Abstract
Due to its sensitivity, immunohistochemistry (IHC) of abnormal prion protein (PrPsc) is used to study experimental and natural cases of transmissible spongiform encephalopathies (TSEs) such as Creutzfeldt-Jakob disease in humans or scrapie and bovine spongiform encephalopathy (BSE) in animals. The limits of detection are particularly critical when PrPsc IHC is used for diagnostic purposes. In this article, we describe for the first time the use of streptomycin sulfate in IHC, providing a novel original and easy way to amplify specifically PrPsc immunohistochemical detection in natural cases of BSE and scrapie, as well as in experimental TSEs in mice models using two different PrP antibodies.
Keywords
I
In addition to several possibilities classically used in IHC procedures to amplify antigen detection such as antigen retrievals and the biotinylated tyramide-based method, we have investigated the possibility of using a molecule demonstrated to have high affinity for the prion protein and capability of bridging and reticulating PrP fragments (Moussa et al. 2006).
Remarkably, streptomycin sulfate binds to prion proteins of different species (bovine, ovine, murine, and human). It precipitates PrPsc in vitro and therefore significantly increases the sensitivity of PrPsc detection by the Western blot method. In view of these reasons, the use of streptomycin sulfate in PrPsc detection by IHC may present a route to increased sensitivity in prion detection. Comparing with classical PrPsc IHC procedures (using two different PrP MAbs), we have studied the effects of streptomycin sulfate introduction in the IHC procedure on brain samples originating from natural BSE or scrapie cases and scrapie or BSE-infected mice, in addition to unaffected animals. We have demonstrated that, in all the species tested, PrPsc detection is amplified in every case without generating any unspecific background staining.
Materials and Methods
Histological Samples
Brain slices of the obex area from BSE-negative and -positive cows (30 slides from three cows), in addition to scrapie-negative and -positive sheep (100 slides from five sheep), were selected for this study on the basis of having been previously diagnosed as TSE affected or not, using routine IHC and Western blot analysis. These samples have also been previously used to test another antigen-retrieval method for PrPsc IHC (Bencsik et al. 2005). Brains slices from C57Bl6 and Tg(ovPrP4) mice inoculated with scrapie or BSE sources were also included in the study (n=45). Briefly, all slides were prepared from formalin-fixed samples (10% in phosphate-buffered saline, 0.1 M for 48 h) that were placed in a bath of formic acid (98-100%; Merck Eurolab, Darmstadt, Germany) for 1 hr at room temperature (RT) to reduce infectivity and then automatically embedded in paraffin (Thermo Electron; Cergy-Pontoise, France). Brain sections of 5-μm thickness were collected on treated glass slides (microslides snow coat, X-tra; Surgipath, Richmond, IL) and kept overnight in an oven at 55C.
PrP Immunohistochemistry
Brain slices were first dewaxed and rehydrated in graded alcohols. We used two different PrP MAbs, SAF84 and 2G11, that require different pretreatments (Andréoletti et al. 2000; Debeer et al. 2002). For SAF84 MAb the pretreatments consisted of placing the sample in a 98-100% formic acid bath for 10 min at RT, followed by hydrated autoclaving for 20 min at 121C in water (Prestige Medical; Blackburn Lane, UK), and finally a proteinase K digestion at 20 μg/ml (Roche Diagnostics; Meylan, France) for 15 min at 37C. For 2G11 MAb, the pretreatment was different and, in particular, the proteinase K digestion step was omitted. Thus, an initial 30-min incubation at RT in a bath of 98-100% formic acid was followed by a 20-min hydrated autoclaving at 121C in a 10-mM citrate buffer (pH 6.1).
After washing in water for 5 min, the slides were ready for the PrP IHC procedure itself, regardless of the PrP MAb used. PrPsc IHC was then performed either as previously described using SAF84 and 2G11 monoclonal primary antibody (0.5 and 2.5 μg/ml, respectively, in PBS 0.1 M, pH 7.4, 0.1% Triton X-100) (Andréoletti et al. 2000; Debeer et al. 2002) or with an additional step using different dilutions (1/20, 1/40, 1/80, 1/160) of streptomycin sulfate (Sigma; Saint Quentin Fallavier, France) at 0.7 M in H2O corresponding to, respectively, 35, 17.5, 8.75, and 4.38 mM. Each concentration was applied just before the primary antibody for 1 hr at RT, followed by three washings at RT using a 0.1 M Tris, 0.384 M glycine, and 0.2% SDS buffer at pH 8.2. Then revelation was achieved using diaminobenzidine intensified with NiCl2 (Biosys; Karben, Germany). Finally, the sections were dehydrated, mounted using Eukitt (Microm; Francheville, France), and observed under a microscope (Olympus; Rungis, France) coupled to an image analysis workstation (Morpho Expert software; Explora Nova, La Rochelle, France). The omission of the primary antibody—substituted by control serum—was used to verify nonspecific background staining in TSE-infected mice, scrapie, and BSE cases. The specificity of positive PrPsc immunolabeling was also assessed using the TSE-negative brains of each species. The protocol was reproducible in all cases.
Results
The intensity of PrPsc immunolabeling using SAF84 MAb was increased when streptomycin sulfate was included in the IHC reaction compared with the labeling intensity in its absence. All concentrations used produced an increased detection of PrPsc signal, without presence of any unspecific background staining. In the majority of cases an 8.75-mM concentration of streptomycin sulfate provided, with excellent contrast, the best enhancement of PrP IHC results, whichever of the three animal species was considered, e.g., in the obex of BSE- (Figures 1A-1D) and scrapie- (Figures 1G-1J) affected animals, as well as in the brain sections of mice experimentally infected with BSE (Figures 1M-1P) or scrapie sources. Compared with this concentration, higher concentrations of streptomycin sulfate did not enhance the detection of PrP. Therefore, this concentration was selected for the combination of streptomycin sulfate with the pretreatments associated with 2G11, another PrP MAb, used for the diagnosis of only the ovine species. Again, when compared with usual technical conditions, the addition of streptomycin sulfate led to an obvious PrPsc IHC signal amplification as shown in the dorsal nucleus of the vagus nerve present in the obex of another scrapie-infected sheep (Figures 1I and 1J), without any unspecific background staining. In the hypothalamus area of a weak-positive ovine case, only five neurons were detectable using 2G11 without streptomycin sulfate (Figure 1G), most often showing pale intracytoplasmic labeling (Figure 1G, inset). Remarkably, the introduction of the streptomycin sulfate allowed clear detection of 25 neurons (Figure 1H), and the labeling of these neurons is clearly illustrated at higher magnification (Figure 1H, inset). The presence of streptomycin sulfate did not induce any unspecific staining as revealed by sections incubated with normal serum, as well as sections from unaffected animals (Figures 1E, 1F, 1K, 1L, 1Q, and 1R). Similarly, the specificity of streptomycin effect was also recognizable at the level of the boundaries between the neuroanatomical areas targeted by PrPsc accumulation and those that are not and that stayed clearly unstained (Figures 1I and 1J).
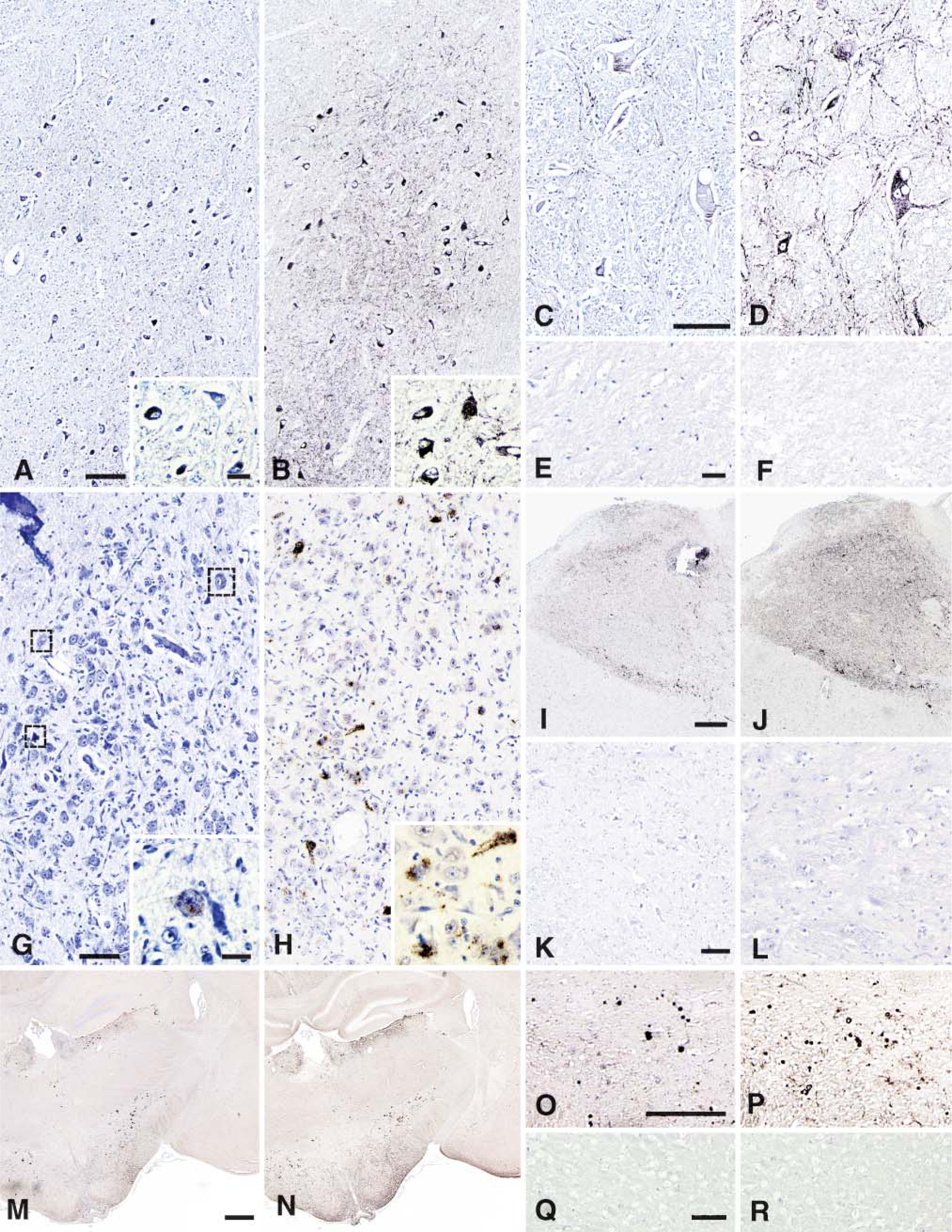
Immunohistochemical detection of PrPsc in three different species: bovine (
Of note, the different types of PrP depositions usually observed using the standard IHC procedures such as granular (Figures 1A-1D, 1G, and 1H), linear (Figures 1D and 1H-1J), intraneuronal (Figures 1A-1D, 1G, and 1H), stellate, perineuronal, or plaques (Figures 1M-1P) were also observed and enhanced in terms of intensity and/or number (i.e., plaques) by the use of streptomycin.
Discussion
The present study clearly indicates that the combination of streptomycin sulfate with the pretreatments currently used in the immunohistochemical diagnosis of animal TSEs as well as in experimental TSE models enables a noteworthy PrPsc signal amplification without any unspecific background staining. This is important, as although various kinds of amplifying systems have been described in combination or used solely, such as antigen-retrieval methods (Shi et al. 1997; Montero 2003; Bencsik et al. 2005) and biotinylated tyramine-based amplification (Köhler et al. 2000; Kim et al. 2003), they are often associated with undesirable nonspecific background staining or loss of tissue adherence. The present amplifying protocol is simple and generates no background staining.
Several dilutions of streptomycin sulfate were successfully used without any background staining. Noticeably, the 8.75-mM concentration regularly gave the most powerful amplification, but further refinement of this parameter may be possible. It is possible and even recommendable to adapt the streptomycin sulfate concentration to be employed in PrP IHC in relation to the nature and the dilution of PrP antibody used, so as to optimize the amplifying effect in each particular case (low positives or TSE atypical cases).
Because in IHC the visualization of an antigen is the result of very complex actions of different parameters such as the state of fixation (chemical bridges binding crucial parts of the proteins), the buffers used (pH, additional components like detergents…), the kind of antigen-retrieval methods used (pH dependent, heated retrieval, enzymatic), and the type of Ab used, it is difficult at this time to propose a simple explanation of the consequential amplifying effect of streptomycin sulfate. The only noticeable data is that the streptomycin-amplifying effect seems to be independent of the PK digestion used in the antigen-retrieval pretreatments. The supposed mechanism of interactions between streptomycin and the prion proteins in vitro is described elsewhere (Moussa et al. 2006) and suggests that it probably occurs through a hydrogen bond transfer between the guanidinium groups present on the streptomycin molecules and negatively charged amino acids present on the surface of the prion protein. If such an interaction between the two occurs in situ, it is plausible to suppose that PrPsc molecules might be locally aggregated by streptomycin sulfate with an improved epitope presentation to the different MAbs used, consequently resulting in an increased detectability of PrPsc in infected tissue sections. Thus, the novel in vitro properties of streptomycin sulfate toward the prion protein allow enhancement of PrPsc IHC sensitivity with either SAF84 or with 2G11 MAbs currently used for the diagnosis and for the analysis of experimental TSEs. As streptomycin sulfate amplified the PrPsc signal in different animal species, and because biochemically it was demonstrated that it binds as well to the human prion protein, it is reasonable to postulate that this amplifying protocol should also be efficient in human prion diseases. Thus, the introduction of streptomycin sulfate in PrP IHC procedure should be used profitably in the diagnosis of the various types of both animal and human TSEs and with particular interest for the detection of subtypes with low levels of PrPsc deposition.
In conclusion, we have established here that it is possible to advance, in a specific manner, the potential of PrPsc IHC beyond its present limitations by use of an attractive and simple amplifying method based on the novel interaction properties between streptomycin sulfate and the prion protein.
Footnotes
Acknowledgements
The authors thank Céline Raynaud and Mikaël Leboidre for excellent histotechnical assistance.
