Abstract
We studied the intra-islet localization of the glucagon-like peptide 1 receptor (GLP-1R) by colocalization studies of the GLP-1R mRNA and protein with islet cell hormones in mice, rats, and humans. In contrast to previous reports, we show that the GLP-1R is selectively located on the β cells. The localization of GLP-1R in islets and ducts was studied using ISH and double and triple fluorescence microscopy. In normal pancreatic tissue from mice and rats, GLP-1R mRNA was only detectable in the β cells. Double and triple immunofluorescence using two different GLP-1R antisera and combinations of insulin, glucagon, pancreatic polypeptide, and somatostatin showed that GLP-1R protein is almost exclusively colocalized with insulin. The same pattern was observed in human pancreas, but the GLP-1R expression was more heterogeneous, with populations of insulin immunoreactive cells with high and low expression. This is the first time that the GLP-1R has been localized in human islets. Furthermore, GLP-1R immunoreactivity was found in the pancreatic ducts in mouse, rat, and human pancreas. As an important confirmation of the specificity of our methods, we found no signals for GLP-1R mRNA or protein in pancreatic tissue from gene-targeted GLP-1R—deficient mice. In conclusion, our data suggest that the GLP-1 receptor is restricted to the pancreatic β cells and the lack of receptor immunoreactivity on δ cells cannot be explained suitably to correspond with published in vivo and in vitro data. This manuscript contains online supplemental material at http://www.jhc.org. Please visit this article online to view these materials.
T
GLP-1 acts through a G protein—coupled receptor (GLP-1R) (Thorens 1992), which is expressed in several tissues including the pancreatic islets (Bullock et al. 1996). Using radioligand binding assays, GLP-1Rs have been identified on multiple lines of pancreatic β cells and a somatostatin cell line (Goke and Conlon 1988; Fehmann and Habener 1991). Autoradiography studies have shown that specific binding sites for GLP-1 could be observed in tissue (Orskov and Poulsen 1991) and in vitro in islets on α, β, and δ cells (Heller and Aponte 1995). Previous studies have addressed GLP-1R expression in vitro in isolated pancreatic islets and shown the presence of GLP-1R in insulin-producing β cells, glucagon-producing α cells, and somatostatin-producing δ cells (Heller et al. 1997). Moens et al. (1996) have shown GLP-1R mRNA expression in β cells but not in α or δ cells. In isolated pancreatic islets, GLP-1 exerts rapid effects to stimulate insulin and somatostatin while inhibiting glucagon secretion (D'Alessio et al. 1989; Heller and Aponte 1995). Using isolated single islet cells and electrophysiological techniques, direct effects of GLP-1 have been shown on purified α and β cells but not δ cells (because of the lack of methods to isolate purified δ cells) (Holz et al. 1993; Gromada et al. 1998; Bode et al. 1999; Suga et al. 2000).
The aim of this study was to determine the localization of GLP-1R mRNA and the GLP-1R protein in normal rat, mouse, and human pancreatic islets using radioactive ISH and immunofluorescence with specific antisera directed against rat GLP-1R or against the human GLP-1R in combination with the main pancreatic hormones in tissue sections. In most previous studies using antiserum, the receptor localization was determined on single cells or purified cells using RT-PCR and rarely in rapidly fixed tissue. Our results suggest that tissue fixation and/or receptor confirmations might explain the difference in the ability to detect GLP-1R on different cell types in the islet under various conditions.
Materials and Methods
Animals
The animals used in these studies were as follows: female CD1 wild-type (wt) mice, 30 weeks (n = 3); female CD1 GLP-1R—deficient mice (30 weeks; n = 3) (Scrocchi et al. 1996); male Sprague-Dawley rats (7–8 weeks; n = 10); and adult human pancreatic specimens (archival material, n = 3). Animal maintenance, handling, and sacrifice were carried out according to regulations specified under the Protection of Animals Act by the Authority in Denmark and Novo Nordisk A/S.
Tissue Preparation and Sectioning
Mice and rat pancreatic specimens were immersion fixed in 4% (w/v) paraformaldehyde in 0.1 M phosphate buffer, pH 7.4, for 24 hr at 4C, dehydrated in increasing concentrations of alcohol, cleared in Estisol (Esti Chem A/S; Køge, Denmark), and embedded in paraffin in a Hypercenter 3000 tissue preparation machine (Leica Meyer Instruments; Houston, TX). Sections (4 μm) were cut on a Leica RM 2165 microtome (Leica; Copenhagen, Denmark). Human pancreatic tissue was fixed either in formalin or Lilly fixative and embedded in paraffin.
Antibodies
Antisera were used at the following dilutions: rabbit anti-rat GLP-1R (Heller et al. 1996), 1:8000; rabbit anti-human GLP-1R, 1:3000 (LS-A1206; Nordic Biosite AB, Taby, Sweden); guinea pig anti-human insulin (651041; ICN Life Science, Holte, Denmark), 1:50 or 1:1000; mouse anti-human glucagon (Glu 001, IgG1; Novo Nordisk A/S, Bagsværd, Denmark), 10 or 40 μg/ml; guinea pig anti-human glucagon 1:500 (4031–01F; Linco, Copenhagen, Denmark); mouse anti-human somatostatin (Som 018, IgG1; Novo), 10 or 40 μg/ml; sheep anti-human somatostatin, 1:500 (Greg Aponte; UC Berkeley, Berkeley, CA); rabbit anti-human pancreatic polypeptide (PP) (A0619; DakoCytomation, Glostrup, Denmark), 1:80; monoclonal mouse IgG1 isotype specific control (X0931; DakoCytomation), 10 or 40 μg/ml; FITC-conjugated rabbit anti-guinea pig (F233; DakoCytomation), 1:40; aminomethylcoumarin acetate (AMCA)-conjugated goat anti-mouse (115-155-146; Jackson ImmunoResearch, TriChem ApS, Skanderborg, Denmark), 1:100; AMCA-conjugated goat anti-rabbit (111-155-144; Jackson Immuno-Research), 1:100; Cy2- and Cy5-conjugated donkey anti-mouse, guinea pig (706-225-148, 706-175-148; Jackson ImmunoResearch) or sheep (713-225-147, 713-175-147; Jackson ImmunoResearch), 1:2000; biotin-conjugated goat anti-rabbit (115-065-144; Jackson ImmunoResearch), 1:2000; Vectastain ABC complex (PK6100; Vector, VWR International, Rødovre, Denmark); biotin-conjugated TSA (700; NEL, PerkinElmer Danmark A/S, Hvidovre, Denmark), 1:50; Texas red—conjugated streptavidin (RPN 1233; Amersham, Hillerød, Denmark), 1:100. Blocking sera were rat (X0912; DakoCytomation); rabbit (X0902; DakoCytomation); goat (X907; DakoCytomation); donkey (D-9663; Sigma-Aldrich, Brøndby, Denmark); and BSA (8577; Sigma-Aldrich).
ISH for GLP-1R mRNA in Mice and Rats
Rat GLP-1R (ACC S75952) cDNA fragment (952–1285 bp) was labeled with 35S, and ISH was performed on paraffin sections as previously described (Kristensen et al. 1991; Willesen et al. 1999). Mouse and rat pancreatic specimens were further subjected to double staining for insulin, glucagon, and somatostatin. After ISH for GLP-1R mRNA, the sections were preblocked in Tris-buffered saline (TBS) containing either 10% rabbit or goat sera and incubated in guinea pig antiinsulin, mouse anti-glucagon, or mouse anti-somatostatin diluted in TBS containing either 7% rabbit and 3% rat sera or 7% goat and 3% rat sera for 1 hr at room temperature. The sections were incubated in either FITC-conjugated rabbit anti-guinea pig or biotin-conjugated donkey anti-mouse for 30 min, followed by Texas red—conjugated streptavidin for an additional 30 min. Sections were dehydrated in increasing concentrations of alcohol, cleared in 3M ammonium acetate, air dried, immersed in autoradiographic emulsion, and exposed for 4 weeks. The sections were counterstained with hematoxylin and mounted with Mounting Medium Fluorescence (MMF; DakoCytomation).
Immunofluorescence Staining for GLP-1R Protein in Mice, Rats, and Humans
Unless otherwise mentioned, all steps were performed at room temperature. Sections were deparaffinized in xylene and rehydrated in decreasing concentrations of alcohols. Antigen retrieval was performed with 10 mM Tris (Sigma-Aldrich) buffer containing 1 mM EDTA (Merck; Glostrup, Denmark), pH 9.0, preheated to 90C for 3 × 5 min at 90% effect in a microwave oven (Polar Patent; Umeå, Sweden). Sections were cooled 10 min in the buffer, and endogenous peroxidase activity was blocked with 0.5% (v/v) H2O2 in 0.05 M TBS, pH 7.6, for 20 min. To block endogenous biotin binding sites, sections were incubated with avidin blocking solution (DakoCytomation) for 10 min, washed in TBS, and incubated with the biotin blocking solution (Dako-Cytomation) for an additional 10 min. After preincubation with TBS containing 10% (v/v) goat serum for 1 hr, the sections were incubated with rabbit anti-GLP-1R in TBS containing 7% (v/v) goat serum and 3% (v/v) species specific serum overnight at 4C. After washing, the sections were incubated with biotinylated goat anti-rabbit Ig in TBS containing 7% (v/v) goat serum and 3% (v/v) species specific serum (DakoCytomation) for 1 hr. Sections were rinsed with TBS and incubated with Vectastain ABComplex for 30 min (Vector; Burlingame, CA), followed by preblocking in 0.1 M Tris buffer containing 0.15 M NaCl and 0.5% (w/v) Du Pont Blocking Reagent and biotinylated tyramide (TSA indirect; NEL 700) diluted 1:50 in amplification buffer (NEN; PerkinElmer Life Sciences, Boston, MA). Sections were incubated with Texas red—conjugated streptavidin and mounted with mounting medium fluorescence (MMF; DakoCytomation). Detergent (Triton X-100) was omitted from preblocking, all antibody incubations, and washing buffers because this dramatically reduces GLP-1R immunoreactivity.
Controls included conventional immunohistochemical staining controls and preabsorption of rabbit anti-rat GLP-1R antiserum with 100 μg/ml of a keyhole limpet hemocyanin (KLH)-conjugated synthetic peptide, identical to the one used to produce the antiserum (Heller et al. 1996), overnight at 4C. No preabsorption was performed on the commercial rabbit anti-human GLP-1R antiserum.
Triple Immunofluorescence Staining for GLP-1R Protein, Insulin, and Glucagon, Somatostatin, or Pancreatic Polypeptide in Mice, Rats, and Humans
GLP-1R immunostaining was performed as described above, followed by preblocking with TBS containing 0.1% (v/v) Triton X-100 (TBS-T) and 10% (v/v) goat serum, for 1 hr. Sections were incubated with guinea pig anti-human insulin, overnight at 4C, followed by FITC conjugated rabbit anti-guinea pig in TBS-T containing 7% (v/v) goat serum and/or 3% (v/v) species-specific serum/BSA for 30 min. After thorough washing with TBS-T, the sections were subjected to a final pre-blocking in goat serum and incubated with either monoclonal mouse anti-glucagon, mouse anti-somatostatin, or rabbit anti-PP diluted in TBS-T containing goat serum and/or species-specific serum/BSA as described above, for 1 hr Finally, sections were incubated with AMCA-conjugated: goat anti-mouse or anti-rabbit in TBS-T containing 7% goat serum and/or 3% species specific serum/BSA, for 30 min, rinsed with TBS and mounted with mounting medium fluorescence (MMF; DakoCytomation).
Controls included (1) conventional immunohistochemical staining controls, (2) specific and cross-preabsorption of rabbit anti-rat GLP-1R antiserum, and guinea pig anti-insulin, with either 100 μg/ml KLH-conjugated synthetic GLP-1R peptide, as described above, or 100 μg/ml human insulin (Novo Nordisk), overnight at 4C, and (3) a mouse monoclonal IgG1-isotype specific control. No preabsorption was performed on the commercial rabbit anti-human GLP-1R antiserum.
All sections were photographed using an Olympus BX51 reflected fluorescence system microscope (Olympus Denmark A/S; Ballerup, Denmark) equipped with darkfield, selective AMCA, FITC, and Texas red filters and a DP50 digital camera. Images presented in Figure 4 (and Supplemental Figures S1 and S2), were taken on a Zeiss AxioImager Meta 510 confocal microscope (Carl Zeiss; Oberkochen, Germany).
Results
Lack of GLP-1R mRNA and Immunoreactivity in Pancreatic Islets From GLP-1R—deficient Mice
To confirm the specificity of our methods, we performed radioactive ISH and immunohistochemical studies of the GLP-1R on pancreatic tissue from normal CD1 mice and gene-targeted GLP-1R—deficient CD1 mice (Figure 1). Demonstration of GLP-1R protein was performed using a highly sensitive immunohistochemical protocol consisting of heat-induced antigen retrieval, lack of detergent in dilution buffers, and tyramide signal amplification (TSA). GLP-1R mRNA could clearly be shown in pancreatic islets from wildtype mice (Figures 1A and 1C, black and white grains, respectively), whereas no mRNA signal was found in pancreatic tissue from GLP-1R—deficient mice (Figures 1B and 1D, lack of grains over the islets). Also, GLP-1R immunoreactivity was clearly shown in pancreatic islets from wild-type mice, whereas pancreatic islets from GLP-1R—deficient mice were totally devoid of any GLP-1R immunoreactivity (Figures 1E and 1F, respectively). Control ISH with the sense probe for GLP-1R mRNA failed to give a signal and preabsorption of the GLP-1R antiserum with a KLH-conjugated peptide identical to the one originally used to produce the GLP-1R antiserum totally abolished the GLP-1R immuno-reactivity (data not shown).
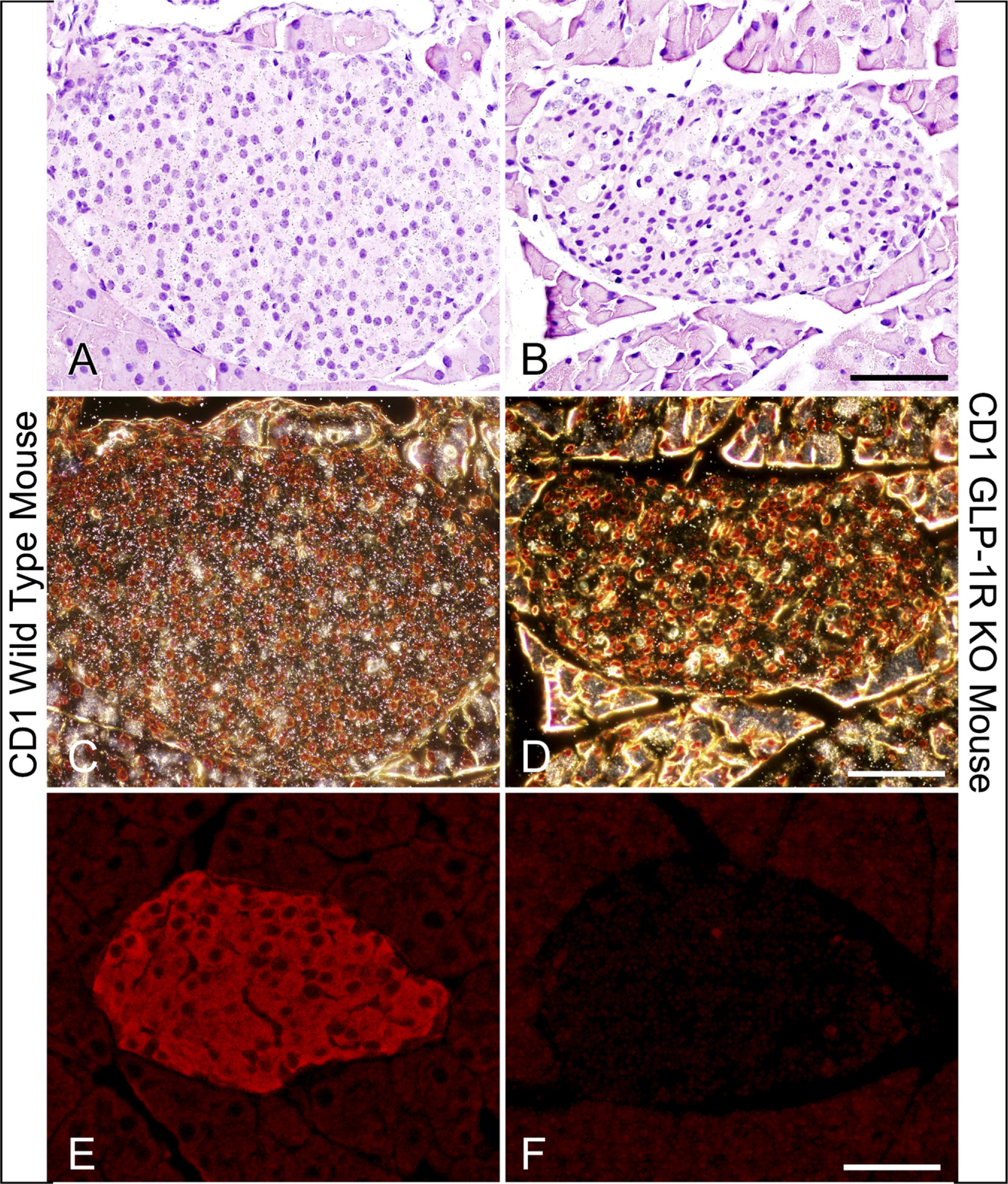
Glucagon-like peptide 1 receptor (GLP-1R) mRNA and GLP-1R immunoreactivity are not detected in pancreatic islets from GLP-1R—deficient mice. GLP-1R mRNA and protein expression in islets from wild-type (
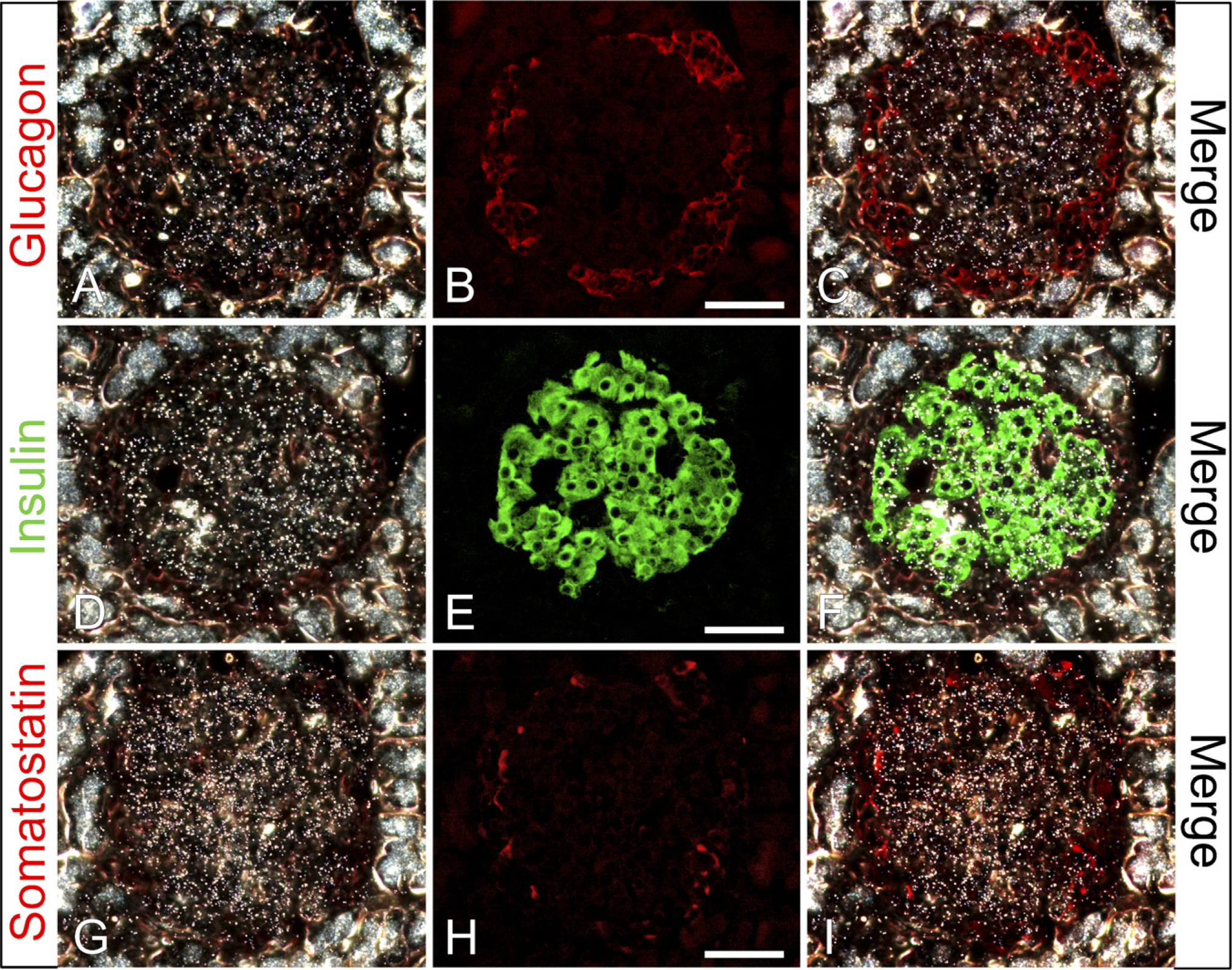
Colocalization of GLP-1R mRNA and insulin but no colocalization with glucagon and somatostatin in normal rat islets. Using radioactive ISH for the GLP-1R mRNA, seen as white grains in the center of the islet (
Colocalization of the GLP-1R mRNA Is Restricted to β Cells in Normal Rat Islets
We studied the intra-islet localization of the GLP-1R by colocalization studies for GLP-1R mRNA using radioactive ISH combined with immunofluorescence for islet cell hormones (Figure 2). GLP-1R mRNA was strongly detectable in the β cells (Figures 2A and 2D–2G, white grains). Grains from GLP-1R mRNA ISH signal were rarely observed to colocalize at a higher density than background on the glucagon- or somatostatin-producing α and δ cells (Figures 2A–2C and 2G–2I, respectively), even after prolonged exposure. Control ISH with the sense probe for the GLP-1R mRNA gave no signal above background levels (results not shown).
Colocalization of the GLP-1R Protein Restricted to β Cells in Pancreatic Islets
The intra-islet localization of the GLP-1R was further studied by triple immunofluorescence using the rabbit anti-rat GLP-1R antiserum (Heller et al. 1996, 1997) with combinations of insulin, glucagon, somatostatin, and pancreatic polypeptide in the rat pancreas (Figure 3). The GLP-1R protein was found exclusively to be colocalized with insulin (Figures 3A, 3B, 3D–3F, and 3H) because no colocalization was found between GLP-1R and glucagon (Figures 3A, 3C, and 3D), somatostatin (Figures 3E, 3G, and 3H), or pancreatic polypeptide (results not shown). The sole exception from this was endocrine cells double immunopositive for insulin and glucagon, which could be found also to be immunoreactive for the GLP-1R (Figures 3A–3D).
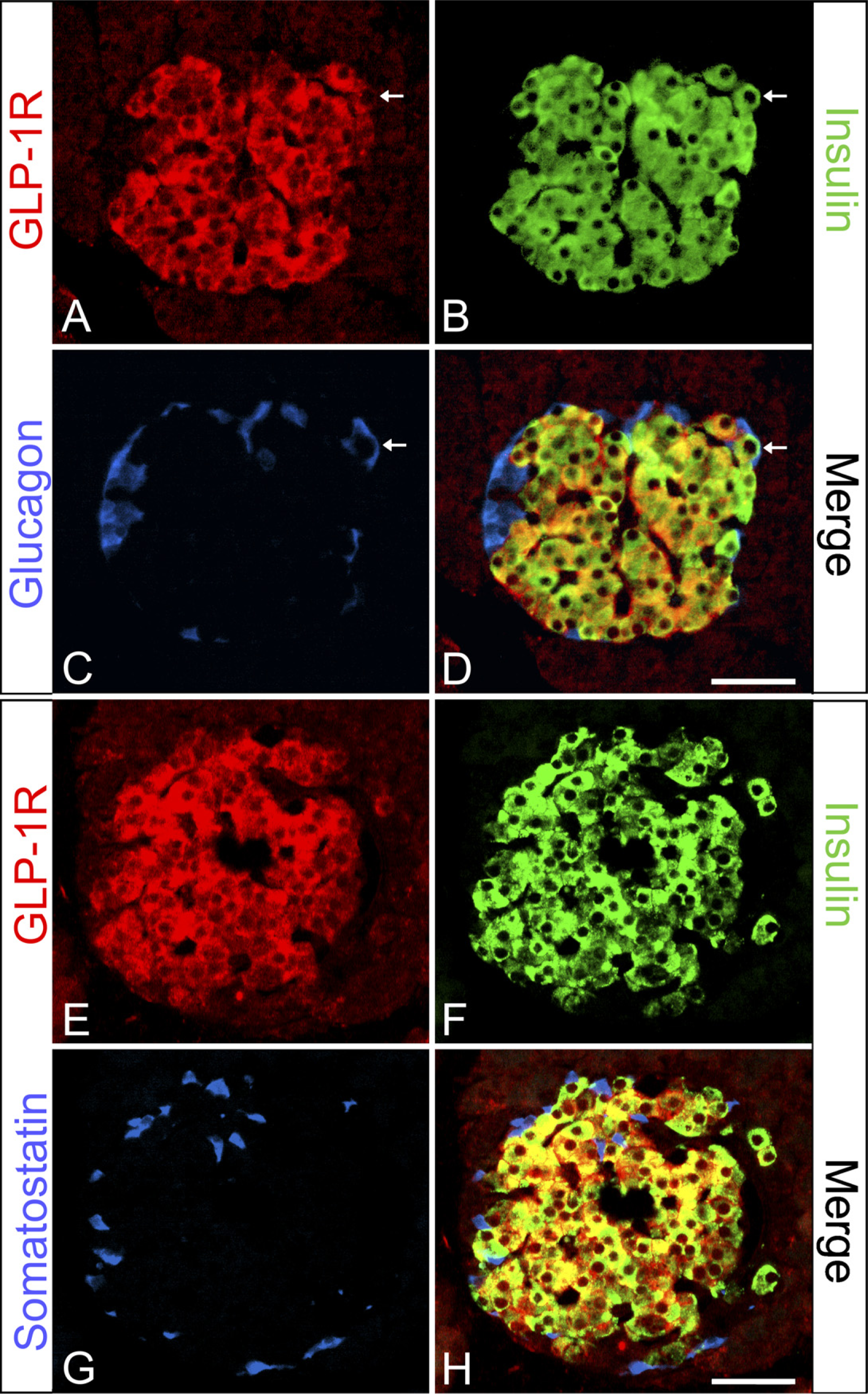
Colocalization of the GLP-1R protein with insulin but not with glucagon or somatostatin in normal rat islets. Triple immunofluorescence for GLP-1R (
As the localization of the GLP-1R using immunohistochemistry in human pancreas has not been previously shown, we made an in-depth analysis (Figure 4). In contrast to what we observed in rats, the GLP-1R immunoreactive cells varied greatly. A subpopulation of the β cells were highly immunoreactive, whereas the great majority showed lower levels of expression (Figures 4A, 4B, and 4D), and there was no colocalization of the GLP-1R and glucagon (Figures 4A, 4C–4F, and 4H) or somatostatin (Figures 4E, 4G, and 4H). Controls consisted of cross-absorption of the GLP-1R and insulin antibodies with either the KLH-conjugated GLP-1R peptide or human insulin. Preabsorption of the GLP-1R antiserum with the GLP-1R peptide and the insulin antibody with human insulin totally abolished the GLP-1R and insulin immunoreactivity, respectively, whereas cross-absorption of the GLP-1R antiserum with insulin and the insulin antibody with the GLP-1R peptide had no effect on either immunoreaction (data not shown). The isotype specific control antibody showed no unspecific staining reactivity. These findings on the intra-islet localization of the GLP-1R protein in the rat and human pancreas were confirmed with the commercial antihuman GLP-1R antiserum, although some cells in the mouse but not in the rat seemed to show colocalization of somatostatin and the GLP-1R (Supplemental Figures S1 and S2).
GLP-1R Is Expressed in the Large Pancreatic Ducts
Although GLP-1 has been shown to have multiple effects on pancreatic ducts in vitro (Hui et al. 2001; Bulotta et al. 2002), only one study has shown GLP-1R immunoreactivity (Xu et al. 1999). Therefore, in the rat, mouse, and human pancreas, the GLP-1R localization was examined. In the large ducts, the GLP-1R immunoreactivity was the strongest, and the receptor was observed on the membrane (Figure 5). Not all of the ducts in the pancreas had strong GLP-1R immunoreactivity, and some were stained quite weakly or not at all (data not shown). All controls were negative.
Discussion
In this study, we showed that GLP-1R mRNA and protein are highly expressed in the pancreatic β cells and that the receptor is rarely observed or below the level of detection using our very sensitive techniques in α and δ cells. In addition, GLP-1R immunoreactivity is found in the large pancreatic ducts in human, rat, and mouse pancreas. GLP-1 has been shown to regulate insulin, glucagon, and somatostatin secretion in vivo and in vitro (Fehmann and Habener 1991; Bulotta et al. 2002). Based on several receptor localization studies, many have assumed that these effects were direct (Goke and Conlon 1988; Orskov and Poulsen 1991).
GLP-1 has rapid and potent effects to regulate insulin secretion in vivo and in vitro. Not only does GLP-1 stimulate glucose-dependent insulin secretion, it also increases insulin biosynthesis, decreases the proinsulin/insulin ratio, stimulates proliferation and decreases apoptosis, and increases glucokinase and the glucose transporter GLUT4 mRNA levels (Holst 2007). All these beneficial effects on β cells, together with other effects (on glucagon, satiety), make GLP-1 analogs very interesting drug candidates for the treatment of type 2 diabetes (Zander et al. 2002; Drucker and Nauck 2006) and potentially for obesity (Raun et al. 2007). Exenatide is a relatively short-acting analog with 53% homology to native GLP-1, marketed as a twice daily injection regimen (Barnett 2007). Liraglutide is a once-daily human analog with 97% homology to native GLP-1 (Vilsbøll 2007). Both analogs have been shown to directly bind to receptors on β cells.
We and others have shown that GLP-1 receptor mRNA and protein are highly expressed in β cells. This point is not controversial. Interestingly, we observed that the GLP-1R was most strongly expressed on the surface of the β cells facing the endothelium in all species studied. This pattern with highest expression near the endothelium could be part of the mechanism behind the physiological action of GLP-1. GLP-1 is released from the intestinal L cells but is rapidly degraded by the DPP-IV enzyme, which is found in the capillaries right outside the L cells (Hansen et al. 1999). Although the main physiological effect of GLP-1 seems to be gastric emptying, GLP-1 is also believed to have an effect on insulin secretion physiologically. Because of the GLP-1 degradation and inactivation by DPP-IV, only very little GLP-1 may reach the pancreas, and it therefore would make sense that there is a higher level of expression of the GLP-1 receptor in the β cells that face the endothelium.
It has been observed in animal studies and humans that the administration of GLP-1 decreases glucagon secretion. Elevated levels of glucagon may play a patho-physiological role in type 2 diabetes and a decrease mediated by GLP-1 could be beneficial and work together with the potential effect of retaining β-cell mass (de Koning et al. 2008). Two studies using direct binding of 125I-GLP-1 have found small numbers of sites on α cells (Orskov and Poulsen 1991; Heller and Aponte 1995), suggesting a direct action of GLP-1 on the α cells. Also, a direct agonistic action of GLP-1 on purified α cells was observed in the form of stimulated secretion as opposed to what would have been expected (Ding et al. 1997). In our study, we were unable to identify GLP-1R mRNA in α cells using a sensitive ISH protocol. Although in a previous study using the same antiserum, Heller et al. (1997) observed immunoreactive GLP-1Rs on 20% of α cells; however, we could not confirm this result using freshly fixed paraffin-embedded pancreatic tissue. The difference observed between the two studies could be caused by the use of in vitro islets in the former study and the use of the freshly isolated whole pancreatic organ in this study. One explanation could be that the receptor on isolated trypsin-treated single cells could be in such a conformation that the receptor antiserum can bind to it. Demasking of antigenic sites with trypsin pretreatment of the sections was not performed; instead, we used a high temperature antigen retrieval (HTAR) method that has been shown to restore many antigens that cannot be unmasked by enzymatic digestion (Shi et al. 2001). Thus, we used an extremely sensitive immunohistochemical method, more sensitive than previous methods used for detecting GLP-1R, yet we observed no receptor immunoreactivity on either α cells or δ cells in the pancreatic islets in vivo. These differences remain unexplained.
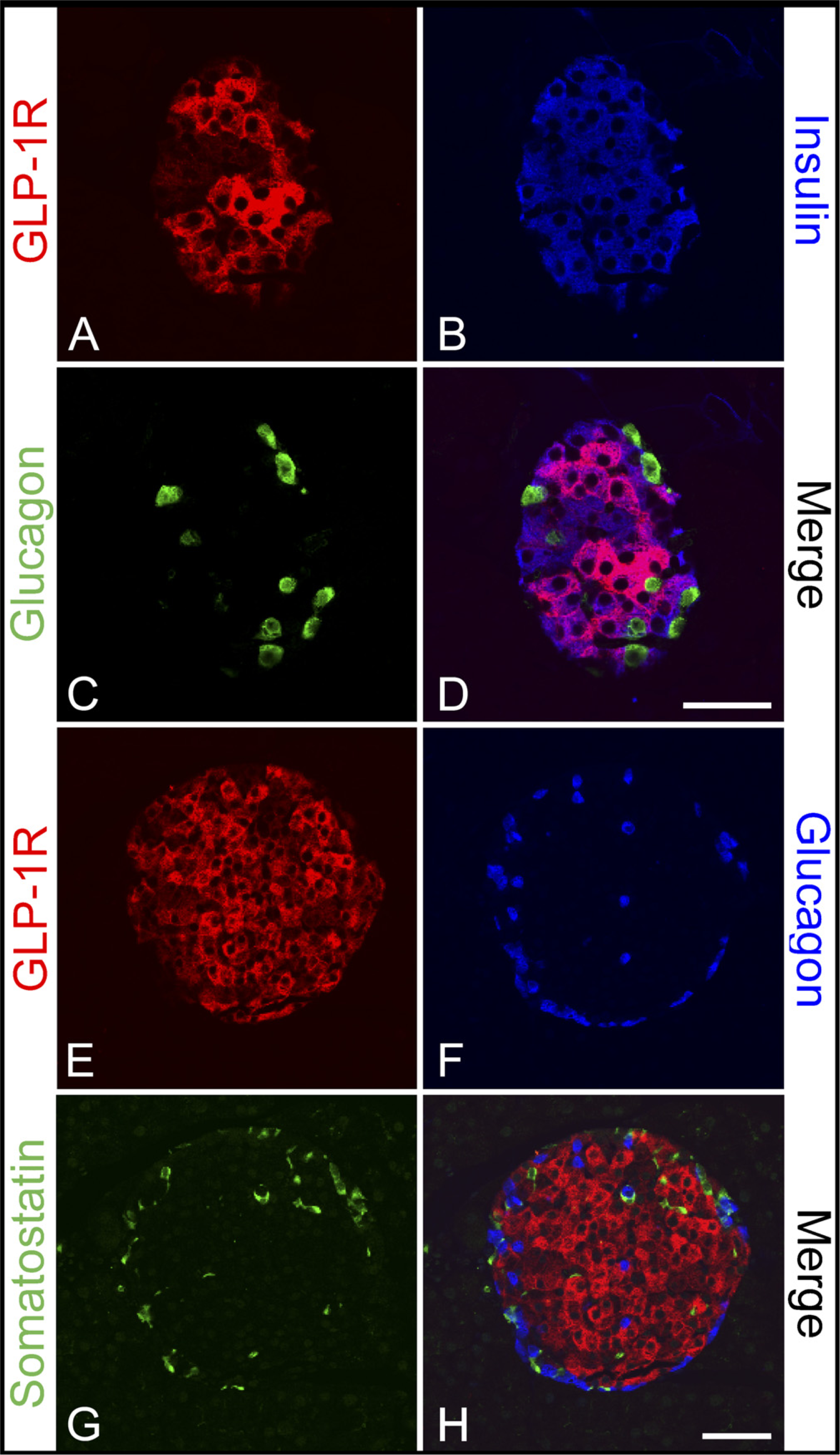
Colocalization of the GLP-1R protein with insulin but not with glucagon or somatostatin in normal human islets. Triple immunofluorescence for the GLP-1R (
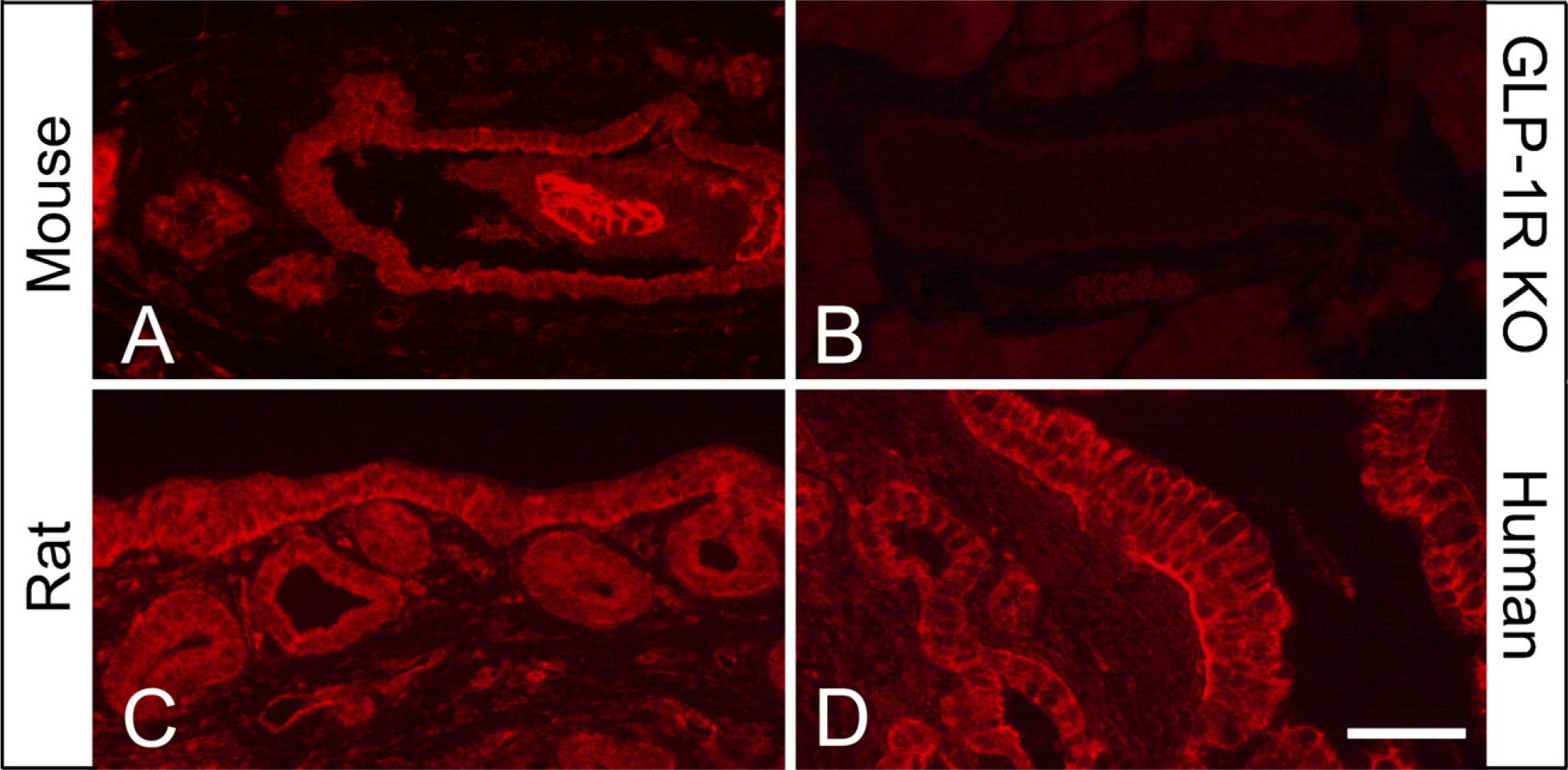
GLP-1R immunoreactivity is found in the large pancreatic ducts. Immunofluorescent staining for the GLP-1R (
GLP-1 has potent in vivo and in vitro effects on somatostatin secretion. Evidence for a direct effect on somatostatin has been provided in both receptor binding studies and secretions studies (D'Alessio et al. 1989; Heller and Aponte 1995). In this study, we did observe GLP-1Rs on a few δ cells in the mouse with one antiserum. This is puzzling as receptor binding on cell lines (Fehmann and Habener 1991) or in vivo injection of 125GLP-1, colocalization of GLP-1, and somatostatin immunoreactivity has been reported (Orskov and Poulsen 1991; Heller and Aponte 1995). Using the same antiserum, it was observed that 80% of the δ cells had GLP-1R immunoreactivity (Heller et al. 1997). In support of the present data, using Western blotting and PCR methods, Moens et al. (1996) failed to detect GLP-1R protein or mRNA in α and δ cells fractions from FACS sorted islets. Moreover, our data support previous in vivo findings from Hörsch et al. (1997) who reported the presence of GLP-1R mRNA in β cells but not in α cells. They furthermore found it most unlikely that δ cells expresses physiological levels of GLP-1R mRNA. It is, however, difficult to understand the very potent effect of GLP-1 on stimulation of somatostatin release if there is not a GLP-1 receptor on δ cells. If there are no receptors on δ cells, how do we get increased somatostatin secretion with the same kinetics as insulin? One explanation is that insulin itself is directly stimulating somatostatin secretion; however, although glucagon has been shown to stimulate somatostatin, insulin has little or adverse effect (Patton et al. 1977; Chiba et al. 1980; Gerber et al. 1981). One possible explanation could involve the intra-islet organization. It is possible that a signal from the β cells to the δ cells is transmitted through gap junction connections that operate to synchronize β-cell secretion in vivo (Nadal et al. 1999; Kanno et al. 2002). There are very few δ cells in the islets so perhaps these are so sensitive that only a few GLP-1R are required for release, if the effect on somatostatin secretion is direct on the δ cells. In humans, δ cells are situated in the core of the islets (Bermúdez-Silva et al. 2008), whereas in rodents they are situated in the rim of the islets. Finally, another possible explanation is the role of sensory nerves in GLP-1 action. Calcitonin gene-related peptide (CGRP) released from sensory nerves in the stomach directly stimulates somatostatin secretion (Inui et al. 1991). GLP-1 activation of sensory nerves (Balkan and Li 2000), which are directly involved in regulation of insulin secretion (Ahren 2004) could also induce somatostatin secretion, thus GLP-1 action on the δ cells could be explained. Unfortunately, this is not supported in the literature, where stimulation induces inhibition of somatostatin secretion (Holst et al. 1983). The small number of δ cells and the species differences in the intra-islet organization of the individual cells makes it difficult to come to full understanding on how GLP-1 stimulates somatostatin release if the receptor is not present.
Using a specific GLP-1R antiserum, we showed that high levels of immunoreactivity are observed in the large ducts in human, rat, and mouse pancreas. It is currently thought that the duct cells could provide a source of islets through their capacity to differentiate into pancreatic β cells under certain experimental conditions (Bonner-Weir et al. 2000; Xu et al. 2008). In further support of direct effects of GLP-1 on ducts, GLP-1 has been shown to induce a rat ductal-like cell line to differentiate in vitro (Hui et al. 2001; Bulotta et al. 2002). In addition, treatment of mice with the GLP-1 analog, exendin-4 induces direct effects on the ducts in vivo (Stoffers et al. 2000), and this effect is not observed in GLP-1R—deficient mice. A recent report has also shown GLP-1R immunoreactivity in rat and human ducts (Xu et al. 2006). Thus, GLP-1 receptors on the pancreatic ducts may play an important role in the formation of endocrine cells in adult murine models and in humans. In humans, it is the belief that only neogenesis and anti-apoptosis, and not proliferation, may contribute to increasing β-cells mass (Butler et al. 2007). In rodents, all three processes are believed to be able to contribute to increase β-cell mass (Hanley et al. 2008). GLP-1 receptor expression in human ducts could potentially be very important in the treatment of type 2 diabetes with GLP-1 analogs. Such an increased mass should, together with direct effects on β cells, help stabilize β-cell function because decreasing β-cell function is one of the two major pathophysiological problems in type 2 diabetes, the other being insulin resistance (Turner et al. 1999).
In conclusion, our data suggest that, in pancreatic tissue sections from mice, rats, and humans, GLP-1 receptor immunoreactivity is restricted to endocrine β cells. The lack of receptor immunoreactivity on δ cells cannot be explained sufficiently to correspond with published in vivo and in vitro data, leaving room for the possibility that few δ cells express the receptor or that the density of receptors on δ cells is too small to be detected. Also, our data show expression of the GLP-1 receptor in mouse, rat, and human duct tissue, underlining a potential importance of neogenesis of β cells from duct cells in humans.
Footnotes
Acknowledgements
We thank Birte Jørgensen, Jette Mandelbaum, and Steen Kryger for superb technical assistance and Ole D. Madsen, Jens Juul Holst, and Charles Pyke for critical reading of this manuscript.
