Abstract
Novel adhesion junctions have been characterized that are formed at the interface between pillar cells and collagen columns, both of which are essential constituents of the gill lamellae in fish. We termed these junctions the “column junction” and “autocellular junction” and determined their molecular compositions by immunofluorescence microscopy using pufferfish. We visualized collagen columns by concanavalin A staining and found that the components of integrin-mediated cell-matrix adhesion, such as talin, vinculin, paxillin, and fibronectin, were concentrated on plasma membranes surrounding collagen columns (column membranes). This connection is analogous to the focal adhesion of cultured mammalian cells, dense plaque of smooth muscle cells, and myotendinous junction of skeletal muscle cells. We named this connection the “column junction.” In the cytoplasm near the column, actin fibers, actinin, and a phosphorylated myosin light chain of 20 kDa are densely located, suggesting the contractile nature of pillar cells. The membrane infoldings surrounding the collagen columns were found to be connected by the autocellular junction, whose components are highly tyrosine-phosphorylated and contain the tight junction protein ZO-1. This study represents the first molecular characterization and fluorescence visualization of the column and autocellular junctions involved in both maintaining structural integrity and the hemodynamics of the branchial lamellae.
Keywords
I
Pillar cells are a type of endothelial cell that delimits a network of vascular compartments within the lamellae of fish gills (Olson 2002b; Wilson and Laurent 2002; Evans et al. 2005). They are spool-shaped and possess a cylindrical cell body connecting two parallel sheets of respiratory epithelium. (See the schematic illustrations in Figures 1A and 2A.) Usually five to eight collagen columns are enfolded by the plasma membrane of a pillar cell. In the peripheral cytoplasm, pillar cells have numerous myofilaments that run parallel to the collagen columns (Bettex-Galland and Hughes 1973). These filaments consist of actin (Mistry et al. 2004; Kudo et al. 2007) and myosin (Smith and Chamley-Campbell 1981; Mistry et al. 2004), which form contractile apparatuses of the pillar cells. Because endothelial cells usually do not have such contractile apparatuses and smooth muscle cells do not form epithelial cell-cell adhesion, we may say that pillar cells are specialized vascular cells that have characteristics of both endothelial and smooth muscle cells.
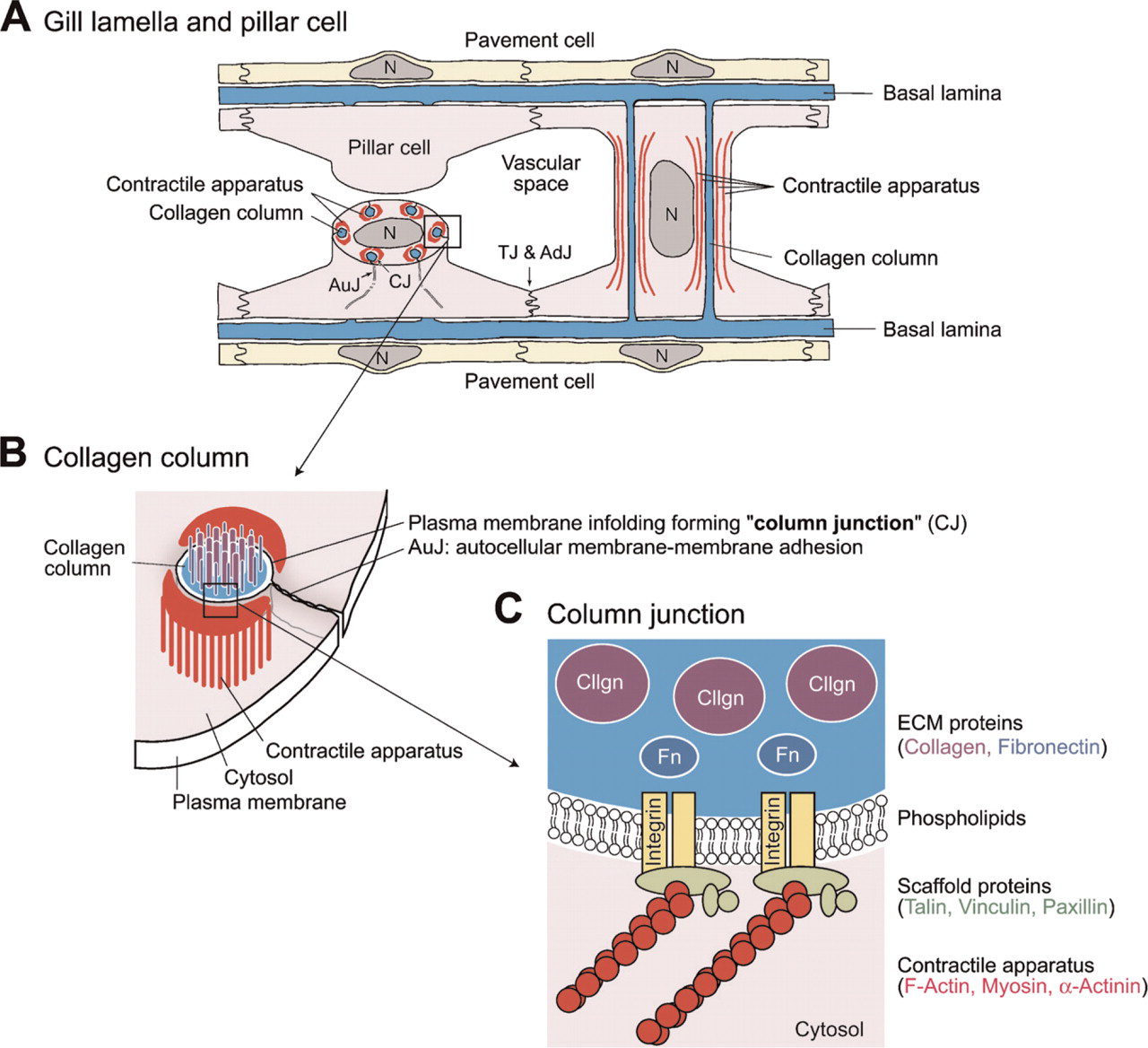
A diagrammatic representation of pillar cells and their intracellular structures. (
Despite their unique and interesting structures, abundant number, and fundamental importance in fish physiology, little is known about the molecular components of collagen columns and/or the contractile apparatus of pillar cells. The following is a brief description of current understanding of the cell-matrix and intercellular junctions, on which our experimental designs are based. In differentiated muscle cells, the connection between the contractile apparatus and ECM proteins via the plasma membrane (cell-ECM adhesion) is important for transmitting tensions. In mammals, actin filaments connect to the cell membrane at dense plaques in smooth muscle cells and at myotendinous junctions in skeletal muscle cells (Burridge et al. 1988; Small et al. 1992; Burkin and Kaufman 1999). These contact structures are believed to be analogous to the focal adhesion of cultured cells. Focal adhesion is the type of cell-ECM contact most thoroughly studied in vitro using cultured mammalian cells, where a selective group of membrane and cytoplasmic proteins are recruited to link ECM proteins and the intracellular actin cytoskeleton. Integrins are the principal plasma membrane receptors of animal cells for binding ECM components, including collagens, fibronectin, vitronectin, and laminins, and are connected to bundles of actin filaments via a characteristic assembly of intracellular accessory proteins, including talin, vinculin, and paxillin, beneath the plasma membrane (Hynes 1992; van der Flier and Sonnenberg 2001). On the other hand, the epithelial and endothelial cell-cell adhesions occur through a junctional complex of adherence junctions mediated by cadherin and tight junctions mediated by claudin, whose cytoplasmic tails are associated with many different peripheral membrane proteins, including α-catenin, β-catenin, and zona occludens-1 (ZO-1) (Tsukita et al. 1992, 1999; O'Brien et al. 2002). The adherence junction has also been demonstrated to be an intercellular membrane-membrane contact site where the level of tyrosine phosphorylation is elevated (Tsukita et al. 1991).
In this study, we found that the molecules involved in cell-ECM adhesions are highly concentrated in the plasma membranes surrounding collagen columns, termed here “column membranes.” This result indicates a tight connection between the ECM materials of collagen columns and the intracellular contractile apparatus at column membranes. We propose to name the cell-ECM adhesion the “column junction” and discuss its importance in pillar cell integrity and contraction. Furthermore, we found that the autocellular membrane-membrane adhesion sites where pillar cells enfold collagen columns are highly tyrosine-phosphorylated and contain ZO-1. We propose this adhesion as “autocellular junction,” which is analogous to tight adherence junctions of epithelial cell-cell adhesion. The methods for molecular characterization and visualization of the intracellular structures of pillar cells developed in this study will be useful for advances in the molecular biology of pillar cells.
Materials and Methods
Antibodies and Fluorescence Probes
TRITC-labeled phalloidin, mouse monoclonal antibody (mAb) raised against purified chicken gizzard talin (clone 8D4, IgG1 isotype), mouse mAb raised against purified human uterus vinculin (clone hVIN-1, IgG1 isotype), mouse mAb raised against α-actinin from cytoskeletal fraction of bovine mammary gland epithelium (clone BM-75.2, IgM isotype), rabbit polyclonal antibody (pAb) raised against purified human fibronectin, and rabbit pAb raised against laminin purified from the basement membrane of mouse sarcoma were obtained from Sigma (St Louis, MO); mouse mAb raised against purified chicken paxillin (clone 349, IgG1 isotype), and rabbit pAb to phosphotyrosine were from BD Bioscience (San Jose, CA); mouse mAb raised against a recombinant protein corresponding to amino acids 334-634 of human ZO-1 (clone ZO1-1A12, IgG1 isotype), Alexa Fluor 488-labeled phalloidin, Alexa Fluor 594-labeled goat pAb to mouse IgG, Alexa Fluor 488-labeled goat pAb to mouse IgG, Alexa Fluor 488-labeled goat pAb to mouse IgM, Alexa Fluor 594-labeled goat pAb rabbit IgG, Alexa Fluor 488-labeled goat pAb to rabbit IgG, and TO-PRO-3 iodide (642/661) were from Invitrogen (Carlsbad, CA); mouse mAb to Ser19 monophosphorylated myosin light chain of 20 kDa (pMLC20) was provided by Dr. Y. Sasaki of Kitasato University (Sakurada et al. 1998).
A database search indicated that the pufferfish proteins involved in the formation of cell-cell and cell-ECM junctions are very similar in their amino acid sequences to those of mammals. The proteins examined, with the scaffold numbers and the identities to the antigen indicated within parentheses, include fibronectin (scaffold 83; 58%), laminin subunits (scaffold 2242, 191, and 317; 52-64%), talin (scaffold 106; 82%), vinculin (scaffold 29; 90%), paxillin (scaffold 116; 70%), α-actinin (scaffold 246; 88%), ZO-1 (scaffold 1; 87%) and MLC20 (scaffold 318; 100%). These high degrees of conservation suggest that antibodies to mammalian counterparts can be used for the immunological detection of the pufferfish adhesion/junction proteins. The same antibodies for fibronectin (Jin et al. 2005), laminin (Parsons et al. 2002), paxillin (Crawford et al. 2003), and ZO-1 (Cibrian-Uhalte et al. 2007) have been used for immunohistochemical analyses of zebrafish. Cross-reactivity of the antibodies to the pufferfish orthologous proteins was confirmed by Western analyses as described below.
Immunohistochemistry
Cultured pufferfish, Takifugu rubripes, weighing 150-200 g were purchased from Numazu Aquaculture Fisheries (Shizuoka, Japan), and were kept in a seawater tank (20C) for 10 days. Pufferfish were perfused under anesthesia through the ventral aorta with ice-cold phosphate-buffered saline (PBS: 137 mM NaCl, 2.7 mM KCl, 6.5 mM Na2HPO4, 1.5 mM KH2PO4, pH 7.4) for 5-10 min. After the perfusion, gill arches were dissected out and immersed in 2% (w/v) para-formaldehyde in PBS, pH 7.4, at 4C for 2 hr, and rinsed in PBS containing 10% (w/v) sucrose. The fixed tissues were cryoprotected by a range of increasing sucrose concentrations, up to 20%, quick-frozen in Tissue Tek optimum cutting temperature compound, and cut on a cryostat at 6 μm. The sections were washed three times in PBS, permeabilized with 0.2% Triton X-100 in PBS at 20C for 10 min, incubated with 5% fetal bovine serum (FBS) in PBS at 20C for 1 hr, and incubated with a mixture of primary antibodies in PBS containing 5% FBS at 20C for 2-16 hr. The primary antibodies used were: anti-talin mouse mAb (1:100 dilution), anti-vinculin mouse mAb (1:400), anti-paxillin mouse mAb (1:100), anti-pMLC20 mouse mAb (1:20), anti-α-actinin mouse mAb (1:100), anti-fibronectin rabbit pAb (1:200), anti-laminin rabbit pAb (1:100), anti-ZO-1 mouse mAb (1:200), and anti-phosphotyrosine rabbit pAb (1:100). The other sets of sections were incubated with 1:1000-diluted normal serums of ICR mice or Japanese White rabbits instead of the primary antibodies as negative controls. After washing with PBS, the sections were incubated with Alexa Fluor-labeled secondary antibody (1:2000 dilution) in PBS containing 5% FBS at 20C for 2 hr. Nuclei were stained with the DNA-binding dye TO-PRO-3 (2 μM) in PBS containing 5% FBS at 20C for 30 min. Actin fibers were labeled with Alexa Fluor 488 or TRITC-labeled phalloidin (0.15 μM). Glycosylated proteins were stained with Alexa Fluor-488-labeled concanavalin A (20 μg/ml) in Tris-buffered saline (TBS: 150 mM NaCl, 10 mM Tris-Cl, pH 7.4). The sections were mounted on antifade glycerol (90% glycerol, 10% 10 × PBS, and 0.1% 1,4-phenylenediamine, pH 7.4). Fluorescence was detected using a confocal microscope (LSM 5; Carl Zeiss, Oberkochen, Germany) or fluorescence microscope (Carl Zeiss) equipped with a confocal laser scanner unit (CSU10; Yokogawa, Tokyo, Japan) or ApoTome (Carl Zeiss). Images were obtained with high-resolution digital charge-coupled device (CCD) cameras and processed with an LSM5 Image Browser (Carl Zeiss), IPLab (Scanalytics, Inc.; Billerica, MA), or AxioVision (Carl Zeiss) software. The brightness and contrast of final images were adjusted with Adobe software Photoshop (Adobe Systems; Mountain View, CA).
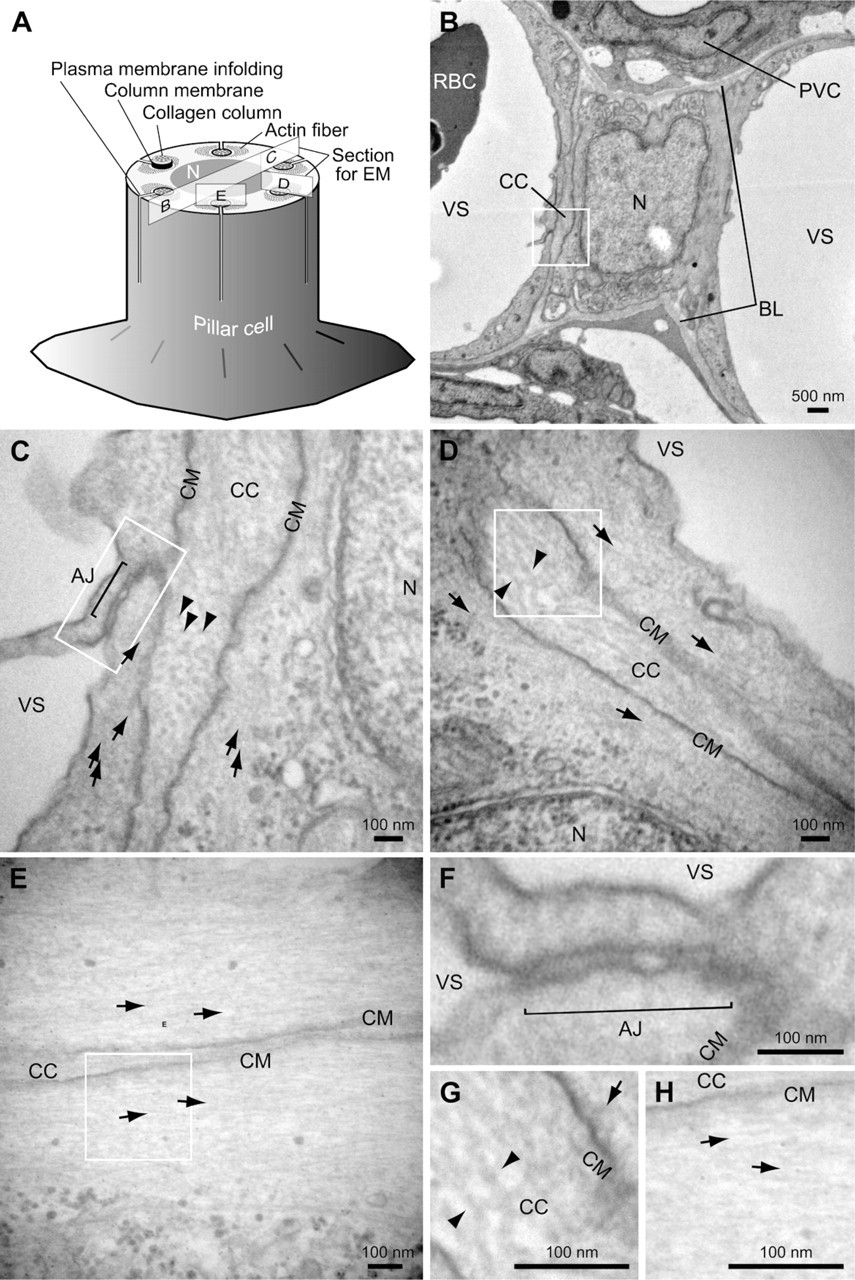
Transmission electron micrograph of pillar cells of pufferfish gills. (
Western Analyses
Samples for Western analyses were prepared as follows. Gills of pufferfish were homogenized in 10 vol 0.25 M sucrose containing 1 mM phenylmethylsulfony fluoride, 10 μg/ml leupeptin, and 10 μg/ml pepstatin A at 4C and used as total gill extract. The total gill extract was centrifuged at 5000 × g for 15 min, and the supernatant (cytosolic fraction) and the pellet (membrane fraction) were separated. The pellet was resuspended in the same buffer, and the protein concentrations were determined using a BCA protein assay kit (Pierce; Rockford, IL). Fifteen μg of protein were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) on 5, 10, and 15% polyacrylamide gel and subjected to Western blotting.
The separated proteins were transferred to a nitrocellulose membrane, blocked with 5% non-fat milk in TBST (TBST: 150 mM NaCl, 10 mM Tris, pH 8.0, and 0.05% Tween 20) at 20C for 1 hr, and incubated with primary antibodies in TBST. The dilution, temperature, and time of the incubation with primary antibodies were: anti-vinculin (1:400), 20C for 1 hr; anti-α-actinin (1:400), 20C for 1 hr; anti-talin (1:200), 37C for 1 hr and then 20C for 12 hr; anti-fibronectin (1:1000 dilution), 20C for 12 hr; anti-laminin (1:1000), 20C for 12 hr; anti-ZO-1 (1:1000), 37C for 1 hr and then 20C for 12 hr; anti-paxillin (1:10,000), 20C for 1 hr; and anti-pMLC20 (1:100), 20C for 1 hr. Bound antibodies were detected using peroxidase-conjugated secondary antibodies (Jackson ImmunoResearch Laboratories; West Grove, PA) and chemiluminescent substrate solution (Immobilon Western, Millipore; Billerica, MA), and the signals were captured using an Image Station 2000R (Kodak; Rochester, NY). Specificities of the signals on the membranes stained by the primary antibodies were confirmed by comparison with those stained in the absence of primary antibodies, which showed no signals (data not shown).
Vascular Casting
Gill vascular casting was carried out using Batson's no. 17 plastic replica and corrosion kit (Polysciences, Inc.; Warrington, PA) according to the user manual. In brief, cultured pufferfish, T. rubripes, weighing ∼150 g were perfused under anesthesia through the ventral aorta with Hank's balanced salt solutions (HBSS: 138 mM NaCl, 2.7 mM KCl, 8.1 mM Na2HPO4, 1.5 mM NaH2PO4, 0.6 mM MgCl2, 1.0 mM CaCl2, 10 mM glucose, pH 7.4) containing 60 U/ml of heparin for 5-10 min, and then with methacrylate corrosion compound. The fish were then immersed in HBSS for 6 hr at 4C. The gills were excised carefully and soaked in 2.5 M sodium hydroxide for 3 days at 20C, soaked in 0.2 M sodium hydroxide containing 1% SDS for 3 days at 20C, rinsed in water six times, and then dried. Dried casts of the gills were mounted on aluminum stubs and sputter-coated with platinum-palladium. Samples were viewed and photographed with a scanning electron microscope (Model S-4700; Hitachi, Tokyo, Japan).
Transmission Electron Microscopy
Cultured pufferfish, T. rubripes, weighing ∼150 g were perfused with HBSS under anesthesia through the ventral aorta. The gills were removed, fixed with 4% (w/v) paraformaldehyde and 2.5% glutaraldehyde, and processed by standard procedures including tannic acid staining. Ultrathin sections were cut and examined with an electron microscope (Model H-7500; Hitachi; Tokyo, Japan).
Results
Transmission Electron Microscopy of Collagen Columns Enfolded in Pillar Cells
We found by electron microscopy that the architecture of the pufferfish pillar cell is essentially identical to that reported for other fish (Newstead 1967) (Figure 2). Several collagen columns traverse the pillar cell within tubes formed by infoldings of the plasma membrane, and each column contains collagenous bundles (arrowheads in Figures 2C, 2D, and 2G). A rich amount of thin filament of actin fibers runs beside collagen columns (arrows in Figures 2C-2E, 2G, and 2H) and anchored to the column membrane (Figures 2G and 2H). Figure 2F shows a high-magnification view of the auto-cellular junction, which is analogous to cell-cell adhesion.
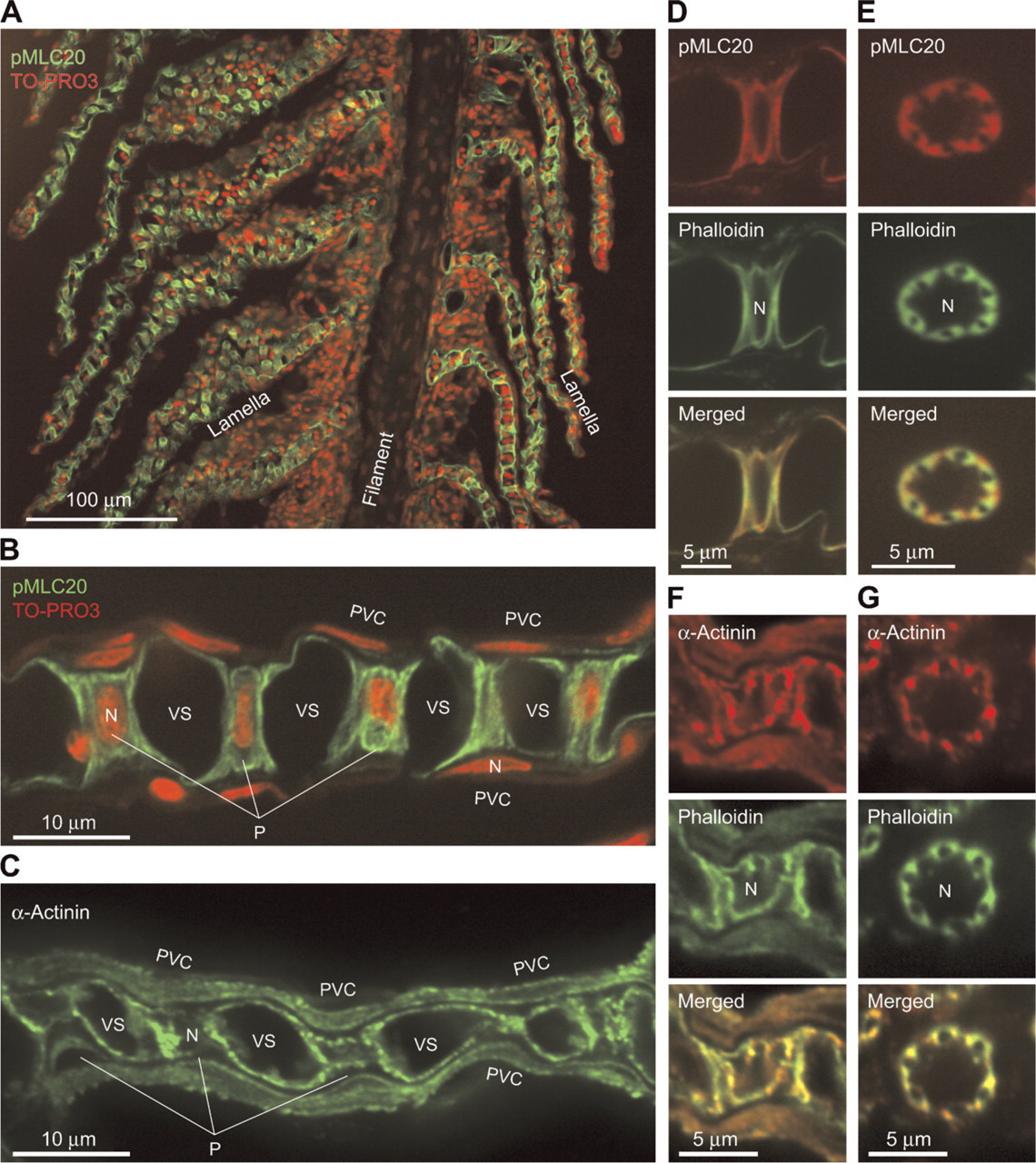
Lamellae Structure of the Pufferfish Gill: Suitable for Pillar Cell Immunofluorescence Microscopy
Vascular casting and scanning electron microscopy revealed the presence of both flat- and sigmoidal-shaped lamellae on a single filament of the pufferfish gill (Figure 3A). This anatomical feature is advantageous for increasing the respiratory surface of the fish and for the structural study of pillar cells, because both longitudinal and transverse images of the lamellae (and therefore of pillar cells) can be obtained easily within a single section. For example, by sectioning along the white line shown in Figure 3A, one can observe both longitudinal (Figures 3C and 3E) and transverse (Figures 3B and 3D) images of pillar cells.
Contractile Apparatuses: Associate “Column Membranes” Surrounding Collagen Columns
We previously demonstrated in the eel Anguilla japonica that the contractile apparatus of branchial pillar cells can be readily visualized by staining with a fluorescent-labeled phalloidin, a toxin from the mushroom Amanita phalloides, which binds specifically to actin filaments (Mistry et al. 2004, Kudo et al. 2007). Similar staining patterns were obtained when pufferfish gill sections were incubated with TRITC-labeled phalloidin (Figures 3B-3E and 4K-4S), in which the presence of two bundles of actin fibers per each collagen column that run parallel and close to the column can be seen (Figures 4K-4S and arrows in Figure 2).
The contractile apparatuses were also stained with an mAb to α-actinin, which is known to mediate bundling of actin filaments, and an mAb to the phosphorylated myosin light chain of 20 kDa (pMLC20) (Figure 5). These signals were not observed when the tissue was stained with non-immune mouse serum as a negative control (Figures 4T and 4U), and the specific cross-reactivity of the antibodies was confirmed by Western analyses (Figures 6B and 6H). These results suggest full functionality of the protein assembly, as visualized by fluorescence microscopy, and thus the appropriateness of the term “contractile apparatus.”
Column Junctions: Composed of Similar Components of Integrin-mediated Cell-ECM Adhesion
The deduced amino acid sequences of pufferfish orthologs of talin, vinculin, and paxillin were found to be highly similar to those of mammals and chicken (accession nos. NM_204523, NM_014000, and NM_204984, respectively). Reflecting this similarity, the commercially available heterologous antibodies used here recognized the corresponding pufferfish proteins (Figures 6A, 6C, and 6G). When we stained pufferfish gill sections with antibodies to talin, vinculin, and paxillin, we observed strong signals in all cases in pillar cells (Figure 4). We did not observe these signals when we stained the tissue with non-immune mouse serum as a negative control (Figures 4T and 4U). Furthermore, staining intensities were as strong as those in vascular smooth muscles (data not shown), which are known to express those molecules at high levels (Small 1995; Graf et al. 1996; Hungerford and Little 1999; Diagne et al. 2003; Yuminamochi et al. 2003). Examination at a higher magnification showed that the staining occurred near the basal lamina membrane and in tube-like structures (five to eight per cell) in pillar cell bodies (Figures 4E-4S). The tube-like structures are surrounded by bundles of actin fibers (Figures 4M, 4P, and 4S); these structures (i.e., talin/vinculin/paxillin-containing structures) in turn embrace the concanavalin A-positive ECM materials of the collagen columns, including fibronectin (Figures 4G and 4J). The peripheral location of fibronectin within the collagen columns (Figures 1C and 4J) is noteworthy. These findings suggest that the tube-like structures represent cell-matrix adhesion sites linking the intracellular contractile apparatuses and ECM proteins of the collagen columns. We propose to name this connection, represented here as a tube-like structure, the “column junction.”
Collagen Column Is Continuous With the Basal Lamina Membrane, but They Differ in Laminin Content
As described previously, concanavalin A, a lectin from the legume Canavalia ensiformis that specifically recognizes the trimannoside core of N-glycosylated proteins, has proven very useful for visualizing the basal lamina membranes and collagen columns (Kudo et al. 2007) (Figures 4D-4G), and for confirming their continuity, previously suggested by electron microscopic observations (Hughes and Grimstone 1965; Newstead 1967).
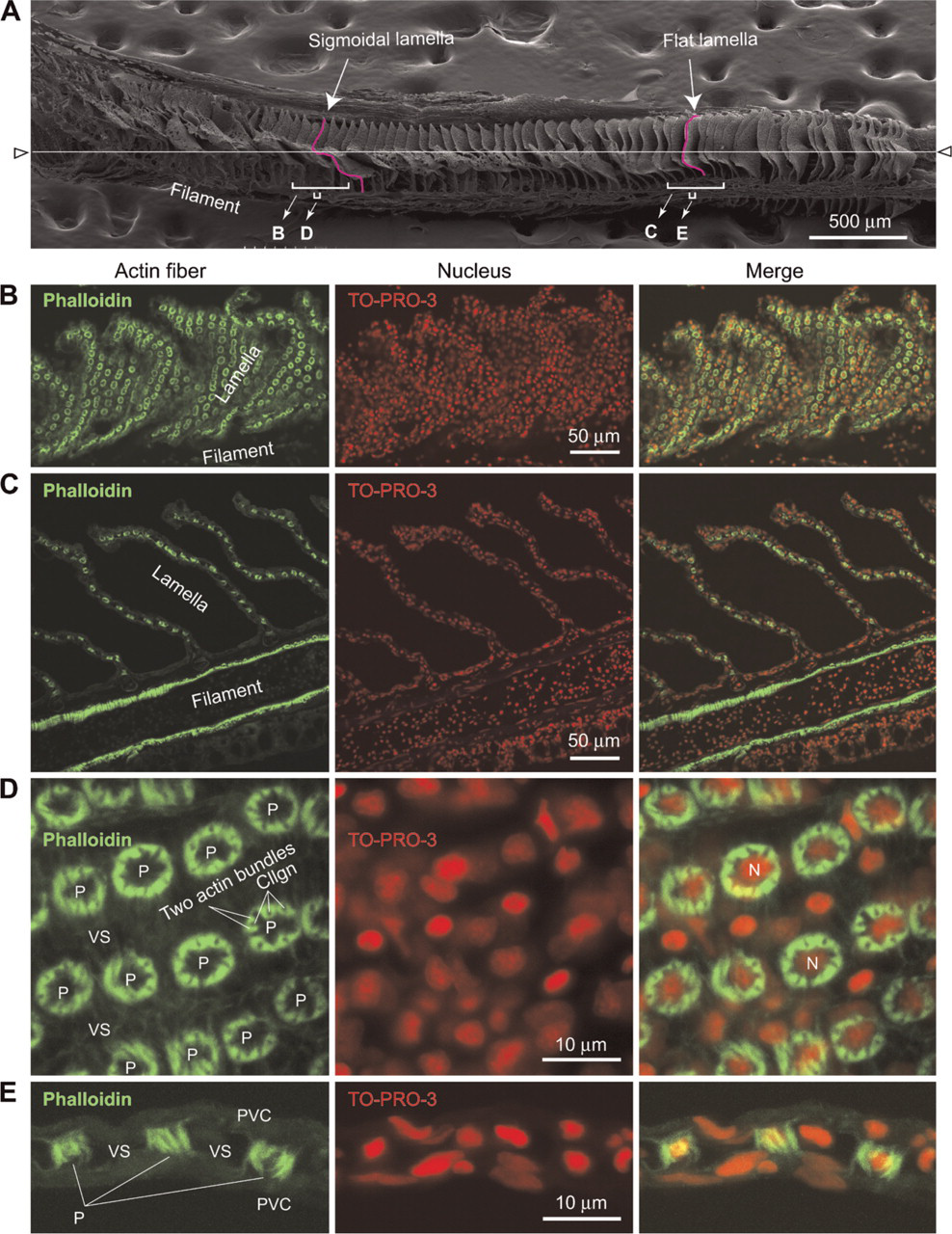
Scanning electron microscope (SEM) and fluorescence microscope images of lamellae of pufferfish gills. (
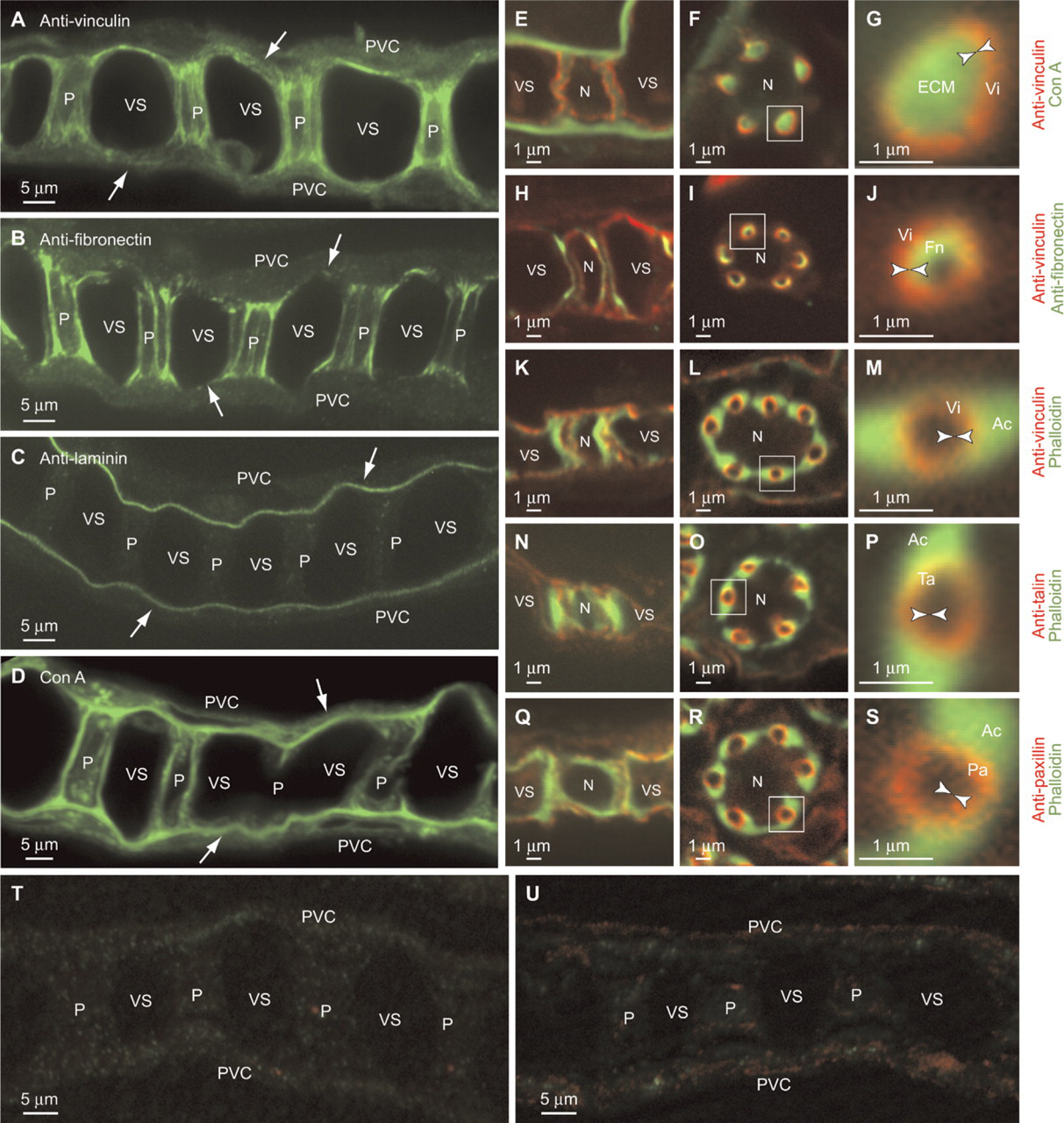
Staining of cell-matrix adhesions at column membranes. (
Staining of contractile apparatuses of pillar cells. (
Anti-fibronectin also stained both the basal laminae and collagen columns, with the latter being much more intensely stained (Figure 4B). Higher magnification images of transverse sections revealed that fibronectin does not localize in the entirety of each collagen column. Instead, it concentrates on its outer surface in close proximity to the column membrane (Figures 4I and 4J), suggesting that fibronectin is one of the major ligands for integrins mediating the cell-matrix adhesion at the column junction (Figure 1C). Another major component of ECM, laminin, had immunoreactivity that was only detected in the basal lamina membrane (Figure 4C). We did not observe these signals when we stained the tissue with normal rabbit serum as a negative control (Figures 4T and 4U). The specific cross-reactivity of the antibodies was confirmed by Western analyses (Figures 6D and 6E).
Fluorescent Visualization of Autocellular Junctions
When we stained vertical sections of the lamellae with anti-pY antibody, the blood side of the column membrane and flange of pillar cells were stained (Figures 7A and 7D). The transverse sections of pillar cells stained by anti-pY demonstrated that the stainings are always located beside the blood side of collagen columns, whose positions are estimated by staining with anti-talin and phalloidin (Figures 7B, 7C, 7E, and 7F). Overlapping anti-pY staining with the localization of the plasma membrane of pillar cells (dotted lines in Figures 7C and 7F), which is estimated according to the model of pillar cells (Figure 1) demonstrates that autocellular junctions are stained with anti-pY. Because tyrosine phosphorylation of cadherin complexes is well documented and widely believed to modulate cell adhesiveness (Tsukita et al. 1991), this observation suggests that the autocellular junction involves a type of adherence junction.
We next stained gill sections with anti-ZO-1 antibody. A mouse mAb raised against a recombinant protein corresponding to amino acids 334-634 of human ZO-1 cross-reacted with the pufferfish ortholog (Figure 6F), and the staining of gill sections was overlapped with that of anti-pY antibody (arrows in Figures 7G-7L). This pattern of localization demonstrates that the auto-cellular junction contains ZO-1 and may involve the claudin-mediated tight junction. The intercellular adhesion sites of vertical sections of pillar cells were also stained with anti-pY and anti-ZO-1 (arrowheads in Figures 7A, 7D, 7G, and 7J).
Discussion
The fine structure of pillar cells, illustrated in Figures 1 and 2A, was established by early studies using transmission electron microscopy (Vialli 1929; Hughes and Grimstone 1965; Newstead 1967; Hughes and Wright 1970; Bettex-Galland and Hughes 1973; Wright 1973) and has been reviewed recently (Olson 2002b; Wilson and Laurent 2002; Evans et al. 2005). We recently constructed a three-dimensional fluorescence image of a single pillar cell (Kudo et al. 2007). In this study, we characterized the column junction and autocellular junction of pillar cells. We used the pufferfish T. rubripes and heterologous antibodies whose cross-reactivities were predicted on the basis of its genomic sequence information and then confirmed experimentally by Western analyses. T. rubripes had four advantages for studying the molecular components of the pillar cells: (1) the genome resource of T. rubripes helped us to estimate the cross-reactivities of heterologous antibodies; (2) transmission electron microscopy demonstrated that the structure of the T. rubripes pillar cell is not special compared with other fish species (Figure 2); (3) sigmoidal-shaped lamellae made it easy to observe both longitudinal and transverse images of pillar cells (Figure 3); and (4) large gill size (>25 mm) helped us to analyze sections under many different conditions of fixation and incubation with primary antibodies.
Electron microscopy revealed that the collagen columns are surrounded by infoldings of the plasma membrane of pillar cells (column membrane), and that bundles of microfilaments terminate in a dark area on the column membrane (Newstead 1967; Wright 1973) (Figures 2D, 2E, 2G, and 2H). These observations suggest a strong interaction between the collagen column and nearby contractile apparatuses via the column membrane. In mammals, cell-matrix adhesion is principally mediated by integrins, the cytoplasmic domain of which connects to actin filaments via a characteristic assembly of intracellular accessory proteins, including talin, vinculin, and paxillin. Although a large number of integrin family members have been found in mammals (Hynes 1992; van der Flier and Sonnenberg 2001), the intracellular accessory proteins (talin, vinculin, and paxillin) are shared among all members. We therefore focused on talin, vinculin, and paxillin in characterizing the cell-ECM adhesion of column junctions. Immunohistochemical analyses demonstrated that talin, vinculin, and paxillin are concentrated on the column membrane (Figures 4E-4S) and form the column junction, and that the column junction is an analog of focal adhesion (Figure 1C).
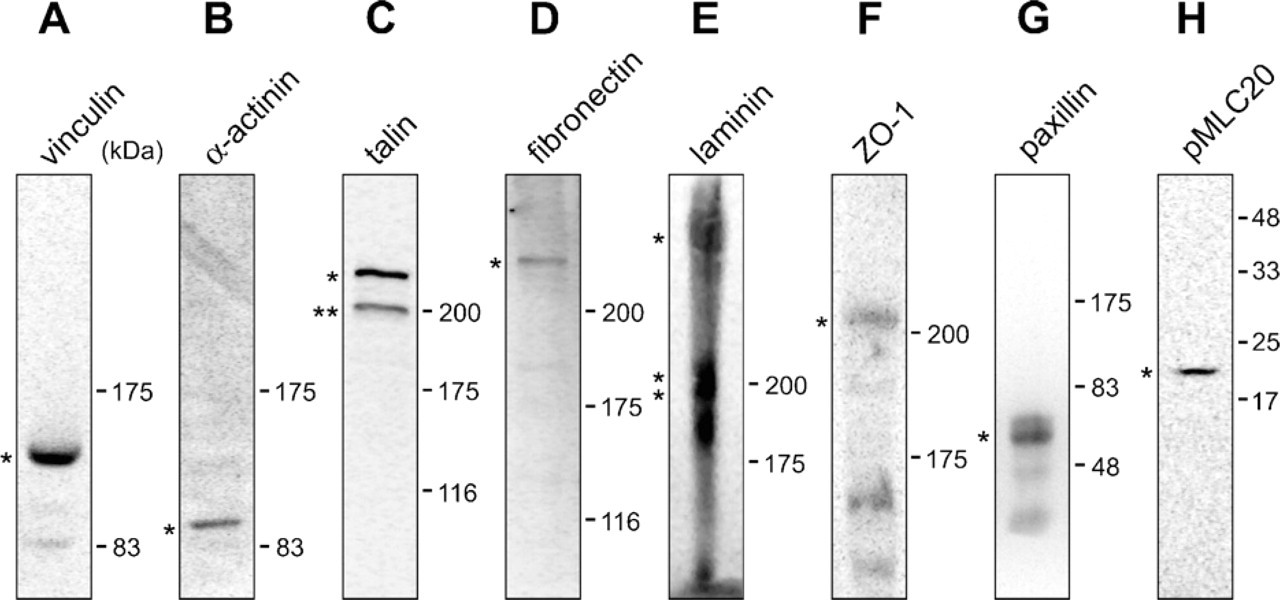
Western analyses. Twenty μg of total gill extract (
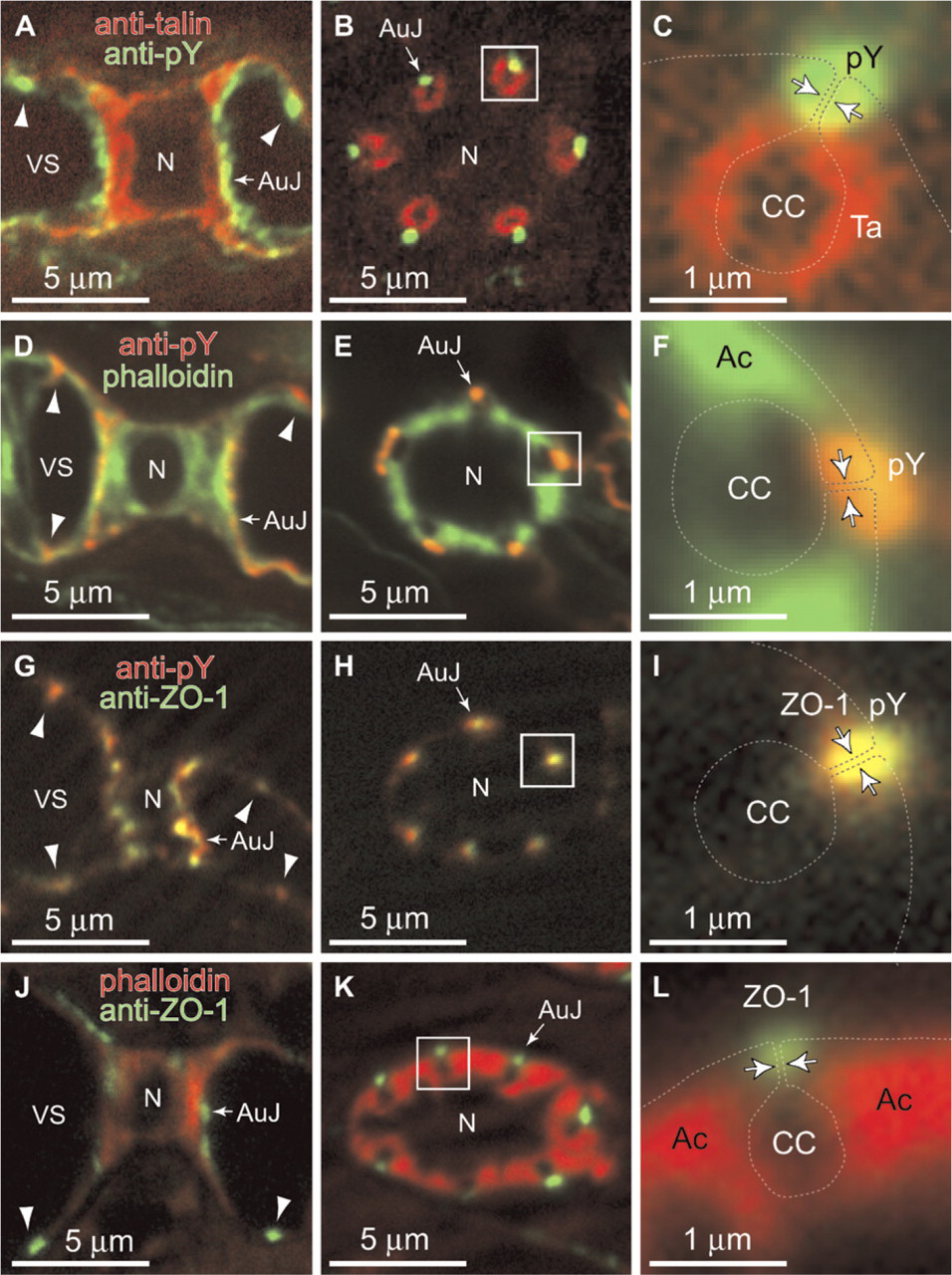
Staining of autocellular membrane-membrane adhesions of pillar cells. (
The column junction characterized here is the third in vivo example of focal adhesion. Two other in vivo examples are the dense plaque of smooth muscle and the myotendinous junction of skeletal muscle (Burridge et al. 1988). These cell-ECM adhesions were identified as the membrane site of tension transmission between the contractile apparatus and the ECM (Draeger et al. 1989). Therefore, the column junction may be the anchoring site to the collagen column for the contractile machinery and the site of tension transmission, which involves the internal force of blood pressure, environmental mechanical stresses, and contraction of the contractile apparatuses stimulated by vasoactive agents.
The column junction may also be a site of signal transduction. It has been demonstrated that cell-ECM adhesion or integrin-mediated adhesion has an important role in the signaling pathway in response to mechanical stress (Shyy and Chien 1997; Davis et al. 2001; Katsumi et al. 2004). The mechanical stresses, as well as the composition of the ECM materials, may affect the assembly of the junctional complex through outside-in signals delivered via integrins (Giancotti and Ruoslahti 1999). In cultured mammalian cells, the increased tension at adhesion sites from the actin stress fibers promotes integrin clustering, which results in the increased fibronectin fibril assembly and the elongation of the adhesion site (Burridge and Chrzanowska-Wodnicka 1996; Katsumi et al. 2004). Therefore, it is likely that the column junction is also linked to those mechanisms and that those signalings are important for maintaining collagen columns and pillar cell differentiation.
Collagen columns are surrounded by infoldings of the plasma membrane in pillar cells, and the membrane infoldings are connected by autocellular membrane-membrane adhesions (Hughes and Grimstone 1965) (Figure 1B). To clarify the nature of the autocellular junctions, we performed immunostaining of sections of the lamellae of the pufferfish gill. We stained the autocellular junction with anti-pY and anti-ZO-1 antibodies. Because ZO-1 is a major component of the claudin-mediated tight junction (Tsukita et al. 1992, 1999; O'Brien et al. 2002) and the cadherin-mediated adherence junction is known to be highly tyrosine-phosphorylated (Tsukita et al. 1991), the autocellular junction is an analog of intercellular adhesions. This characteristic of the autocellular junction is consistent with the estimation by electron-microscopic observation of pillar cells (Hughes and Grimstone 1965; Newstead 1967; Wright 1973) (Figure 2F). Recently, whole-genome analyses of claudin genes of T. rubripes revealed 56 claudin genes in pufferfish; 32 of these genes are expressed in the gill (Loh et al. 2004). Therefore, some of these claudin genes may constitute the autocellular junction. Further analyses are necessary to identify claudin genes on the autocellular junction.
Functionally, pillar cells play dual roles as endothelial cells and smooth muscle-like cells and are equipped with: (1) membrane-bound enzymes involved in metabolic clearance or in conversion of circulating toxins and hormones, as generally seen in endothelial cells (Olson 2002a); (2) intercellular and autocellular junctions that form an epithelial sheet (Figures 2 and 7); (3) large amounts of smooth-muscle myosin (Smith and Chamley-Campbell 1981; Mistry et al. 2004), actin (Mistry et al. 2004) (Figure 3), an actin-binding protein (FHL5) (Mistry et al. 2004), actinin, and pMLC20 (Figure 5); (4) receptors for vasoactive agents, such as endothelin (Sultana et al. 2007) and adrenomedullin (Nag et al. 2006), which can potentially regulate pillar-cell contraction; and (5) a well-developed column junction (Figure 4), which is the indirect demonstration of the presence of mechanical force.
The contractile nature of pillar cells was previously suggested by electron microscopic observation of myofilament-like fibrils in the pillar cell cytoplasm (Newstead 1967; Bettex-Galland and Hughes 1973; Wright 1973) and by direct observation of changes in the diameter of pillar cells in living gills by epi-illumination microscopy, which is accompanied by an increase in ventral aortic blood pressure, reduction in cardiac output, increase in gill vascular resistance, and reduction in oxygen partial pressure of the venous blood (Sundin and Nilsson 1998; Stenslokken et al. 1999). Recently, the presence of carbonic anhydrase (Sender et al. 1999; Gilmour et al. 2007), ammonia transporter (Nakada et al. 2007), and a member of the winged helix/forkhead transcription factor FoxF1 (Hidaka et al. 2004), which is required for normal development of lung and foregut in mammals (Mahlapuu et al. 2001), have been demonstrated in pillar cells. These observations, as well as the results of this study, have highlighted the importance of pillar cells in respiration and nitrogen metabolism.
Footnotes
Acknowledgements
This work was supported by grants-in-aid for scientific research (14104002, 16710145, and 19770057) from the Ministry of Education, Culture, Sport, Science and Technology of Japan MEXT) and the 21st Century Center of Excellence (COE) Program of MEXT.
We thank Yasuharu Sasaki for the the anti-monophospho-MLC20 antibody; Jun Koki and Satoshi Genzeki for performing the scanning electron microscopy; Atsushi Suzuki, Keiko Mizuno, and Shigeo Ohno for the anti-ZO-1 antibody; Katsuhiro Tosaya and the staff of Numazu Aquaculture Cooperative for the supply and transport of the pufferfish; and Setsuko Sato for her secretarial assistance.
