Abstract
Methylmethacrylate (MMA) embedding of undecalcified bone is routinely employed for histomorphometric analyses. Although MMA-embedded bone has been used for immunolabeling at the light microscopic level after removal of the resin, there are no such reports for electron microscopy. The aim of the present study was to determine whether MMA embedding can be used for ultrastructural immunolabeling and how it compares to LR White (LRW), an acrylic resin frequently used for immunocytochemistry of bone. Rat tibiae were fixed by vascular perfusion with aldehyde and embedded either in MMA or LRW resin. Thin sections were processed for postembedding protein A-gold immunolabeling with antibodies to rat bone sialoprotein (BSP) and osteopontin (OPN). The density of gold particles over bone was quantified. The density and distribution of immunolabeling for BSP and OPN respectively, were comparable between MMA and LRW. These results indicate that MMA performs as well as LRW for the ultrastructural immunolabeling of noncollagenous bone matrix proteins.
A
The glycolated form of methacrylate (glycolmethacrylate, GMA) is employed for both light and electron microscopic immunolabeling. GMA is a bifunctional methacrylate that forms crosslinks during polymerization. Consequently, it cannot be dissolved out of sections (Wolf et al. 1992), limiting its application for conventional histomorphometric analyses. This is not the case for MMA. However, this resin has been mainly used for light microscopic immunolabeling, and its application in electron microscopy is not well documented (Baskin et al. 1992). Therefore, the aim of the present study was to investigate whether MMA embedding can be used for ultrastructural immunolabeling of two predominant bone matrix proteins, bone sialoprotein (BSP) and osteopontin (OPN), and to compare the results with those obtained with LR White (LRW) resin, under tissue processing conditions conventionally used with these resins. The latter acrylic-based resin allows good performance in immunocytochemistry (Bendayan et al. 1987) and has been used for cytochemical characterization of the presence and distribution of noncollagenous matrix proteins in calcified tissues (Nanci et al. 1989; McKee and Nanci 1995). The results indicate that MMA is applicable to the ultrastructural immunolocalization of BSP and OPN in bone matrices.
Materials and Methods
Tissue Processing
Male Wistar rats weighing 100 ± 10 g (Charles River Canada; St-Constant, QC, Canada) were anesthetized with chloral hydrate (0.4 mg/g body weight). For embedding in LRW (London Resin Company; Berkshire, UK), rats were perfused for 20 min with a mixture of 4% paraformaldehyde and 0.1% glutaraldehyde in 0.1 M phosphate buffer, pH 7.2, and tibiae were dissected out, cut in half along their length, and immersed in the same fixative overnight. The samples were dehydrated in graded ethanol and then embedded in resin that was polymerized at 58C for 48 hr, as described previously (Nanci et al. 1989). For embedding in MMA (J-T Baker; Phillipsburg, NJ), just after anesthesia, tibiae were dissected, split in half, placed directly in ethanol, and subsequently impregnated with a MMA mixture. Briefly, samples were infiltrated for 4 days at 4C in a clear glass scintillation vial containing a mixture of 80% (v/v) MMA, 20% (v/v)
For light microscopy, semithin sections (1 μm) of the primary spongiosa were cut with a glass knife on a Reichert Ultracut E microtome and stained with toluidine blue or by the von Kossa method. Thin sections of regions near the center of the growth plate were then prepared with a diamond knife, mounted on Formvar-carbon-coated nickel grids, and processed for postembedding protein A-gold immunocytochemistry (reviewed in Bendayan 1995).
Immunolabeling for BSP and OPN
Grid-mounted tissue sections of LRW- and MMA-embedded undecalcified bone were floated for 15 min on a drop of 0.01 M PBS containing 1% ovalbumin (Oval; Sigma Chemical, St Louis, MO) to saturate nonspecific binding sites, and then were transferred to and incubated for 3 hr on a drop of polyclonal chicken egg yolk anti-rat OPN antibody (Nanci et al. 1996) diluted 1:150 with PBS. After incubation, sections were rinsed with PBS, placed on PBS-Oval for 15 min, and then transferred for 1 hr onto a drop of rabbit antichicken IgG (Cappel Research Products; Scarborough, ON, Canada), diluted 1:2000 with PBS. Finally, they were rinsed with PBS, placed on PBS-Oval for 15 min, and incubated for 30 min with protein A-gold complex (8–10 nm diameter) prepared in house according to Bendayan (1995). Other grid-mounted sections were blocked with PBS-Oval and incubated for 1 hr on a drop of polyclonal anti-rat BSP antibody (diluted 1:10 with PBS; the LF87 was courtesy of Dr. L.W. Fisher, National Institutes of Health, Bethesda, MD; Fisher et al. 1995). In the latter case no secondary antibody was used and grids were washed, blocked, and directly incubated with the protein A-gold complex. As controls, sections were incubated with protein A-gold alone. All incubations were performed at room temperature in a moist environment. The grids were then washed thoroughly with PBS, rinsed with distilled water, and air-dried. All sections were routinely stained with uranyl acetate and lead citrate and examined using a JEOL JEM 1200EX II transmission electron microscope operated at 60 kV.
Quantitative Analysis of the Immunolabeling
Images were printed at a final magnification of X38,000 and digitized using a Sony CCD 7002P camera (Sony Corporation; Tokyo, Japan). The number of gold particles per print, over interfibrillar accumulations of noncollagenous matrix (patches), and over collagenous regions of bone was quantified and compared using a Q500 image analysis system (Leica; Cambridge, UK). Based on the fact that NCPs accumulate mainly as interfibrillar patches (reviewed in Nanci 1999), a morphometric program was set up to recognize and measure the area of regions in which there was a concentration of gold particles, as determined by a threshold distance between particles. For each print, a visual control by the same operator was made to ensure that the regions selected did indeed correspond to interfibrillar areas. For statistical analyses, Systat version 10 software (SPSS; Chicago, IL) was used. Because the data did not show a normal distribution, the number of particles obtained after incubation of MMA and LRW sections with BSP or OPN was compared using the Mann-Whitney
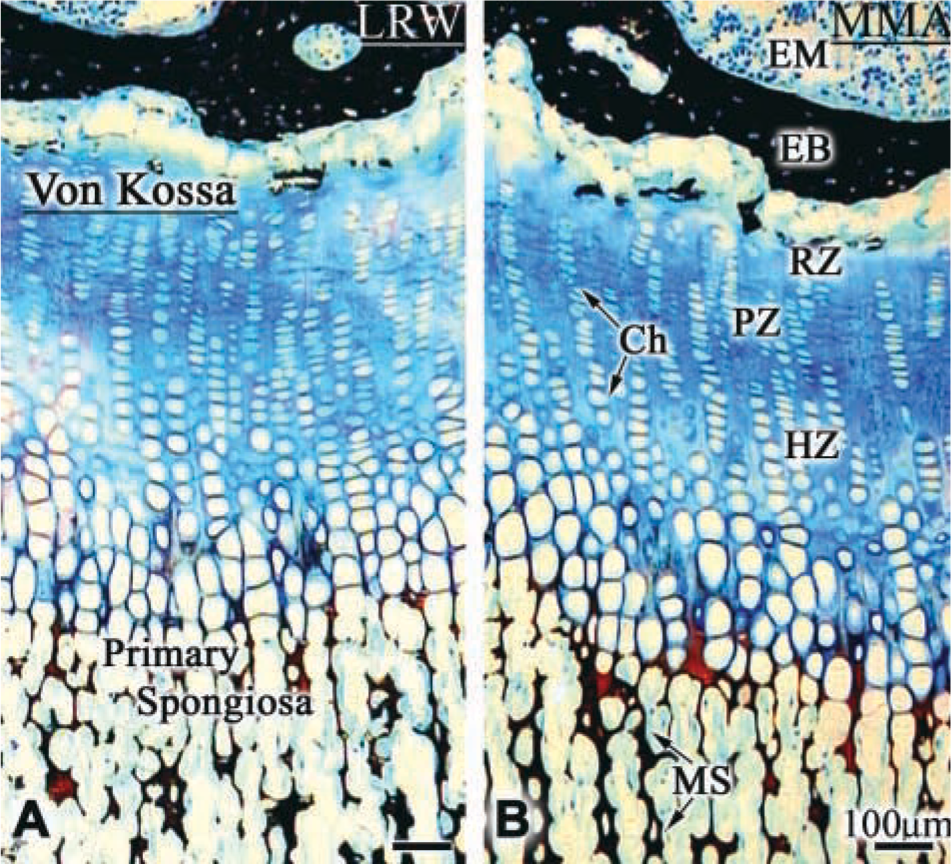
Endochondral ossification in rat tibia. (
Results
Structural Features
Von Kossa staining did not reveal any difference in the staining pattern and intensity observed over bone and calcified cartilage embedded in MMA or LRW (Figures 1A and 1B). Ultrastructural preservation was generally better and more consistent with LRW processing, but MMA processing also permitted visualization of cellular features in some regions (compare Figures 2A and 2B). Likewise, there was no significant difference in the appearance of the matrix (compare Figures 3A and 3B, and Figures 3C and 3D).
Immunolocalization of BSP and OPN
The distribution of immunolabeling for BSP and OPN was similar for MMA- and LRW-embedded specimens. With both resins, gold particles were found mainly over the calcified bone matrix; some were also seen over osteoid. The labeling was mostly associated with electron-dense portions of the matrix, i.e., over cement lines and interfibrillar accumulations (patches) of granular/reticular-textured organic matrix (Figure 3). Incubation of tissue sections with protein A-gold alone resulted in the occasional presence of particles throughout sections.
Quantitative Analysis of the Immunolabeling
Numerical parameters for the number of gold particles obtained after incubation of tissue sections with BSP and OPN antibodies are summarized in Table 1. For both BSP and OPN incubations, the distribution of patches/surface area within the three arbitrarily defined density categories was comparable for bone samples embedded in either MMA or LRW. For both resins, about 44% of the bone surface examined fell within the 8–16 patches/20 μm2 category and nearly 28% in the >17 patches category (Figure 4). With both antibodies, there was no significant difference in mean numbers of total gold particles/micrograph observed over the two resins (Figure 5A). Power analysis indicated that the number of samples studied was sufficient to allow an 80% chance of detecting a difference of 102.2 particles for BSP and 109.6 particles for OPN at a 0.05 level of statistical significance.
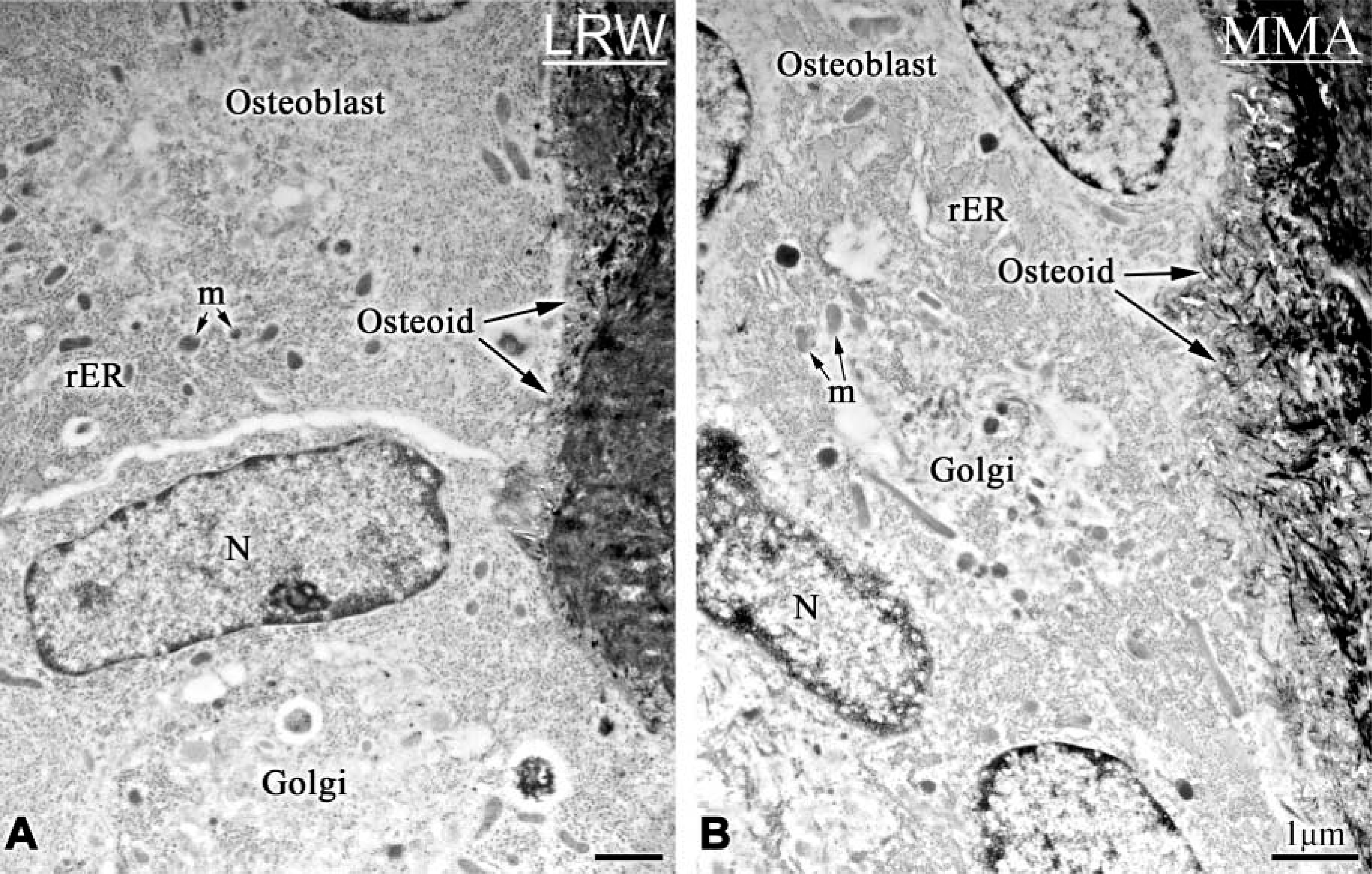
Electron micrographs from the primary spongiosa of rat tibiae, illustrating the ultrastructural preservation in (
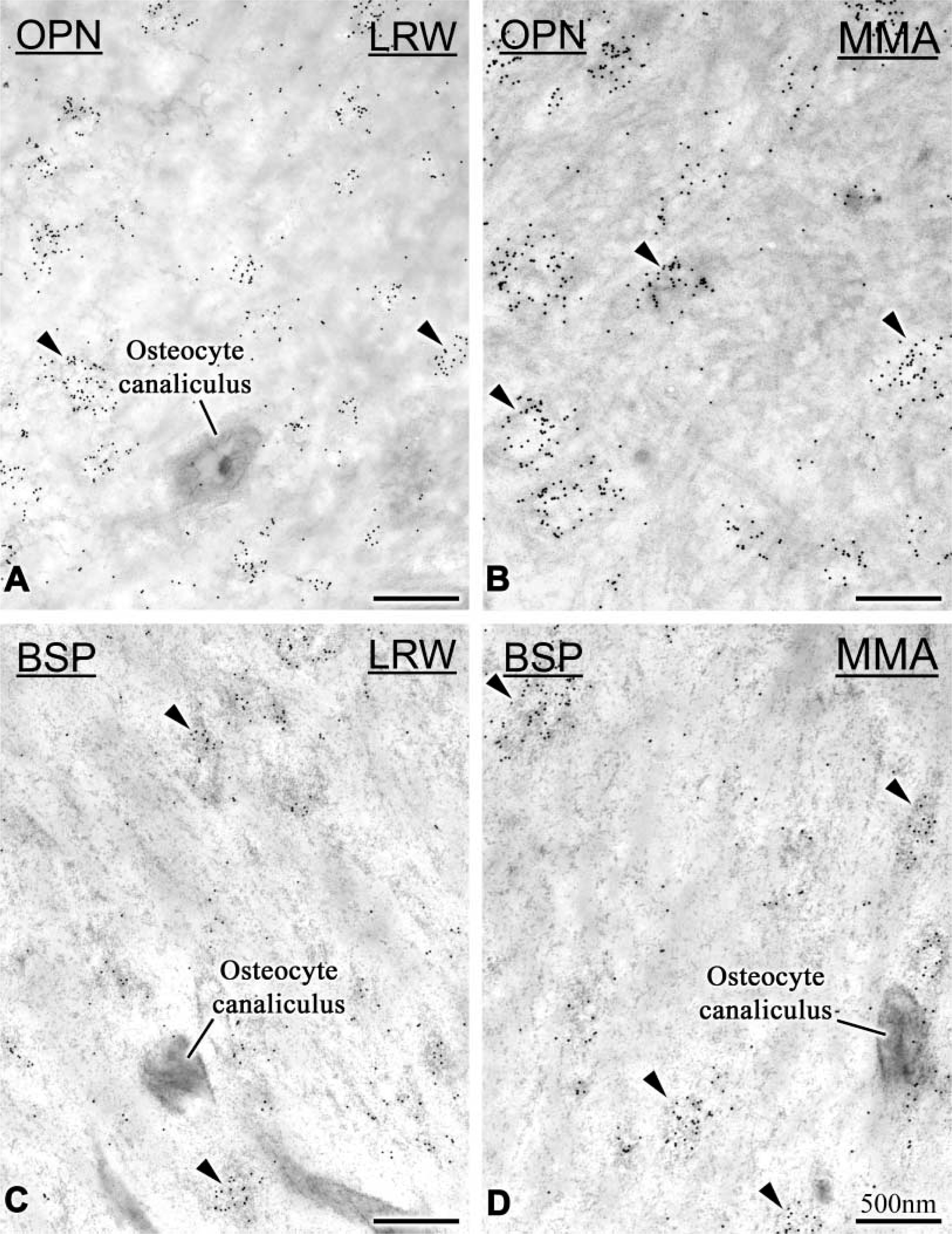
Immunocytochemical preparations with anti-rat osteopontin (OPN) and anti-rat bone sialoprotein (BSP) antibodies. (
The majority of the immunolabeling was found over the patches, and there was no significant difference in labeling density over them between the two resins (Figure 5B). Patches represented an average of 19% of the observed bone surfaces but an average of 88% of the labeling was found over them (Table 1). The relationship between labeling density and the number of patches was linear for both BSP and OPN (Figure 6).
Quantitative analysis of immunolabeling over LR White- and methylmethacrylate-embedded bone a
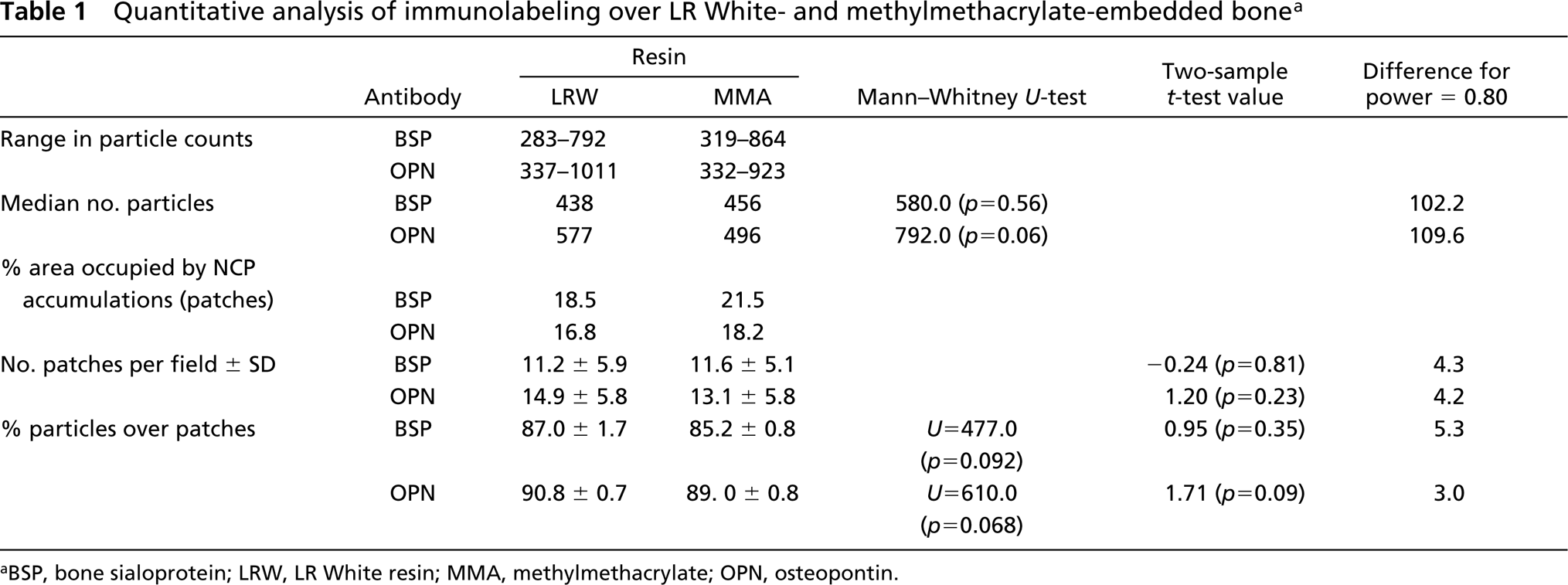
aBSP, bone sialoprotein; LRW, LR White resin; MMA, methylmethacrylate; OPN, osteopontin.
The mean number per area of gold particles obtained with both BSP and OPN over osteoid was 4.3 for MMA and 6.25 for LRW, over nuclei 2.5 and 3.5, respectively, and over cartilage 0 and 0.44, respectively (data not shown).
Discussion
Whereas MMA is widely used for histomorphometric analysis of bone biopsy specimens, (Theuns et al. 1993), its application for immunolabeling has been limited. Indeed, it is generally believed that the high temperature generated by reactive radicals during polymerization of the resin is not compatible with retention of antigenic determinants (Cole 1982; Ashford et al. 1986; Baskin et al. 1992; Wolf et al. 1992; Erben 1997). Nevertheless, the use of conventional processing yielded inconsistent and suboptimal cell prevervation, and distribution of immunolabeling and density were similar to those obtained with LRW, a resin polymerized at 58C. It has been determined that the maximal temperature reached by MMA when polymerized at room temperature does not exceed 60C (Theuns et al. 1993). Therefore, when polymerized at 32C, as in the present case, it is likely to go above this temperature. However, compared with LRW, the MMA embedding procedure does not significantly alter antigenic properties, at least for BSP and OPN, and the temperature generated during polymerization certainly does not dramatically affect antigenicity of typical noncollagenous matrix proteins in calcified bone. This may be due to the fact that these proteins are intimately associated with the mineral phase, which may prevent their denaturation. However, lower temperatures may, however, be important for more labile antigens such as cell proteins and enzymes.
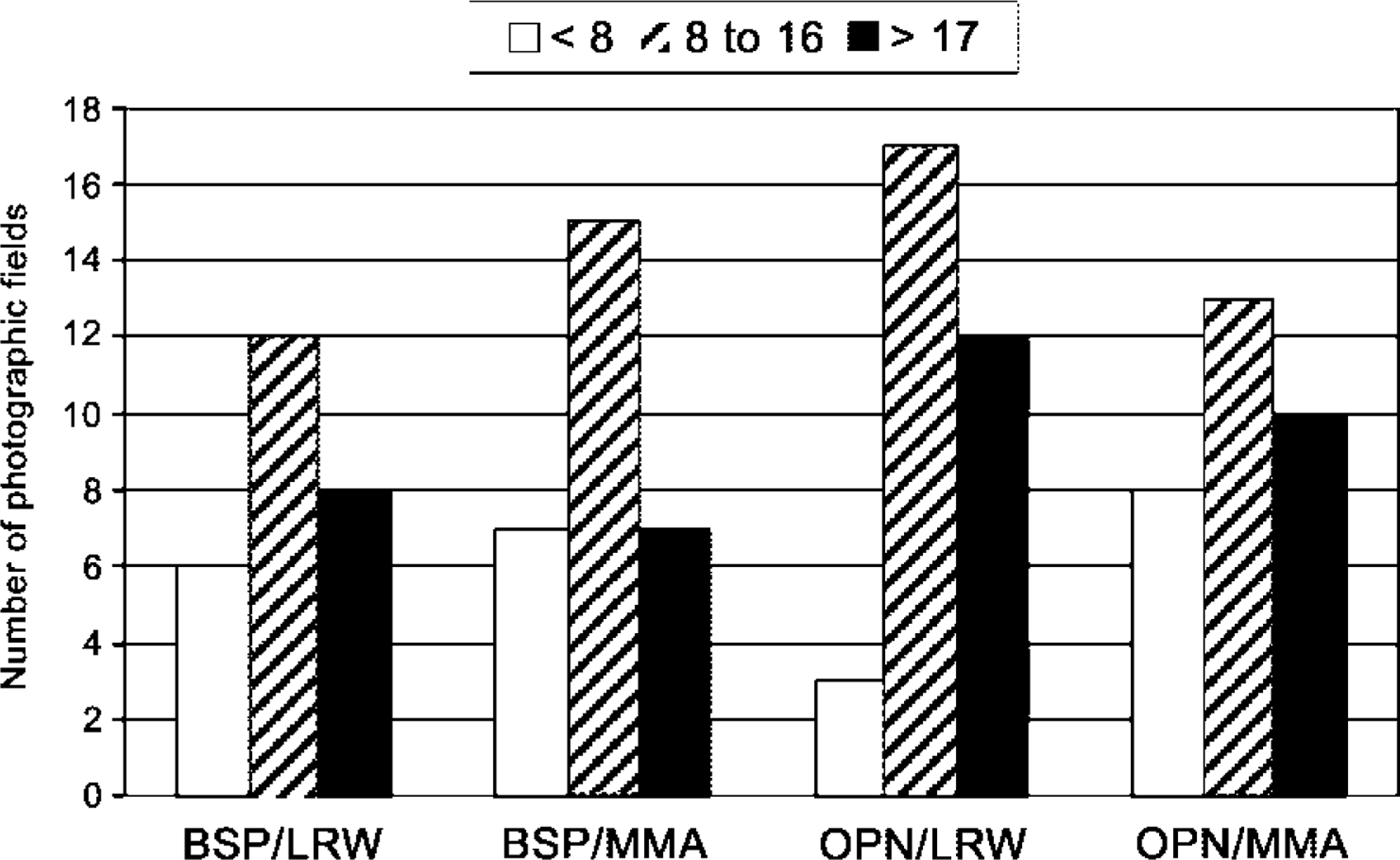
Classification of bone surfaces according to frequency of noncollagenous matrix patches. Nearly half of the surfaces contain 8–16 patches/20 μm2.
The labeling for BSP and OPN, respectively, obtained with undecalcified sections of MMA- and LRW-embedded bone (fixed as conventionally used with these resins) shows similar distributions and densities. In addition, the BSP:OPN labeling density ratios are essentially similar, indicating that MMA and LRW affect the antigenic properties of both BSP and OPN in a similar manner.
Matrix organization and relative proportions of collagenous and noncollagenous components in bone represent important parameters that vary with development stage and anatomic site, and reflect local tissue dynamics (Bianco 1992; Nanci 1999). In the region of the rat primary spongiosa examined, interfibrillar accumulations of noncollagenous matrix proteins occupy 16–21% of the bone surface area, and 85–90% of the BSP or OPN immunolabeling is found over them. The observed variability in density of non-collagenous matrix protein patches within photographic fields is likely associated with microanatomic variations in the relative proportions of the collagenous and noncollagenous components. In this context, it has been proposed that the amount of interfibrillar matrix depends on the packing density of collagen which is, in part, determined by the speed at which it is deposited (discussed in Nanci 1999; Bosshardt and Nanci 2000).
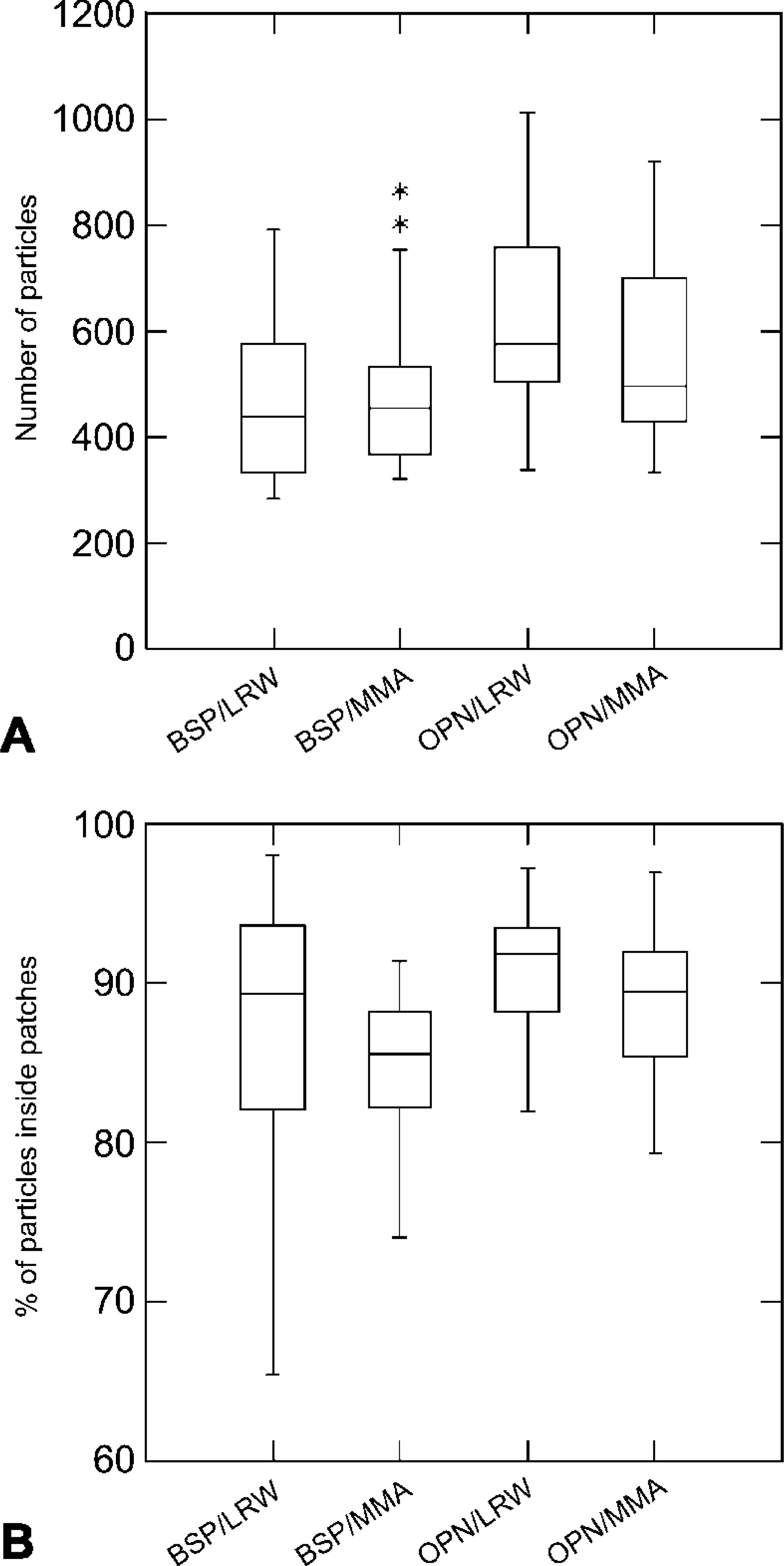
Box plots comparing structural and immunocytochemical parameters in LR White (LRW) and methylmethacrylate (MMA). (
Although the number of gold particles over cartilage was almost nonexistent with both resins, osteoid showed roughly twice as many particles as over nuclei. Labeling over nuclei is used as an indication of background (Bendayan 1995). The presence of a higher number of gold particles over osteoid, albeit at a very low density, is consistent with a diffusion of secreted molecules from the osteoblast surface towards the mineralization front; a similar observation has been made for osteocalcin (Camarda et al. 1987).
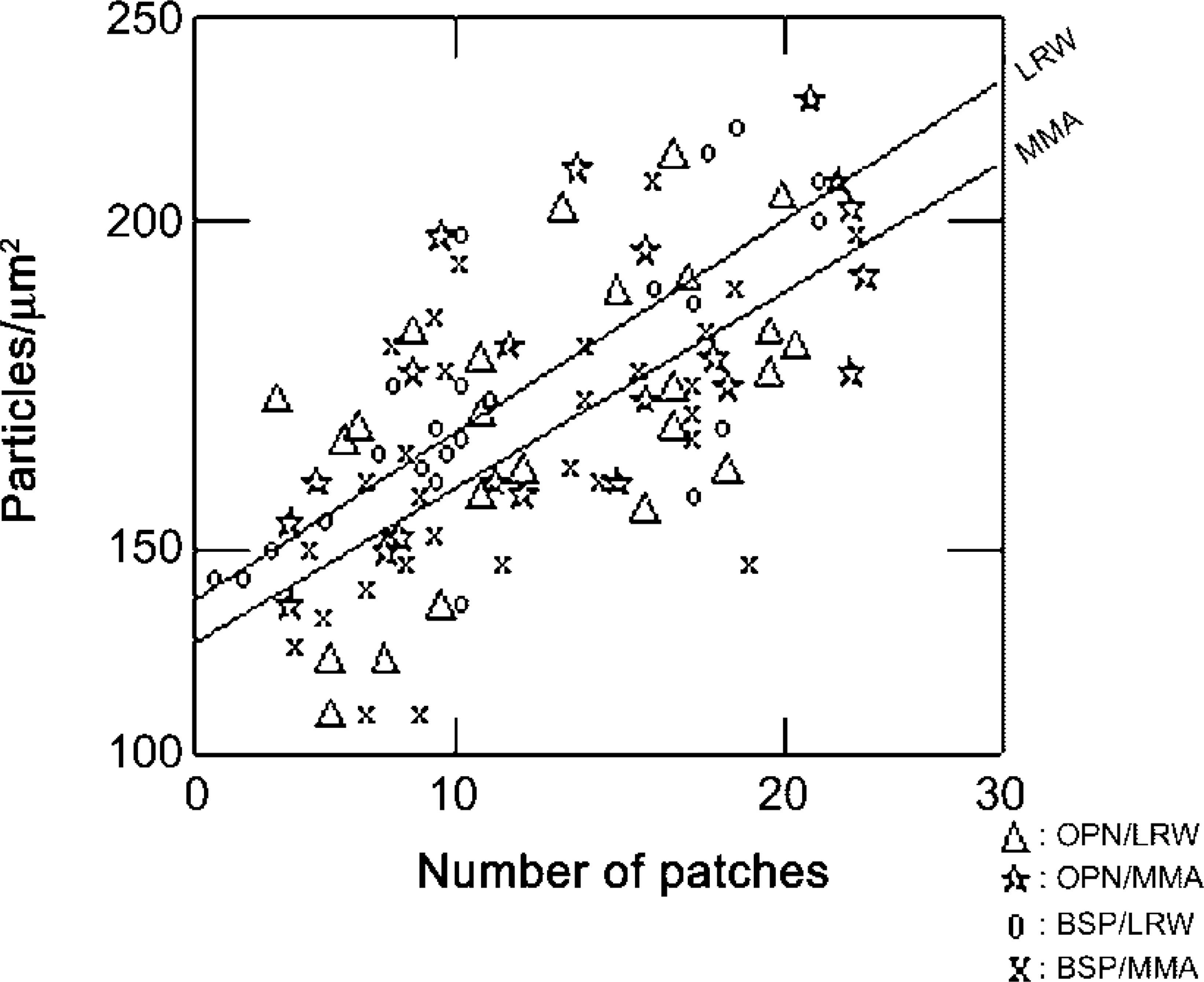
Both resins show a linear relationship between the density of gold particles and the number of patches (
In conclusion, our study shows that it is possible to detect and immunolocalize BSP and OPN in sections of alcohol-fixed rat bone embedded in MMA from which the resin is not removed. This finding is important because it highlights the feasibility of processing experimental and clinical bone samples for parallel light microscopic and histomorphometric analyses, and for compositional studies at the ultrastructural level. In addition, it offers the possibility to further exploit valuable MMA-embedded bone samples from previous studies by determining whether the morphological and histomorphometric alterations observed are associated with changes in the content and/or distribution of major matrix proteins.
Footnotes
Acknowledgements
Supported by the Canadian Institutes of Health Research.
We are grateful to Natalie Dion (Hôpital St Luc, Montréal) for MMA embedding, Sylvia Zalzal (Université de Montréal) for advice with immunolabeling, Micheline Fortin (Université de Montréal) for assistance with tissue sectioning, Pierre Rompré (Université de Montreal) for statistical analysis, and Paul Pilet (Inserm 99–03, Nantes) for adapting the gold particle counting software.
