Abstract
A new method is described for performing fluorescence in situ hybridization (FISH). FISH signals are enhanced by microwave pulses applied during the DNA-DNA hybridization process. It is the first description of FISH with a single/low-copy probe done more efficiently by application of microwave; the latter leads to quick results or enhancement of weak signals. Microwave FISH has been compared systematically with normal FISH, and we could demonstrate the efficiency of microwave irradiation especially in the first 100 min of hybridization.
Keywords
Here we present a new microwave-FISH protocol to enhance FISH signals on chromosomes in which microwave pulses were applied during the DNA-DNA hybridization process. Using this approach, quick results or enhancement of weak signals can be achieved.
The described microwave-FISH protocol (see the following section) was tested in comparison to normal FISH without microwave (Liehr et al. 1995) using the BAC probe b35B4. This probe derives from chromosome 1q21 and contains 143 kb of nonrepetitive DNA; however, it produces three specific FISH signals in 1q21, 1p12, and 1p36.1 (Figure 1). A total of 123 kb of b35B4 sequence is present in at least five copies in 1q21, leading to a strong FISH signal there. In 1p36.1, 59 kb of b35B4 sequence are present twice in tandem order. Additionally, 21.5 kb of b35B4 sequence are located once in 1p12. This probe was chosen because its application of one FISH probe leads to three signals of different intensity (Figure 1) (Weise et al. 2005).
FISH and microwave FISH were done for 2–24 hr. Microwave beams were applied in 5- or 20-min intervals (Figure 2). The FISH results were evaluated using the ISIS System (MetaSystems; Altlussheim, Germany). The autoexposure time necessary for image acquisition was taken as standard for hybridization efficiency. The microwave FISH needed half exposure times between 20 min and 100 min (Figure 2A). Additional experiments showed that between 5 min and 20 min, the difference in exposure time was the same (Figure 2B), whereas between 120 min and 8 hr, the difference remains low, and, afterwards, the hybridization efficacy was no longer influenced by microwave (Figure 2C). As shown in Figure 1, microwave affects the hybridization even after 5 min and only with one microwave beam. Although in normal FISH, the signal to background ratio was low, all six signals are clearly visible in microwave FISH without background in all analyzed 20 metaphases (Figures 1B-1C). The probe b35B4 was labeled with TexasRed for these experiments. The probe b35B4 was applied biotin-labeled to test the effects of microwave on the effectiveness of the detection step using avidin-FITC. No differences in autoexposure time were found after incubation times of 5 or 10 min (Figure 2).
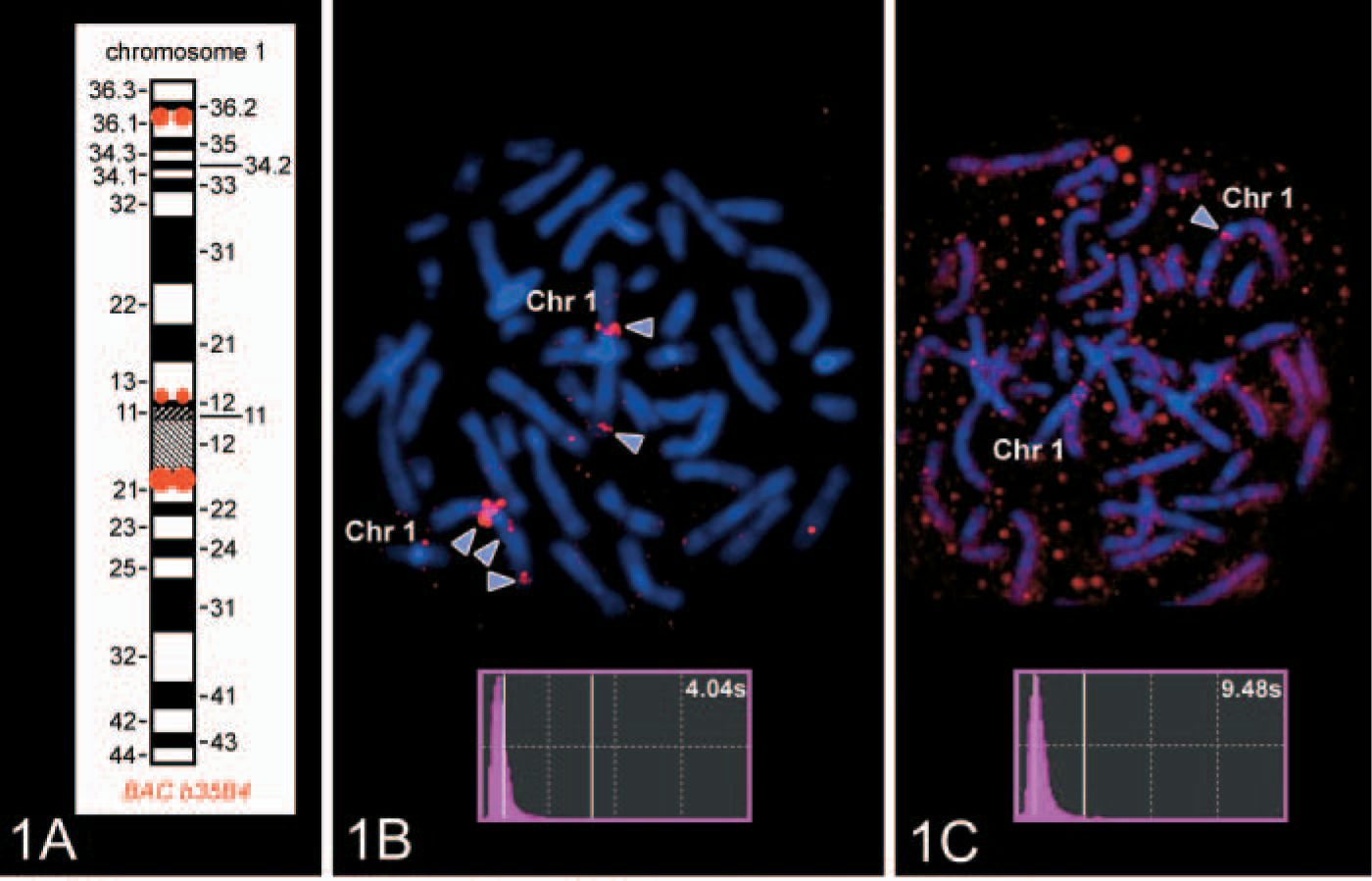
Schematic drawing of the BAC b35B4 hybridization pattern on chromosome 1.
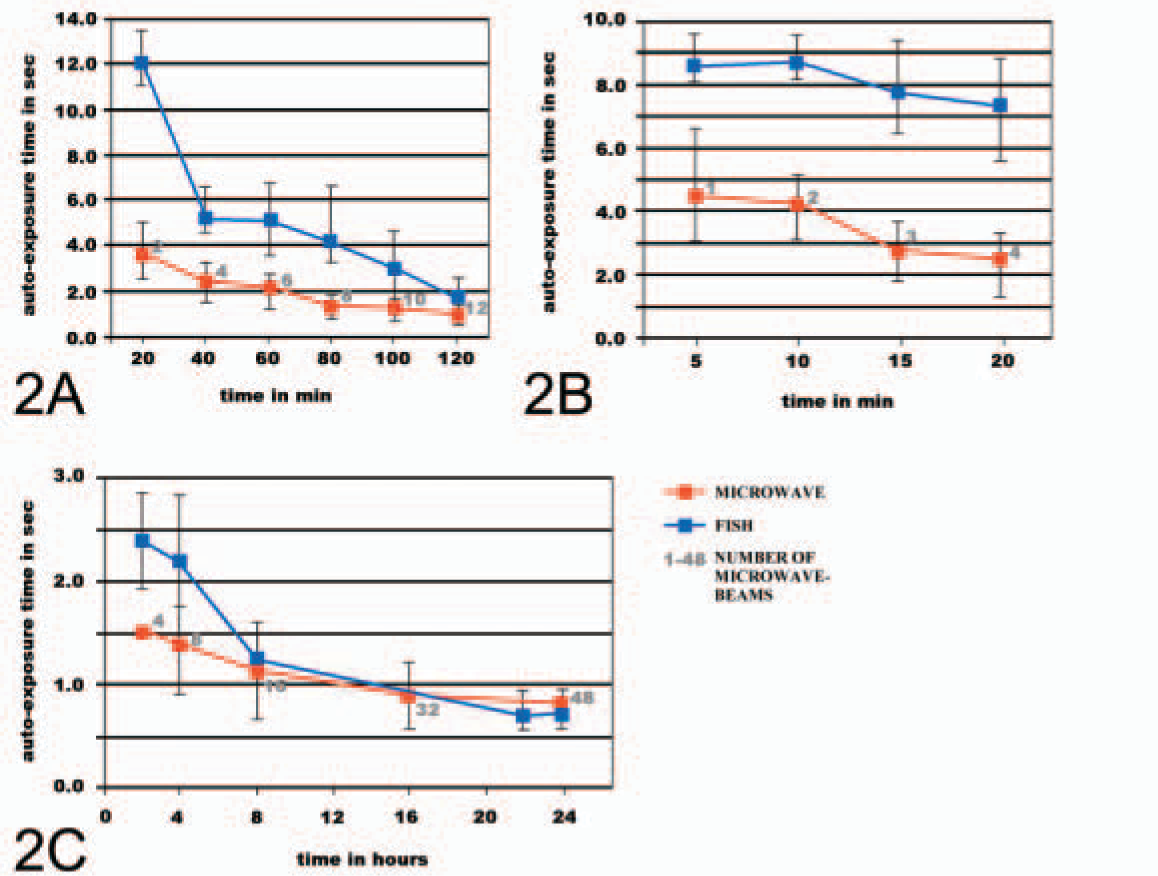
Comparison of autoexposure times between microwave FISH (microwave, red line) and normal FISH slides (FISH, blue line). Number of microwave beams are indicated in gray along the microwave data line. Slides were evaluated every 20 min within 2 hr
Furthermore, autoexposure times after one microwave beam with and without subsequent 10-min hybridization at 37C were compared. Because the autoexposure times showed no differences (data not shown), it can be speculated that microwave irradiation may act as a steric modifier, allowing the DNA probe to find the DNA target in a more efficient and faster way than in a normal FISH assay in which it depends on diffusion of probes to target.
In summary, the described microwave technique here is the first to enhance the efficiency of a single/low-copy DNA probe; all previous publications using microwave were dealing with centromeric probes (Kitayama et al. 2000; Ko et al. 2001). The technique is applied routinely in our laboratory for subcentromere-specific, multicolor FISH probe sets (Starke et al. 2003); in prenatal cases with small supernumerary marker chromosomes, this time-saving possibility was used obtain results within 2 hr. Moreover, we compared the dynamics of hybridization with and without application of microwave, demonstrating the efficiency of microwave irradiation especially in the first 100 min of hybridization. In practice, we recommend a treatment approximately 4–5 microwave beams at 600 W within 30 min to get a clear effect.
The used BAC DNA was isolated from Escherichia coli and adjusted to a final concentration of 100 ng/μl. Subsequently, 2 μl were used for degenerated oli-gonucleotid primers-PCR amplification (amp-PCR: 50 μl volume; 30 cycles) and labeling (Texas Red/Biotin labeling-PCR: 20 μl; 20 cycles). Finally, the probe was dissolved in 30 μl hybridization solution (for PCR details, see Telenius et al. 1992; Nietzel et al. 2001).
1. Perform preparation of metaphase spreads from peripheral blood lymphocytes according to standard protocols. Slides should be allowed to age 3–10 days at room temperature before use. On the same day, pretreat the slides, denature the chromosome spreads (see Liehr et al. 1995), and place a Gene Frame (Abgene House; Epsom Surrey, UK), maximal volume 65 μl) on the slide.
2. For each slide mix, 5 μl of labeled and dissolved probe plus 30 μl hybridization puffer plus 15 μl of 1 μg/μl Cot1-DNA (Gibco, vacuum dried; Invitrogen GmbH, Karlsruhe, Germany). Denature probe solution at 75C for 5 min, at 4C for 2 min, and at 37C for 30 min in a thermocycler.
3. Add the denatured probe solution in the Gene Frame area and seal with the provided polyester cover.
4. Put the prepared slide in a Coplin jar within a water bath standing in a microwave oven (M 752, Miele; Gütersloh, Germany). Place the microwave temperature sensor in the water bath to control the hybridization temperature. Apply microwave beams of 600 W when the temperature is lower than 37C. For faster microwave irradiation, cool the water bath with ice.
5. Remove the Gene Frame from the slide and wash according to conventional FISH protocols.
6. Counterstain with diaminophenylindol and evaluate under a fluorescence microscope.
Footnotes
Acknowledgements
Supported in part by the Deutsche Krebshilfe/Mildred Scheel Stiftung für Krebsforschung (70–3125-Li1) and the IZKF together with the TMWFK (TP 3.7 and B307–04004), and in part by the DFG (PO284/6–1).
The continuous support of the Carl Zeiss GmbH (Jena, Germany) is gratefully acknowledged. Dr. M. Rocchi (Bari, Italy) kindly provided the BAC probe b35B4.
