Abstract
We investigated the effects of pH and ionic strength of solutions used for antigen retrieval to elucidate the mechanism of heat-induced antigen retrieval (HIAR) in immunohistochemistry. The immunostaining intensity of nuclear, cytoplasmic, cell membrane, and extracellular matrix antigens with 17 different antibodies was evaluated in formaldehyde-fixed and paraffin-embedded mouse and human tissues. Deparaffinized sections were autoclaved for 10 min in buffers with different pH values ranging from 3.0 to 10.5. To test the influence of ionic strength on immunoreactions, the sections were autoclaved for 10 min in 20 mM Tris-HCl buffers (TB) at pH 9.0 and 10.5 with or without 25, 50, and 100 mM NaCl. There were two immunostaining patterns for pH dependency of HIAR. First, the majority of antibodies recovered their antigenicity when heated in the buffers with both acidic pH (pH 3.0) and basic pH (pH 9.0 and 10.5). Second, some antibodies showed strong immunostaining only at basic pH values (pH 9.0 and 10.5). When the sections were autoclaved in TB at pH 9.0, immunostaining of all eight antibodies examined decreased as the NaCl concentration increased. On the other hand, when the sections were treated with TB at pH 10.5, all antibodies yielded stronger reactions in the buffer containing NaCl than in the buffer without NaCl; five antibodies exhibited the strongest immunoreaction at concentrations from 25 to 50 mM. These results suggest that the extended polypeptides by heating are charged negatively or positively at basic or acidic pH, and that an electrostatic repulsion force acts to prevent random entangling of polypeptides caused by hydrophobic attractive force and to expose antigenic determinants, during cooling process of HIAR solution.
Keywords
A
Investigators have tried to select the most suitable conditions (temperature, pH, buffers, and additives) for heating each antigen, because the mechanisms of HIAR are not well understood. Based on studies in which purified proteins treated with formaldehyde were analyzed by SDS-PAGE after heating, Rait et al. (2004) and we (Yamashita and Okada 2005a) have recently demonstrated that heat treatment cleaves the intra- and intermolecular crosslinks (i.e., methylene bridges) induced with formaldehyde. We have also demonstrated by Western blotting that β-actin and fibronectin can be extracted from autoclaved paraffin sections (Yamashita and Okada 2005a). These studies suggest that the main mechanism of HIAR is disruption of methylene bridges.
Citrate buffer (pH 6.0) is the most popular solution for HIAR (Shi et al. 1993; Brown and Chirala, 1995), but several investigators have reported that the efficiency of HIAR is highly dependent on the pH of the retrieval solutions (Shi et al. 1995; Evers and Uylings 1997; Ferrier et al. 1998; Brorson 2002). Shi et al. (1995) reported three patterns of pH-influenced HIAR immunostaining: type A, almost constant staining, with only a slight decrease in staining intensity between pH 3.0 and pH 6.0; type B, a dramatic decrease in staining intensity between pH 3.0 and pH 6.0; and type C, increasing intensity of immunostaining correlated with increasing pH values of the HIAR solution. Yamashita and Okada (2005a) showed that intensity of immunoreactions obtained by heating in buffer is reversibly changed by successive heating in another buffer with different pH. Nine of 10 antigens exhibited much stronger immunoreaction in sections heated in Tris-HC1 buffer (TB) at pH 9.0 than in those heated in TB at pH 6.0 (Yamashita and Okada 2005a). A second heating at pH 6.0 significantly decreased immunostaining of the antigens that had been boiled at pH 9.0, but the immunostaining was restored after a third heating at pH 9.0. These findings suggest that the pH of the buffers is a factor that is essential for proper refolding of antigens or exposure of antigenic determinants so that they are capable of reacting with antibodies.
In the present study, we investigated the effect of the pH and ionic strength of the retrieval solution on HIAR in formaldehyde-fixed and paraffin-embedded specimens to elucidate the mechanisms of HIAR in detail.
Materials and Methods
Reagents and Tissue Preparation
Table 1 shows the antibodies used in this study and their sources, abbreviations, and dilutions. Tissues from mature CD-I mice (8 weeks old) were fixed with 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4) for 6 hr or with 10% formalin for 24 hr, and washed with PBS (10 mM phosphate buffer, pH 7.4 containing 0.85% NaCl) overnight at 4C. They were then dehydrated with graded alcohols, cleared in xylene, and embedded in paraffin. Routinely formalin-fixed (20% formalin), paraffin-embedded sections of human colon and uterus were obtained from file at Department of Pathology, School of Medicine, Keio University.
Influence of pH in Immunohistochemistry
Heat Treatment. Paraffin sections (4 μm) mounted on a new silane-coated slide glass (Muto Pure Chemicals Co., Ltd.; Tokyo) were deparaffinized and washed with distilled water. They were then soaked and autoclaved for 10 min in the following buffers at 120C: 20 mM glycine-HCl buffer (pH 3.0), 20 mM citrate buffer (pH 4.5 or pH 6.0), or 20 mM TB buffer (pH 7.5, pH 9.0, or pH 10.5). They were allowed to cool to room temperature and were washed with PBS. Some sections were immunostained without autoclaving. Sections were also stained with hematoxylin and eosin after autoclaving to assess the preservation of tissue structure.
Immunohistochemical Staining. The sections were treated with PBS containing 1% BSA and 10% block ace (Dainippon Pharmaceutical Ltd.; Osaka, Japan) at room temperature for 1 hr, and then with the primary antibodies overnight at 4C. After washing with PBS, the sections were incubated with labeled polymer horseradish peroxidase anti-rabbit or labeled polymer horseradish peroxidase anti-mouse (DakoCyto-mation; Carpinteria, CA) for 1 hr at room temperature. Peroxidase enzyme activity was visualized with imidazole-3, 3'-diaminobenzidine tetrahydrochloride solution. Normal rabbit IgG or mouse IgG was used for the controls in place of the primary antibodies. For the control of estrogen receptor (ER)α, ERβ, glucocorticoid receptor (GR), and androgen receptor (AnR) immunostaining, antibodies absorbed with the respective antigenic peptides were used. Some sections were counterstained with hematoxylin.
Primary antibodies used
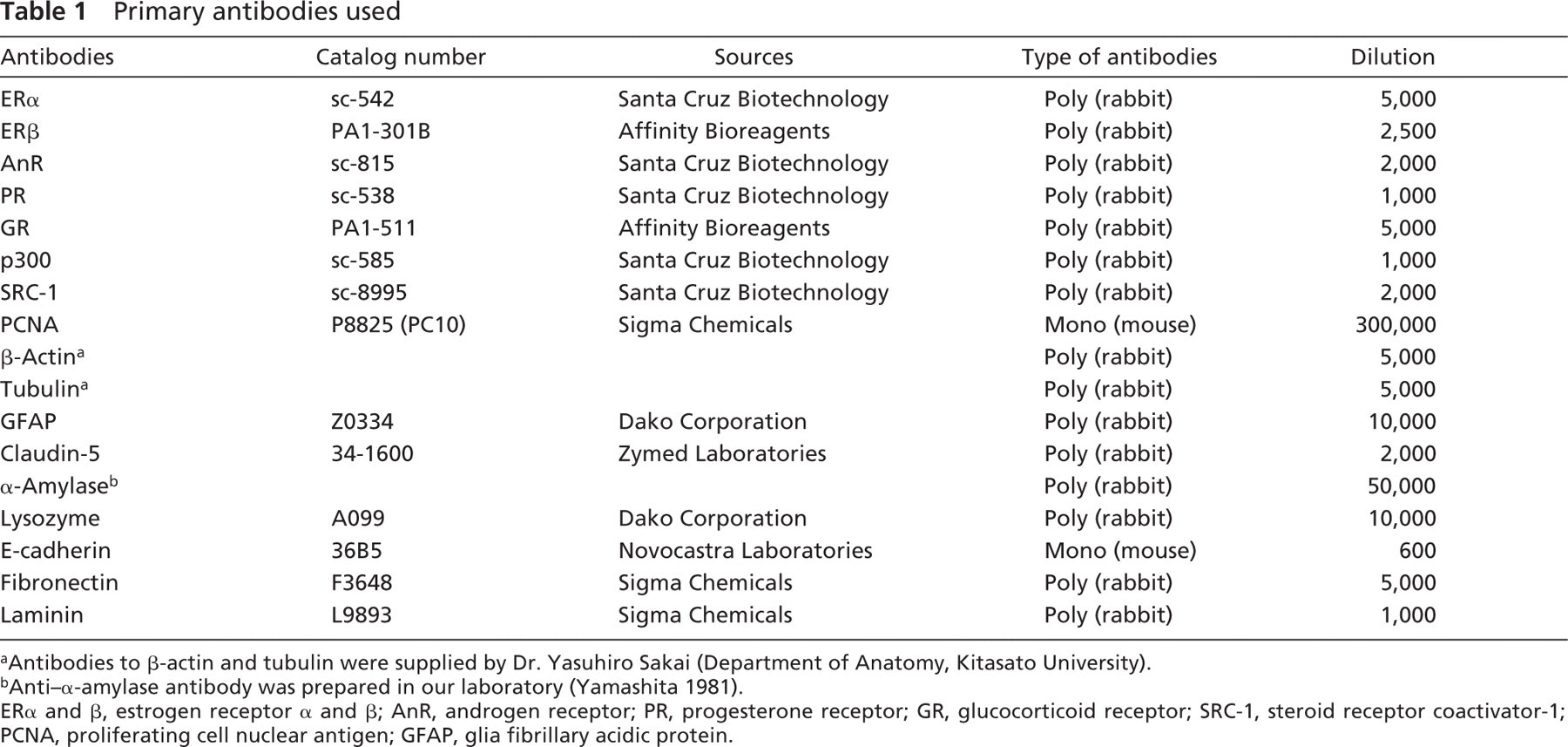
Antibodies to β-actin and tubulin were supplied by Dr. Yasuhiro Sakai (Department of Anatomy, Kitasato University).
Anti-α-amylase antibody was prepared in our laboratory (Yamashita 1981).
ERα and β, estrogen receptor α and β; AnR, androgen receptor; PR, progesterone receptor; GR, glucocorticoid receptor; SRC-1, steroid receptor coactivator-1; PCNA, proliferating cell nuclear antigen; GFAP, glia fibrillary acidic protein.
Influence of Ion Strength in Immunohistochemistry
Tissues fixed with 4% paraformaldehyde for 6 hr were used in this examination. Deparaffinized sections were auto-claved for 10 min in 20 mM TB (pH 9.0 and pH 10.5) containing 0 mM, 25 mM, 50 mM, or 100 mM NaCl, and immunohistochemical staining was carried out according to the procedures described previously. Staining for eight antigens located in the nucleus, cytoplasm, cell membrane, or extracellular matrix was assessed in this study: ERα, AnR, GR, β-actin, tubulin, claudin-5, fibronectin, and laminin.
Results
Effects of pH on Cell Structures
When sections were autoclaved in glycine-HCl buffer, pH 3.0, the nuclear components of several cell types were extracted from the tissues fixed with 4% paraformaldehyde for 6 hr; in particular, the nuclei of the epithelial cells of the genital tract and intestine and of the liver cells were severely damaged (Figure 1B): in the tissues fixed with formalin for 24 hr, partial destruction of nuclear structure was observed in these cells. Small amounts of secreted materials remained in the lumens or ducts of the coagulating gland, seminal vesicles, and pancreas, and the cytoplasmic staining with eosin in many cell types was weak in the autoclaved sections at pH 3.0 compared with unautoclaved sections (Figures 1A and 1B). The nuclear components of some cell types were partially extracted in the specimens fixed for 6 hr after autoclaving in TB at pH 10.5 (Figure 1F). In the human specimens, the nuclear structure was preserved after autoclaving in any buffers. As the pH of the buffers was more basic, staining with hematoxylin became weaker than in sections treated with buffers at pH 4.5, pH 6.0, and pH 7.5 (Figures 1B-1F).
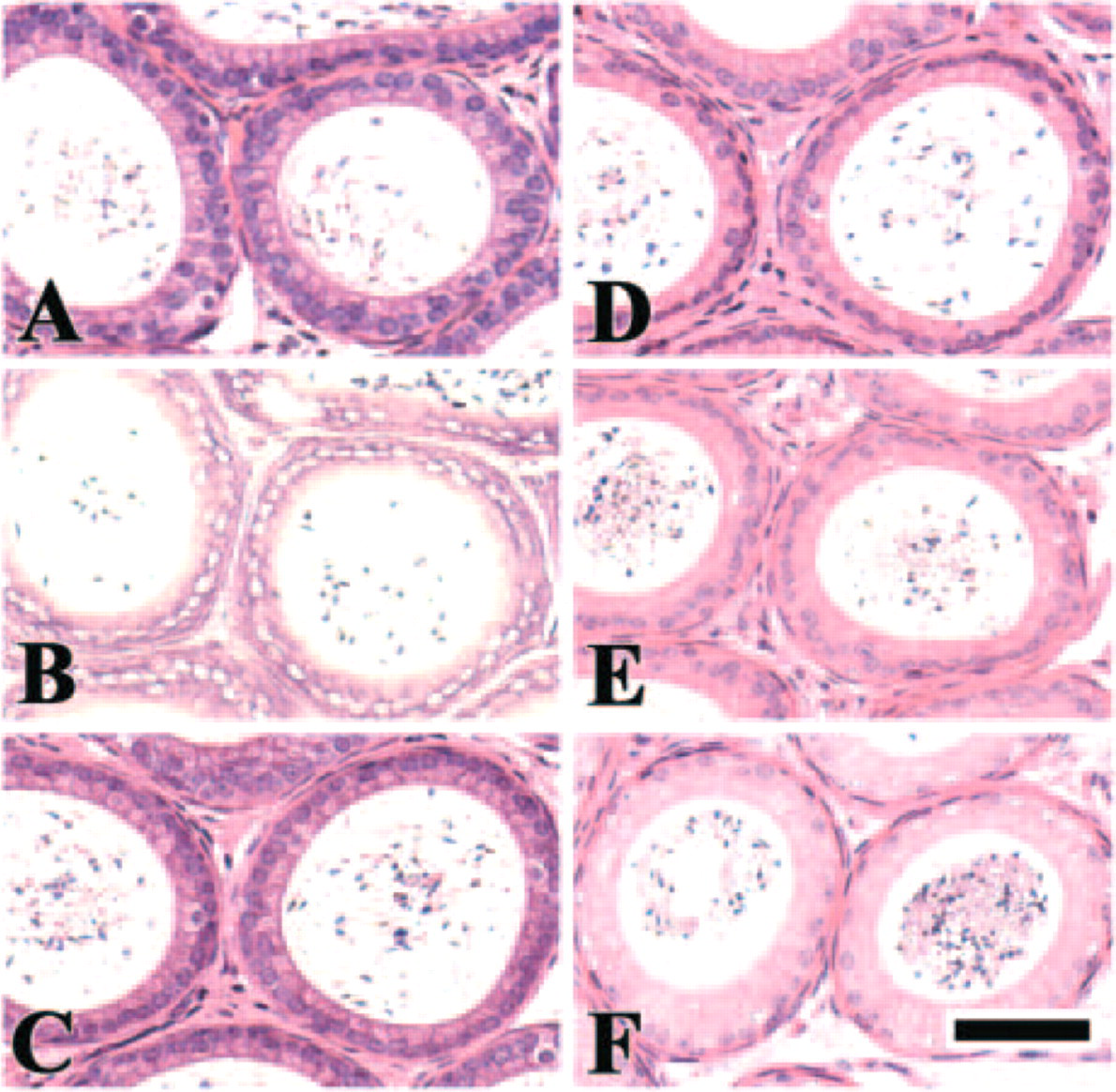
Effects of pH on cell structures. Mouse epididymis was fixed with 4% paraformaldehyde for 6 hr and embedded in paraffin. Deparaffinized sections were autoclaved at 120C for 10 min in the following buffers: 20 mM glycine-HCl buffer, pH 3.0 (
Effects of pH on HIAR
The effects of pH on HIAR in the mouse tissues are summarized in Table 2. All antigens examined recovered their antigenicity after heating in the tissues fixed with 4% paraformaldehyde for 6 hr. However, in the tissues fixed with 10% formalin for 24 hr, antibody to lysozyme yielded the strongest reaction in the sections without autoclaving. In the tissues fixed for 6 hr, immunoreactions for E-cadherin, lysozyme (Figures 2P-2T), and fibronectin were positive when HIAR was carried out in buffers at pH 9.0 and pH 10.5, and immunoreaction for p300 was detected only after HIAR at pH 9.0 (Table 2). Immmunoreaction for steroid receptor coactivator-1 was positive in the buffers at pH 6.0 and 7.5, but remarkably increased when autoclaved at pH 9.0 (Table 2). However, the immunoreactions for other 12 antigens were strong when heated in buffers at pH 3.0, pH 9.0 and pH 10.5, and the reactions for these antigens were negative to weak when the sections were autoclaved in the buffers at pH 4.5, pH 6.0, or pH 7.5 (Figures 2A-2O, Table 2). In the tissues fixed for 24 hr, the pattern of pH-dependent HIAR in each antigen was almost the same as that in the tissues fixed for 6 hr, although, at pH 6.0, immunostaining of most antigens was stronger than in those fixed for 6 hr.
Effects of pH on HIAR in Human Tissues
ERα and progesterone receptor (PR) were immunostained in the human uterus, and proliferating cell nuclear antigen (PCNA), glia fibrillary acidic protein (GFAP) and E-cadherin were localized in the colon. Antibody to ERα exhibited no reaction in any cell types in the human uterus. However, PR, PCNA, GFAP, and E-cadherin showed similar pH-dependent HIAR patterns to those found in mouse tissues: they showed no immunoreaction without heating. Immunostaining for PR, PCNA, and GFAP was retrieved when autoclaved at pH 3.0, pH 9.0, and pH 10.5 (Figures 3A-3G), and a small peak of immunostaining was recognized at pH 6.0. E-cadherin immunoreaction was detected when heated only at basic pH (pH 9.0 and pH 10.5) (Figures 3H-3N).
pH dependency of HIAR
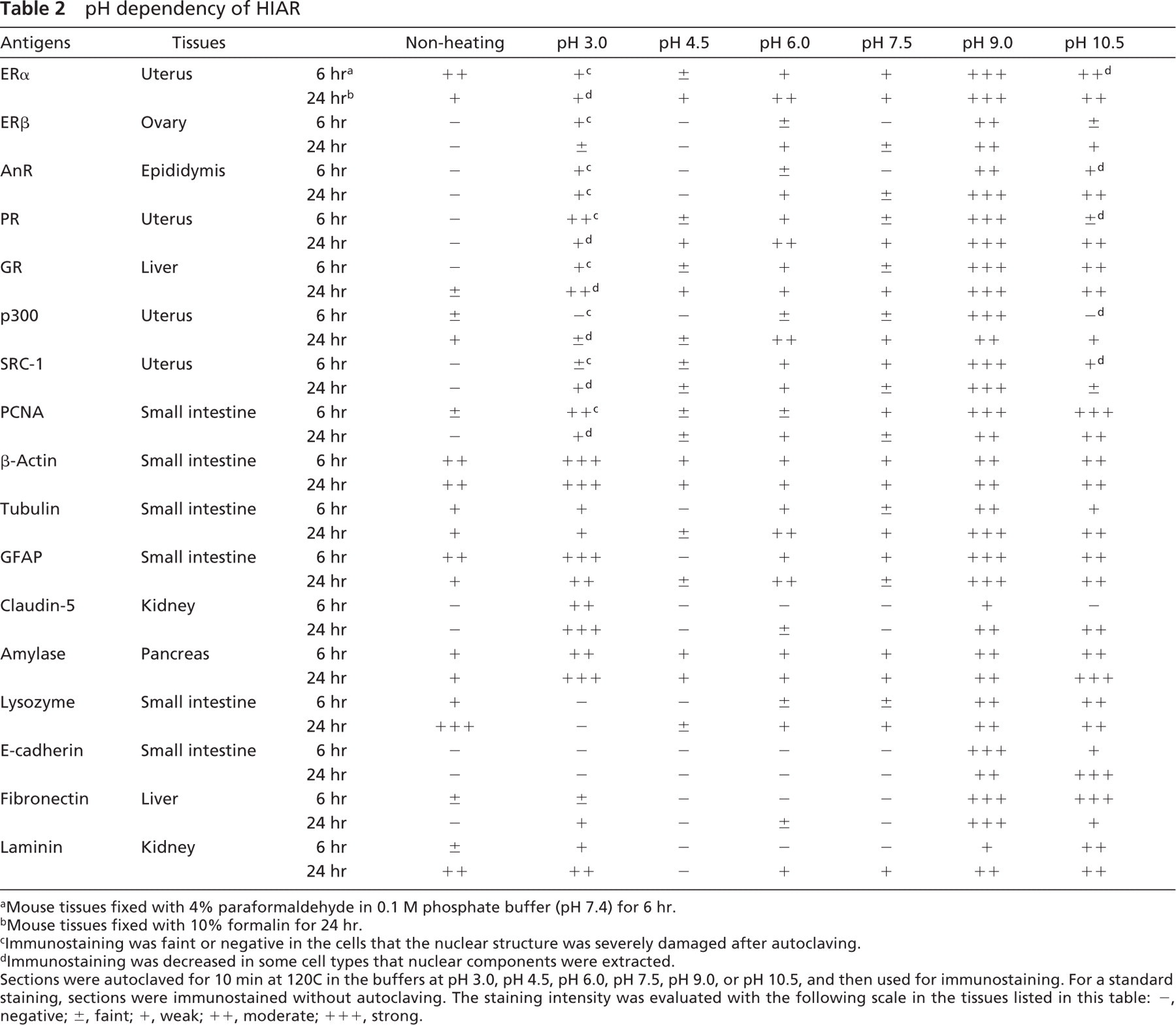
Mouse tissues fixed with 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4) for 6 hr.
Mouse tissues fixed with 10% formalin for 24 hr.
Immunostaining was faint or negative in the cells that the nuclear structure was severely damaged after autoclaving.
Immunostaining was decreased in some cell types that nuclear components were extracted.
Sections were autoclaved for 10 min at 120C in the buffers at pH 3.0, pH 4.5, pH 6.0, pH 7.5, pH 9.0, or pH 10.5, and then used for immunostaining. For a standard staining, sections were immunostained without autoclaving. The staining intensity was evaluated with the following scale in the tissues listed in this table: -, negative; ±, faint; +, weak; ++, moderate; +++, strong.
Effects of Ionic Strength on HIAR
The effects of NaCl concentration in 20 mM TB at pH 9.0 and pH 10.5 on HIAR was investigated in the tissues fixed with 4% paraformaldehyde for 6 hr in regard to eight antigens: ERα, GR (Figures 4I-4P), AnR, β-actin, claudin-5 (Figures 4A-4H), tubulin, laminin, and fibronectin. Most antigens exhibited stronger immunoreaction when heated in NaCl-free TB at pH 9.0 than at pH 10.5. All antigens in the sections autoclaved in TB buffer (pH 9.0) reacted most strongly when treated with the buffer that did not contain NaCl (Figure 5). The staining intensity of all antigens in 20 mM TB at pH 9.0 decreased as the concentration of NaCl rose (Figure 5). In the sections heated in TB at pH 10.5, the reactions for all antigens were the strongest when heated in the buffer containing NaCl (Figure 5). The staining intensity of five antigens—GR (Figure 4O), β-actin, claudin-5 (Figure 4G), laminin, and fibronectin—peacked when autoclaved in the buffer containing 25–50 mM NaCl (Figure 5). Immunoreaction of ERα increased as the concentration of NaCl rose, whereas that of AnR and tubulin was almost constant from 25 mM to 100 mM NaCl (Figure 5). When NaCl was added to TB at pH 10.5, nonspecific background staining also tended to increase.
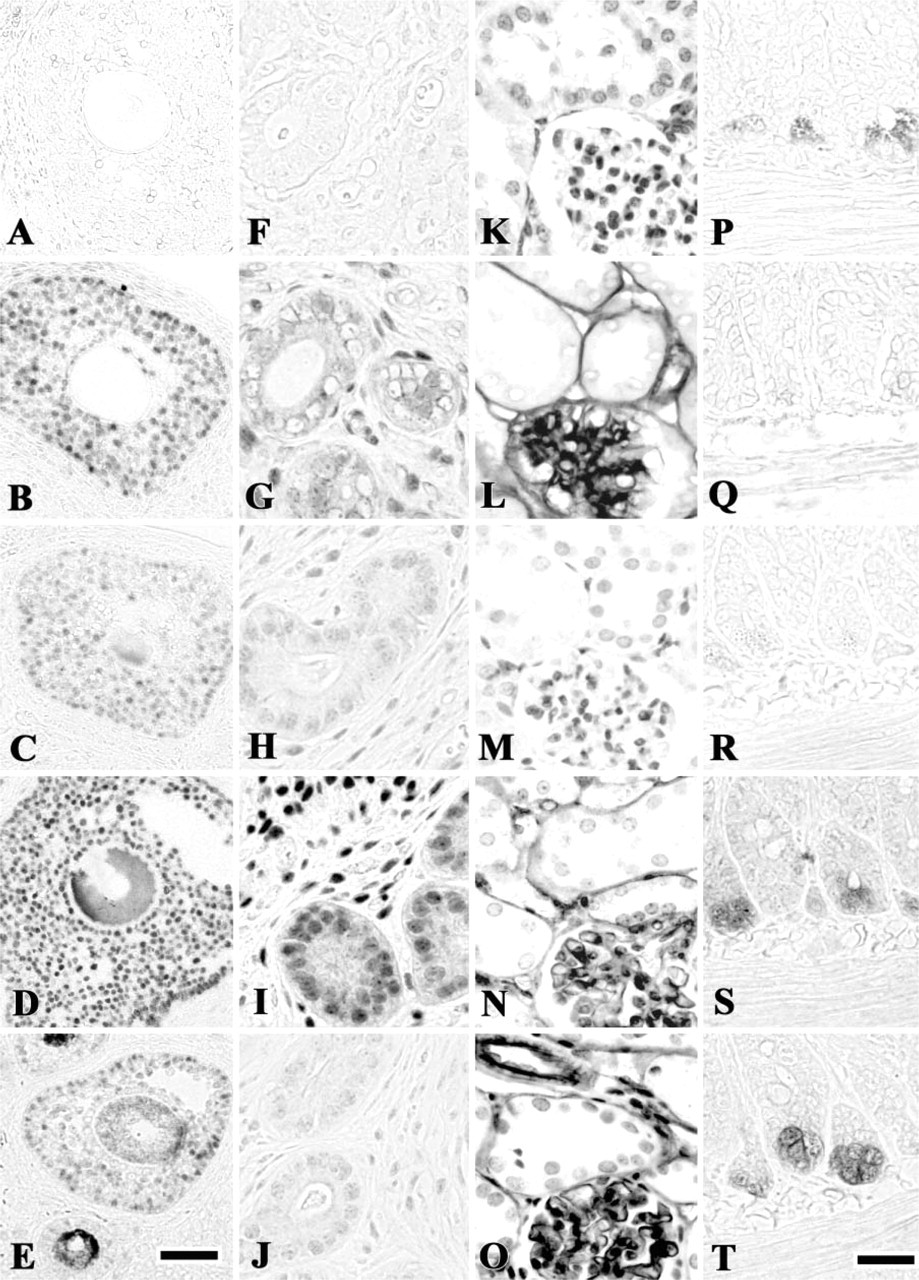
Effects of pH on heat-induced antigen retrieval. Sections from tissues fixed with 4% paraformaldehyde for 6 hr were autoclaved at 120C for 10 min in the following buffers and then immunostained: 20 mM glycine-HCI buffer (pH 3.0) (
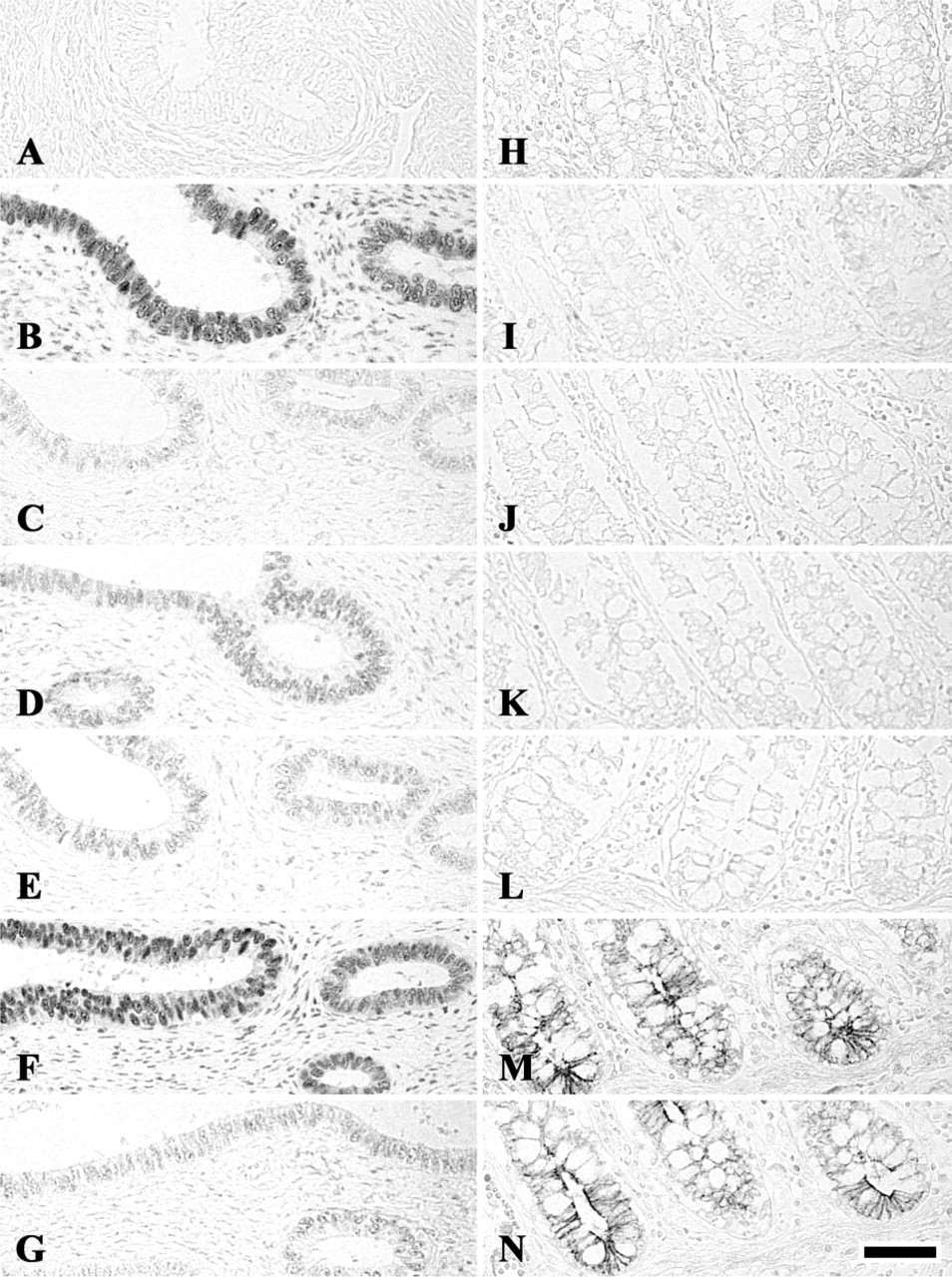
Effects of pH on heat-induced antigen retrieval in the human tissues. Progesterone receptor (
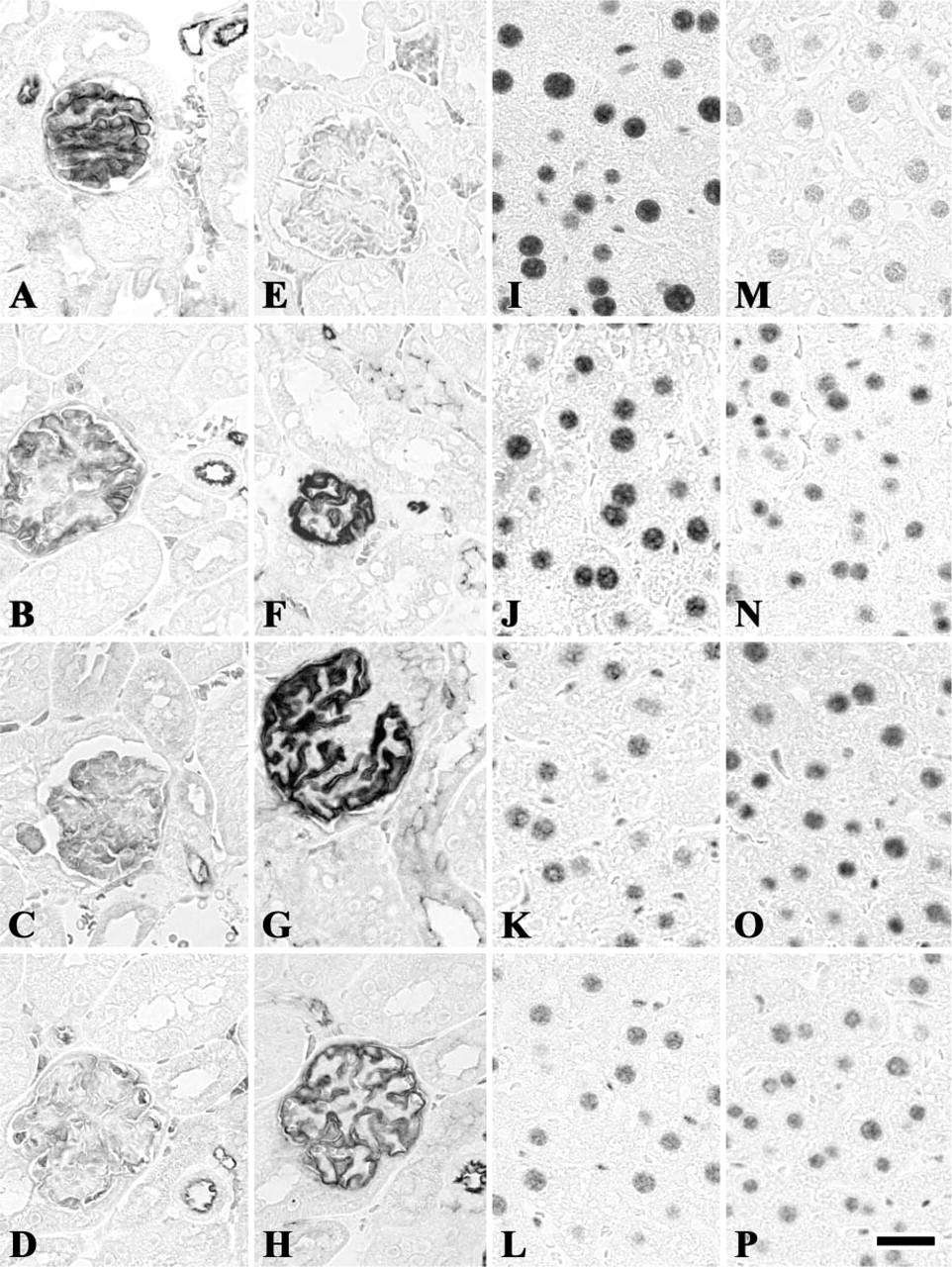
Effects of ionic strength on heat-induced antigen retrieval. Claudin-5 (
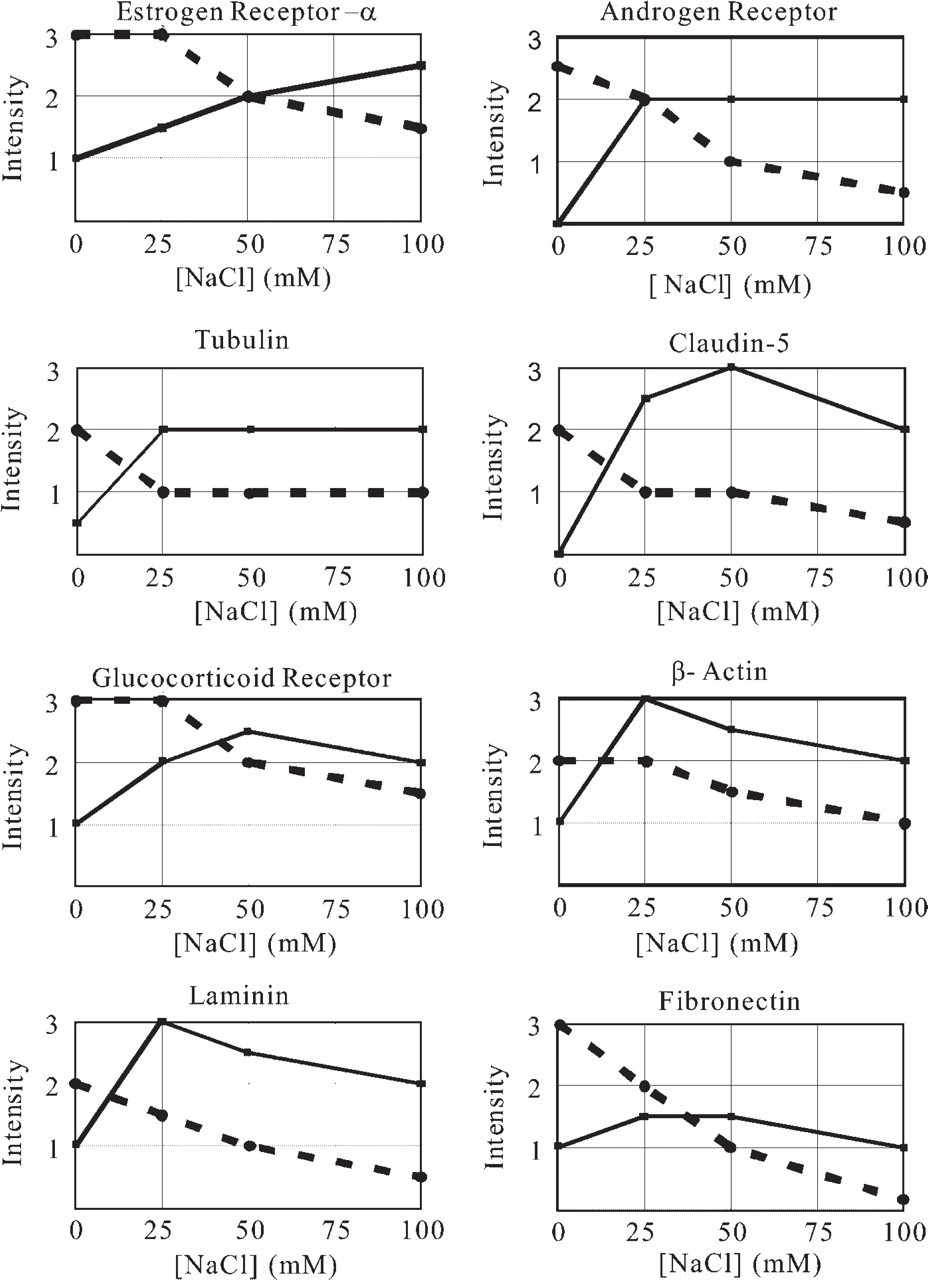
Profile of effects of ionic strength to heat-induced antigen retrieval. Sections were treated according to the procedure described in Figure 4. The following antigens were immunostained: estrogen receptor α in the uterus; androgen receptor in the epididymis; tubulin in the small intestine; claudin-5 in the kidney; glucocorticoid receptor in the liver; β-actin in the small intestine; laminin in the kidney; and fibronectin in the liver. Assessment of immunostaining with a scale (0–3) was used: 0, negative; 1, weak; 2, moderate; 3, strong. Dotted and solid lines display immunostaining intensity of antigens autoclaved in TB (pH 9.0) and TB (pH 10.5) containing various concentrations of NaCl, respectively. All antigens show the strongest immunoreaction when sections were autoclaved in NaCl-free TB at pH 9.0 (dotted lines), but they yield the strongest staining when sections were heated in TB containing NaCl at pH 10.5 (solid lines).
Discussion
The antigenicity of the majority of antigens was effectively restored when the specimens fixed with 4% paraformaldehyde for 6 hr or 10% formalin for 24 hr were heated in the buffers at pH 3.0 and pH 9.0 (Table 2). This pattern of HIAR may correspond to the type-B pattern (V-form type) in the classification by Shi et al. (1995). E-cadherin and lysozyme exhibited strong immunoreaction when heated in basic buffers, showing a pattern similar to type C (ascending type) noted by Shi et al. (1995) (Table 2). Ferrier et al. (1998) reported that immunostaining of tissue-type plasminogen activator and plasminogen activator inhibitor type 1 is positive only after heating in retrieval solution at pH 2.5 and pH 10.0. Evers and Uylings (1997) recommended TB at pH 9.0–9.5 for HIAR of several neuronal antigens. In our present study, type B was the primary pattern in HIAR, although Shi et al. (1995) did not describe the proportion of antigens showing this pattern. The α-amylase immunostaining was almost constant regardless of the pH value of the buffers used for HIAR when the antiserum was diluted 5000-fold (Yamashita and Okada 2005a), but it exhibited the type B pattern when diluted 50,000-fold in the present study. If highly diluted antibodies were used for immunohistochemistry in the study by Shi et al. (1995), the type A (stable type) likely became equivalent to type B. In the human tissues routinely fixed with 20% formalin and embedded in paraffin, PR, PCNA, and GFAP showed type B pattern, and E-cadherin exhibited type C pattern. These results suggest that the pH dependency of HIAR of each antigen is maintained over species when the tissues are immunostained with the same antibody.
Many biochemical studies have demonstrated that unfolded polypeptides treated with denaturants, such as urea or guanidine hydrochloride, readily self-associate or randomly associate with other proteins in the solution on removal of the denaturants. Hydrophobic force is thought to be the principal driving force for the protein aggregation (Jaenicke and Seckler 1997; Fink 1998; Hanson and Gellman 1998). Polypeptides are tightly packed and neighboring polypeptides crossly contact each other in the tissues, particularly in the nuclei, secretory granules, and fibrous structures. We hypothesized that the pH dependency of HIAR is based on the following mechanisms (Figure 6). When methylene bridges are cleaved by heating, the higher order structure of antigen molecules is destroyed and polypeptide chains extend to expose both hydrophobic and hydrophilic portions. Antigenic determinants are concealed at around neutral pH, because attractive forces caused by the hydrophobic and ionic forces act strongly and entangle neighboring polypeptides randomly as the solutions cool. At basic or acidic pH values, however, the majority of extended polypeptides are charged negatively or positively and an electrostatic repulsion force acts to prevent random aggregation of polypeptides based on the hydrophobic force.
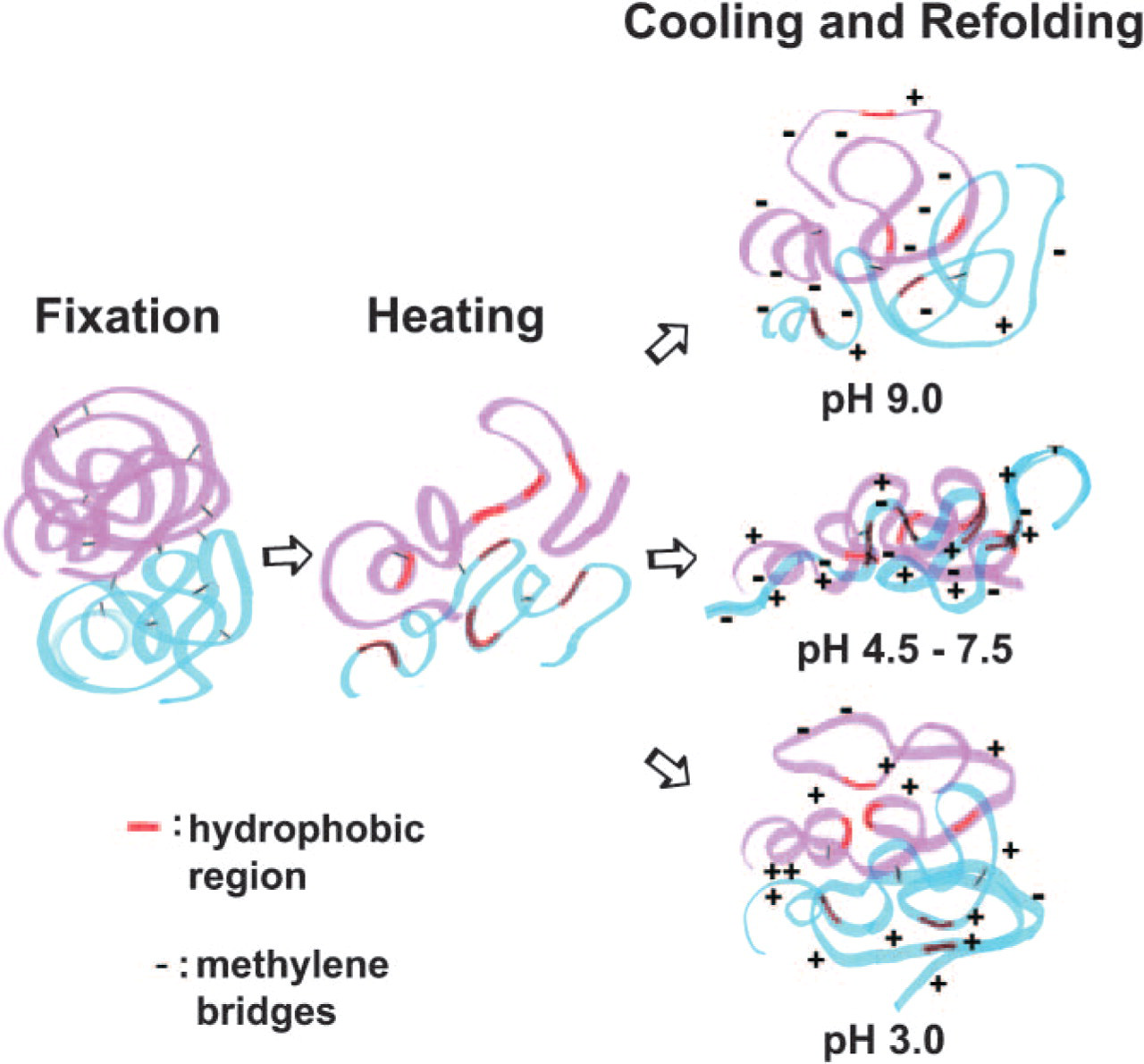
Schematic of heat-induced antigen retrieval mechanisms. After heating, methylene bridges produced by formaldehyde are cleaved and polypeptide chains extended to expose their hydrophobic regions. During cooling process, the polypeptide chains rapidly refold. At pH 3.0 and pH 9.0, hydrophobic attractive force and electrostatic repulsion force based on positively or negatively charged polypeptides may balance to prevent intertwining of polypeptide chains and expose antigenic determinants for antigen-antibody interaction. On the other hand, at pH 4.5–7.5, ionic and hydrophobic attractive forces may cooperatively act and neighboring polypeptides entangle each other to hidden antigenic determinants.
Citrate buffer at pH 6.0 is the most popular solution for HIAR. It showed relatively good HIAR efficiency for many antigens; in particular, in the tissues fixed with 10% formalin for 24 hr, whereas it was not optimal buffer for all antibodies examined. Similar results were reported by Shi et al. (1996). Citrate buffer may reduce the ionic or hydrophobic forces at pH 6.0, because citrate buffer yielded stronger immunoreaction for many antibodies than Tris-HCl buffer at pH 6.0 (Yamashita and Okada 2005a).
To test the hypothesis described previously, we immunostained sections heated in TB at pH 9.0 or pH 10.5 containing 25–100 mM NaCl to reduce the electrostatic forces. At pH 9.0, all antigens exhibited the strongest immunostaining when autoclaved in TB without NaCl. At pH 10.5, on the other hand, all antigens showed stronger immunostaining when heated in TB containing NaCl than in NaCl-free TB. Five antigens exhibited the strongest immunostaining when heated in TB containing 25–50 mM NaCl. Stempfer et al. (1996) reported similar results using in vitro system in which α-glucosidase with extended hexa-arginine peptide was denatured with 8 M urea and refolded on a polyanionic matrix. Refolding was only feasible at moderate salt concentrations (30–40 mM NaCl) at pH 7.6. Refolding of the enzyme was prevented by strong ionic attraction between the protein and the matrix, when the denaturant was removed from the solution at very low salt concentration. At high salt concentrations (>100 mM NaCl), refolding was inhibited by hydrophobic interactions between unfolded or partially folded protein and the matrix. The refolding process of polypeptides in the tissues may more closely resemble the system with the matrices rather than in solution.
These biochemical findings support our hypothesis concerning pH-dependent HIAR. At around pH 9.0, there may be a balance between the hydrophobic force and the ionic repulsion force by negatively charged proteins that prevents intertwining of unfolded polypeptide chains and maintains a suitable conformation of many epitopes to interact with antibodies in the tissues. At pH 9.0, increasing ionic strength reduces electrostatic repulsion and the hydrophobic force causes the polypeptide to become entangled and subsequently reduces immunostaining. At pH 10.5, on the other hand, reduction of electrostatic force in the buffer containing NaCl probably moderates the denaturation of polypeptides based on the highly negatively charged polypeptide chains and yields stronger immunoreactions than in buffer that does not contain NaCl. Recently, Namimatsu et al. (2005) have demonstrated that heating in citraconic anhydride solution at neutral pH is a universal antigen retrieval method. Their results may also support our hypothesis. Citraconic anhydride modifies ∊-amino groups of lysine residues, places numerous negative charges on proteins at neutral pH, and elicits conformational changes of proteins (Dixon and Perham 1968; Batra 1991; Mir et al. 1992). In their antigen-retrieval method, it is postulated that heating exposes ∊-amino groups of lysine modified with formaldehyde and the ∊-amino groups are citraconylated, and that the proteins in the tissues are charged negatively at neutral pH. Then, the citraconyl groups may be gradually removed from proteins. Therefore, electrostatic repulsion by negative charges and hydrophobic attraction may have a balance to expose epitopes during citraconylation at neutral pH.
Footnotes
Acknowledgements
This work was supported in part by a Grant-in-Aid (16590154) for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
We are grateful to Satoshi Kusakari, Hitoshi Abe, Minako Suzuki, Kiyora Nakajima, and Yuko Hashimoto for their technical assistance.
