Abstract
We studied the effectiveness of heat-induced antigen retrieval (HIAR) in conventionally processed, epon-embedded specimens and the mechanisms of HIAR in the specimens. Frozen sections were first immunostained to examine the possibility of using HIAR for 18 antigens to avoid the effects of epoxy resin embedment. The antigenicity of 7 out of 18 antigens was retrieved with glutaraldehyde fixation followed by osmium tetroxide treatment whereas none were retrieved with glutaraldehyde fixation without post-osmication. Six antigens also exhibited positive immunostaining in semi-thin epon sections when the sections were deplasticized with sodium ethoxide followed by autoclaving. In the immunoelectron microscopy with the post-embedding method, positive reactions with fine ultrastructures were obtained using HIAR without deplasticization. These results suggested that osmium tetroxide binds to ethylene double bonds (which are introduced into protein crosslinks by glutaraldehyde) and forms an extremely stable resonance interaction with the Schiff bases, thus destabilizing the protein crosslinks. Heating also further degrades these crosslinks. The present study demonstrated that archival epon blocks can be useful resources for immunohistochemical studies for both light and electron microscopy.
Keywords
Introduction
Immunoelectron microscopy is a powerful technique for localizing antigens in cell organelles and for studying the functions of antigens by demonstrating the relationship between antigens and other macromolecules. The post-embedding method provides more reproducible and reliable immunostaining results than the pre-embedding method, which can obscure antibody access to the antigenic sites in the tissues, because immunoreactions occur on the surfaces of ultrathin sections. Specimens fixed with aldehyde and embedded in acryl resins such as LR-White and Lowicryl K4M have been used for post-embedding immunoelectron microscopy to preserve the antigenicity and provide easy access for antibodies to antigens; although, suitable fixatives and fixation procedures should be selected for each antigen (Roth et al. 1981; Newman and Hobot 1987; Yamashita et al. 2009).
Archives of materials embedded in epoxy resins are collected in many histology and pathology laboratories and in hospitals for morphological analyses, and these archives are expected to provide valuable data if they are applicable for immunohistochemical studies. However, conventionally processed specimens for transmission electron microscopy are thought to be inadequate for immunoelectron microscopy with post-embedding methods for the following reasons. 1) Glutaraldehyde strongly induces crosslinks in proteins and significantly suppresses antigen-antibody interactions. In addition, heat-induced antigen retrieval (HIAR) is ineffective for most antigens, because the glutaraldehyde-induced crosslinks are very stable compared with those induced by formaldehyde (Kawahara et al. 1997; Marquie 2001). 2) Osmium tetroxide severely destroys the antigenicity of most antigens by cleaving polypeptides in tryptophan residues and oxidizing methionine to methionine sulfone and cysteine to cysteic acid (Spande et al. 1970; Nielson and Griffith 1979; Deetz and Berhrman 1981). Furthermore, the adduction of osmium tetroxide to these and other functional groups of amino acids likely inhibits antigen-antibody interactions. 3) Proteins reportedly react with monomers of epoxy resins and co-polymerize with the resin (Kellenberger et al. 1987). Therefore, the frequent separation of epitopes at the cutting faces of ultrathin sections is assumed to occur. 4) Epoxy resins produce tight three-dimensional crosslinks of a hydrophobic nature, which may suppress antigen-antibody interactions on the surface of the cutting face of ultrathin sections.
Despite these disadvantages, attempts have been made to use conventionally processed materials for immunoelectron microscopy, and successful results have been obtained for some antigens. The removal of epoxy resin can be performed by treating the sections with hydrogen peroxide aqueous solutions or sodium/potassium ethoxide in ethanol (Bettica and Johnson 1990; Röcken and Roessner 1999; Coleman et al. 2006). The oxidation and removal of osmium is performed by treatment with a sodium metaperiodate aqueous solution (Bendayan and Zollnger 1983; Hearn et al. 1985; D’Alessandro et al. 2004). HIAR is reportedly useful for ultrathin sections (Röcken and Roessner 1999; Brorson 1998; Yano et al. 2003; Brorson 2004; Fossmark et al. 2005), since HIAR has been introduced to formalin-fixed, paraffin-embedded tissues as the most common method for antigen-retrieval (Shi et al. 1991; Cattoretti et al. 1993; Pileri et al. 1997; Yamashita 2007). The combined use of these procedures has also been examined for light and electron microscopy (Mar et al. 1987; D’Alessandro et al. 2004; Coleman et al. 2006; Zhai et al. 2007). The application of immunohistochemical techniques to semi-thin sections of epoxy resin-embedded specimens would also help to improve antigen localization because of the excellent preservation of morphological details compared with paraffin-embedded specimens.
Immunohistochemistry using epon-embedded materials is difficult, as mentioned above, and optimization is still being attempted for each antigen at both the light and electron microscopic levels. In the present study, the effectiveness of HIAR in the post-embedding method using conventionally processed epon-embedded specimens was systematically examined for 18 antigens, and the mechanisms of HIAR in the specimens were investigated through the following studies. First, frozen sections fixed with glutaraldehyde or with glutaraldehyde and osmium tetroxide in succession were dehydrated with ethanol and then rehydrated, and HIAR was then examined to avoid the effects of epoxy resin embedment. Second, the most effective procedure for the frozen sections was applied to semi-thin sections from conventionally processed specimens after partial removal of resin with sodium ethoxide. Third, HIAR was applied to ultrathin sections for immunoelectron microscopy, in which the procedure used for the preparation of light microscopy specimens was modified to conserve the ultrastructure with a good image contrast and to obtain a strong immunoreaction. Fourth, proteins (ribonuclease A [RNase A] and ovalbumin) were crosslinked with glutaraldehyde, glutaraldehyde and osmium tetroxide, or osmium tetroxide and then analyzed using SDS-PAGE after heat treatment to elucidate the mechanisms of HIAR in the sections fixed with glutaraldehyde and post-fixed with osmium tetroxide.
Materials & Methods
Reagents
RNase A (R-5250) and ovalbumin (A-2512) were purchased from Sigma-Aldrich Chemicals (St. Louis, MO). Glutaraldehyde (25%), osmium tetroxide (4%), epoxy resin (Quetol 812 set), and Immunosaver were purchased from Nissin EM Corp. (Tokyo, Japan). Can Get Signal A (CGS-A) and Can Get Signal B (CGS-B) were purchased from Toyobo Co., Ltd. (Osaka, Japan). Saturated sodium ethoxide was prepared by dissolving sodium hydroxide in ethanol. The solution was stored at room temperature in the dark and was used for the etching of epon sections after 10–40 days.
Table 1 shows the antibodies used in this study and their sources, abbreviations, and dilutions for immunostaining on frozen sections, semi-thin epon sections and ultrathin sections. Envision plus/horseradish peroxidase for rabbit and mouse IgG were purchased from Dako (Carpinteria, CA). Anti-mouse IgG rabbit antibody, anti-rabbit IgG goat antibody, and anti-goat IgG rabbit antibody were obtained from MP Biochemicals (Solon, OH). Colloidal gold (about 14 nm) was prepared using the method reported by Slot and Geuze (1985) and anti-mouse IgG rabbit antibody or anti-rabbit IgG goat antibody was conjugated with the colloidal gold particles as described previously (Yamashita et al. 2009).
Primary Antibodies.
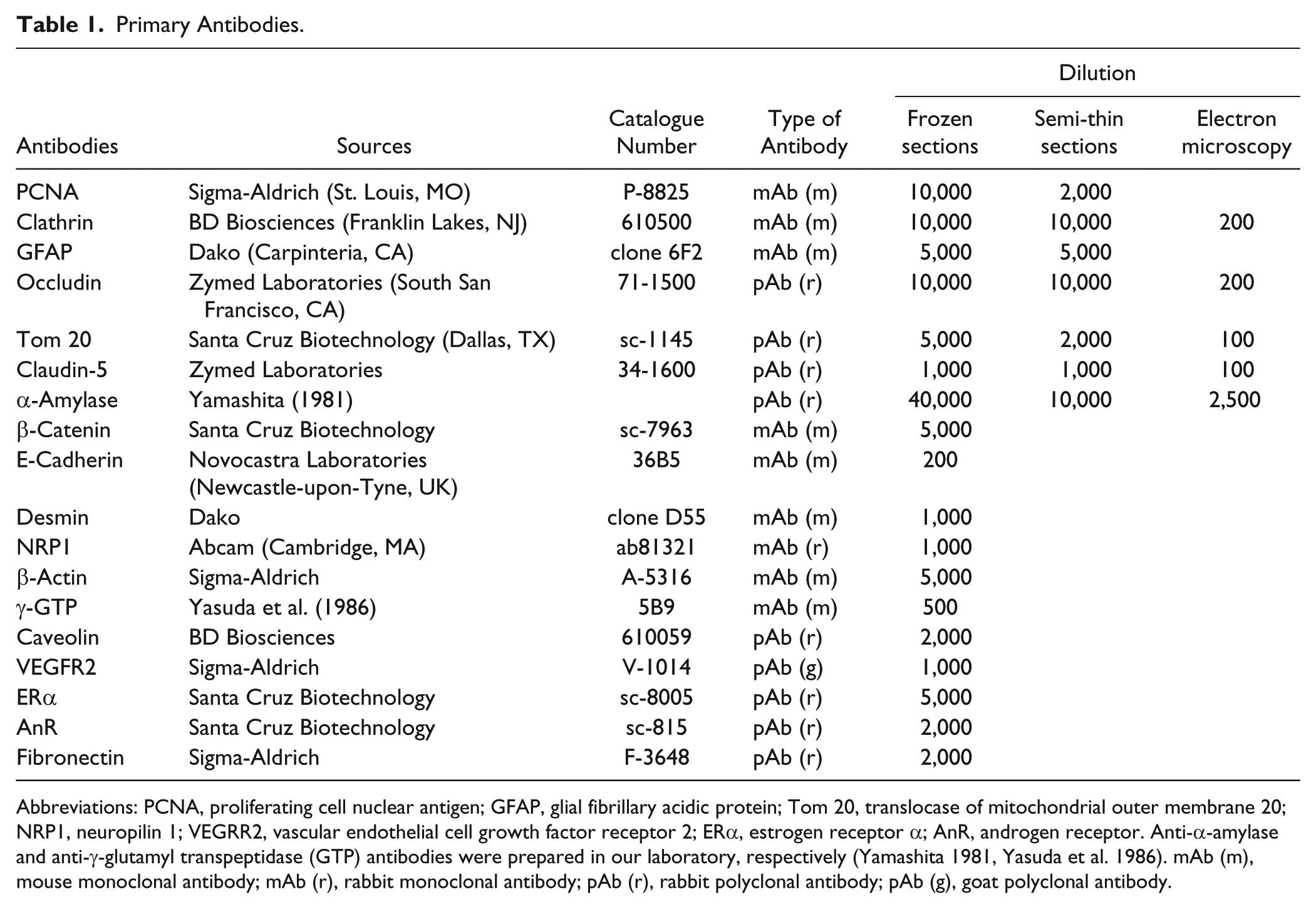
Abbreviations: PCNA, proliferating cell nuclear antigen; GFAP, glial fibrillary acidic protein; Tom 20, translocase of mitochondrial outer membrane 20; NRP1, neuropilin 1; VEGRR2, vascular endothelial cell growth factor receptor 2; ERα, estrogen receptor α; AnR, androgen receptor. Anti-α-amylase and anti-γ-glutamyl transpeptidase (GTP) antibodies were prepared in our laboratory, respectively (Yamashita 1981, Yasuda et al. 1986). mAb (m), mouse monoclonal antibody; mAb (r), rabbit monoclonal antibody; pAb (r), rabbit polyclonal antibody; pAb (g), goat polyclonal antibody.
Tissue Preparation
ICR mice (10–12-week-old) and Wistar rats (10-week-old) were obtained from Clea Japan, Inc. (Tokyo, Japan). All animals were maintained and treated according to a protocol approved by the Keio University Animal Care Committee. To prepare the fresh-frozen sections, small pieces of tissues were mounted in OCT compound (Miles Inc., Elkhart, IN) and frozen in dry ice-quenched isopentane. For electron microscopy, tissues were fixed with 2% glutaraldehyde/0.1 M phosphate buffer (PB) (pH 7.4) for 3 hr at 4C, washed with PBS, and then post-fixed with 1% osmium tetroxide/0.1 M PB (pH 7.4) for 1 hr on ice. The tissues were washed with PBS, dehydrated with graded ethanol, infiltrated in QY-1, embedded in epoxy resin, and then polymerized at 60C for 3 days.
Immunohistochemistry for Light Microscopy using Frozen Sections
Frozen sections (6 µm) were mounted on a new silane-coated slide glass (Muto Pure Chemicals, Tokyo, Japan). The sections were fixed in the following fixatives for 30 min at room temperature: 4% formaldehyde containing 25 mM calcium chloride in 0.1 M HEPES buffer (pH 7.4) or 2% glutaraldehyde in 0.1 M PB (pH 7.4). Some sections fixed with glutaraldehyde were further treated with 1% osmium tetroxide in 0.1 M PB (pH 7.4) for 30 min at room temperature, and then washed with distilled water. Some sections fixed with glutaraldehyde and osmium tetroxide were further treated with 1% sodium metaperiodate aqueous solution for 5 min, washed with distilled water, incubated with 1% sodium tetrahydroborate aqueous solution for 5 min, and then washed with distilled water. These sections were then dehydrated in graded ethanol, kept in 100% ethanol for 30 min, and then rehydrated. Sections were then incubated with 20 mM Tris-HCl buffer (TB) (pH 9.0) at room temperature or at 95C for 30 min, or autoclaved at 120C for 10 min. The sections were treated with 1% BSA/TBS (10 mM TB, pH 7.4, containing 150 mM sodium chloride) for 1 hr and incubated with the primary antibodies dissolved in the blocking buffer overnight at 4C. For the immunohistochemistry of VEGFR2, the sections were successively treated with anti-goat Ig rabbit IgG (1:500) for 30 min. After washing with TBS, the sections were incubated with Envision plus/horseradish peroxidase for mouse or rabbit for 1 hr at room temperature and the peroxidase enzyme activity was detected with imidazole-DAB (Yamashita and Okada, 2005). Some sections were counterstained with hematoxylin.
The effects of heat treatment on morphology were estimated using hematoxylin and eosin stained frozen sections without immunostaining.
Immunohistochemistry in Semi-thin Epon Sections
Semi-thin sections (1.5 µm) were floated on a droplet of distilled water placed on a new silane-coated slide glass, dried on a hot plate, and incubated at 60C overnight in an oven. The sections were treated with saturated sodium ethoxide in ethanol for 1 hr at room temperature, washed with ethanol three times, incubated with 500 mM TB (pH 7.4), and then washed with distilled water. Some deplasticized sections were further treated with 1% sodium metaperiodate for 5 min and then with 1% sodium tetrahydroborate for 5 min. To determine which antigen retrieval buffer provided the strongest immunoreaction, the sections were autoclaved in the following solutions for 10 min at 120C: Immunosaver; 20 mM, 100 mM and 500 mM TB (pH9.0); 20 mM and 100 mM TB (pH 10.0); 20 mM and 100 mM Bicin buffer (pH 9.0); and 20 mM CHES (N-cyclohexyl-2-aminoethanesulfonic acid) buffer (pH 9.5). The following dilution buffers for the primary antibodies were examined to obtain the strongest immunoreaction for each antigen: 10 mM TB (pH 7.4) containing 50 mM sodium chloride and 1% BSA; TBS containing 1% BSA; CGS-A containing 1% BSA; and CGS-B containing 1% BSA. Immunostaining was performed according to the procedures described above and some sections were counterstained with toluidine blue solution.
Immunoelectron Microscopy
Ultrathin sections were mounted on a nickel grid pre-treated with Neoprene W (Nissin EM), dried, and stored at room temperature until use. Semicircular tubes were prepared from a Teflon tube slit longitudinally along the center of the inner walls and two to six grids were placed in the slits (Yamashita et al. 2009); etching, HIAR, immunoreactions, and electron staining were then performed in the tubes. HIAR was performed using ultrathin sections without treatment, or those treated with1% aqueous sodium metaperiodate for 15 min, or partially deplasticized with sodium ethoxide. For etching, ultrathin sections were treated with sodium ethoxide (20-fold dilution with ethanol) for 30 to 120 sec at room temperature and then washed sequentially with 100% ethanol three times, 70% ethanol twice, 500 mM TB (pH 7.4), and finally distilled water. The ultrathin sections were placed in a HIAR solution in an Eppendorf tube (1.5 ml; Eppendorf, Hamburg, Germany) and were heated for 1–3 hr at 95C or were autoclaved for 10 min at 120C. After cooling to room temperature, the sections were treated with TBS containing 1% BSA and 0.1% Tween-20 for 30 min at room temperature. The sections were then incubated with primary antibodies overnight at 4C. The sections were then washed with 10 mM TB (pH 7.4) containing 0.4 M sodium chloride and incubated with colloidal gold-labeled antibodies for 1 hr, as described previously (Yamashita et al. 2009). After washing with the washing solution and then with distilled water, the sections were treated with 4% glutaraldehyde in 0.1 M PB (pH 7.4) for 5 min and then washed with distilled water. The sections were electron-stained with saturated uranyl acetate aqueous solution for 5 min and Reynold’s lead citrate solution for 2 min, 30 sec, and then examined with a Hitachi H-7650 transmission electron microscope (Hitachi High-Technologies Corp., Tokyo, Japan) at 80 kV.
Protein Crosslinking and Analyses with SDS-PAGE
RNase A and ovalbumin (10 mg/ml) dissolved in 0.1 M PB (pH 7.4) were mixed with an equal volume of 0.5% glutaraldehyde/0.1 M PB (pH 7.4) or 2% osmium tetroxide/0.1 M PB (pH 7.4) and incubated for 30 min at room temperature, then dialyzed against distilled water at 4C. A portion of the dialysate of glutaraldehyde-treated proteins was mixed with an equal volume of 2% osmium tetroxide/0.2 M PB (pH 7.4) and incubated for 30 min at room temperature, then dialyzed against distilled water at 4C. To one part of the dialysate, a 1/10 volume of 5% sodium metaperiodate was added and incubated for 30 min at room temperature, then dialyzed against distilled water at 4C. All samples were dissolved or suspended in 20 mM TB (pH 9.0) containing 1% SDS. The samples with or without autoclaving at 120C for 10 min were mixed with an equal volume of 2-mercaptoethanol-free SDS buffer solution for SDS-PAGE and were analyzed using SDS-PAGE without further heat treatment: RNase A and ovalbumin were separated on 14% and 10% gels, respectively, both with 3.5% stacking gels. After electrophoresis, the proteins in the gels were stained with Coomassie Brilliant Blue R-250.
Results
Effects of Heat Treatment on Morphology in Frozen Sections
Frozen sections fixed with formaldehyde containing calcium chloride, glutaraldehyde, or glutaraldehyde and osmium tetroxide showed excellent morphology after heating in 20 mM TB (pH 9.0) at 95C for 30 min. Frozen sections fixed with formaldehyde containing calcium chloride showed a good morphology after autoclaving in 20 mM TB (pH 9.0) (Fig. 1A), and sections fixed with glutaraldehyde maintained excellent tissue structure after autoclaving (Fig. 1C). On the other hand, autoclaving degraded the tissue structure in sections fixed with glutaraldehyde and post-fixed with osmium tetroxide (Fig. 1E). Sections fixed with glutaraldehyde and osmium tetroxide that appeared black faded after autoclaving. Autoclaving yielded no significant effects on tissue structures when the sections fixed with formaldehyde or glutaraldehyde were further treated with sodium metaperiodate aqueous solution (Fig. 1B and 1D). However, autoclaving severely destroyed tissue structures when sections that had been fixed with glutaraldehyde and osmium tetroxide were further treated with sodium metaperiodate aqueous solution (Fig. 1F); the sections exhibited an excellent morphology without autoclaving.
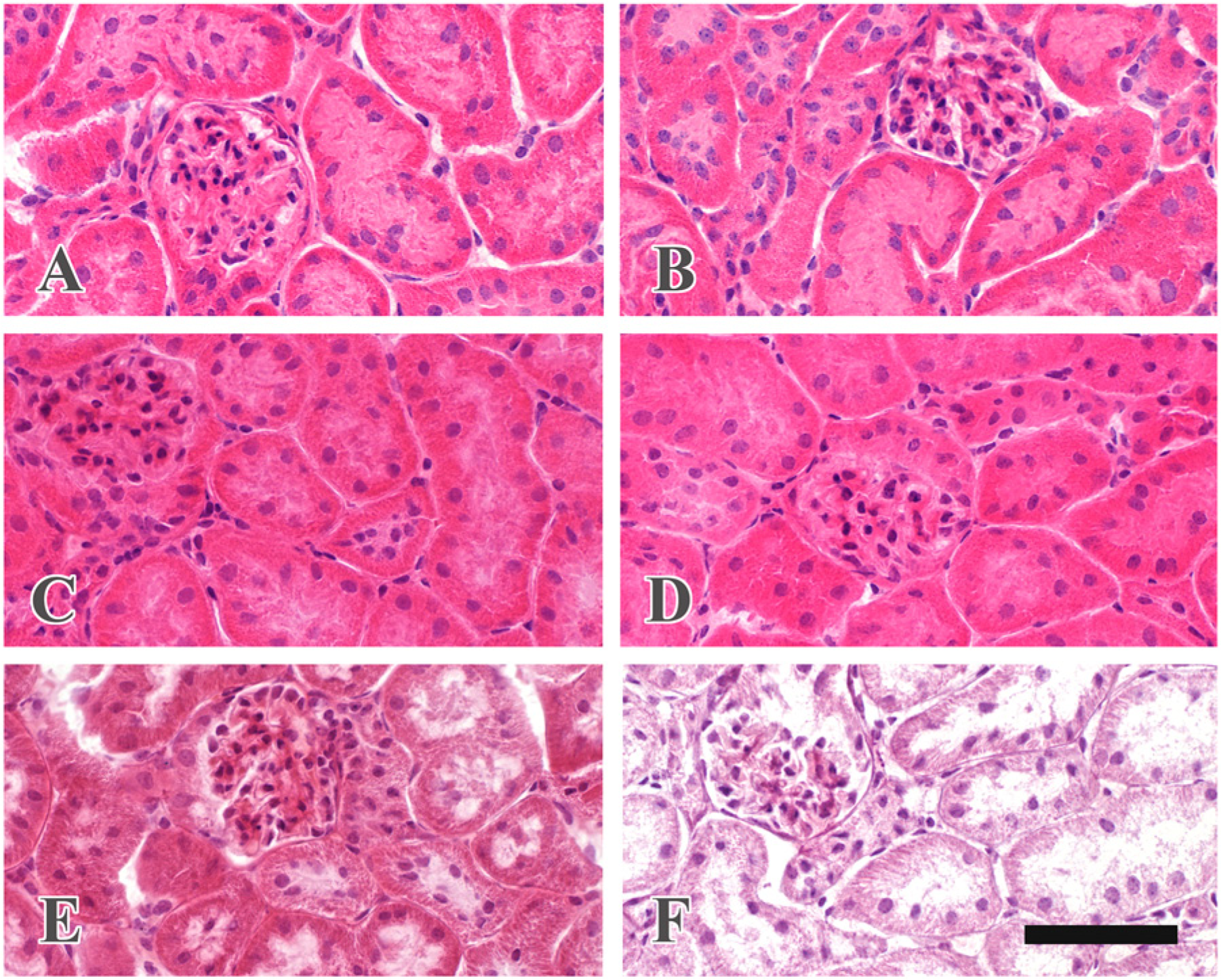
Effect of autoclaving on tissue structure in frozen sections. Frozen sections were fixed with 4% formaldehyde containing 25 mM calcium chloride for 30 min (A and B), 2% glutaraldehyde for 30 min (C and D), or 2% glutaraldehyde and 1% osmium tetroxide for 30 min each (E and F). The sections were further treated with 1% sodium metaperiodate for 5 min and then with 1% sodium tetrahydroborate for 5 min (B, D and F). All sections were autoclaved in 20 mM TB (pH 9.0) at 120C for 10 min and stained with hematoxylin and eosin. Scale bar = 100 µm.
HIAR in the Frozen Sections
The effects of heat treatment on immunostaining in frozen sections fixed with various fixatives are summarized in Table 2. Heat treatment was effective for 17 out of 18 antigens except for fibronectin. The antigenicity of seven antigens—PCNA (Fig. 2A, 2B and 2C), clathrin (Fig. 2D, 2E and 2F), GFAP, α-amylase, occludin, Tom 20 (Fig. 2G, 2H and 2I), and claudin-5—was retrieved when the sections were fixed with glutaraldehyde and osmium tetroxide and then heated, whereas a negative reaction was observed when the sections were fixed with only glutaraldehyde and then heated. Autoclaving at 120C for 10 min yielded stronger immunoreactions than heating at 95C for 30 min. These seven antigens exhibited positive immunoreactions when the fixed sections were treated with sodium metaperiodate and sodium tetrahydroborate followed by autoclaving, whereas the immunoreaction of PCNA, GFAP and claudin-5 was weakened after these treatments. On the other hand, eight antigens recovered their immunoreactivity after heat treatment when the frozen sections were fixed with formaldehyde containing 25 mM calcium chloride, although HIAR had almost no effect when the sections were fixed with glutaraldehyde or glutaraldehyde and osmium tetroxide (Fig. 2J, 2K and 2L). Two antigens, γ-glutamyl transpeptidase (GTP) and β-actin, showed a stronger immunoreaction in tissues fixed with glutaraldehyde than in those fixed with formaldehyde containing calcium chloride when the sections were heated at 95C for 30 min (Fig. 2M, 2N and 2O).
Effect of Heat-induced Antigen Retrieval in Frozen Sections.
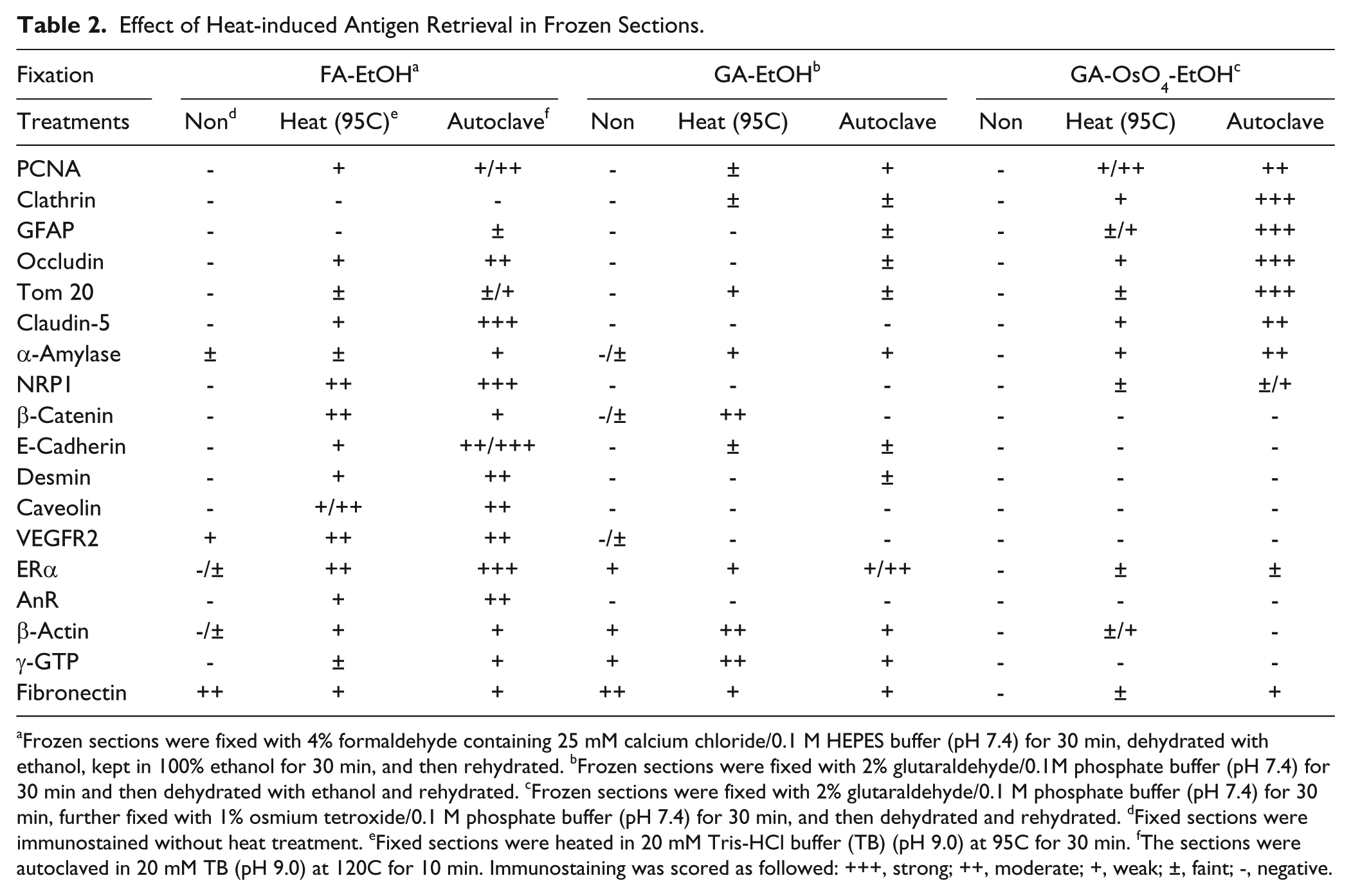
Frozen sections were fixed with 4% formaldehyde containing 25 mM calcium chloride/0.1 M HEPES buffer (pH 7.4) for 30 min, dehydrated with ethanol, kept in 100% ethanol for 30 min, and then rehydrated. bFrozen sections were fixed with 2% glutaraldehyde/0.1M phosphate buffer (pH 7.4) for 30 min and then dehydrated with ethanol and rehydrated. cFrozen sections were fixed with 2% glutaraldehyde/0.1 M phosphate buffer (pH 7.4) for 30 min, further fixed with 1% osmium tetroxide/0.1 M phosphate buffer (pH 7.4) for 30 min, and then dehydrated and rehydrated. dFixed sections were immunostained without heat treatment. eFixed sections were heated in 20 mM Tris-HCl buffer (TB) (pH 9.0) at 95C for 30 min. fThe sections were autoclaved in 20 mM TB (pH 9.0) at 120C for 10 min. Immunostaining was scored as followed: +++, strong; ++, moderate; +, weak; ±, faint; -, negative.
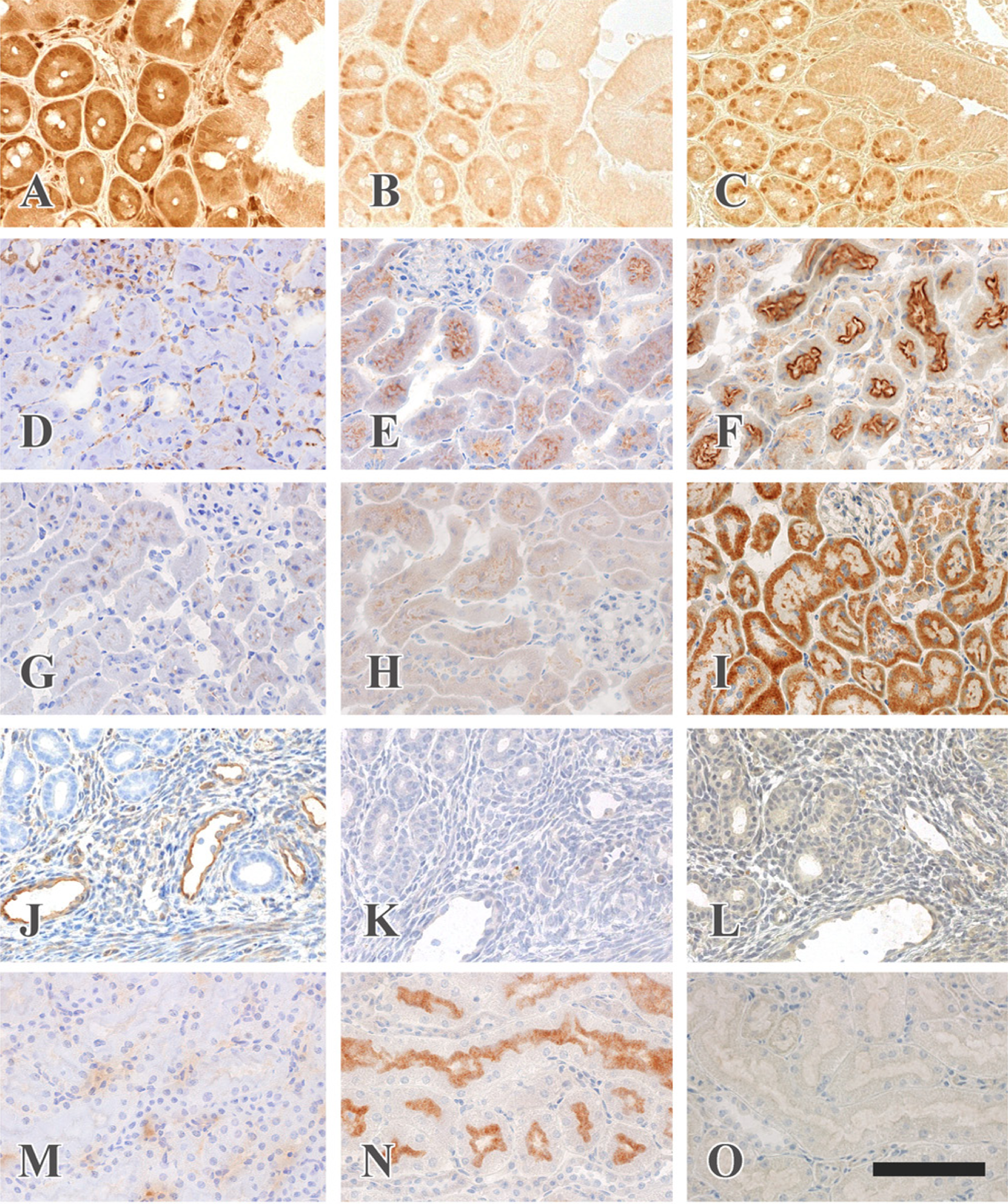
Heat-induced antigen retrieval in frozen sections. Frozen sections (6 µm) were fixed with 4% formaldehyde containing 25 mM calcium chloride (A, D, G, J and M), with 2% glutaraldehyde (B, E, H, K and N), or with 2% glutaraldehyde and 1% osmium tetroxide (C, F, I, L and O), and then autoclaved in 20 mM TB (pH 9.0) at 120C for 10 min (A, B, C, D, E, F, G, H and I), or heated in 20 mM TB (pH 9.0) at 95C for 30 min (J, K, L, M, N and O). The sections were then immunostained with anti-PCNA antibody in the small intestine (A, B and C), anti-clathrin antibody in the kidney (D, E and F), anti-Tom 20 antibody in the kidney (G, H and I), anti-caveolin antibody in the uterus (J, K and L), or anti-γ-GTP antibody in the kidney (M, N and O). PCNA is localized in the nuclei of intestinal glandular cells locating in the basal portion (C). Immunostaining for endogenous mouse IgG is recognized in the cytoplasm of intestinal glandular cells and plasma cells and in the connective tissues (A). Clathrin is detected in the apical cytoplasm of proximal tubules (F) and Tom 20 immunoreactivity is seen in the cytoplasm of proximal and distal tubules (I). Caveolin immunoreactivity is present in the endothelial cells of blood vessels (J). γ-GTP is localized in the brush border of proximal tubules (N). Scale bar = 200 µm.
HIAR in Semi-thin Epon Sections
HIAR was applied to semi-thin epon sections to demonstrate recovery of antigenicity for the seven antigens. The effects of deplasticization, buffers for HIAR, pre-treatment with sodium metaperiodate, and diluents for the primary antibodies were examined to obtain reproducible immunoreactions of a high sensitivity. Six antigens revealed a positive immunostaining in semi-thin epon sections after treatment with sodium ethoxide for 1 hr followed by autoclaving (Fig. 3), whereas PCNA showed a negative immunoreaction. Antigen retrieval solutions, 100 mM TB (pH 9.0), 100 mM TB (pH 10.0) and 500 mM TB (pH 9.0), yielded stronger immunostaining than the other solutions for the six antigens (Fig. 3A and 3B), although 20 mM TB (pH 9.0) showed stronger immunoreaction for these antigens than 100 mM TB (pH 9.0) in the frozen sections. Treatment with sodium metaperiodate preceding autoclaving yielded no significant effect on the immunostaining of most antigens. TBS containing 1% BSA revealed a strong immunoreaction with a low background staining for clathrin (Fig. 3B), GFAP (Fig. 3C), occludin (Fig. 3D), and α-amylase (Fig. 3F).
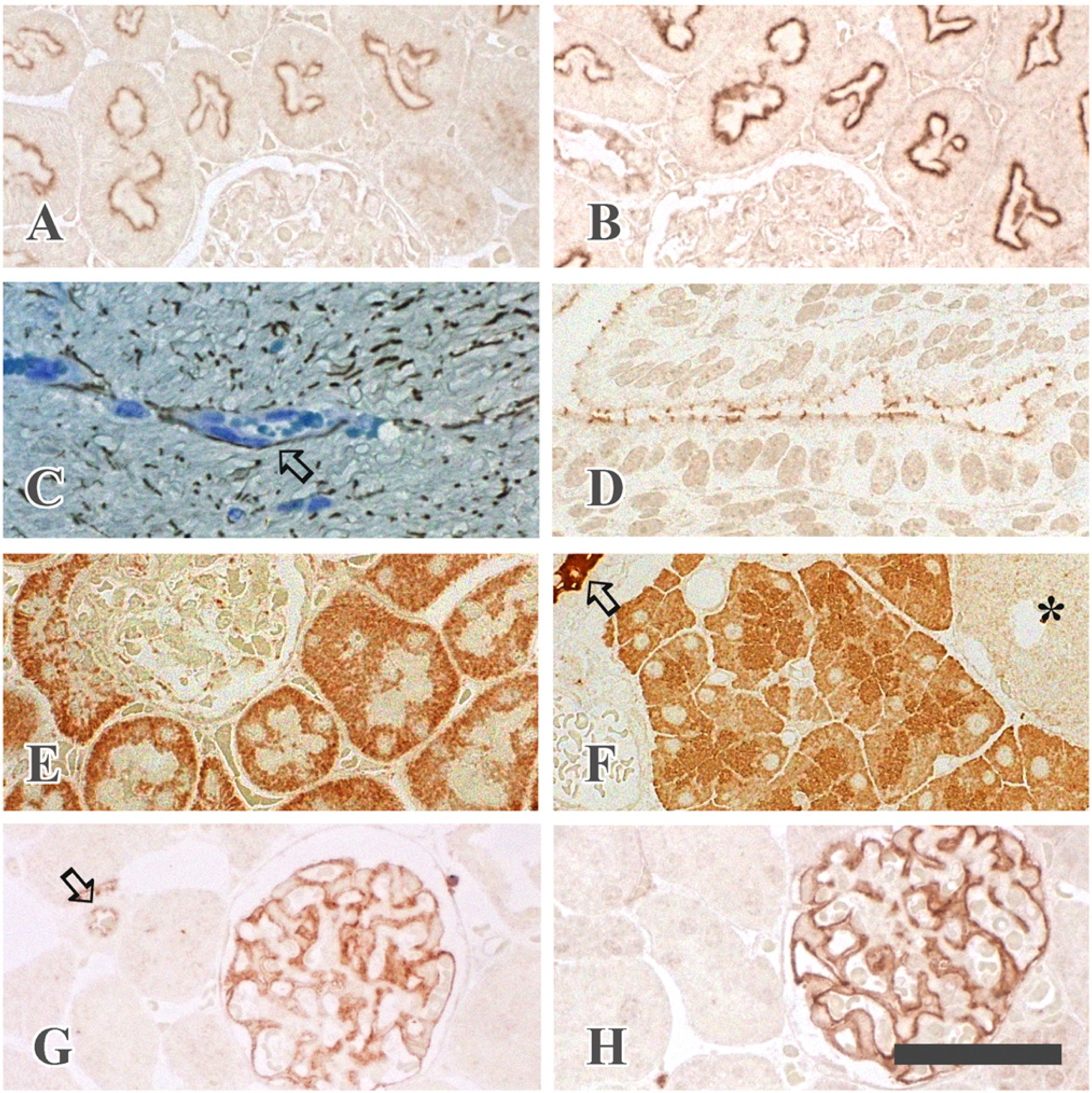
Heat-induced antigen retrieval in semi-thin epon sections. Semi-thin sections (1.5 µm) were prepared from specimens fixed with glutaraldehyde, post-fixed with osmium tetroxide and embedded in the epoxy resin. The sections were treated with saturated sodium ethoxide in ethanol for 1 hr, and then autoclaved in 100 mM TB (pH 9.0) (B, C, D, E, F, G and H) or in 20 mM TB (pH 9.0) (A) at 120C for 10 min. Antibodies to clathrin (A and B), GFAP (C), occludin (D), Tom 20 (E) and α-amylase (F) were diluted with 1% BSA/TBS. Anti-claudin-5 antibody was diluted with 1% BSA/TBS (G) or 1% BSA/CGS-A (H). A clathrin immunoreaction is seen beneath the brush border of the renal proximal tubules: autoclaving in 100 mM TB yields stronger immunoreaction than that in 20 mM TB (A and B). GFAP-immunoreaction is detected in the cytoplasm of astrocytes and along the blood vessels in the spinal cord: the section is counterstained with toluidine blue; arrow indicates a vein (C). Occludin is detected in the junctional complexes of uterine epithelial cells (D). Rod-like Tom 20 immunostaining is observed in the cytoplasm of the proximal and distal tubules in the kidney (E). α-Amylase localization is demonstrated in the secretory granules in the rat exocrine pancreas: arrow, intercalated duct; *, Langerhans island (F). Claudin-5 immunoreaction is shown along the glomerular capillaries and the endothelial cells of an artery (arrow) in the kidney (G and H). The antibody diluted with 1% BSA/CGS-A (H) shows stronger immunostaining than that diluted with 1% BSA/TBS (G). Scale bar = 50 µm.
Anti-Tom 20 diluted with 10 mM TB (pH 7.4) containing 50 mM sodium chloride and 1% BSA or CGS-B containing 1% BSA exhibited stronger immunoreaction than those diluted with TBS containing 1% BSA or CGS-A containing 1% BSA (Fig. 3E). Claudin-5 showed the strongest immunoreaction when the antibody was diluted with CGA-A containing 1% BSA (Fig. 3G and 3H).
HIAR for Immunoelectron Microscopy
Although heat treatment was essential for the detection of antigens on ultrathin sections, heat treatment reduced the electron density of ribosomes, chromatins, intracellular membranes and secretory granules in the exocrine pancreas (Fig. 4A and 4B). Autoclaving at 120C for 10 min and heating at 95C for 60 min showed almost the same effects on the recovery of antigenicity and preservation of fine structures. Treatment with sodium ethoxide preceding autoclaving yielded no significant and reproducible effects for the intensity of immunoreactions, and excess etching made small holes in the sections and reduced the contrast and fine structure of the cellular organelles. Heating in 500 mM TB (pH 9.0) yielded better ultrastructure than that in 100 mM TB (pH 9.0) and 100 mM TB (pH 10.0).
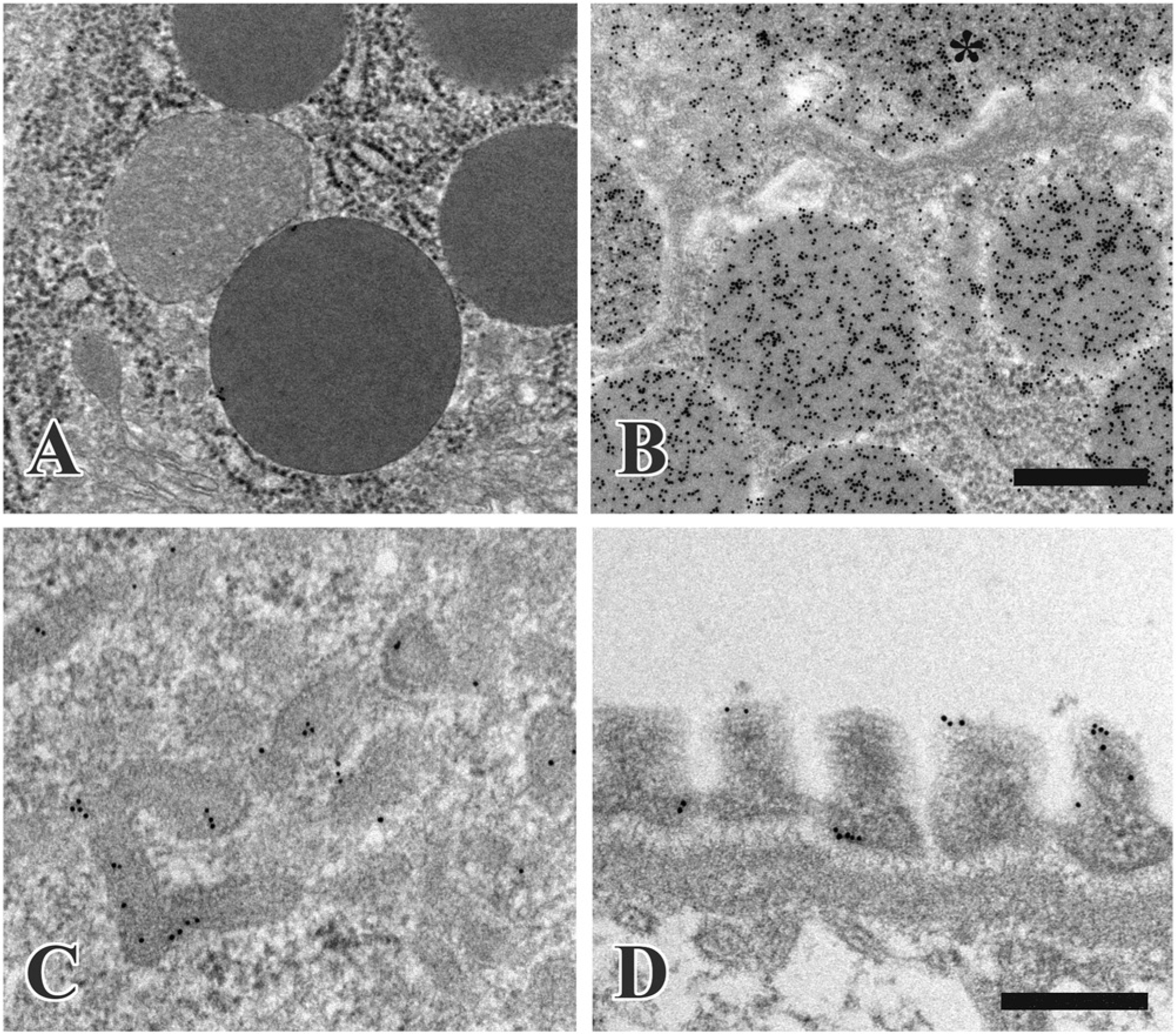
Heat-induced antigen retrieval in immunoelectron microscopy with the post-embedding method. α-Amylase is localized in the rat exocrine pancreas. Ultrathin sections were immunostained without heating (A) or after heating in 500 mM TB (pH 9.0) for 1 hr (B): *, lumen of the intercalated duct. Clathrin is detected in the proximal tubule of the kidney: the ultrathin section was heated in 500 mM TB (pH 9.0) for 2 hr (C). Claudin-5 localization is shown in the renal glomerulus: the section was heated in 500 mM TB (pH 9.0) for 2 hr (D). Scale bars = 500 nm (A and B), 300 nm (C and D).
α-Amylase was localized in the secretory granules, the Golgi apparatus and rough-surfaced endoplasmic reticulum in the exocrine pancreas: the antibody was diluted with TBS containing 1% BSA and 0.1% Tween-20 (Fig. 4B). Tom 20 was localized along the outer mitochondrial membrane in the proximal and distal tubules in the kidney; for this staining, the primary antibody was diluted with 10 mM TB (pH 7.4) containing 50 mM sodium chloride, 1% BSA and 0.1% Tween-20. Clathrin was detected in the coated vesicles of apical canaliculi in the proximal tubules; here, the antibody was diluted with TBS containing 1% BSA and 0.1% Tween-20 (Fig. 4C). The immunoreaction of claudin-5 was demonstrated in the foot processes of podocytes in the glomeruli of the kidney after the antibody was diluted with CGS-A containing 1% BSA (Fig. 4D).
Protein Crosslinking with Fixative Regents and Effect of Heating
Autoclaving of native proteins yielded no significant changes in the electrophoretic pattern of each protein in SDS-PAGE, although the proportion of dimers of RNase A and ovalbumin slightly increased (Fig. 5A and 5B; lanes 1 and 2). RNase A treated with glutaraldehyde formed a light yellow precipitate and the precipitate turned to black upon further osmium tetroxide treatment. These precipitates did not dissolve after autoclaving in 20 mM TB (pH 9.0) containing 1% SDS. When RNase A treated with glutaraldehyde and osmium tetroxide was further incubated with sodium metaperiodate, the black precipitate turned white, and the majority of the precipitate dissolved in the buffer after autoclaving. Polypeptides from the RNase A samples with precipitates did not enter into either the stacking or separating gels but polypeptides were seen at the top of the stacking and the separation gels in samples treated with sodium metaperiodate followed by autoclaving (Fig. 5A; lane 8). Osmicated RNase A appeared as a faint brown color and exhibited no significant changes in electrophoretic patterns except for a slight increase in the proportion of dimers and slight degradation after autoclaving (Fig. 5A; lanes 9 and 10). Glutaraldehyde crosslinked ovalbumin and formed dimers, trimers, tetramers and oligomers, and autoclaving yielded no significant change in the electropherogram (Fig. 5B; lanes 3 and 4). Polypeptides were hardly detected in the ovalbumin sample after treatment with glutaraldehyde and osmium tetroxide or samples that were further treated with sodium metaperiodate independent of autoclaving (Fig.5B; lanes 5, 6, 7 and 8). Osmicated ovalbumin showed no clear polypeptide bands, except for the band in the buffer front (Fig. 5B; lanes 9 and 10).
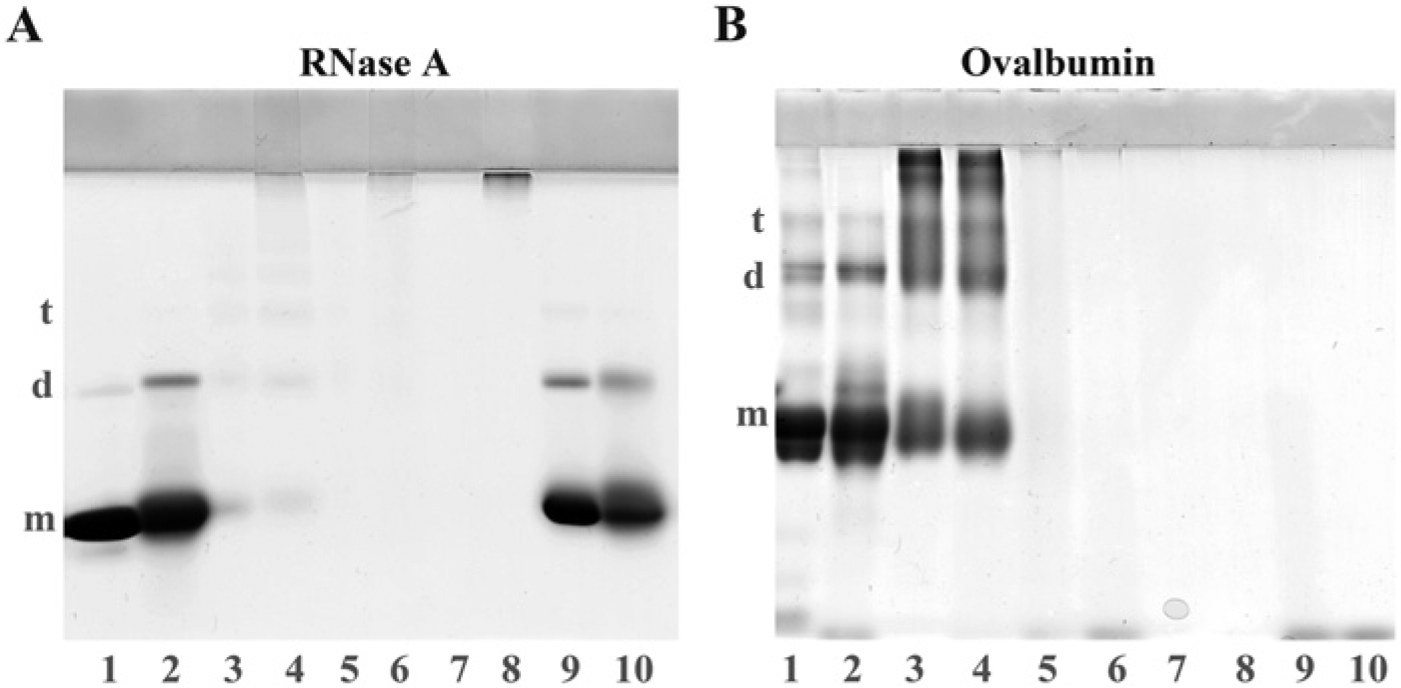
Effects of autoclaving on proteins treated with various fixatives. Because osmium tetroxide is reported to cleave tryptophan residues and degrade polypeptides, the effects of glutaraldehyde and osmium tetroxide treatment and autoclaving on RNase A (which does not contain tryptophan) and ovalbumin (which contains three tryptophan residues) were examined. Lanes 1 and 2: native proteins without treatment by fixing reagents. Lanes 3 and 4: RNase A and ovalbumin solutions (10 mg/ml) were mixed with an equal volume of 0.5% glutaraldehyde/0.1 M phosphate buffer (PB, pH 7.4) for 30 min. Lanes 5–8: Glutaraldehyde-treated proteins were mixed with an equal volume of 2% osmium tetroxide/0.2 M PB (pH 7.4) (lanes 5 and 6), and further treated with sodium metaperiodate (lanes 7 and 8). Lanes 9 and 10: Native RNase A and ovalbumin solutions were mixed with an equal volume of 2% osmium tetroxide/0.1 M PB (pH 7.4) for 30 min. The samples were analyzed using SDS-PAGE after autoclaving in 20 mM TB (pH 9.0) at 120C for 10 min (lanes 2, 4, 6, 8 and 10), or without autoclaving (lanes 1, 3, 5, 7 and 9). RNase A and ovalbumin were separated on 14% and 10% gels, respectively, with a 3.5% stacking gel. Glutaraldehyde polymerizes RNase A, a lysine-rich protein (lanes 3) but osmium tetroxide shows low crosslinking activity for RNase A (lane 9). After autoclaving, RNase A sample treated with glutaraldehyde and osmium tetroxide and further incubated with sodium metaperiodate is depolymerized and polypeptides are detected at the top of the stacking and separation gels (lanes 7 and 8). Ovalbumin forms oligomers after glutaraldehyde treatment (lane 3) and the oligomers are stable for autoclaving (lane 4). Osmium tetroxide degrades ovalbumin into fragments (lane 9). RNase A: m, monomer (13.7 kD); d, dimer (27.4 kD); t, trimer (41.1 kD). Ovalbumin: m, monomer (42.5 kD); d, dimer (85 kD); t, trimer (127.5 kD).
Discussion
The present study clearly demonstrates that HIAR is effective for many antigens in tissues fixed with glutaraldehyde and post-fixed with osmium tetroxide and then embedded in epoxy resin, although the detection of antigens has been thought to be a special case in these specimens. First we used frozen sections fixed with glutaraldehyde and osmium tetroxide to study the possibility and mechanism of HIAR and exclude the effect of deplasticization of the embedding media. Furthermore, the fixed sections were dehydrated and rehydrated prior to heat treatment, as dehydration is reported to expose the hydrophobic portion of proteins and accelerate protein crosslinking with aldehydes (Fowler et al. 2008). After autoclaving, 7 out of 18 antibodies exhibited a strong immunoreaction in frozen sections fixed with glutaraldehyde and osmium tetroxide, although heating had almost no effect on antigen retrieval in sections fixed with glutaraldehyde alone. The following results indicate that crosslinkages formed by glutaraldehyde are modified with osmium tetroxide and become unstable for heating. The morphology in the frozen tissues fixed with glutaraldehyde and osmium tetroxide showed a higher degree of disruption after autoclaving, as compared with those fixed with glutaraldehyde or formaldehyde containing calcium ions. Autoclaving changed the precipitates of RNase A formed by the treatment with glutaraldehyde and osmium tetroxide to soluble polypeptides after sodium metaperiodate treatment.
Osmium tetroxide showed low protein crosslinking activity. Osmium tetroxide treatment had almost no effect on RNase A, which does not contain tryptophan residues. On the other hand, osmium tetroxide degraded ovalbumin, which contains three tryptophan residues, into small fragments, as reported by Deetz and Berhrman (1981). Although the mechanisms of tissue fixation by glutaraldehyde are still debatable, α, β-unsaturated oligomers/polymers produced through aldol condensation of glutaraldehyde monomers, the formation of quaternary pyridinium compounds, and crosslinks between lysine and other amino acids, such as arginine and cysteine, are reported to be important for inter- and intra-protein crosslinking (Migneault et al. 2004). In particular, several investigators have stated that α, β-unsaturated oligomers/polymers play a central role in the crosslink reaction of proteins by producing Schiff bases, as the crosslinking efficiency of glutaraldehyde is remarkably higher than that of other bi-functional, crosslinking reagents, and because glutaraldehyde forms extremely stable crosslinks against acid hydrolysis, with the crosslinks being stable in 6 N hydrochloric acid at 110C for 24 hr (Monsan et al. 1975; Migneault et al. 2004). In contrast, Kawahara et al. (1992) demonstrated that the aldol condensation of glutaraldehyde monomers and protein crosslinking by Schiff bases should occur in parallel and should form a linear aldol-condensed glutaraldehyde oligomer with several Schiff base linkages branching off, as the glutaraldehyde solution showed no absorbance at 235 nm caused by the α, β-unsaturated bonds in the absence of amines (Fig. 6(b)). Both hypotheses yield similar final reaction products, and a Schiff base and an adjacent ethylene bond constitute a conjunctive system, with resonance interactions reportedly making the Schiff base linkages stable to hydrolysis. Because osmium tetroxide is well known to bind with the ethylene bonds (Nielson and Griffith 1979) and RNase A and ovalbumin treated with glutaraldehyde form osmium black after osmium tetroxide treatment, osmium tetroxide presumably adducts the ethylene bonds and crosslinks the double bonds (Fig. 6(c)). Heat treatment may partially dehydrate the additives and form 1, 2-diols, as the black color fades in frozen sections fixed with glutaraldehyde and osmium tetroxide and electron density reduces in the ultrathin sections after autoclaving. The additives and 1, 2-diols may disrupt the resonance interaction and destabilize the Schiff bases against heating (Fig. 6(d)). In addition, the formation of 1, 2-dioles may recover the flexibility of the polypeptide, which is prevented by double bonds, and facilitate an epitope-paratope interaction. In tissues or proteins fixed with glutaraldehyde and osmium tetroxide, sodium metaperiodate treatment probably oxidizes the osmium additives completely and forms the 1, 2-dioles and cleaves the diols to aldehydes, thus inducing further degradation of the protein-glutaraldehyde co-polymers. The results of the present study support the hypothesis of Kawahara et al. (1992; 1997) concerning the mechanisms of fixation by glutaraldehyde.
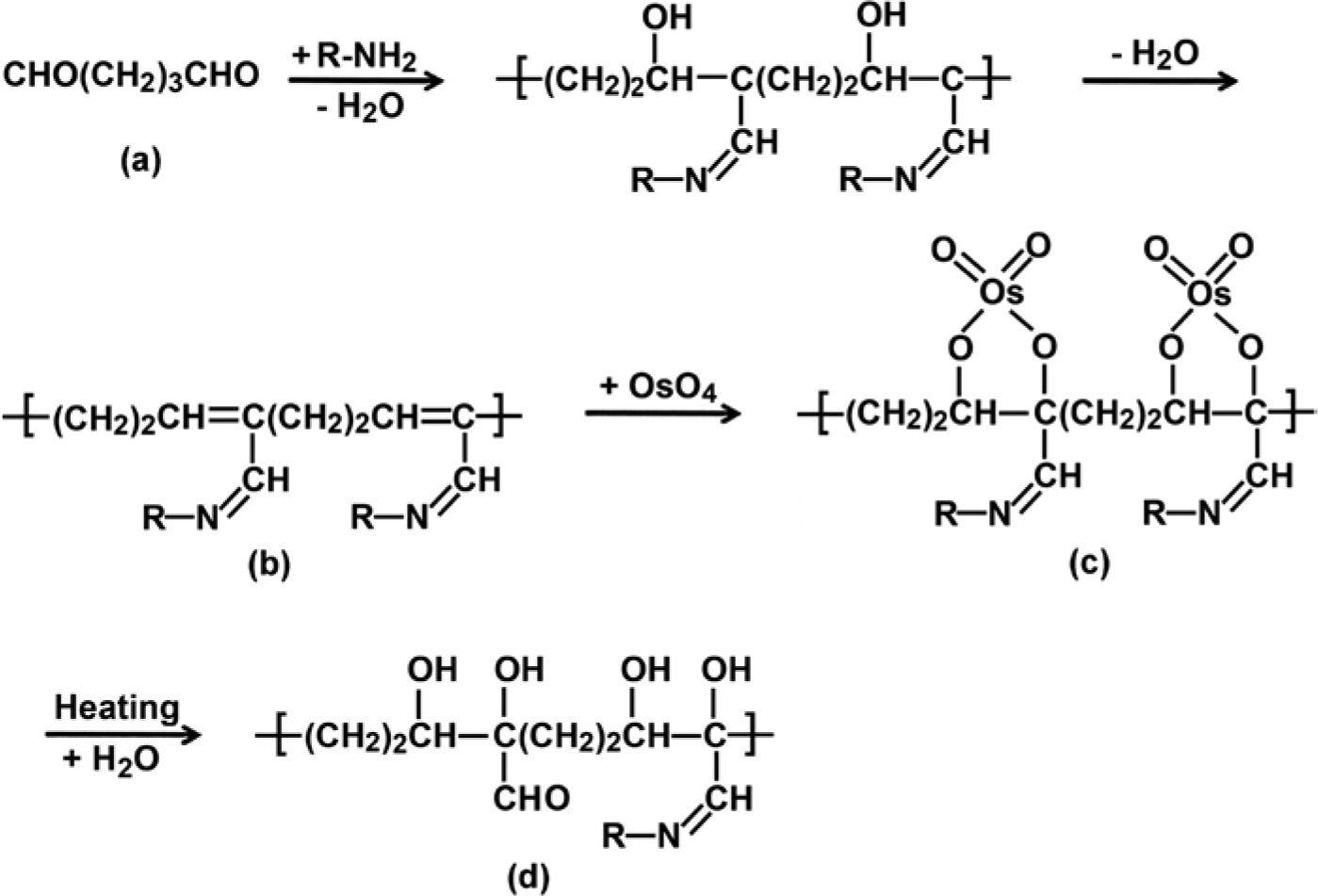
Mechanism of heat-induced antigen retrieval in specimens fixed with glutaraldehyde and osmium tetroxide.
Although the formation of covalent bonds between epoxy resin and proteins has been reported (Kellenberger et al. 1987), it is unclear whether amino acids that react with epoxy resin monomers are present in the tissues fixed with glutaraldehyde and post-fixed with osmium tetroxide, which highly modify various amino acids. Six antigens localized in frozen sections (other than PCNA) were also detectable in semi-thin epon sections; thus, sodium ethoxide treatment appears to conserve the antigenicity of most antigens. Similarly, Brorson (1997) demonstrated that the immunoreactivity of most antigens is not affected by sodium ethoxide treatment, as ultrathin sections prepared from specimens fixed with formaldehyde and embedded in LR-White, a sodium ethoxide resistant resin, showed no significant difference in immunostaining after sodium ethoxide treatment. Mar and Wight (1988) reported similar results using epon-embedded tissues fixed with a formaldehyde and glutaraldehyde mixture. Whether the great reduction in PCNA-immunostaining by sodium ethoxide treatment is due to the direct modification of the PCNA epitope by ethoxide or the extraction of PCNA because of the destruction of crosslinks between PCNA and neighboring nuclear proteins remains unclear. Although etching with sodium ethoxide was essential to obtain a positive immunoreaction in the semi-thin sections, the treatment yielded no significant effect for the immunoreaction using ultrathin sections; this is probably because etching exposes antigens in epoxy resins but also extracts them during heat treatment.
Ultrathin sections were heated at 95C for 1–3 hr for immunoelectron microscopy to easily control experimental conditions as compared with autoclaving. The heating time may depend on block hardness, the types of epoxy resin, and the composition of the epoxy resin mixture. Brorson and Skjorten (1996) pointed out that the concentration of the accelerator (DMP-30) is an important factor for HIAR of epon-embedded materials to reduce copolymerization between proteins and epoxy resin and to obtain a strong immunoreaction. In addition, the type of antigen retrieval buffer is an important factor for maintaining acceptable amounts of fine structures and to expose epitopes. A suitable choice of diluents for the primary antibody should also yield sensitive immunoreactions with a low background staining on ultrathin sections, because the epitope-paratope interactions and antibody-resin interactions highly depend on the types of buffers, ionic strength and the presence of detergents in the solutions.
The present study demonstrated that HIAR is effective for many antigens in conventionally processed epon blocks, contrary to presumptions. Post-fixation with osmium tetroxide destabilizes protein crosslinks induced by glutaraldehyde, and heating further degrades these crosslinks. Heat treatment is essential for immunoelectron microscopy using epon-embedded materials, and partial deplasticization followed by heating is required for light microscopy using semi-thin epon sections. These results indicate that archival epon-embedded specimens can be useful resources for immunohistochemical studies at both the light and electron microscopy levels, as they both provide excellent morphology and detailed antigen localization as compared with paraffin-embedded materials.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Supported in part by a
