice-cold PBS for 2 min through intraarterial infusion to remove blood, and the gills were immediately removed, frozen in Tissue-Tek OCT compound, and stored at −80C. Seven-μm frozen sections were cut on a cryostat at −20C, mounted on glass slides, and air dried at room temperature for 30 min. Sections were incubated with 50 to 200 μl transglutaminase assay buffer (50 mM Tris-HCl, pH 7.5, 1 mM dithiothreitol, and 5 mM CaCl2) containing 0.1 μg/μl of His6-Xpress-GFP in the presence or absence of transglutaminase inhibitor (50 mM EDTA) at room temperature for 1 hr (Furutani et al. 2001). Sections were washed three times in excess TBS. Sections were then incubated with TBS containing Alexa 594-labeled ConA (50 μg/ml; Molecular Probes) and Hoechst 33342 (0.1 μg/ml; Molecular Probes) at room temperature for 1 hr, washed three times in excess TBS, and mounted on antifade glycerol (90% glycerol, 10% 10X TBS, and 0.1% 1,4-phenylenediamine, pH 7.4). Samples were analyzed using a fluorescence microscope as previously described.
Results
Visualization of Basal Lamina and Collagen Columns by Staining With Fluorescence-labeled ConA
Figures 1A-1D show a section perpendicular to the sheet of lamella of eel gill stained with fluorescent probes. The nuclei of pillar and pavement cells were stained with Hoechst 33342 (Molecular Probes) and are indicated by single and double asterisks, respectively (Figure 1C). Actin bundles of contractile apparatuses of pillar cells were strongly stained with TRITC-labeled phalloidin and outline the vascular space (Figure 1A). When the sections were stained with fluorescencelabeled ConA, a strong signal appeared in parallel with the signal obtained with phalloidin (Figures 1B and 1D). At the flange of the pillar cell, sheet-like structures between the pillar and pavement cells were stained with ConA (Figure 1B, arrows); these are considered to comprise the basal lamina of the lamella. In the body of the pillar cell, thin lines perpendicular to the plane of the lamella were observed when stained with ConA (Figure 1B; arrowhead). To identify this structure, we prepared horizontal sections of the plate of lamellae (Figures 1E-1H). In the bodies of the pillar cells, ConA staining showed five to eight dot-like structures surrounding the nucleus (Figures 1F and 1H). These dot-like ConA-positive structures were, in turn, surrounded by actin fibers (Figures 1E and 1H). We therefore concluded that this dot-like structure represents the cross-sectional surface of a collagen column in a pillar cell. In sum, we found that fluorescence-labeled ConA stains basal lamina and collagen columns at gill lamellae.
Figures 1I-1L show the three-dimensional image of a single pillar cell stained with phalloidin, ConA, and Hoechst 33342 (Molecular Probes). In this image, seven collagen columns surround the nucleus. Figure 1J illustrates the continuous connection between basal lamina and collagen columns, and this has also been established by previous electron microscopic observations (Hughes and Grimstone 1965; Newstead 1967). Abundant amounts of actin fibers wrap around the collagen columns (Figure 1L). At the flange region, a thin sheet of the pillar cells stained by phalloidin is followed by the ConA-positive basal membrane. The original video sequence for the three-dimensional image is presented in Supplementary Material (Movie SM1).
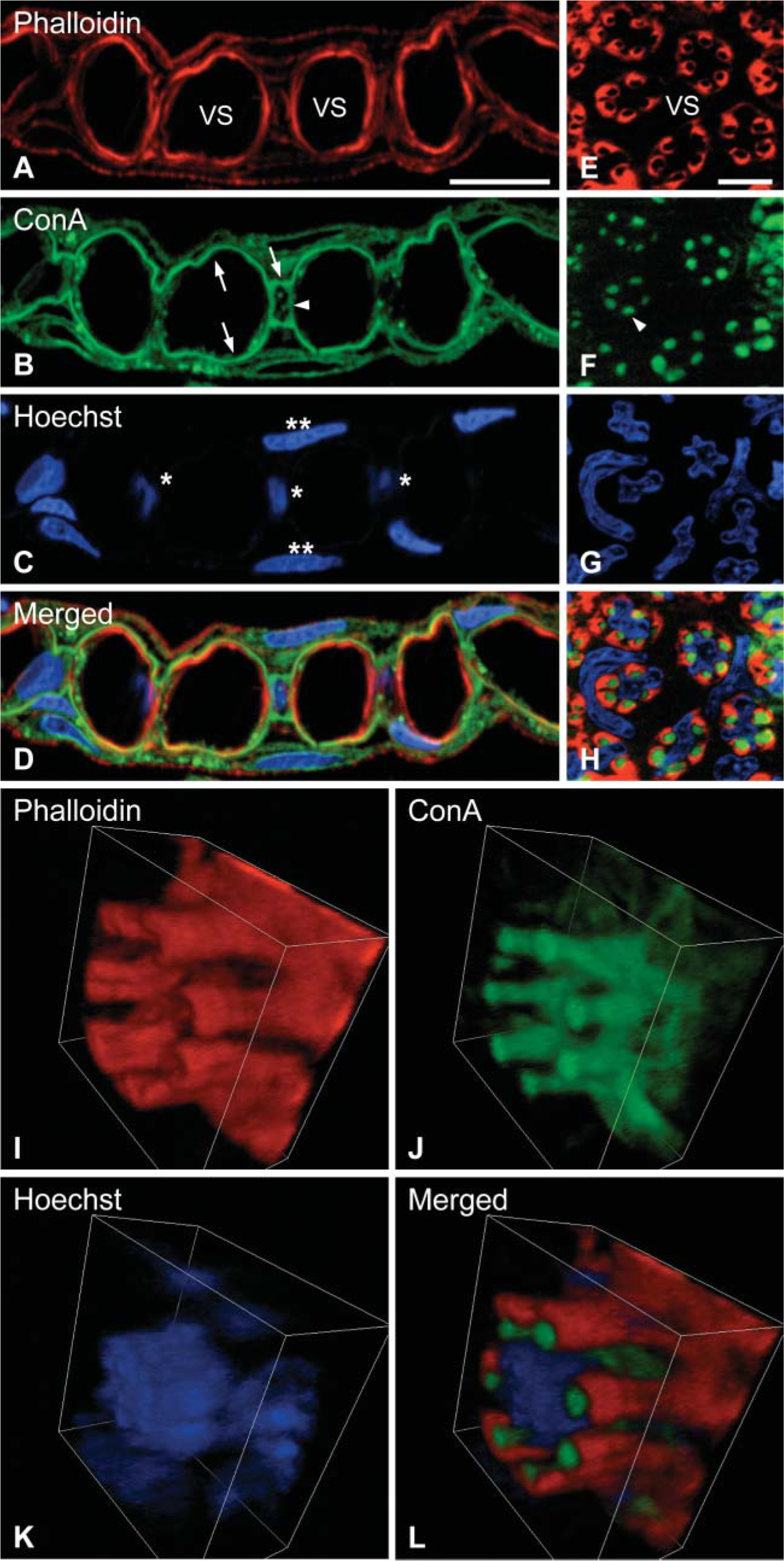
Collagen columns visualized by ConA. Vertical (
Visualization of Collagen Columns by Activity Staining of Transglutaminase
Figure 2 shows a horizontal section of the plate of lamellae of eel gill stained with His6-Xpress-GFP, a fluorescent substrate for transglutaminase. At the cell bodies of pillar cells, the staining showed five to eight dot-like structures that overlapped the collagen columns visualized with ConA. His6-Xpress-GFP staining disappeared completely in the presence of transglutaminase inhibitor (50 mM EDTA, data not shown). These results, therefore, suggest that transglutaminase has a role in maintaining the integrity of the collagen columns.
Discussion
In the gill lamella, collagen columns cross the vascular space and connect two parallel sheets of basal lamina. To avoid blood coagulation caused by contact between blood and the ECM materials, collagen columns are enfolded by the plasma membrane of the pillar cells (Hughes and Grimstone 1965; Newstead 1967). Despite the excellent structure and functional beauty of the lamellae, composition of the collagen columns is still unknown. How are they formed and enfolded by pillar cells, how do pillar cells proliferate, and how do gill lamellae develop? To answer these questions, simple methods for visualizing collagen columns are required. In this study we found probes for the visualization of collagen columns that may be useful in the further study of these phenomena.
The pillar cell is a type of endothelial cell that delimits a network of vascular compartments within the lamellae of fish gills. Pillar cells also share a characteristic of muscle cells. During the 1960s and 1970s, electron microscopic analyses of branchial pillar cells of teleost fish (Oligocottus maculosus, Oncorhynchus mykiss, etc.) and elasmobranch (Scyliorhinus canicula) have demonstrated that numerous thin filaments are situated parallel to the collagen columns in the cytoplasm and resemble myofilaments of smooth muscle from artery or gut (Newstead 1967; Bettex-Galland and Hughes 1973; Wright 1973). Smooth muscle-like characteristic of the pillar cells had been proposed initially by these studies. In pillar cells, some of the thin filaments continue into the cytoplasmic flanges. The others anchor directly to the plasma membrane surrounding collagen columns and form electron-dense structures. Our fluorescence images obtained with phalloidin are consistent with these thin filaments observed by electron microscopy and indicate that they are F-actin. Electron microscopic analyses have also suggested the presence of myosin in pillar cells (Bettex-Galland and Hughes 1973), and this was later confirmed by immunohistochemical analyses of the gills of Chrysophrys auratus and Anguilla japonica (Smith and Chamley-Campbell 1981; Mistry et al. 2004). Interestingly, phalloidin and anti-myosin antibodies stained the pillar cells as strongly as vascular smooth muscle cells (Mistry et al. 2004), and these results are consistent with the electron microscopic observation that demonstrated the similarity in the density of the thin filament between pillar cell and smooth muscle cells (Bettex-Galland and Hughes 1973). Furthermore, our recent analyses on four-and-a-half-LIM protein 5 (FHL5), an actin-binding protein, showed that FHL5 is specifically expressed in pillar cell, heart, and skeletal muscle (Mistry et al. 2004). Because endothelial cells usually do not have such contractile apparatuses, we may conclude that pillar cells are specialized vascular cells that have characteristics of both endothelial and smooth muscle cells. Strong interaction with ECMs such as collagen columns and basal laminae is also a characteristic of muscle cells, and smooth and skeletal muscles are known to have well-developed ECMs that are tightly connected to each cell to transmit tension (Burridge et al. 1988; Small et al. 1992; Burkin and Kaufman 1999). It has been demonstrated that the ECM of muscle is stained by ConA (Pena et al. 1981). Therefore, ECM of pillar cells and that of muscle may share components that bind ConA with high affinity.
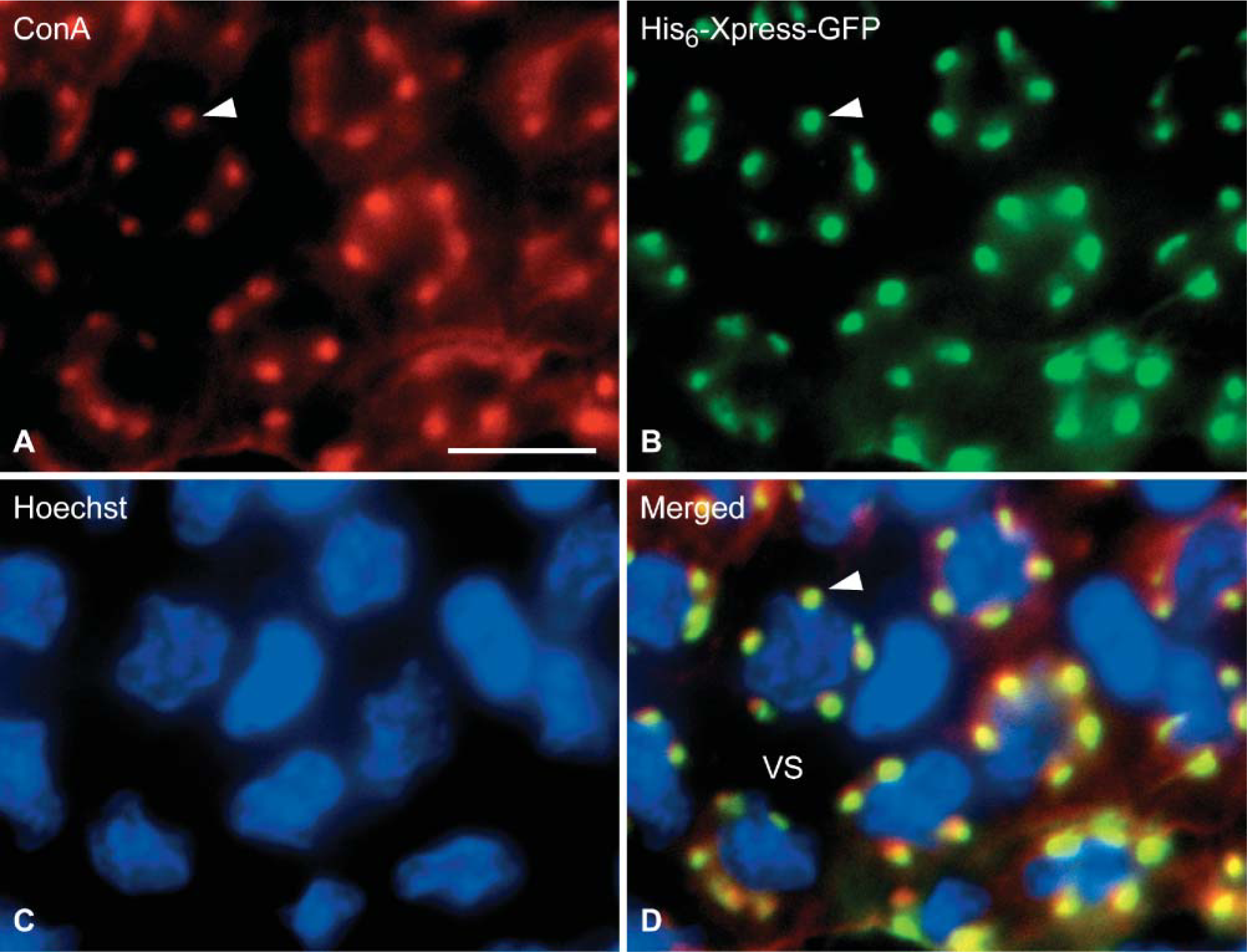
Visualization of collagen columns by activity staining of transglutaminase. Gill section was stained for glycoprotein with ConA-Alexa 594 (red,
Structural similarity between the pillar cell and muscle cell raises the question about their functional similarity: Are pillar cells really contractile or not? If contractile, what is the meaning of the contraction? Some electron microscopic images of the pillar cells have demonstrated that the collagen columns are not straight but rather show a spring-like shape (Newstead 1967; Bettex-Galland and Hughes 1973; Wright 1973). This shape of collagen columns suggests that the bundles of F-actin, which are anchored to the plasma membrane surrounding collagen columns, and myosin may give a force to contract the spring-shaped collagen columns. In 1973, Bettex-Galland and Hughes glycerinated gills of rainbow trout (O. mykiss), incubated them in the presence or absence of ATP, and then found that actomyosin-like thick filaments were seen only in the pillar cells incubated with ATP (Bettex-Galland and Hughes 1973). In 1998, Sundin and Nilsson observed that intraarterial injection of endothelin-1 (ET-1), a strong vasoconstrictor peptide, into rainbow trout (O. mykiss) and Atlantic cod (Gadus morhua), resulted in increased pillar cell diameter and redistribution of blood flow from the pillar cell region to the marginal channels of the lamellae, indicating contraction of pillar cells (Sundin and Nilsson 1998; Stenslokken et al. 1999). Finally, in vivo videomicroscopy has demonstrated that ET-1 reduces blood flow to the lamellae of sculpin (Myoxocephalus) (Evans et al. 2005; Stenslokken et al. 2006). These results, in combination with the structural and compositional similarities between the pillar and muscle cells, provide strong evidence for the contractile nature of pillar cells.
There are two possible meanings of the contraction of pillar cells. The first one is that the contractile apparatuses prevent collagen columns from being stretched and provide plasticity to the vascular network of the lamella against changes in blood pressure. This is conceivable because lack of a contractile nature in the pillar cells would cause the collagen columns to be constantly stretched and so changes in blood pressure in the lamella would not be cushioned. The second possibility is that contraction of the pillar cells narrows the vascular lumen of the lamellae, and this contributes to the regulation of blood flow through the lamellae. Stenslokken et al. (1999) demonstrated that injecting ET-1 into Atlantic cod causes pillar cell contraction, increase in ventral aortic blood pressure, reduction in cardiac output, increase in gill vascular resistance, and reduction in oxygen partial pressure of the venous blood. These results suggest that the contraction of pillar cells and related arteries may be involved in the regulation of respiratory gas exchange at the gill with minimum influence on body fluid homeostasis by controlling blood flow through the lamellae.
As mentioned above, actin is concentrated in the part of the pillar cell that wraps around the collagen columns. This localization pattern suggests that actin may also play a part in keeping the cell membranes wrapped around the collagen columns, thereby minimizing the thrombogenic potential of collagen. Electron microscopic analyses have shown that the collagen columns are surrounded by infoldings of plasma membrane in pillar cells, and the membrane infoldings are zipped by autocellular membrane-membrane adhesion similar to the intercellular membrane-membrane adhesions in tight and adherence junctions (Hughes and Grimstone 1965; Newstead 1967). The molecular mechanism that establishes the complete isolation of collagen columns from the circulation would be clarified in future studies on proteins involved in the formation of tight and adherence junctions.
Although the components of collagen columns and the basal laminae of fish gills have not been clarified, strands of collagen have been clearly visualized by transmission electron microscopy (Newstead 1967; Bettex-Galland and Hughes 1973; Wright 1973). About 70-100 collagenous fibrils are present within a single collagen column of Oligocottus (Newstead 1967). Our current results demonstrate that transglutaminase is also present in collagen columns. There are nine genes for transglutaminases in the mammalian genomes: coagulation factor XIIIa, transglutaminases types 1 to 7, and erythrocyte protein band 4.2 (Grenard et al. 2001; Lorand and Graham 2003). Transglutaminases are involved in blood coagulation, keratinization of the skin, stabilization of ECMs, production of the vaginal plug by clotting of rodent seminal plasma, activation of cytokines, and apoptosis (Ichinose et al. 1990; Greenberg et al. 1991; Aeschlimann and Paulsson 1994; Lorand and Graham 2003). These functions of transglutaminases depend on protein cross-linking activity. On the other hand, transglutaminases act as ligands of integrins and have some role in cell-ECM interactions that are not dependent on protein cross-linking activity (Ueki et al. 1996; Takahashi et al. 2000; Lorand and Graham 2003; Janiak et al. 2006). The transglutaminase in the collagen columns permeates the entire body of the column and is not confined to the surface, where the ECM interacts with the plasma membrane of pillar cells (Figure 2). This pattern of localization suggests that the transglutaminase in the collagen columns may help to stabilize ECM proteins by cross-linking rather than by being involved in the interaction between collagen columns and the plasma membrane of pillar cells.
Footnotes
Acknowledgements
This work was supported by Grants-in-Aid for Scientific Research (14104002, 16710145) from the Ministry of Education, Culture, Sport, Science and Technology of Japan (MEXT) and the 21st Century COE Program of MEXT.
We thank Kentaro Miyamoto and Manami Matsuwra for the purification of His6-Xpress-GFP, Yutaka Furutani and Tsutomu Nakada for discussion, and Setsuko Sato for secretarial assistance.
