Abstract
Prolonged hyperoxia, as may be used to treat patients with severe hypoxemia, can lead to lung injury, respiratory failure, and death. Resident mast cells play important roles in regulating the lung response to changing environmental conditions, as evidenced by their roles in asthma and airway hyperresponsiveness. In this study we evaluated the effect of prolonged hyperoxia on the number and distribution of mast cells in the rat lung. In rats maintained in normoxia, mast cells were distributed primarily in the loose connective tissue surrounding large bronchioles and vessels of the lung. In rats exposed to normobaric hyperoxia for 72 hr, mast cell number in lung sections increased significantly, and mast cells were found preferentially accumulated around vessels throughout the lung. Notably, mast cells around smaller vessels were abundant in hyperoxic lungs but rare in normoxic lungs. Also, mast cells were increased in the pleura of lungs exposed to hyperoxia. These changes in mast cell number and distribution in response to hyperoxia were evident in aged (22-month-old) rats as well as young (3-month-old) rats. As mast cell-derived mediators have many effects, e.g., on vascular leak and vascular tone, positioning of increased mast cell numbers throughout the lung vasculature may be an important contributor to changes in lung function subsequent to persistent hyperoxia.
P
One consequence of prolonged exposure to hyperoxic conditions involves lung remodeling. For example, chronic hyperoxic exposure leads to thickening of the airway epithelial and smooth muscle layers (Hershenson et al. 1992; Burghardt et al. 1996). In addition, chronic hyperoxia affects the pulmonary vascular bed, leading to remodeling of the distal pulmonary veins via increased smooth muscle number and thickness (Hu and Jones 1989). In addition to remodeling of the airway and microvasculature, prolonged hyperoxia causes airway hyperreactivity (Burghardt et al. 1996; Denis et al. 2001) and pulmonary hypertension (Gore and Jones 1989). These structural and physiological changes suggest that persistent hyperoxia may cause fundamental changes in regulatory signaling within the lung.
Mast cells are resident immune cells that typically can be found in the larger airways of the lung, predominantly in the lamina propria of the bronchi and the supporting tissue of the bronchioles. Upon activation, mast cells release a variety of mediators including leukotrienes, prostaglandins, histamine, serine proteases, and cytokines. Mast cells are known to contribute to airway hyperreactivity (Page et al. 2001), edema (White 1990), and lung inflammation (Maezawa et al. 2003). In addition, airway smooth muscle proliferation is strongly promoted by tryptase (Brown et al. 1995), a secretory granular protein in mast cells (Caughey 1997). Mast cells have also been shown to increase in the trachea of neonatal rats exposed to prolonged moderate hyperoxia (Denis et al. 2001). Given that mast cells can be associated with many of the features resulting from prolonged hyperoxia, we investigated if increased numbers of mast cells could be found in adult rats in sites that would support their role in the response of lungs to hyperoxia.
Materials and Methods
Animals and Treatments
Following previously published protocols (Di Giulio et al. 1998), experiments were performed on male Wistar rats, age 3 months (young) or 22 months (aged), maintained in specific pathogen-free conditions. One set of each age group, five animals per age, was exposed for 72 hr to normobaric hyperoxia 98-100% O2 (760 torr) for 3 days in a large Plexiglas chamber with continuous monitoring by oxymeter. The chamber air was recirculated with a pump: CO2 was removed from the chamber air with baralyme and was continuously monitored with a capnograph. Boric acid was mixed with the litter to minimize emission of urinary ammonia. Temperature was maintained at 25 ± 2C. Additional sets of each age group were maintained similarly, breathing air in the same room (21% oxygen; 156 torr of oxygen). Under these conditions, 12% of young rats and 7% of aged rats died of pulmonary edema during the 72 hr of hyperoxia, whereas none of the rats, young or old, died of pulmonary edema when maintained in room air. Surviving rats were anesthetized IP with 30 mg/kg Nembutal (Abbott Laboratories; Chicago, IL), animals were perfused with saline solution, and the organs were dissected, washed, and fixed in formalin. All experimental protocols were approved by the University of Michigan Committee on the Use and Care of Animals.
Toluidine Blue Staining and Immunohistochemistry
As previously described (Brock et al. 2001), fixed tissues were embedded in paraffin, sectioned, mounted, dewaxed in Americlear (Stephens Scientific, Richard-Allan; Riverdale, NJ) and rehydrated through decreasing concentrations of ethanol. For toluidine blue staining, tissues were stained with 0.25% acidified toluidine blue for 5 sec, washed, and mounted. For immunohistochemistry, sections were quenched of endogenous peroxidase activity by treatment with 0.3% hydrogen peroxide for 30 min, washed, and blocked with Powerblock (InnoGenex; San Ramon, CA). Rabbit polyclonal antibody against 5-LO (a generous gift from Dr. J. Evans; Merck Frosst Center for Therapeutic Research, Pointe Claire-Dorval, Quebec, Canada) was prepared in PBS containing 0.1% BSA (titer 1:750) and applied overnight at 4C. After washing with 0.1% BSA in PBS, slides were probed with secondary antibody (biotinylated goat anti-rabbit, 1:250) for 30 min at 37C, washed again, then treated with avidin-biotinylated peroxidase complex (Vectastain Elite ABC kit; Vector Laboratories, Burlingame, CA) for 30 min atroom temperature. 3,3î-DAB was used as peroxidase substrate, and preparations were counterstained with Harris' hematoxylin.
Quantitative and Statistical Analysis
Lung sections from three young animals maintained in normoxia and three in hyperoxia were stained with toluidine blue. From each section, 10 adjacent microscopic fields measuring 0.254 mm2 through alveolar and bronchiolar tissue but excluding major bronchi and pleura were photographed. Toluidine-positive cells per field and toluidine-positive cells per vessel (defined by the presence of reticulocytes) were counted. Mean values per animal were calculated, and normoxic mean values were compared with hyperoxic mean values using an unpaired Student's
Results
The presence of mast cells in lung tissues was first evaluated using toluidine blue staining, as has been used to quantify mast cell numbers in lungs (Vignola et al. 1998). With this method, mast cells could be identified in the loose connective tissue surrounding large vessels (Figure 1A) and large bronchioles (Figure 1B) of lungs from young (3-month-old) rats maintained in normoxic conditions. In contrast, numerous mast cells were detected around larger vessels in lungs of young rats exposed to hyperoxic conditions, as indicated by meta-chromatic staining with toluidine blue (Figures 1C and 1D). Closer inspection showed few to no mast cells around bronchioles and smaller vessels from lung sections of normoxic rats (Figures 1E and 1F), compared with numerous mast cells lining the vessels in lung sections of hyperoxic rats (Figures 1G and 1H). Quantitatively, lung sections from young rats maintained in normoxia had 13.0 (SE = 1.36) toluidine blue cells per mm2, whereas sections from hyperoxic rats had 24.9 (SE = 2.64) cells per mm2, a statistically significant difference (
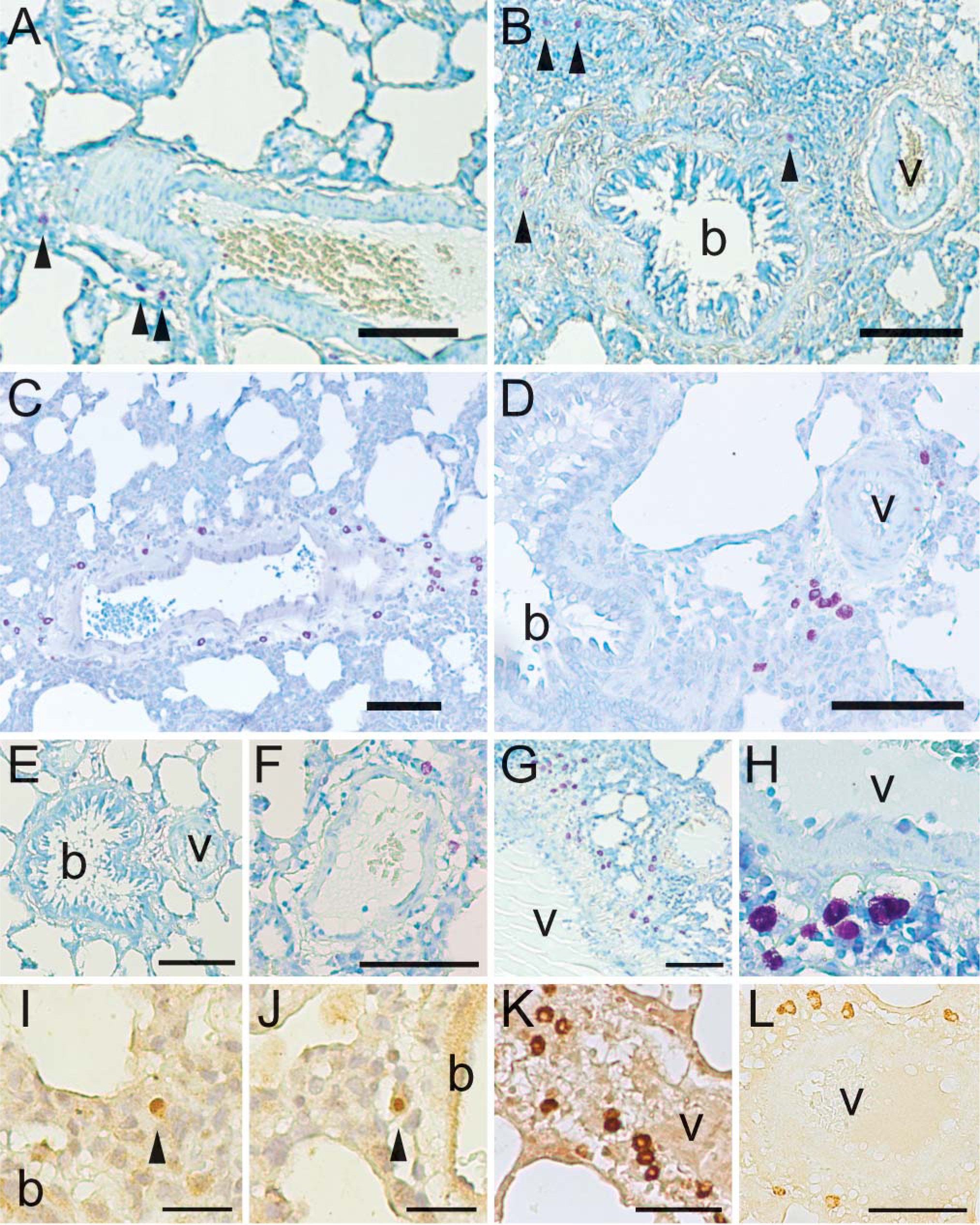
Hyperoxia increases mast cell number around lung vasculature. Young (3-month-old) rats were raised under normoxic (
Edema, or vascular leak, is an important symptom associated with exposure to hyperoxia, and cysteinyl leukotrienes are mediators produced by mast cells that can cause edema. The enzyme 5-lipoxygenase (5-LO) initiates the synthesis of cysteinyl leukotrienes from arachidonic acid, and the subcellular localization of 5-LO can regulate leukotriene synthesis (Luo et al. 2003). Immunohistochemical staining of serial lung sections for 5-LO revealed nuclear 5-LO in mast cells in the loose connective tissue surrounding larger bronchioles of normoxic lungs (Figures 1I and 1J). In hyperoxic lungs, positive staining for 5-LO was also found in mast cells, but the nucleus was conspicuously negative in many cells (Figures 1K and 1L). These results indicated that in many of the mast cells associated with vessels in the lungs of rats exposed to hyperoxia the subcellular distribution of 5-LO was shifted from the nucleus to the cytoplasm.
In addition to the larger vessels, smaller vessels measuring <100 μm in diameter were also scattered across lung sections. In lung sections from young rats maintained under normoxic conditions, zero to two mast cells could be found associated with these smaller vessels (Figures 2A-2C). Positive staining of mast cells along the pleura (Figure 2A) as well as along larger vessels (Figure 1) demonstrated that the staining was effective in these sections. In contrast to the pattern observed in normoxic lungs, multiple mast cells were commonly observed around the smaller vessels of lung sections from young animals exposed to hyperoxia (Figures 2D-2F). Quantitatively, sections from normoxic rats had 0.195 (SE = 0.100) mast cells per vessel, whereas sections from hyperoxic animals averaged 2.88 (SE = 0.532) mast cells per vessel, which was statistically different (
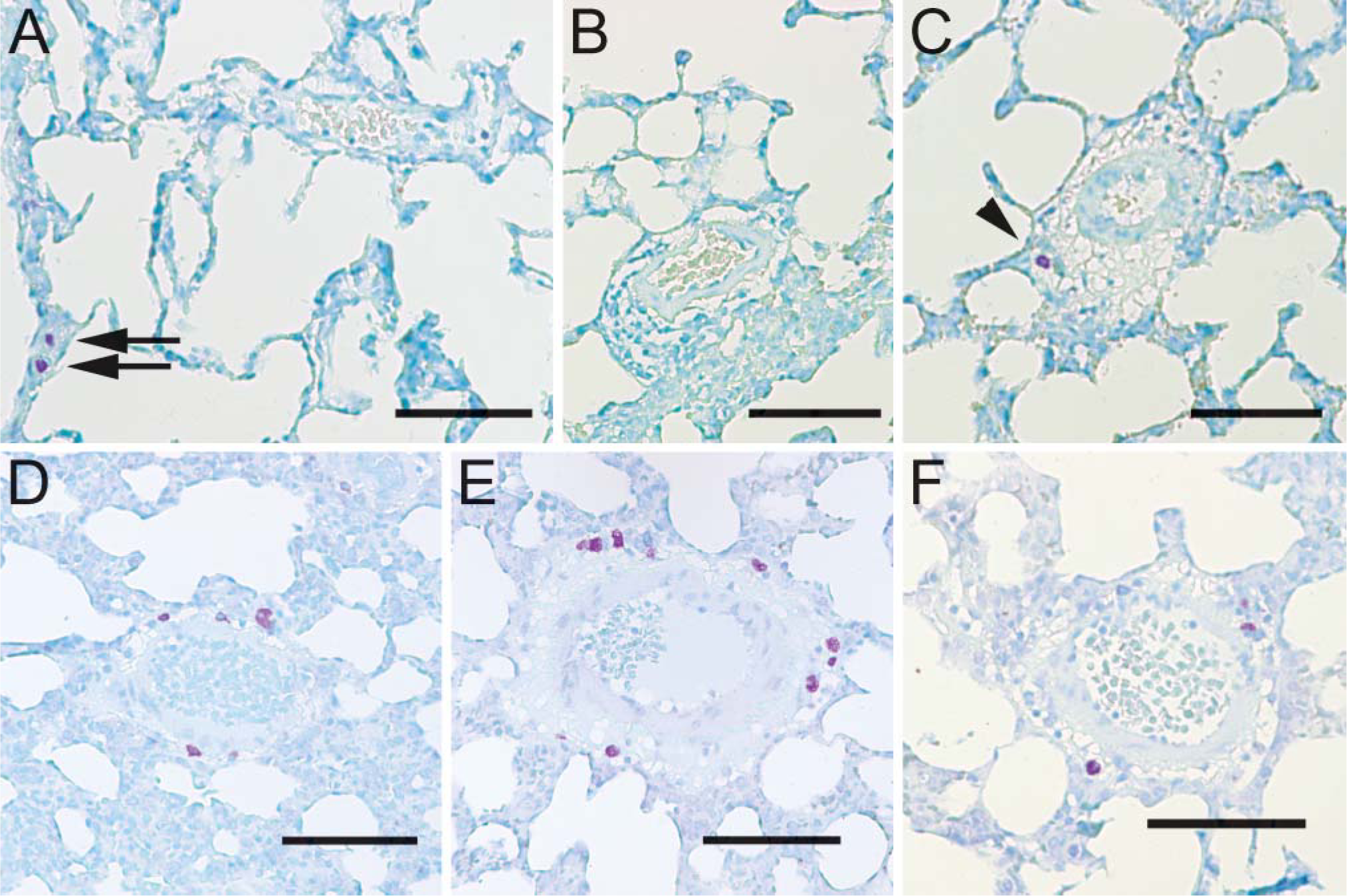
Hyperoxia increases mast cell number around small vessels. In lungs from young rats maintained in normoxia, few mast cells are evident near smaller vessels (arrowhead), although mast cells are detected along the pleura (arrows) (
Mast cells were also evident within the pleura of all lung sections. Mast cells were evident in the pleura of lung sections from young normoxic rats (Figure 3A), with fewer lining the pleura of oblique fissures between adjacent lobes (Figure 3B). The number of mast cells in peripheral pleura of hyperoxic lungs from young rats was increased (Figure 3C). Pleura surfaces of oblique fissures between adjacent lung lobes showed abundant mast cells by staining with either toluidine blue (Figure 3D) or 5-LO (Figure 3E). Surprisingly, many mast cells in the pleura had nuclear 5-LO (Figure 3E, inset), whereas mast cells at vessels had cytoplasmic 5-LO (e.g., Figure 1K). Thus, exposure to hyperoxia led to an increase in mast cell number along the lung pleura as well as around vessels, although redistribution of 5-LO to the cytoplasm, evident near vessels, was not found at the pleura.
It was not clear whether older rats would be more or less affected by exposure to hyperoxia. Examination of lung sections from 22-month-old rats exposed to hyperoxia indicated abundant mast cells around large vessels (Figures 4A and 4B) and smaller vessels (Figures 4C and 4D). As in young animals, many mast cells had cytoplasmic 5-LO, as indicated by the lighter, non-staining cell center (Figure 4D, inset). The number and distribution of mast cells in hyperoxic lungs from older animals were virtually indistinguishable from those of younger animals. Abundant mast cells were also present in the pleura of lungs from old, hyperoxic animals (Figure 4E). Lung sections from 22-month-old rats maintained in room air, like those from young rats, had few masts cells, with their distribution mostly restricted to the largest vessels and bronchioles (not shown). These results indicate that mast cells increase in number, particularly around vessels, in response to hyperoxia in aged animals as well as in young animals.
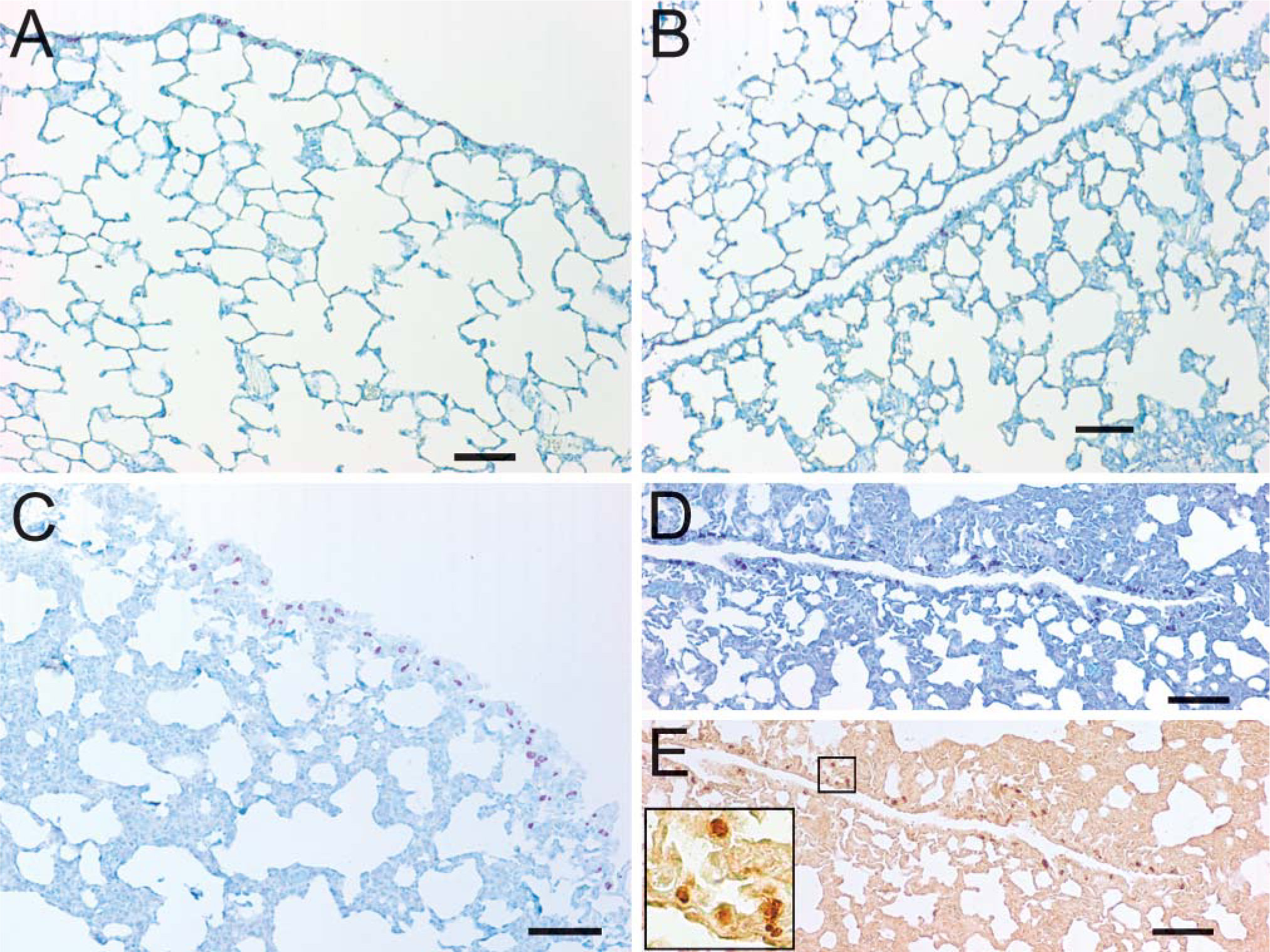
Hyperoxia increases mast cell number in lung pleura. (
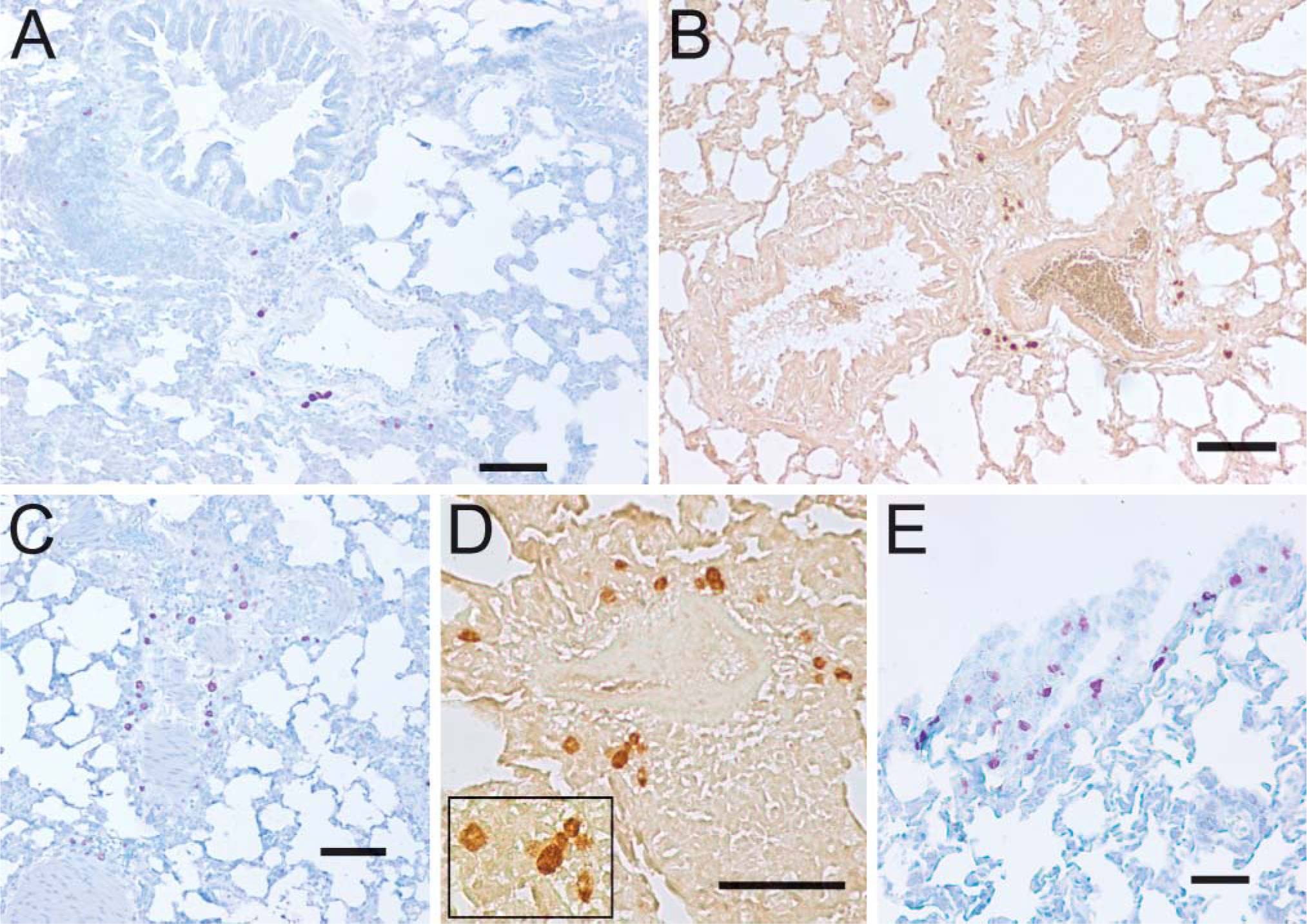
Hyperoxia increases mast cell number in aged rats. Using toluidine blue (
Discussion
Increased inspired oxygen fractions may be used to treat premature infants and patients with acute lung injury or severe hypoxemia. Physiologic manifestations of oxygen toxicity include decreases in vital capacity, diffusing capacity, and lung compliance, with pathologic changes resembling those of the adult respiratory distress syndrome (Jackson 1985). We report here that hyperoxia produces an increase in mast cell number, with accumulation of mast cells around both large and small vessels throughout the lung. Mast cell numbers also increase at the pleura and the fissures between lobes. In short, mast cells increase at the major interfaces between the lung and the body. These changes occurred in both young and aged rats.
The mechanism behind the increase in toluidine-positive cells is unclear. It seems likely that mast cells generated from precursors in the bone marrow during persistent hyperoxia migrated through the vasculature and into the lung, where they matured. However, it is also possible that hyperoxia triggered the differentiation of immature, toluidine-negative mast cells already present in the lung. Finally, it has been reported that hyperoxia induces mast cell hyperplasia (Subramaniam et al. 2003). Immature mast cell progenitors can proliferate within the lung (Abonia et al. 2006), and these may have migrated through the loose connective tissue to their observed positions near vessels. Hyperplasia of mast cells followed by migration has been proposed to account for the appearance of mast cells at new sites in the lung during acute lung injury in experimental hemorrhagic shock (Kasacka et al. 2003). The roles and sites of proliferation, migration, and differentiation remain to be determined.
The most remarkable aspect of this study is the increase in mast cell number, specifically in the loose connective tissue around large and small vessels. In most cases, mast cells were accumulated just outside the muscle layer. In addition to increasing mast cell number, hyperoxia induces an increase in smooth muscle in the lung, particularly in the airways (Hershenson et al. 1992; Szarek et al. 1995; Shenberger et al. 1997). It is possible that mast cells affect smooth muscle development as well as function. Tryptase, a major component in mast cell granules, is a potent smooth muscle mitogen (Brown et al. 1995). Our observation of increased mast cell numbers near vascular smooth muscle suggests that, during persistent hyperoxia, mast cell-mediated remodeling of the pulmonary vasculature may occur, with consequences, e.g., on pulmonary compliance.
In addition to tryptase, mast cells secrete a broad range of other mediators. Notably, mast cells release histamine, cysteinyl leukotrienes, and prostaglandin D2 (PGD2). Both histamine and cysteinyl leukotrienes increase vascular permeability resulting in edema, an effect associated with chronic hyperoxia. Histamine and cysteinyl leukotrienes promote smooth muscle constriction, whereas PGD2 relaxes vascular smooth muscle. We also find that the enzyme 5-LO, which initiates leukotriene synthesis, is present in the mast cells, but that its subcellular localization changes, being nuclear in normoxic conditions and cytoplasmic in hyperoxic conditions. As cytoplasmic localization of 5-LO can allow greater cysteinyl leukotriene synthesis than nuclear localization (Brock et al. 1999), this change may be important. An increase in the production of cysteinyl leukotrienes following chronic hyperoxic exposure has been reported (Burghardt et al. 1996), suggesting that mast cell-derived leukotrienes may contribute to edema and pulmonary hypertension associated with prolonged hyperoxia (Jones et al. 1984). Mast cell mediators are also powerful chemoattractants and activators of other leukocytes, so they may contribute to pulmonary inflammation following hyperoxia. Thus, the positioning of the increased mast cells may be significant. In addition to mast cells known to line the major airways, mast cells surrounding vessels may significantly impact lung function in a variety of ways.
The presence of mast cells at the pleura of the lung has been previously reported (Li et al. 1994), although their role at this location is not clear. In tissue sections from rats maintained in normoxic conditions, mast cells were patchy in their distribution along the pleura, with presence ranging from rare to common. An increase in the number of mast cells at the pleura following prolonged hyperoxia was striking, at fissures as well as peripherally. In these tissues, mast cells were common to abundant. As the lung pleura contains both blood and lymphatic vessels, it seems likely that the increased number of mast cells at this site might have effects on both of these systems.
The presence of mast cells does not necessarily indicate their impact on lung function: they typically need to be activated to release mediators and granule contents. Furthermore, the magnitude of their response to activation can be modified by priming agents (Boyce 2003). For example, Th2-type cytokines (interleukins −3, −4, −5, −6, and −9) can promote mast cell survival and augment their release of mediators upon cell activation. In addition, the response of mast cells to activation can furthermore depend on the stimulus. Thus, both lipopolysaccharide from gram-negative bacteria and peptidoglycan from gram-positive bacteria can activate mast cells to produce Th2-type cytokines, but histamine release is much greater in response to peptidoglycan (Varadaradjalou et al. 2003). Together the effect of increased pulmonary mast cells in patients treated with increased oxygen will depend on whether and how those mast cells may be primed and activated.
In this study we used a relatively high level of oxygen for 3 days and observed profound effects on mast cell number and distribution. Levels of hyperoxia, as well as duration of exposure, are important variables that may affect these outcomes. For example, exposure of rats to moderate hypoxia (FiO2, 50%) for 15 days increased airway reactivity and tracheal smooth muscle surface area in neonatal rats but not in adult rats (Denis et al. 2001). Changes in lung function are dependent on the length of exposure to hyperoxia (Szarek et al. 1995; Arkovitz et al. 1997). In addition, recovery of lung function after return to normoxia is time dependent (Hershenson et al. 1994). Effects of different degrees of hyperoxia, different lengths of exposure, and return to normoxia on mast cell number and distribution will be important to understand.
In summary, prolonged hyperoxia may contribute to bronchopulmonary dysplasia in neonates as well as to decreased pulmonary compliance, edema, remodeling, and inflammation in adults. Hyperplasia of mast cells and their migration to vessels throughout the pulmonary vasculature, as well as the pleura, in response to chronic hyperoxia positions these important immunoregulatory cells in key positions. Once accumulated at these sites, mast cells may become activated to secrete mediators that will have profound effects on pulmonary function. Future studies will examine the effects of varying levels and durations of increased oxygen exposure on mast cell accumulation, as well as the effects of bacterial infection on mast cell activation and change in lung function.
Footnotes
Acknowledgements
This study was supported by Grant AI-43574 from the National Institutes of Health, Bethesda, MD.
