Abstract
Biological effects of substance P (SP) are mediated by the neurokinin-1 (NK1) receptor that exists as a full-length and as a carboxy-terminally truncated isoform in humans. Although NK1 receptor mRNA and binding sites have been detected in certain malignancies, little is known about the cellular and subcellular localization of NK1 receptor protein in human neoplastic tissues. We developed and characterized a novel anti-peptide antibody to the carboxy-terminal region of the human full-length NK1 receptor. Specificity of the anti-serum was demonstrated by (1) detection of a broad band migrating at molecular mass 70,000-90,000 Da in Western blots of membranes from NK1-expressing tissues; (2) cell-surface staining of NK1-transfected cells; (3) translocation of NK1 receptor immunostaining after SP exposure; and (4) abolition of tissue immunostaining by preadsorption of the antibody with its immunizing peptide. Distribution of NK1 receptors was investigated in 72 formalin-fixed, paraffin-embedded human tumors showing that NK1 receptors were frequently expressed in glioblastomas and breast and pancreatic carcinomas. Immunoreactive NK1 receptors were clearly confined to the plasma membrane and uniformly present on nearly all tumor cells. Development of this novel NK1 receptor antibody allows the efficient localization of NK1 receptor protein in human formalin-fixed, paraffin-embedded tissues. NK1 receptor visualization with this simple and rapid immunohistochemical method will facilitate identification of tumors with a sufficient receptor overexpression for diagnostic or therapeutic intervention using SP analogs.
S
NK1 receptor mRNA and binding sites have been detected in a relatively high percentage in glioblastomas and breast and pancreatic cancers (Johnson et al. 1991; Hennig et al. 1995; Winkler et al. 1995; Ehlers et al. 2000; Singh et al. 2000; Friess et al. 2003). Activation of NK1 receptors by SP triggers signaling pathways, e.g., extracellular signal-regulated protein kinases (ERK1/2), which are potentially relevant for tumor growth and progression (Luo et al. 1996; Fukuhara et al. 1998; Palma et al. 1999b; Palma and Maggi 2000; Koon et al. 2004). Conversely, administration of NK1 receptor antagonists has been reported to inhibit tumor cell growth in vitro and in vivo (Palma et al. 1999a,2000; Munoz et al. 2004,2005).
Although expression of NK1 receptors in human tumors has been well documented, the exact cellular and subcellular sites of NK1 receptor in human neoplastic tissues still need to be fully elucidated. Expression of NK1 receptors has previously been detected using binding autoradiography or reverse transcription-polymerase chain reaction (RT-PCR) (Hennig et al. 1995; Ehlers et al. 2000; Singh et al. 2000). However, anatomical resolution of autoradiographic receptor binding is limited, and RT-PCR does not discriminate between receptor transcripts originating from individual target cells.
In the present study we have generated and characterized antibodies directed to the carboxy-terminal tail of the NK1-Fl receptor. We have also developed an immunohistochemical (IHC) protocol that allows efficient detection of this receptor in formalin-fixed, paraffin-embedded human tissues. Generation of this novel antibody enabled us to determine the cellular distribution of NK1 receptor proteins in a variety of human tumors.
Materials and Methods
Patients, Tumors, and Tissue Preparation
Seventy two tumor specimens were retrieved from the archives of the Department of Pathology. All tissue specimens had been fixed in formalin and embedded in paraffin. The following tumors were investigated: colorectal adenocarcinoma (n=5), ductal pancreatic adenocarcinoma (n=5), ductal invasive breast carcinoma (n=5), ovarian carcinoma (n=10), prostate cancer (n=4), thyroid carcinoma (n=6), carcinoid (n=15), pancreatic insulinoma (n=8), growth-hormone-producing pituitary adenoma (n=4), pheochromocytoma (n=2), glioblastoma (n=4), and meningioma (n=4). In addition, several fresh tumor specimens were immediately frozen in liquid N2 and stored at −70C until Western blot analysis. The following tumors were investigated: breast carcinoma (n=4) and ovarian carcinoma (n=4).
Generation and Purification of Antipeptide Antibodies
Polyclonal antisera were generated against the carboxy-terminal tail of the NK1 receptor. Identity of the peptide was SSRSDSKTMTESFSFSSNVLS, which corresponds to amino acids 387-407 of the NK1-Fl receptor. The peptide was synthesized, purified, and coupled to keyhole limpet hemocyanin as described (Schulz et al. 2000). The conjugate was mixed 1:1 with Freund's adjuvant and injected into groups of three rabbits {9040-9042} for NK1 antisera production. Animals were injected at 4-week intervals, and serum was obtained 2 weeks after immunizations beginning with the second injection. Specificity of the antisera was initially tested using immunodot-blot analysis as described (Schulz et al. 2000). For subsequent analysis, antibodies were affinity purified against their immunizing peptide using the Sulfo-Link coupling gel according to manufacturer's instructions (Pierce; Rockford, IL).
Immunocytochemistry
Human embryonic kidney 293 (HEK-293) cells were stably transfected with the human NK1 receptor. Cells were grown on coverslips overnight and either not exposed or exposed to 1 μM SP (Bachem; Weil am Rhein, Germany). Cells were then fixed and incubated with 1 μg/ml anti-NK1 {9042} antibodies followed by cyanin 3.18-conjugated secondary antibodies (Amersham; Braunschweig, Germany). Specimens were mounted and examined using a Leica TCS-NT laser scanning confocal microscope (Leica; Wetzlar, Germany) as described (Pfeiffer et al. 2001).
Western Blot Analysis
Membranes were prepared from stably transfected HEK-293 cells as well as from fresh tumor specimens. Cells and tissues were lysed in homogenization buffer (5 mM EDTA, 3 mM EGTA, 250 mM sucrose, 10 mM Tris-HCl, pH 7.6, containing 1 mM phenylmethylsulfonylfluoride, 1 μM pepstatin A, 10 μg/ml leupeptin, and 2 μg/ml aprotinin), and membranes were pelleted at 20,000 × g for 30 min at 4C. Membranes were then dissolved in lysis buffer (150 mM NaCl, 5 mM EDTA, 3 mM EGTA, 20 mM Hepes, pH 7.4, containing 4 mg/ ml dodecyl-β-maltoside and proteinase inhibitors as above) and incubated with 150 μl wheat germ lectin agarose beads (Amersham) for 90 min at 4C. Beads were washed five times in lysis buffer, and adsorbed glycoproteins were eluted with SDS sample buffer for 20 min at 60C. Samples were then subjected to 8% SDS polyacrylamide gel electrophoresis and immunoblotted onto nitrocellulose. Blots were incubated with 1 μg/ml anti-NK1 {9042} antibodies followed by peroxidase-conjugated secondary antibodies and enhanced chemiluminescence detection (Amersham). For adsorption controls, antibody was preincubated with 10 μg/ml of its cognate peptide for 2 hr at room temperature.
Immunohistochemistry
Seven-μm paraffin sections were cut and floated onto positively charged slides and IHC stained as described (Schulz et al. 2000,2004,2005; Mundschenk et al. 2003). Briefly, sections were dewaxed, microwaved in 10 mM citric acid (pH 6.0) for 20 min at 600 W, and subsequently incubated with anti-NK1 {9042} antibodies in concentrations ranging from 0.5 to 2 μg/ml overnight at 4C. Staining of primary antibody was detected using biotinylated goat anti-rabbit IgG followed by incubation with avidin-biotinylated peroxidase solution. Tissue was then rinsed and stained with 3,3′-diaminobenzidine-glucose oxidase for 15 min. Cell nuclei were lightly counterstained with hematoxylin. For IHC controls, primary antibody was omitted, replaced by preimmune sera, or adsorbed with several concentrations ranging from 1 to 10 μg/ml of homologous or heterologous peptides for 2 hr at room temperature. A tumor known to stain positively was included in each batch of staining as positive control.
Assessment of Staining Patterns
All slides were evaluated by two independent investigators. The presence or absence of staining and the depth of color were noted, as well as the number of cells showing a positive reaction and whether or not the staining was localized to the plasma membrane. Tumors were only categorized as positive when they exhibited a moderate to strong plasma membrane and/or cytoplasmic staining in the majority of tumor cells, which was easily visible with a low-power objective.
Results
Characterization of NK 1 Receptor Antibodies
Specificity of the antisera was monitored using Western blot analysis. When membrane preparations from stably transfected cells were electrophoretically separated and blotted onto nitrocellulose, the anti-NK1 {9042} antiserum detected a broad band migrating at molecular mass 70,000 to 90,000 Da only in cells transfected with its cognate receptor but not in cells transfected with an empty vector (Figure 1). Antisera were further characterized using immunofluorescent staining of transfected cells. When HEK-293 cells stably expressing NK1 were stained with the anti-NK1 antibody {9042}, prominent immunofluorescence localized at the level of the plasma membrane was detected (Figure 2A). After incubation with SP, NK1 immunoreactivity (ir) was translocated from the plasma membrane into the cytosol indicating that the NK1 receptor was rapidly endocytosed in an agonist-dependent manner (Figure 2B). Next, the NK1 receptor antisera were tested for possible cross-reactivity with other proteins present in human tissues. When membrane preparations from human breast or ovarian carcinomas were electrophoretically separated and blotted onto nitrocellulose, the anti-NK1 antibody {9042} detected a broad band migrating at molecular mass 70,000 to 90,000 Da (Figures 3A and 3B). A few cases were observed in which this broad receptor band consisted of several narrower bands, suggesting that the NK1 receptor may exist in differently glycosylated forms (Figure 3B). All ir bands were completely abolished by preadsorption of the antibody with 10 μg/ml of its immunizing peptide (Figures 3A and 3B).

Western blot analysis of the specificity of anti-NK1 antibodies. Membrane preparations from HEK-293 cells transfected with NK1 or empty vector (MOCK) were separated on 8% SDS-polyacrylamide gels and blotted onto nitrocellulose membranes. Membranes were then incubated with affinity-purified anti-NK1 {9042} antibodies at a concentration of 1 μg/ml. Blots were developed using enhanced chemiluminescence. Two additional experiments gave similar results. MOCK, empty vector; ordinate, migration of protein molecular mass markers (molecular mass × 10-3).
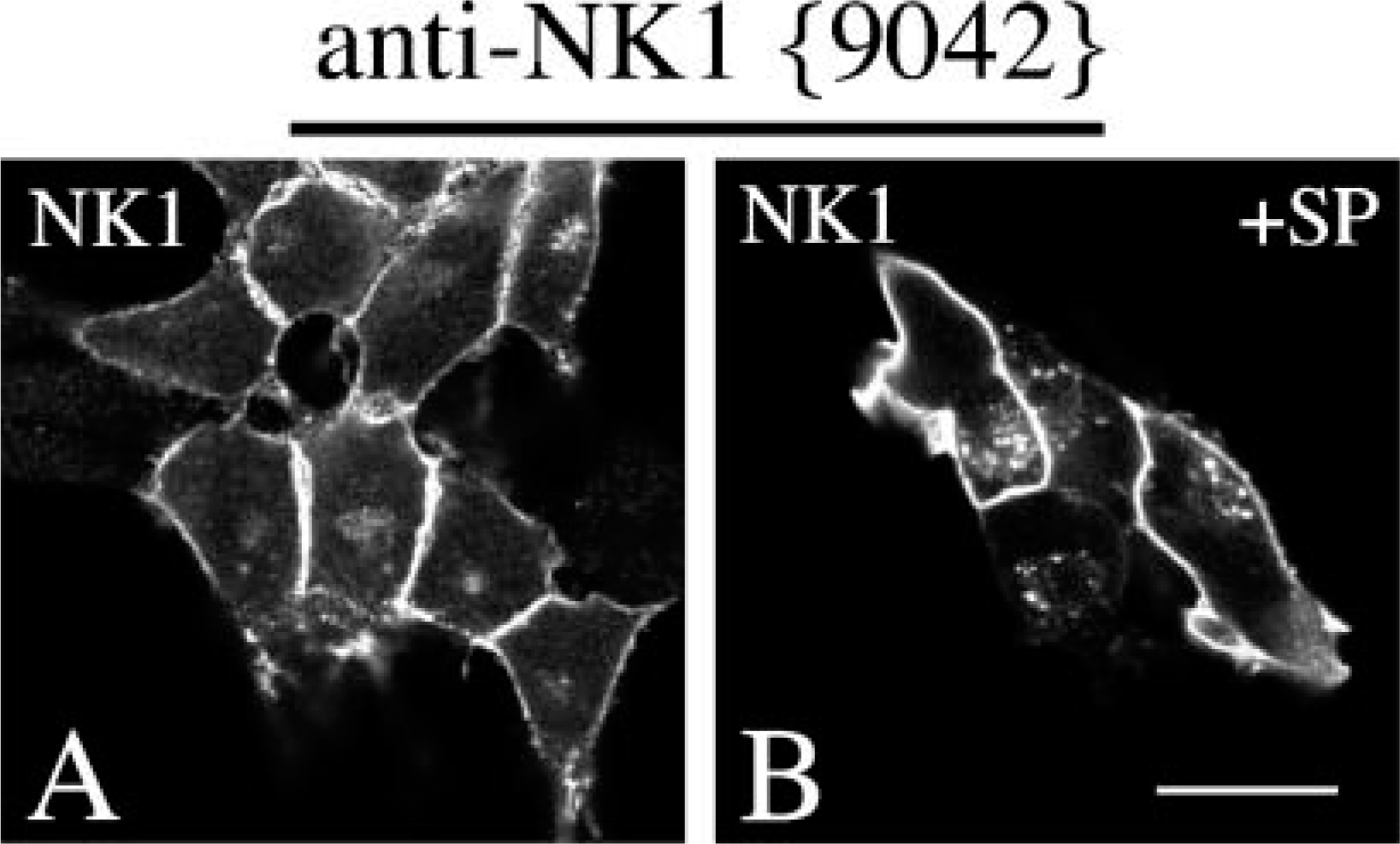
Characterization of anti-NK1 antibodies by immunofluorescent staining of transfected cells. (
NK 1 Receptor IHC Staining in Neoplastic Human Tissues
The anti-NK1 antibodies were then subjected to IHC staining of human tissues. Initial experiments showed that heat-induced epitope retrieval is required for efficient IHC staining of paraffin-embedded tissues (not shown). All three anti-NK1 antisera yielded essentially identical IHC staining patterns with prominent ir predominantly localized to the plasma membrane of the tumor cells, although with different staining intensity (Figure 4). Immunostaining for each antiserum was completely abolished by preadsorption with 10 μg/ml of the immunizing peptide (Figure 4B). The anti-NK1 antibody {9042} produced the most prominent immunostaining and was therefore used throughout the study. Prevalence of NK1 receptors in human neoplasms is summarized in Table 1. Immunoreactive NK1 receptors were frequently detected in pancreatic adenocarcinomas (80%), breast carcinomas (75%), and glioblastomas (75%). In most of these tumors, ir NK1 receptors were homogeneously present on nearly all tumor cells (Figures 4A, 4C, 4F). However, a few tumors were observed with heterogeneous NK1 expression, i.e., tumor cells with strong to moderate NK1 receptor staining were seen next to tumor cells lacking ir NK1 receptors (Figures 4D and 4E). Immunoreactive NK1 receptors were also found in 4/10 ovarian carcinomas (40%), 2/6 thyroid carcinomas (30%), and 1/4 growth hormone-producing pituitary adenomas (25%). In contrast, ir NK1 receptors were not detected in neuroendocrine tumors including carcinoid or insulinoma. NK1 receptors were also not observed in meningiomas, pheochromocytomas, or prostate and colorectal cancer.
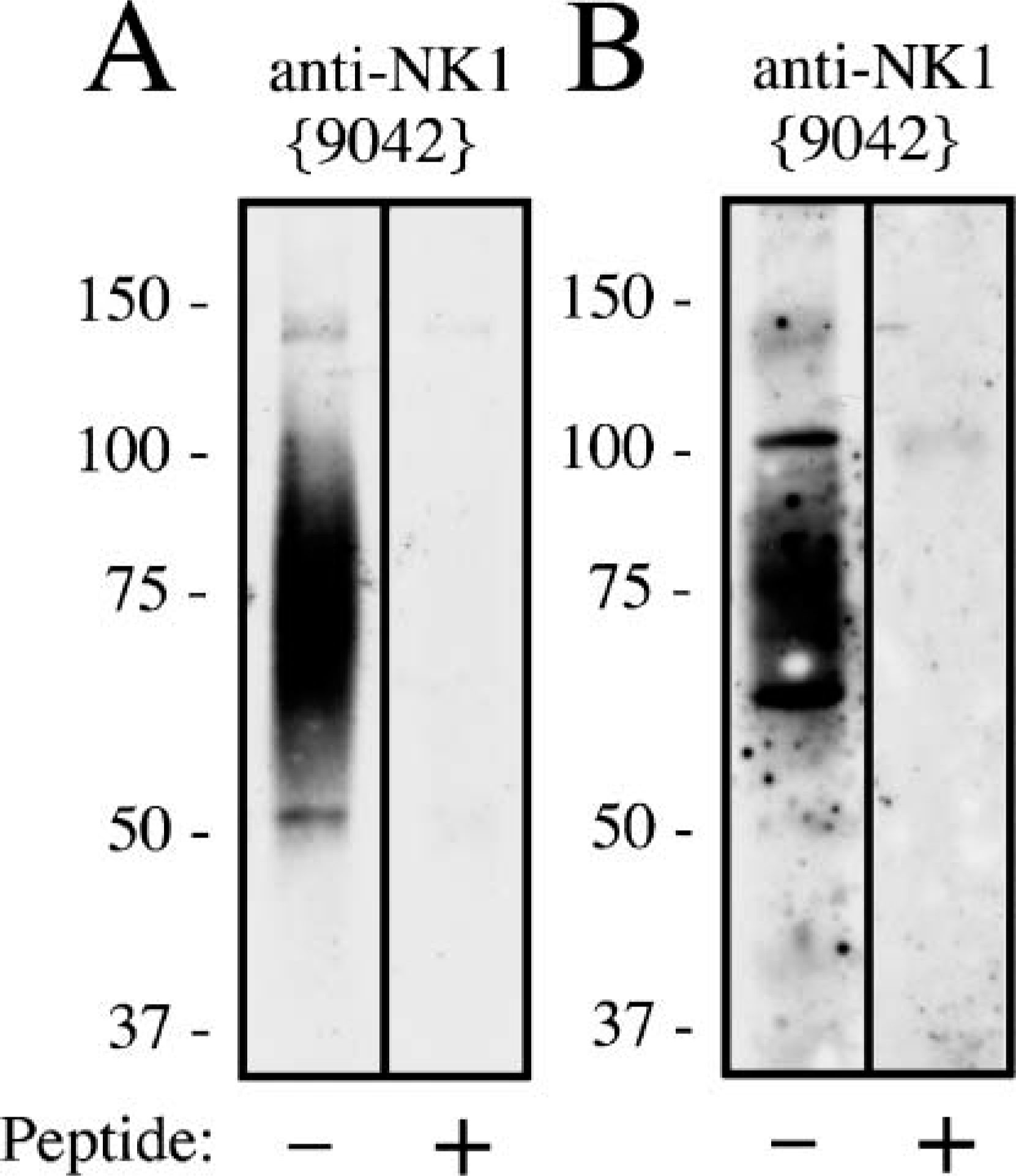
Western blot analysis of the specificity of anti-NK1 antibodies in human tumors. (
Discussion
It is well established that NK1 receptors are overexpressed in certain malignancies including glioblastomas and breast and pancreatic carcinomas; however, little is known about their cellular and subcellular localization in human neoplastic tissues. We therefore generated antibodies that exert selective specificity for the human NK1-Fl receptor. We show that the cytoplasmic tail of this receptor can serve as an epitope for the generation of antisera that effectively stain formalin-fixed, paraffin-embedded human tissues. Several lines of evidence indicate that this antibody specifically detects its targeted tachykinin receptor and does not crossreact. First, in Western blots of membranes from transfected cells, the anti-NK1 antibody detected a band migrating at molecular mass 70,000 to 90,000 Da only in NK1-transfected cells but not in cells transfected with an empty vector. Second, the anti-NK1 antibody revealed prominent cell surface staining of NK1-transfected cells. This immunostaining translocated from the cell surface into the cytosol after agonist exposure indicating rapid endocytosis of the NK1 receptor. Third, in Western blots of membranes from receptor-expressing tumors, the anti-NK1 antibody detected a broad band migrating at molecular mass 70,000 to 90,000 Da, which corresponds to a glycosylated form of the receptor. Fourth, tissue immunostaining of the anti-NK1 antiserum was completely abolished by preadsorption with homologous but not heterologous peptides. Finally, it should be noted that three of three NK1 antisera yielded similar results.
Availability of NK1 receptor antibodies will facilitate further basic morphological investigation of NK1 expression in human normal and neoplastic tissues. The IHC NK1 receptor evaluation offers several major advantages. This method can analyze NK1 receptors in routinely processed archival paraffin-embedded material of any diagnostic pathology center. It requires only an immunopathological laboratory to perform the test, which can be carried out without costly and time-consuming receptor autoradiography (Hennig et al. 1995). IHC evaluation of the complete NK1 receptor status of a given tumor specimen can be accomplished in <24 hr. Although one could expect the IHC NK1 receptor determination to be less sensitive than receptor autoradiography with 125I-labeled SP, sensitivity of the anti-NK1 {9042} antibody generated and characterized in this study was sufficiently high to detect this receptor in human tumors known to express NK1 receptors in a high percentage, e.g., glioblastomas and breast and pancreatic carcinomas (Hennig et al. 1995; Ehlers et al. 2000; Singh et al. 2000; Friess et al. 2003). In addition, the IHC NK1 receptor determination provided a better histological quality and cellular resolution compared with receptor autoradiography. Thus, previously unappreciated cellular localizations of NK1 receptors were uncovered, i.e., heterogeneous expression of NK1 receptors in a subpopulation of pancreatic carcinomas.
SP can affect the growth of human tumor cells in vivo and in vitro. Whereas growth-promoting activities have been reported for SP, growth-inhibiting properties have been found for NK1 antagonists in various tumor models (Luo et al. 1996; Fukuhara et al. 1998; Palma et al. 1999a,b,2000; Palma and Maggi 2000; Koon et al. 2004; Munoz et al. 2004,2005). It is therefore very tempting to suggest the use of NK1 antagonists for treatment of a subset of human malignancies. The present findings also suggest that NK1 receptors could be promising molecular targets for tumor imaging and targeted radiotherapy using radiolabeled SP analogs (Breeman et al. 1996; Reubi et al. 2005).
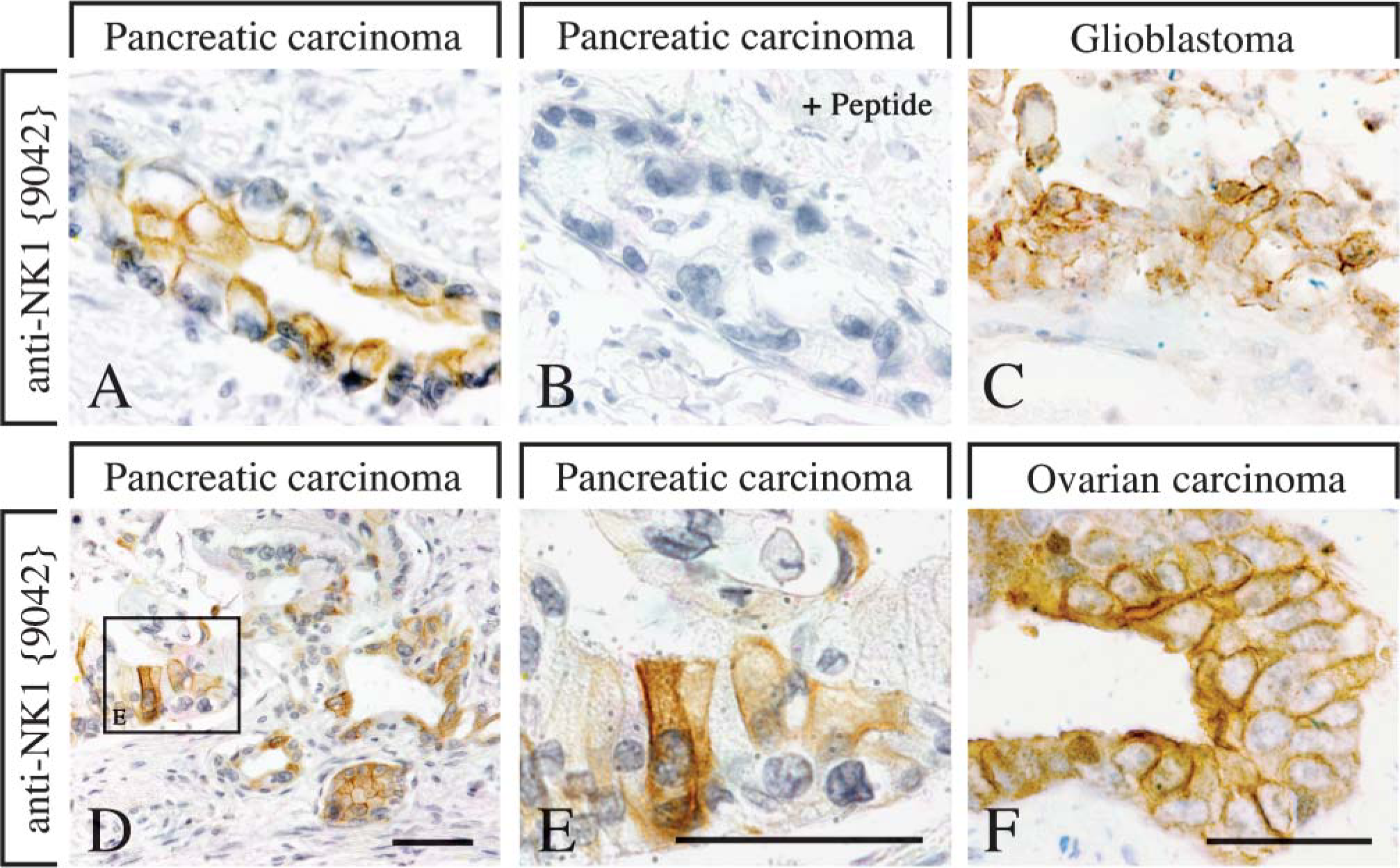
NK1 immunohistochemical (IHC) staining in human neoplastic tissues. NK1 IHC staining in pancreatic carcinoma (
It is believed that the human NK 1 gene could generate NK1-Fl and NK1-Tr transcripts (Fong et al. 1992). NK1-Fl and NK1-Tr have identical ligand-binding domains but differ in length of their cytoplasmic tails. Specifically, the cytoplasmic tail of NK1-Tr lacks 100 residues, a region that functions as a substrate for phosphorylation by G-protein-coupled receptor kinases in NK1-Fl. Consequently, NK1-Tr shows a reduced capacity to undergo SP-induced internalization and desensitization (Fong et al. 1992). Interestingly, overexpression of NK1-Tr has recently been shown to promote malignant transformation of breast cells (Patel et al. 2005). Given that the antibody raised and characterized here is directed to the end of the cytoplasmic tail of the NK1-Fl receptor, it was not possible to detect NK1-Tr in the present study.
Prevalence of NK1 receptors in human tumors

In conclusion, we have generated and extensively characterized anti-NK1 antibodies. Development of this novel antibody enabled us to visualize NK1-Fl receptors in human formalin-fixed, paraffin-embedded tissues. It is now possible to determine the exact cellular and sub-cellular sites of NK1 receptor protein in normal human and neoplastic tissues. Rapid immunocytochemical NK1 receptor visualization may also be helpful to identify those tumors with sufficient receptor overexpression for diagnostic or therapeutic intervention.
Footnotes
Acknowledgements
We thank Beate Peter and Dana Mayer for skillful technical assistance.
