Abstract
Our aim was to examine the effects of androgen administration on breast tissue histology of female-to-male transsexuals and to study the immunohistochemical expression of three human tissue kallikreins, hK3 (PSA), hK6, and hK10. We studied 23 female-to-male transsexuals who were treated with injectable testosterone for 18-24 months. We also used 10 control female breast tissues. All tissues were fixed in buffered formalin, embedded in paraffin, and examined by hematoxylin-eosin staining and immunohistochemical staining for PSA, hK6, and hK10. Females treated with androgens exhibited similar involutionary changes as those seen in breast of menopausal women, such as marked reduction of glandular tissue, involution of the lobuloalveolar structures, and prominence of fibrous connective tissue, but presence of only small amounts of fat tissue. Fibrocystic lesions were generally not observed. In immunohistochemistry, in control breast tissues, we found moderate to strong cytoplasmic immunoexpression of hK6 and hK10 in the epithelial ductal and lobuloalveolar structures, but myoepithelial cells were negative. Luminal secretions were also positive. In menopausal breast, the immunoexpression of hK6 and hK10 was weaker and focal. No control case showed immunoexpression for PSA. In female-to-male transsexuals, one case showed focal PSA cytoplasmic immunoexpression in the epithelium of moderately involuting lobules. Long-term administration of androgens in female-to-male transsexuals causes marked reduction of glandular tissue and prominence of fibrous connective tissue. These changes are similar to those observed at the end-stage of menopausal mammary involution.
T
Human tissue kallikreins are members of a multigene family that includes 15 genes (KLK1-KLK15 for genes, hK1-hK15 for proteins), all located on chromosome 19q13.4 (Yousef and Diamandis 2001; Borgono et al. 2004; Borgono and Diamandis 2004). All kallikreins, except hK2 and hK3, which are almost prostate tissue-specific, are expressed in many normal, mainly glandular tissues, among them the breast epithelium (Black and Diamandis 2000; Diamandis et al. 2000; Yousef and Diamandis 2001; Borgono et al. 2004; Borgono and Diamandis 2004; Clements et al. 2004). We previously examined the immunohistochemical expression (IE) of some hKs in normal and pathologic human tissues (Howarth et al. 1997; Petraki et al. 2001, 2002,2003). There are suggestions that some kallikreins may function as tumor suppressors and that they are downregulated during breast cancer progression (Goyal et al. 1998; Yousef et al. 2000,2002; Dhar et al. 2001).
We examine here the effects of androgen administration on breast tissue histology of female-to-male transsexuals and on IE of hK3 (PSA), hK6, and hK10.
Materials and Methods
Patients and Tissue Samples
Twenty-three female-to-male transsexuals were included in this study. All subjects were treated with injectable testosterone esters every 2 weeks (Sustanon-250; Organon, Oss, The Netherlands), for 18-24 months before radical mastectomy was performed. As controls, we used 10 female breast tissues free of any disease (3 in proliferative phase, 4 in secretory phase, and 3 in menopause). All breast tissues were fixed in buffered formalin and paraffin-embedded tissue sections, 4 μm thick, were used for hematoxylin-eosin staining and the performance of the immunohistochemical staining for PSA, hK6, and hK10.
Immunohistochemistry
The streptavidin-biotin-peroxidase protocol, using the DAKO LSAB+Kit Peroxidase, was performed (DAKO; Mississauga, ON, Canada). A monoclonal antibody for PSA (MONOSAN 1:40) and specific polyclonal antibodies for hK6 (1:150) and hK10 (1:150), raised by immunizing rabbits with full-length recombinant hK6 (produced in mammalian cells) and hK10 (produced in yeast), respectively, were used. Staining procedures included deparaffinization in warm xylene for 5 min with two changes of xylene at room temperature, followed by rehydration by transfer through graded alcohols. Endogenous peroxidase activity was blocked with 0.5% H2O2 in methanol for 10 min. The sections were pretreated with 10 mmol/L citrate buffer (pH 6.1) in microwave for 5 min and incubated overnight at 4C with the primary antibodies in 3% BSA. After two washes of the sections in 50 mM Tris buffer (pH 7.6), the biotinylated link (DAKO Corporation) was applied for 15 min and a streptavidinperoxidase conjugate followed for another 15 min. The enzymatic reaction was developed in a freshly prepared solution of 3,3î-diaminobenzidine tetrahydrochloride using DAKO Liquid DAB Substrate-Chromogen Solution for 10 min (brown color). The sections were then counterstained with hemalum, dehydrated, cleared in xylene, and mounted.
(
A cytoplasmic immunoexpression was evaluated for all three hKs. Absence of IE or weak focal staining of hK6 and hK10 were considered negative. Positive immunostaining was classified as moderate (focal or extensive) and strong (focal or extensive).
Statistics
Statistical analysis was carried out with the SPSS 10.0 software (SPSS, Inc.; Chicago, IL), using the
Results
Hormonal Changes
After testosterone administration, serum testosterone levels of female-to-male transsexuals rise by approximately 10-15-fold (Spinder et al. 1989). Serum 17β-estradiol luteinizing hormone and follicle-stimulating hormone levels fall only moderately (approximately 1.5 to 2-fold) (unpublished data).
Histological Study
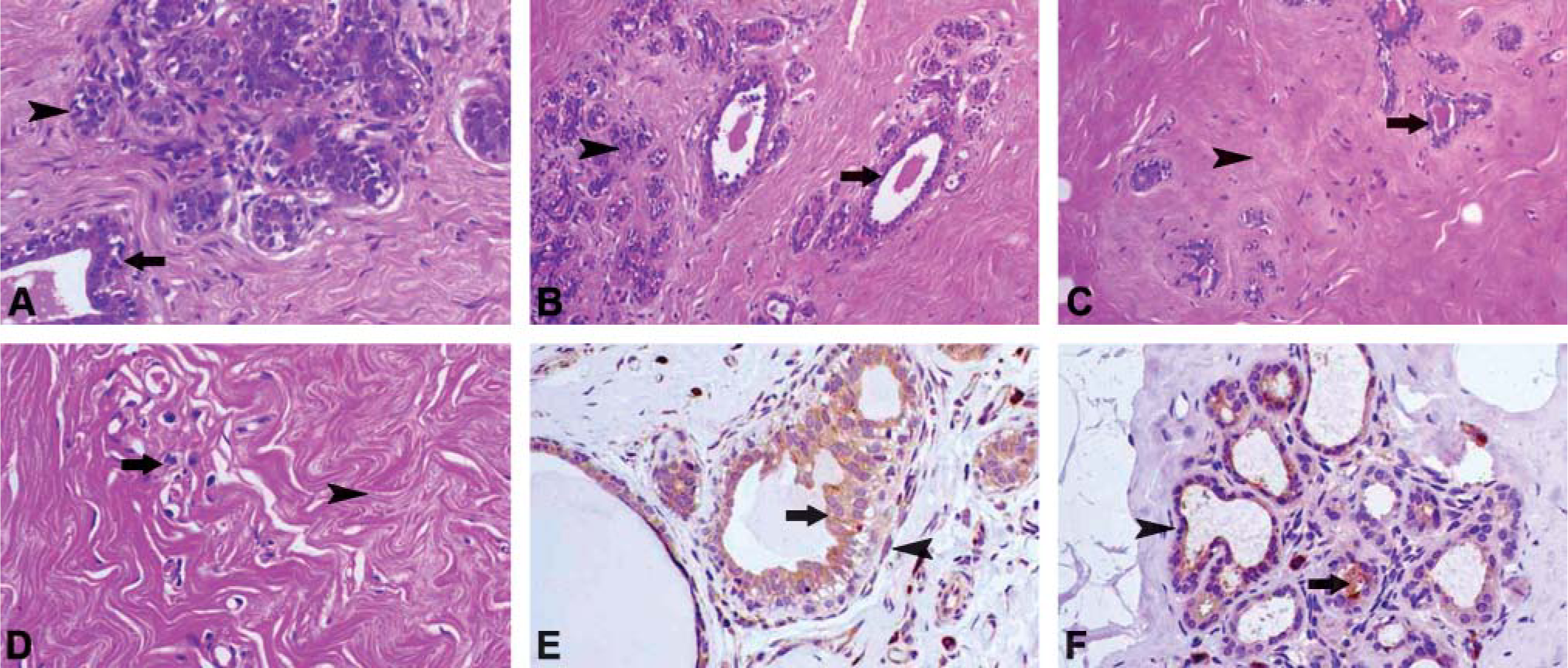
It should be mentioned that fibrocystic lesions were not observed with the exception of focal ductal epithelial hyperplasia in two cases and focal apocrine metaplasia in three cases (very low percentage in comparison with the results of the control group and the general population). Additionally, an intraductal papilloma was observed in one case (Figures 1B-1D, Table 1).
Immunohistochemical and histological findings of the 23 breast tissues of female-to-male transsexuals after testosterone administration
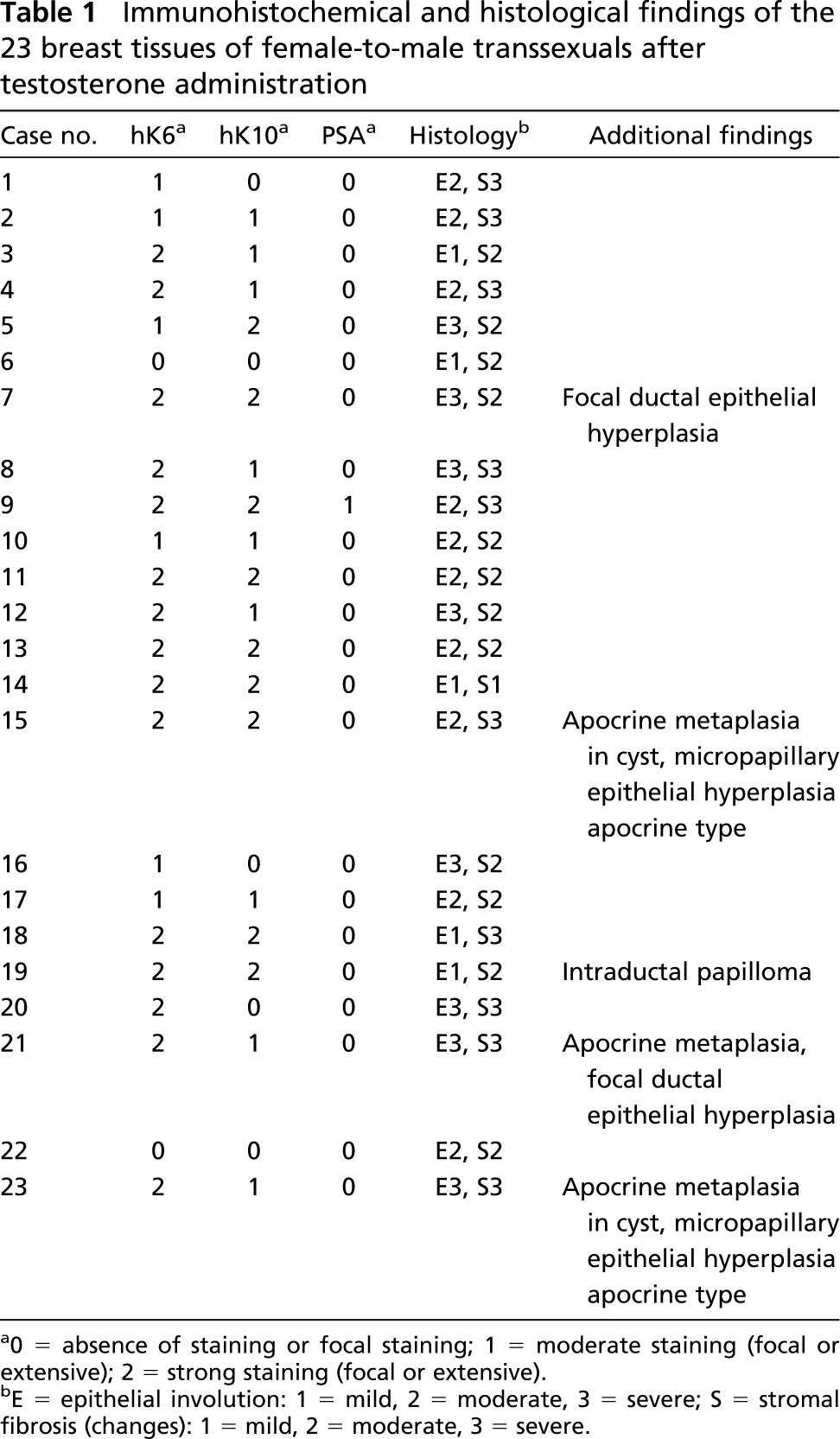
0 = absence of staining or focal staining; 1 = moderate staining (focal or extensive); 2 = strong staining (focal or extensive).
E = epithelial involution: 1 = mild, 2 = moderate, 3 = severe; S = stromal fibrosis (changes): 1 = mild, 2 = moderate, 3 = severe.
(
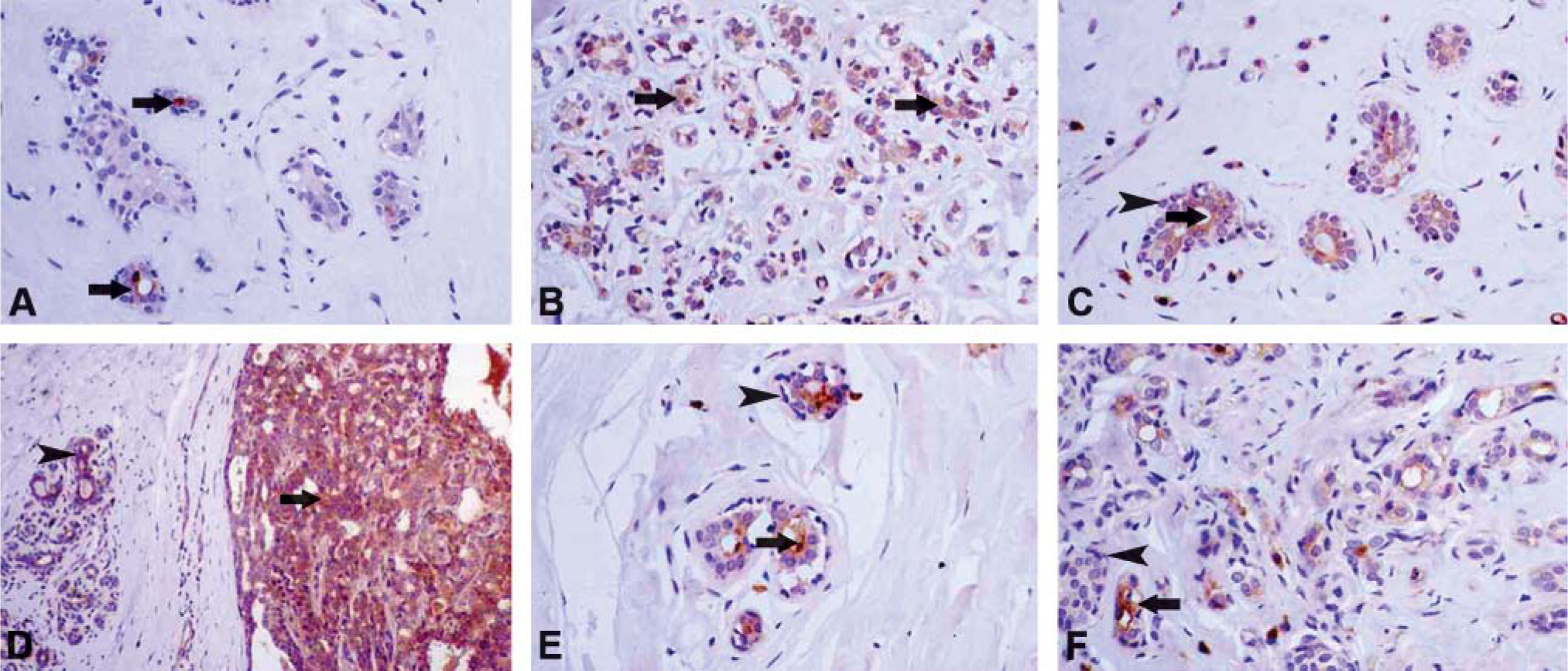
Immunohistochemical Study (hK6, hK10, PSA)
Discussion
Three earlier studies have reported findings of androgen effects on the breasts (Futterweit and Schwartz 1988; Sapino et al. 1990; Burgess and Shousha 1993).
The first two studies addressed histological aspects. The effects varied strongly from one subject to the other: most subjects showed intralobular fibrous stroma and some extralobular fibrous stroma; approximately half showed lobular atrophy. The effects on breasts probably reflected the simultaneous action of androgens and estrogens generated by peripheral aromatization of the high levels of androgens in these subjects. The third study, which also used histochemical techniques, did not find major differences in comparison to the breast tissue of normal women except that there were more microcalcifications than normal.
Relation of hK6 immunohistochemical expression with the degree of epithelial and stromal changes in the 23 breast tissues of female-to-male transsexuals after testoster one administration
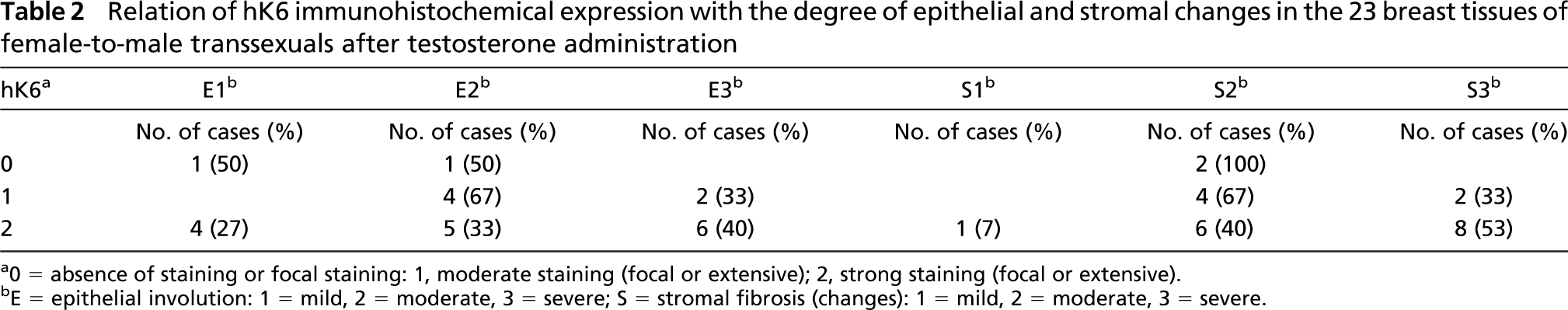
0 = absence of staining or focal staining: 1, moderate staining (focal or extensive); 2, strong staining (focal or extensive).
E = epithelial involution: 1 = mild, 2 = moderate, 3 = severe; S = stromal fibrosis (changes): 1 = mild, 2 = moderate, 3 = severe.
Relation of hK10 immunohistochemical expression with the degree of epithelial and stromal changes in the 23 breast tissues of female-to-male transsexuals after testosterone administration
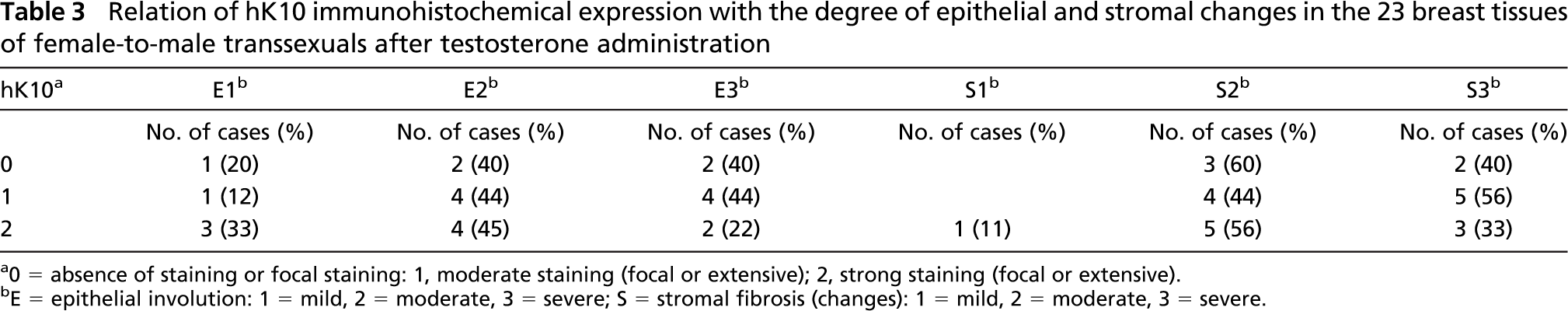
0 = absence of staining or focal staining: 1, moderate staining (focal or extensive); 2, strong staining (focal or extensive).
E = epithelial involution: 1 = mild, 2 = moderate, 3 = severe; S = stromal fibrosis (changes): 1 = mild, 2 = moderate, 3 = severe.
The histological findings in the breasts of the androgen-treated females in our study were characterized by epithelial involution and stromal fibrosis (similar to findings in earlier studies) and showed a high degree of similarity with the histological picture of breasts of postmenopausal women. Similar involutionary changes as in breasts of menopausal women were observed in most cases: marked reduction of glandular tissue (involution of the lobuloalveolar structures) and prominence of dense, hyalinized, fibrous connective tissue. It is noteworthy that only small amounts of fat tissue were observed in all cases, comparable with the fat tissue found in menopausal women. From an endocrine viewpoint, this is a remarkable finding. 17β-estradiol (E2) levels in postmenopausal women are well below 50 pmol/L. In female-to-male transsexuals receiving androgen treatment, substantial amounts of circulating levels of E2 are generated from peripheral aromatization of testosterone. In our patients, we observed a positive correlation of plasma levels of E2 with plasma levels of testosterone. In a recent study of testosterone-treated female-to-male transsexuals, peak plasma E2 levels of more than 300 pmol/L were found, which never fell below 85 pmol/L and averaged 131 ± 33 pmol/L (mean ± SD) (Spinder et al. 1989). Another consideration concerning the impact of estrogens with this treatment regime is the decline of sex hormone-binding globulin levels after androgen administration, which leads to increases of free concentrations of both testosterone and estradiol. An explanation for the lack of biological effects of the high levels of circulating estradiol on mammary tissue might be found in the endocrine mechanism similar to that governing the male breast. Postpubertal men have significant levels of circulating E2, but their effects on mammary tissue are prevented by the simultaneous presence of high levels of testosterone (Rochefort and Garcia 1983). Testosterone is capable of inhibiting the E2-induced proliferation of mammary epithelial cells and abolishes E2-induced augmentation of ER-α expression (Zhou et al. 2000). Testosterone further promotes pro-apoptotic effects in breast cancer cells (Kandouz et al. 1999). Therefore, the simultaneous presence of high circulating testosterone and estrogens in these subjects might have prevented the biological action of E2, causing a histological picture of postmenopausal involution of the mammary tissue.
Another aspect of our study is the immunohistochemical expression of three tissue kallikreins. In menstruating women, immunohistochemical expression of hK10 was moderate and of hK6 was strong, whereas no immunohistochemical expression of hK3 (PSA) was detected. In the breast tissue of androgen-treated female-to-male transsexuals, focal production of hK3 (PSA) was found in one case but not in others. The immunohistochemical expressions of hK6 and hK10 were slightly downregulated, more so of hK10 than of hK6. Recent studies indicate that testosterone treatment of female-to-male transsexuals increases serum and urine levels of hK3 (PSA) and of other kallikreins (Goh 1999; Obiezu et al. 2000; our unpublished data). It has been speculated that the breast was the source of PSA (hK3) (Goh 1999). Our inability to detect PSA by immunohistochemistry in breast tissue of most transsexuals is likely to the result of the very low levels of PSA in this tissue in comparison to the prostate (Howarth et al. 1997; Black and Diamandis 2000).
Another aim of our study was to determine, as much as possible, whether exposure to high levels of androgens in these women promotes the initiation or progression of breast cancer. Epidemiological studies suggest that circulating testosterone is associated with the risk of developing breast cancer in postmenopausal women (Key et al. 2002; Missmer et al. 2004). We found no evidence of in situ breast carcinomas in our subjects in this short-term study. Almost all long-term follow-up studies of women with hyperandrogenism from polycystic ovarian disease (which is endocrinologically characterized by the simultaneous presence of high circulating androgen and estradiol levels) do not provide evidence of higher breast cancer risk in these women. However, the incidence of endometrial cancer and probably, ovarian cancer, is higher in these women (Solomon 1999; Wild et al. 2000; Balen 2001; Somboonporn and Davis 2004a). Further, two recent reviews with regard of the risk that androgens pose for the development of breast cancer in women are reassuring (Somboonporn and Davis 2004a,b). Animal studies in primates receiving exogenous testosterone also indicate that androgens may limit the mitogenic and cancer promoting effects of estrogens on mammary epithelium (Somboonporn and Davis 2004a). Epidemiological studies in women regarding the significance of endogenous testosterone are inconclusive, but are in any case not strongly suggestive for cancer promoting effects on breast tissue (Somboonporn and Davis 2004a,b).
In conclusion, we report here that long-term administration of androgens in young females causes marked reduction of glandular tissue and promotion of fibrous connective tissue, changes similar to those seen in women in menopause.
