Abstract
Bone sialoprotein (BSP) is an anionic phosphoprotein expressed in mineralizing connective tissues that binds to hydroxyapatite and nucleates its formation in vitro. Two polyglutamic acid regions (poly [E]) are believed to participate in these activities. The aim of this study was to evaluate the contribution of these acidic regions to the binding of prokaryote recombinant BSP (prBSPE) within an actual in vivo environment. Full-length prBSPE and prBSPE in which the poly [E] domains were replaced by polyalanine (prBSPA) were tagged with dinitrophenol (DNP). Tagged preparations comprised intact molecules and some fragmented forms. They were infused through a surgically created hole in the bone of rat hemimandibles and detected using immunogold labeling with anti-DNP antibodies. prBSPE-DNP was consistently immunodetected along exposed mineralized bone surfaces and osteocyte canaliculi at the surgical site. Few gold particles were observed on these surfaces when prBSPA-DNP was infused. Quantitative analyses showed significant differences in labeling between prBSPE-DNP (5.04 ± 0.73 particles/μm2) and prBSPA-DNP (1.37 ± 0.35 particles/μm2). These results indicate that poly [E] domains influence binding of prBSPE to surfaces presenting a mixture of mineral and proteins bathed by tissue fluids and suggest that they may similarly mediate the interaction of native BSP in the bone environment.
I
Bone sialoprotein (BSP) is a NCP member of the Small Integrin-Binding LIgand N-linked Glycoprotein (SIBLING) family of proteins (Fisher et al. 2001) whose pattern of expression is temporally and spatially associated with initial mineralization events (Chen et al. 1991a,b). Like other members of this family, it contains an integrin binding site and numerous posttranslational modifications including N- and O-linked glycosylation, tyrosine sulfation, and serine and threonine phosphorylation (Ganss et al. 1999). These posttranslational additions are believed to mediate matrix-matrix and cell-matrix events, for example, phosphorylation influences crystal growth, whereas glycosylation can affect cell attachment (reviewed in Qin et al. 2004).
Structural characterizations of BSP and in vitro assays have revealed two glutamic acid-rich regions (poly [E]) in the N-terminal half of the molecule (Hunter and Goldberg 1993,1994; Harris et al. 2000; Tye et al. 2003). These domains are believed to be partially responsible for the nucleating activity of BSP and could also be involved in the binding capacity of BSP to hydroxyapatite (HA) (Oldberg et al. 1988; Stubbs et al. 1997; Goldberg et al. 2001).
Our current understanding of the properties, activities, and molecular structure of BSP derives mainly from in vitro assays and in silico predictions. The objective of this study was to test, in the biological environment of the body, the hypothesis that poly [E] domains contribute to the binding capacity of BSP to mineral. These poly [E] domains were mutated for polyalanine (poly [A]) in BSP because
Materials and Methods
Site-directed Mutagenesis
Full-length recombinant rat BSP (prBSPE) and a mutated form referred to herein as prBSPA were both prokaryotically expressed and purified as described in Tye et al. (2003). For prBSPA, the two poly [E] domains on prBSPE were replaced by poly [A] groups by site-directed mutagenesis (Figure 1). For both recombinant molecules, the signal sequence at the N-terminal was removed and replaced by two vector-derived amino acids. A thrombin-cleavable pentahistidine site was added to the carboxyl-terminus of the cDNA for purification purposes.
Dinitrophenylation
Full-length prBSPE and prBSPA were tagged with DNP on lysine groups (Figure 1) using the method of Little and Eisen (1967). Labeling efficiency was estimated at 38% or 5/13 lysine groups available in prBSPE or prBSPA based on yields obtained when preparing albumin-DNP (Nanci et al. 2004). At maximum labeling, five DNP groups would increase the base molecular mass of prBSPEor prBSPA by no more than 835 Da.
SDS-PAGE and Western Blotting
prBSPEand prBSPA (with and without DNP tag) were solubilized into sample preparation buffer containing 62.5 mM Tris (pH 6.8), 2% SDS, 15% glycerol, 40 mM dithiothreitol, and 0.005% bromophenol blue (final concentrations). Approximately 5 μg of protein was applied to individual lanes of minigel format (8.6 cm × 6.8 cm × 1 mm) 12% polyacrylamide slab gels. Broad-range molecular mass proteins (Bio-Rad; Mississauga, ON, Canada) were also loaded. Polyacrylamide gel electrophoresis in the presence of SDS was carried out under discontinuous conditions as described by Laemmli (1970). Silver staining of some gels was done using the Blum method described by Rabilloud et al. (1994). For other gels, proteins were transferred onto nitrocellulose membranes (BA-85; Schleicher and Schuell, distributed by Mandel Scientific Company, Guelph, ON, Canada) and probed with anti-DNP antibody (1:10,000; Dako, Carpinteria, CA) or anti-BSP antibody (1:500, LF-100; courtesy of Dr. L.W. Fisher, NIDCR, NIH, Bethesda, MD) as described previously (Chen et al. 1995). Detection was performed with a secondary goat anti-rabbit antibody IgG-peroxidase conjugate (1:30,000; Sigma-Aldrich Canada, Oakville, ON, Canada) and using the ECL plus Western blotting detection system (Amersham Biosciences, Baie d'Urfe, QC, Canada) as recommended by the manufacturer. Molecular mass markers were visualized by staining with Ponceau S (Sigma-Aldrich Canada).
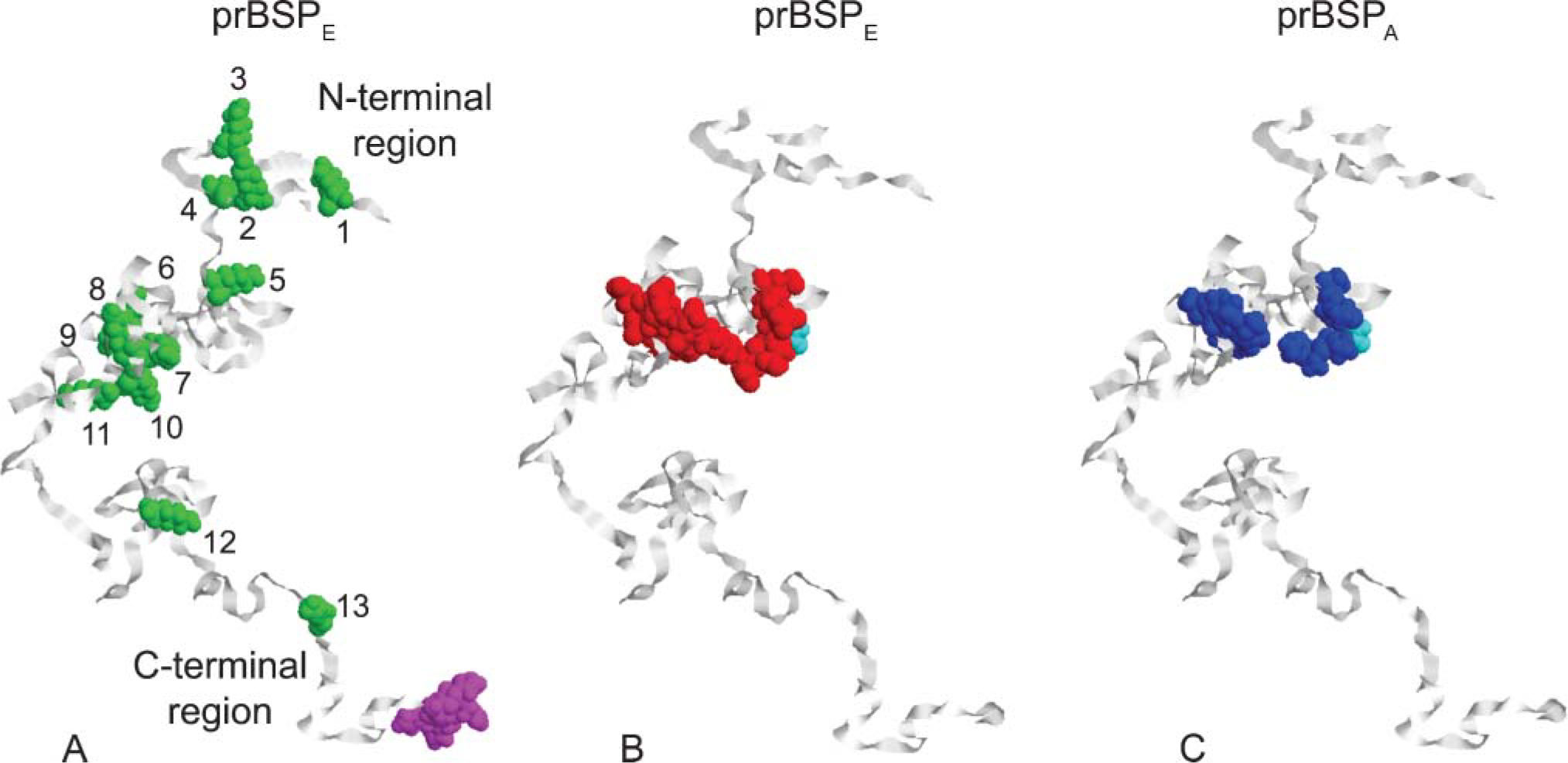
Schematic models of (
Surgical Procedure
Holes were surgically created in the bone on the buccal aspect of the right hemimandible of male Wistar rats weighing 100 ± 10 g (Charles River Canada; St-Constant, QC, Canada), as previously described (Vu et al. 1999). Alzet osmotic minipumps (model 2001D, 200 μl, 8.0 μl/hr; Alza Corporation, Palo Alto, CA) filled with prBSPE-DNP or prBSPA-DNP were slipped under the skin of the back of the animal. Estimated delivery rate was 0.6 μg/hr over 1 day, for a total of 14 μg of protein infused into each rat. Three-day Alzet osmotic minipumps (model 1003D, 100 μl, 1.0 μl/hr; Alza Corporation) filled with prBSPE-DNP were also placed in some animals. Estimated delivery rate was 0.35 μg/hr, for a total of 25 μg of protein delivered. Negative control rats were sham operated or infused with 0.9% sodium chloride (JT Baker; Phillipsburg, NJ). Each experimental group contained three animals. The rats received an injection of Temgesic (buprenorphine hydrochloride; Reckitt and Colman, Hull, UK) and following surgery they were fed with soft food containing Temgesic. X-rays were taken to verify the positioning and stability of catheters. All experimental protocols and animal handling described above were done in accordance with guidelines set forth by the Comité déontologie de l'expérimentation sur les animaux of Université de Montréal.
Tissue Processing
Animals were anesthetized with chloral hydrate (0.4 mg/g body weight; Sigma-Aldrich Canada) and fixed by intracardiac perfusion with 4% paraformaldehyde (BDH; Toronto, ON, Canada) and 0.1% glutaraldehyde (Electron Microscopy Sciences; Washington, PA) buffered with 0.08 M sodium cacodylate (Electron Microscopy Sciences) containing 0.05% calcium chloride (Sigma-Aldrich Canada), pH 7.2, for 20 min. Treated and control contralateral hemimandibles, as well as knees, were dissected and further fixed by immersion in the same fixative overnight at 4C. Tissues were decalcified for 14 days at 4C in 4.13% disodium EDTA (Fisher Scientific; Whitby, ON, Canada) (Warshawsky and Moore 1967). Decalcified tissues were washed for 24 hr in 0.1 M cacodylate buffer, pH 7.2, dehydrated in graded ethanol, embedded in LR White resin (London Resin Company; Berkshire, UK), and polymerized for 48 hr at 58C.
Immunocytochemistry
Light microscopic observations were done with 1-μm semithin sections cut with glass knives on a Reichert Jung Ultracut E ultramicrotome (Leica Microsystems; Wetzlar, Germany) and stained with toluidine blue. Ultrathin sections 80-100 nm in thickness were cut with a diamond knife and transferred onto Formvar-coated (polyvinyl formate; Meca Ltée, Montréal, QC, Canada) 200-mesh nickel grids. Immunolocalizations of proteins were done as previously described (Nanci et al. 1996) using the postembedding colloidal gold method (reviewed by Bendayan 1995). Anti-DNP antibody (1:200, 3 hr; Dako) was used to reveal the DNP-protein complexes and anti-BSP antibody (1:500, 3 hr, LF-100; courtesy of Dr. L.W. Fisher) to detect both endogenous and tagged molecules. All grids were stained with 4% aqueous uranyl acetate for 8 min and with lead citrate for 2 min and examined under a transmission electron microscope (80 kV, JEM-2010; JEOL, Tokyo, Japan).
Quantification of Immunolabeling
Random fields of exposed bone surfaces at the periphery of surgical areas were photographed at X15,000 magnification in the transmission electron microscope. A rectangular grid representing 1 μm height × 2 μm width (2 μm2) was superimposed over appropriately scaled printed micrographs, and the number of gold particles falling within the rectangle was scored. Seventy two rectangular areas were counted for prBSPE-DNP, 51 for prBSPA-DNP, and 14 for sham-operated animals. Subsequent data analyses were done using Version 7.1 of Statistica for Windows (Statsoft; Tulsa, OK) and results expressed as mean ± standard deviation. Particle counts for each group did not follow normal distributions and the non-parametric Kruskal-Wallis ANOVA and median test followed by multiple comparisons of mean ranks for all groups were used to define levels of significance; p<0.05 was considered significant.
Molecular Modeling
Molecular modeling (Figure 1) was done using Version 2.05 of Alchemy 2000 for Windows (a simplified version of the SYBYL software package supplied by Tripos; St Louis, MO. SYBYL is still sold commercially but the Windows-based program is not). Full-length sequences for prBSPE and prBSPA (Tye et al. 2003), both 314 amino acid residues in length, were entered into the protein sequence editor and modeled as random coils using program defaults (filled valences and hydrogen atoms present in each model). These random coil models reflected possible folding arrangements in solution based on the inherent physicochemical properties of the amino acids that comprise prBSPE and prBSPA (Figure 1) and are consistent with existing NMR data indicating that BSP is unstructured in solution (Fisher et al. 2001). We chose to represent prBSPE and prBSPA in an extended, rather than a compact, random coil arrangement to facilitate illustration of the location of certain amino acid groups like lysine across the length of the protein molecule (Figure 1).
Results
Schematic random coil models of prBSPE and its alanine-substituted mutant form (prBSPA) are shown in Figure 1. Both prBSPE and prBSPA contain 13 lysine groups in identical positions (numbered 1-13 in Figure 1A), any one or more of which can react with DNP during the dinitrophenylation procedure. Eleven (85%) of the lysines are located in the N-terminal half of the molecule (Figure 1A), and five of these (45%) are within the same globular domain where the two polyglutamic acid motifs were replaced by polyalanine motifs in prBSPA (Figures 1B and 1C). As expected, substitution of 17 glutamic acid residues by an equal number of alanine residues alters both the isotopic molecular mass (from 34,854 to 33,868 Da) and electronegativity (pI rises from 4.20 to 4.38) of the protein. At pH 7.4, prBSPE is estimated to carry a charge of −55.7, whereas prBSPA would have a charge of −38.2. The models shown in Figures 1B and 1C suggest that these differences in residue type and charge do not, however, cause major changes in the conformation of the protein in solution at least in terms of extended random coil models.
Silver-stained SDS-PAGE gels showed that uncomplexed prBSPE and prBSPA exhibit a major band near 66 kDa (Figure 2), a finding consistent with the previously reported apparent molecular mass of these recombinant proteins (Tye et al. 2003). Some very faintly stained bands that likely represent degradation products were also detected at lower molecular mass. Consistent with the small differential in deduced molecular mass and electronegativity, the main prBSPA band migrated slightly lower than the one for prBSPE (Figure 2). The silver-staining pattern was similar after dinitrophenylation, indicating that the addition of DNP residues did not substantially alter the electrophoretic mobility of these proteins. On immunoblots, anti-BSP antibody reacted with both DNP-complexed and -uncomplexed versions of prBSPE and prBSPA (Figure 2). The major staining was observed near 66 kDa. Noteworthy, most of the lower molecular mass bands reacted with the anti-BSP antibody, indicating that they represent fragments of the recombinant proteins and not unrelated contaminants (Figure 2). Except for the presence of a few, faintly reactive higher molecular mass bands, which likely result from aggregation during dinitrophenylation, and some other lower molecular mass bands of ∼31 kDa, the staining patterns before and after tagging were similar, indicating no effect on the antigenicity (Figure 2). Immunoblots with anti-DNP revealed that the most intense staining was also associated with the major bands near 66 kDa (Figure 2). The higher and lower molecular mass bands were also reactive, indicating they also contain dinitrophenol residues.
To investigate binding potential, mineralized bone surfaces at the surgical site were continuously exposed to either prBSPE-DNP or prBSPA-DNP by infusion for 24 hr. The reason for tagging the two forms of prBSP with DNP was to be able to discriminate infused prBSP from endogenous BSP. Tagged molecules were immunodetected mainly over the surface layer of bone surrounding the drill site (Figure 3A). Dinitrophenylated proteins were occasionally found in deeper regions along the surface of microfractures created during the surgical procedure. On the other hand, fewer gold particles were associated with the bone matrix when prBSPA-DNP was infused (Figure 3B). Incubation with anti-BSP showed labeling along exposed bone surfaces as well as at sites where endogenous molecules normally concentrate, such as cement lines and interfibrillar matrix accumulations (Figure 3C). In animals infused for 3 days with prBSPE-DNP, some gold particles were also found over matrix accumulations in newly formed bone near the drill site (Figure 4). Hardly any gold particles were observed over tissue sections from sham-operated animals when incubated with anti-DNP antibody (Figure 3D). No significant immunolabeling was seen over bone in contralateral hemimandibles or in the tibias of animals infused with prBSPE-DNP or prBSPA-DNP (data not shown).
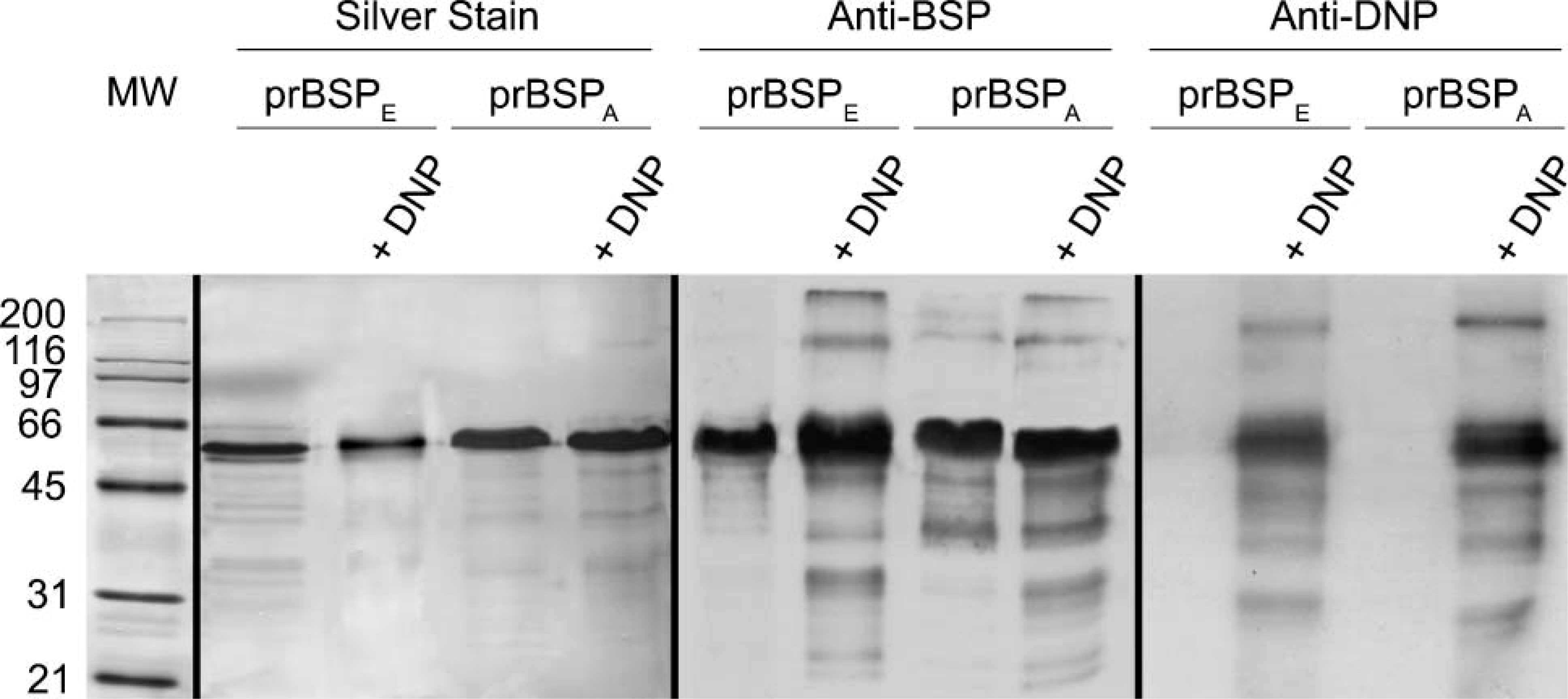
Characterization of the prokaryote recombinant BSP preparations. The first lane contains standard broad-range molecular mass proteins (in kDa) stained with Ponceau S. Protein profile of prBSPE and prBSPA with or without DNP as revealed by silver staining (Lanes 2-5). Immunoblots of prBSPE and prBSPA with or without DNP probed with antibodies for BSP (Lanes 6-9) and DNP (Lanes 10-13). Lanes 2, 6, and 10: prBSPE, Lanes 3, 7, and 11: prBSPE-DNP, Lanes 4, 8, and 12: prBSPA, Lanes 5, 9, and 13: prBSPA-DNP.
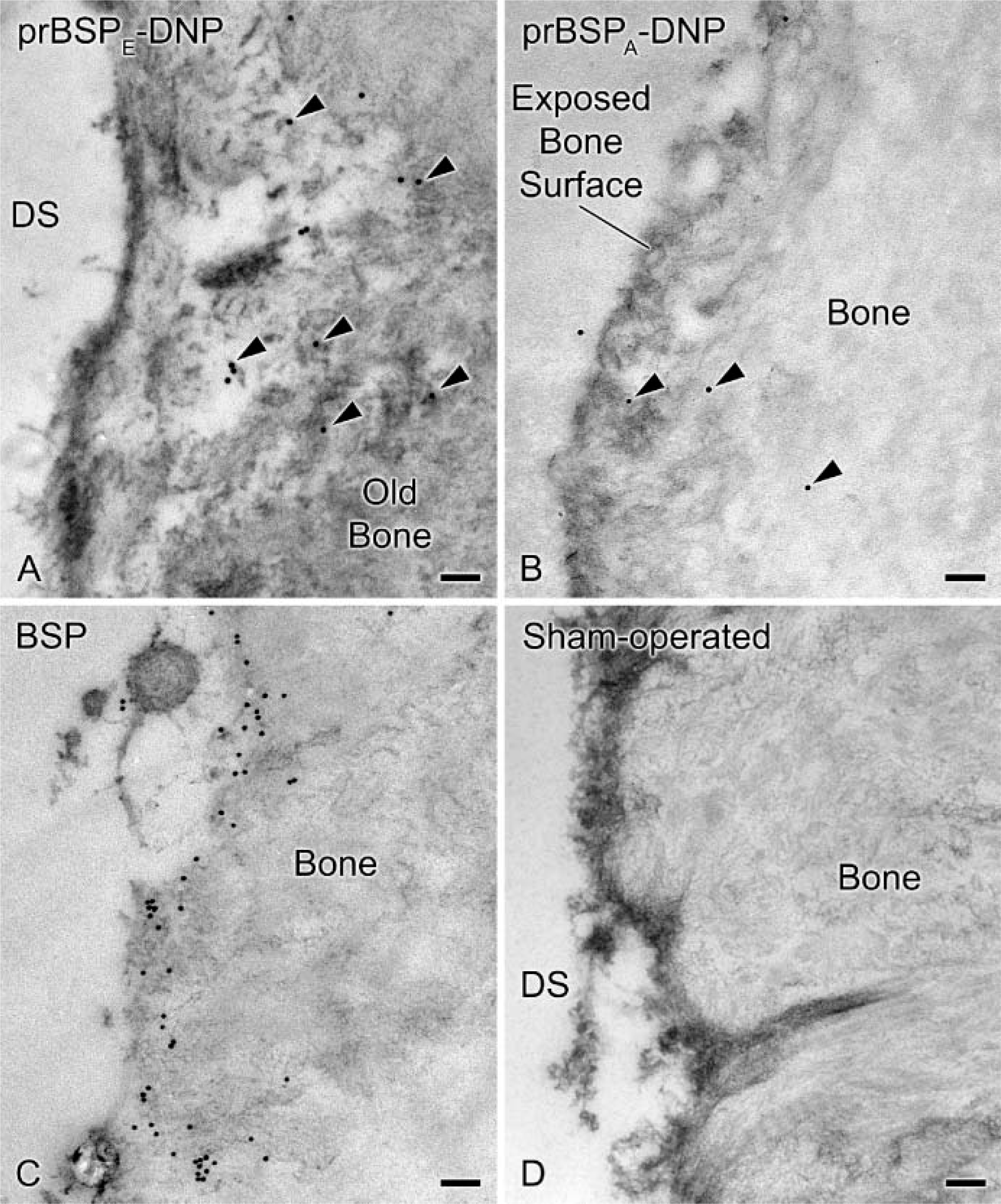
Transmission electron micrographs illustrating the mandibular bone at the drilling site(DS). Dinitrophenylated prBSPE (prBSPE-DNP) (arrowheads in
Quantitative analyses following 1-day infusions indicated labeling indices of 5.04 ± 0.73 gold particles/μm2 of exposed bone surface for prBSPE-DNP and 1.37 ± 0.35 gold particles/μm2 of exposed bone surface for prBSPA-DNP. The background level was evaluated by incubating sham-operated animals with anti-DNP and resulted in a labeling index of 0.14 ± 0.23 gold particles/μm2 of exposed bone surface. The difference in labeling between prBSPE-DNP and prBSPA-DNP, as well as between both these proteins and background, was statistically significant (p<0.05) (Figure 5).
Discussion
Pertinent functional information can be derived from in vitro assays but, ultimately, results must be validated in vivo where the physiological environment is complex. In this context, a recent study on the binding of salivary proteins to HA questioned the biological relevance of adsorption data obtained from in vitro studies using single proteins (Yin et al. 2005). To our knowledge, our investigation is the first study attempting to evaluate in a multifactorial animal model the effects of altering functional domains on the binding capacity of a prokaryote recombinant form of BSP to a mineralized substrate. Results show that substitution of poly [E] domains by poly [A] has a major impact and reduces by 73% its binding to mineralized bone. Our results do not allow distinguishing whether the effect is on binding to the mineral or to the organic phase because the tissues were decalcified for histological processing. However, retention of tagged molecules in such preparations can only occur if they are close to bulk endogenous organic matrix constituents to which they are cross-linked by the fixative. In addition, BSP has been shown to interact with collagen (Fujisawa and Kuboki 1992; Tye et al. 2005). Organic constituents are very closely associated to the mineral and can therefore influence any interaction with it. These possibilities must be taken into consideration when investigating the function and mineral binding capacity of proteins within the biological environment of the organism.
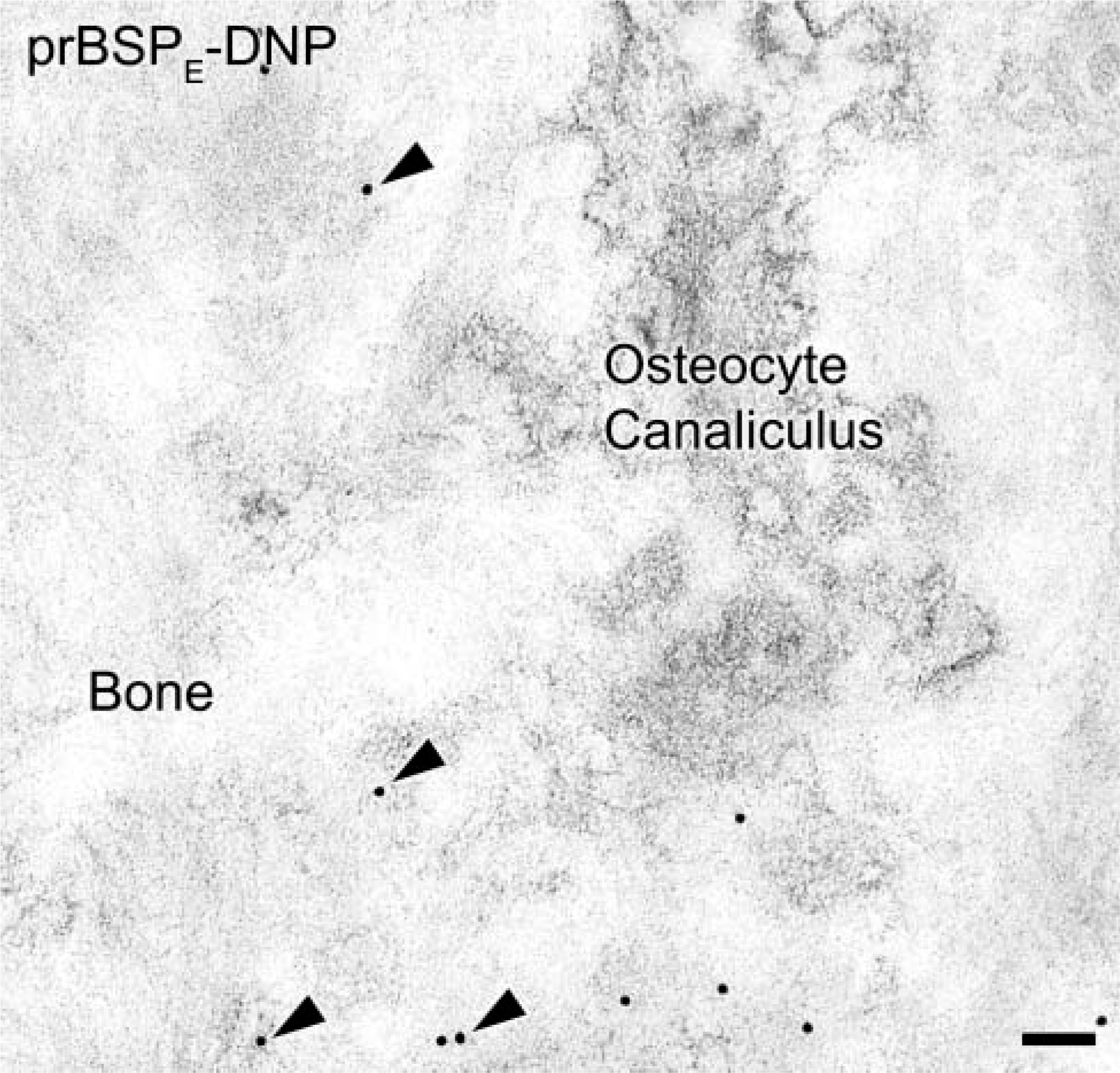
Transmission electron micrographs of mandibular bone following a 3-day infusion with prBSPA-DNP. Tagged molecules were observed incorporated into newly formed bone matrix (arrowheads). Bar = 100 nm.
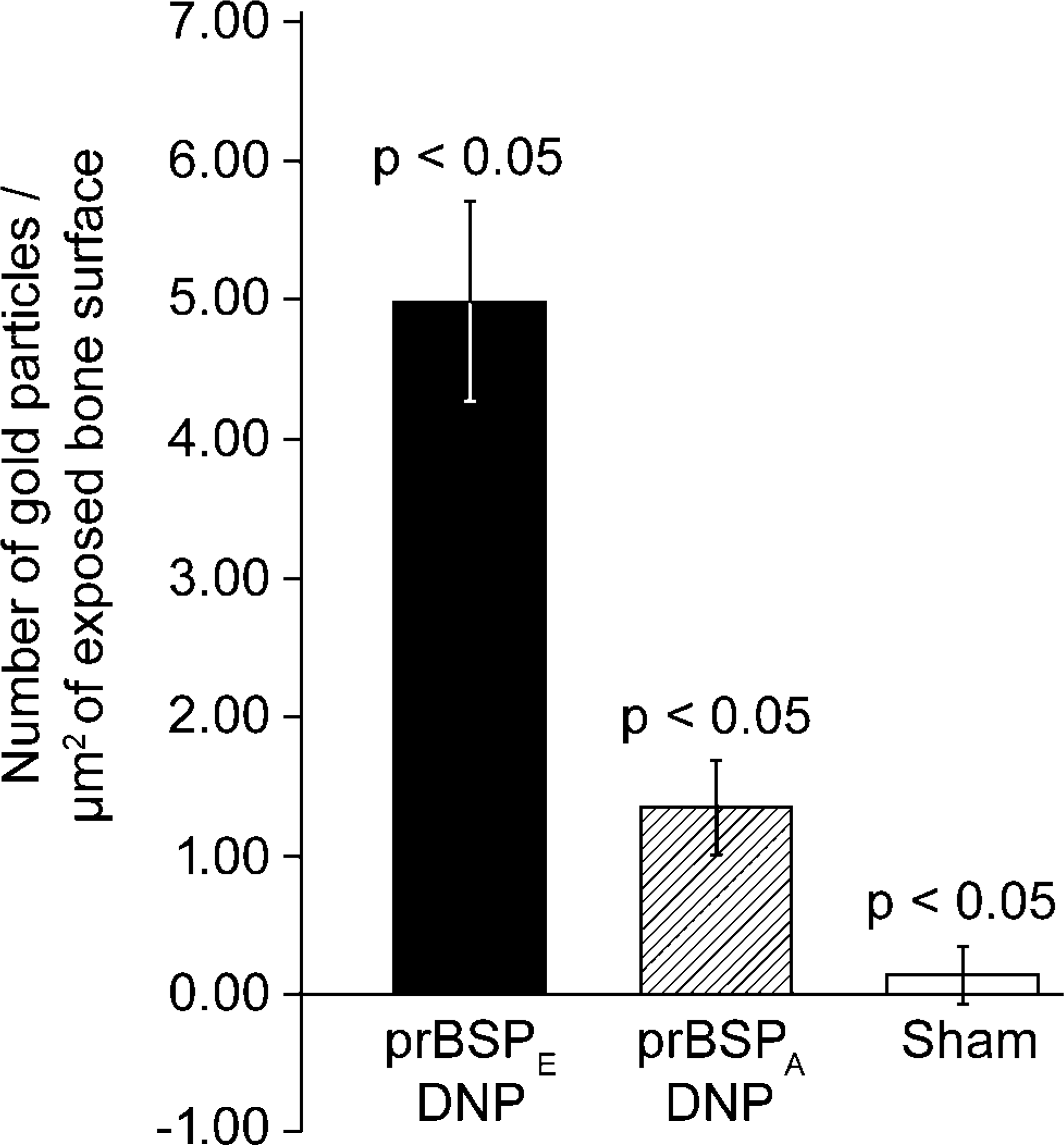
Quantitative analysis of the differential binding among prBSPE-DNP, prBSPA-DNP, and sham-operated animals after a 24-hr infusion period (expressed as mean ± standard deviation). Differences among the three groups are significant (p<0.05).
There have been in vitro studies investigating the role of poly [E] domains in the nucleating activity and mineral-binding capacity of BSP (Hunter and Goldberg 1994; Stubbs et al. 1997; Goldberg et al. 2001; Tye et al. 2003). In a recent report, it was demonstrated that the replacement of both contiguous poly [E] domains with poly [A] resulted in a significant reduction, but not complete loss, of the nucleation capacity of prBSPE (Tye et al. 2003). Similarly, synthetic homopolymers of poly [E] do not completely inhibit the binding of native BSP to HA (Goldberg et al. 2001). It was therefore suggested that additional domains or specific conformational motifs on BSP are involved in HA binding and nucleation (Tye et al. 2003). The fact that prBSPA still exhibits some binding to mineralized bone matrix (Figure 5) is consistent with an implication of other molecular determinants on the protein backbone in the interaction of BSP with HA.
Immunoblotting indicates that both tagged protein preparations comprised the intact molecules (major band near 66 kDa) but also some smaller and higher molecular mass peptides. However, these must clearly represent minor constituents because they are barely visible on silver-stained gels. This is consistent with a previous report that similar prokaryote recombinant BSP preparations had an apparent 99%+ purity based on amino analysis and mass spectrometry (see Tye et al. 2003). Furthermore, it is doubtful that any of them represent contaminating bacterial products because most of the bands are recognized by the anti-BSP antibody. Therefore, the low molecular mass protein bands most likely correspond to degradation products, present in very low amounts in the original recombinant protein preparations and/or that formed during handling of the purified protein for biochemical analyses and dinitrophenylation. In the case of those with high molecular mass, they probably represent aggregates that formed during the dinitrophenylation process because these are not present in the uncomplexed proteins. Noteworthy, the anti-DNP immunoblots show that both prBSPA-DNP and prBSPA-DNP have very similar banding patterns, suggesting that both preparations have been similarly affected. The presence of more than one labeled form in infusates complicates interpretation of the results because it is not possible to unequivocally attribute the observed difference in binding solely to the substitution of poly [E] by poly [A] domains. However, together the data strongly suggest that the differential binding observed has a limited contribution from these other minor dinitrophenylated BSP products and is likely due to the basic difference between the two proteins, that is, substitution of poly [E] domains by poly [A], which interestingly does not seem to significantly affect breakdown. Irrespectively, the data unequivocally demonstrate that the two preparations behave differently in an in vivo environment.
Based on the equal number of lysine residues present in prBSPE and prBSPA and the suggestion from random coil modeling that both proteins share similar inherent conformations in solution, there is little reason to suspect that the differential binding observed would be due to differences in degree of dinitrophenylation, especially considering that the disparity in binding between prBSPE and prBSPA was almost 80%. Another possible cause would be the charge differential between prBSPE and prBSPA. At physiological pH, the estimated charge ratio of prBSPE/prBSPA is ∼4.45, whereas the prBSPE/prBSPA binding ratio determined in this study was 3.69. If the observed difference in binding was due solely to charge, then both the binding ratios and charge ratios should be identical. Because the ratios actually differ by >2.5X, it must include other components such as the nature of the amino acid residues that make up the binding sites.
As previously demonstrated with osteopontin-DNP, NCPs infused through a minipump effectively integrate into bone compartments where endogenous proteins are normally found (Nanci et al. 2004). Detection of tagged prBSPE in the interfibrillar matrix accumulations following a 3-day infusion shows that recombinant BSP can also integrate into newly formed bone. The abundant immunodetection of endogenous BSP along exposed surfaces demonstrates that the observed binding of tagged molecules onto these surfaces is a normal event and is not induced by dinitrophenylation or by the infusion process itself. The body seems to respond to bone damage by quickly sequestering and accumulating NCPs such as BSP and osteopontin (Nanci et al. 2000,2004) on exposed mineralized surfaces. Absence of osteoblasts on these surfaces, paucity of inflammatory cells such as macrophages (McKee and Nanci 1996) in their immediate vicinity, and incorporation of tagged molecules administered at a distance indicate that circulating molecules from blood or tissue fluid microenvironment are likely sources for these proteins. Indeed, it has been proposed that circulating NCPs also contribute to the formation of cement lines at natural bone interfaces (Nanci et al. 2000,2004). The abundance of endogenous BSP also suggests that the native, posttranslationally modified molecule may have a higher affinity for exposed bone matrix than either version of the recombinant proteins used in this study. In this context, it has been shown that although posttranslational modifications are not necessary for the nucleating activity of BSP, they increase the potency of the molecule (Tye et al. 2003).
No binding of either prBSPE or prBSPA was observed at a distance away from the infusion site. This may reflect the possibility that (1) no naturally exposed bone surfaces were available and (2) concentrations may be high near the site of infusion but reach levels below the detection limit as the molecules diffuse away. Alternatively, molecules may bind to complement factor H and thus be sequestered to limit their functional ranges (Fedarko et al. 2000). Our results suggest that if prBSP binds to factor H, this must occur over a time frame that allows the molecules to also interact with the exposed mineralized matrix or that the protein prefers a calcified matrix (Nanci et al. 2004). Interestingly, factor H is known to bind sialic acid-containing proteins (Meri and Pangburn 1994; Fedarko et al. 2000; Pangburn et al. 2000). The potential sequestration of prBSP by factor H would therefore imply that sialic acid or other posttranslational modifications may not be necessary for this interaction.
Infusion of DNP-tagged molecules through surgically created holes in bones permits experiments to be carried out at near physiological concentrations, to distinguish the administered molecules from endogenous ones, and to follow them over time (Nanci et al. 2004). This approach can be applied to a number of functional studies such as determining the behavior of different isoforms and evaluating the activity of predicted functional groups and bioactive peptides.
In conclusion, results of this study show that substitution of poly [E] by poly [A] domains reduces the ability of a preparation of prBSP comprising intact molecules and fragments to bind and accumulate at exposed surfaces of mineral/organic matrix bone composite in the real tissue microenvironment. It should be noted that the behavior of a protein in vivo will not only depend on its physicochemical properties (e.g., affinity for HA) but will also reflect the modulation of these properties by the various tissue components it is exposed to. For example, the effect of proteins on mineralization can vary depending on whether they are in solution or bound to collagen (Saito et al. 1998,2000). Also, affinity of a protein for HA can be enhanced through interactions with other proteins (Yin et al. 2005), as would be found in the organic matrix generally associated with HA and in tissue fluids. In addition, it has recently been shown that adsorption of BSA to HA is influenced by the degree of complexing of calcium ions to the protein (Kandori et al. 2005), a factor which is certainly pertinent in vivo. Indeed, regional interactions may be one way by which proteins exert multiple behaviors. The approach we have used here offers a simple system in which amino acid motifs, posttranslational modifications, and derived peptides can be tested for function and therapeutic potential in an environment that reflects the complex nature of the body.
Footnotes
Acknowledgements
This work was supported by the Canadian Institutes of Health Research (CIHR).
The authors are grateful to Dr. Pierre Moffatt and Dr. Henry C. Margolis for critical reading of the manuscript, Ms. Micheline Fortin for assistance with sectioning, and Ms. Sylvia Francis Zalzal for preparing the protein A-gold complexes.
