Abstract
Fluorescence in situ hybridization (FISH) on human chromosomes in meta-and interphase is a well-established technique in clinical and tumor cytogenetics and for studies of evolution and interphase architecture. Many different protocols for labeling the DNA probes used for FISH have been published. Here we describe for the first time the successful use of Photoprobe biotin-labeled DNA probes in FISH experiments. Yeast artificial chromosome (YAC) and whole chromosome painting (wcp) probes were tested.
T
The Photoprobe biotin labeling system (Linaris; Wertheim-Bettingen, Germany) is based on an aryl azide derivative of biotin with a positively charged spacer arm between the biotin and the azide group. Using this system, biotin is directly conjugated to the nucleic acid (single- or double-stranded) in just 20 min using heat. Incorporation is random and is not base-specific.
This approach has been tested successfully on microdissection-derived and DOP-PCR-amplified wcp probes (Senger et al. 1998). Examples for chromosomes 2 and 6 are shown in Figures 1A and 1B, respectively. In addition, YAC probes for chromosome 2 have been labeled using the Photoprobe biotin-labeling system (an example is depicted in Figure 1C). The detection of biotin was done with an avidin-FITC/biotinylated antiavidin/avidin-FITC sandwich system (Linaris). The time for image acquisition in the FITC channel was between 0.6 and 1.8 sec for wcp and YAC probes. This is two to three times shorter than for the same probes labeled with biotin-dUTP by DOP-PCR.
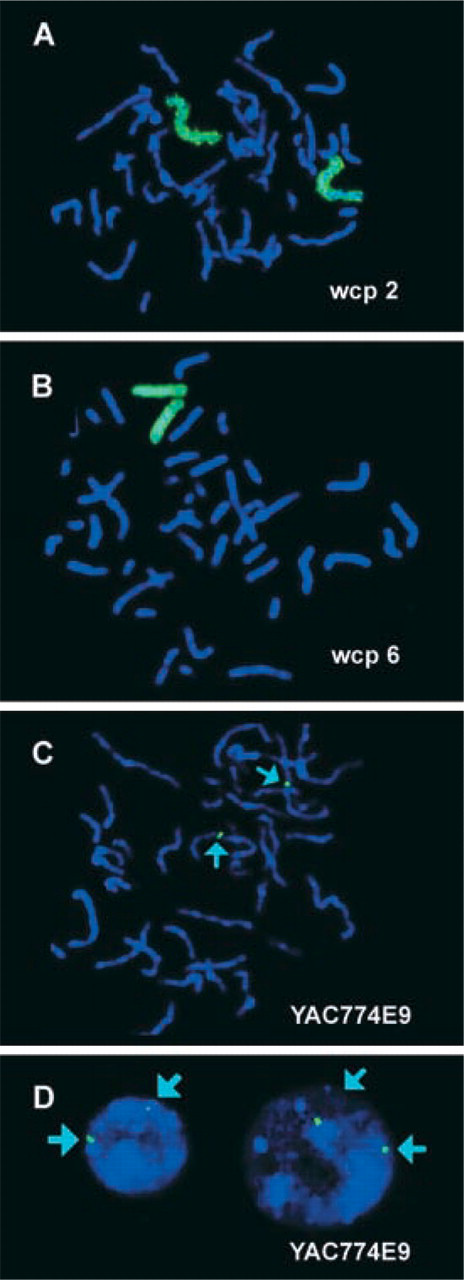
Images were captured with the ISIS digital FISH imaging system (MetaSystems; Altlussheim, Germany) using a XC77 CCD camera with on-chip integration (Sony). FISH experiments were performed on chromosomes of a healthy female donor. The used probes were labeled with biotin using the Photoprobe biotin-labeling system (Linaris) and detected with an avidin-FITC/biotinylated anti-avidin/avidin-FITC sandwich system (green signals; Linaris). FISH results of a whole chromosome painting (wcp) probe for chromosome 2 (
Protocol
Two μg probe DNA [in our case probes were amplified by DOP-PCR (Telenius et al. 1992)] dissolved in 20 μl of 1 X TE buffer (1 mM Tris-HCl, 0.1 mM EDTA, pH 8.0) is mixed with 20 μl of Photoprobe biotin solution (Linaris; cat. no. LSP1000).
For thermal coupling the reaction mixture is overlaid with 20 μl of mineral oil and heated at 95C for 20 min in a heating block or a thermocycler. Afterwards the work is continued at room temperature.
Add 20 μl deionized water and 80 μl Tris buffer (pH 9.5) to the mixture.
To separate the phases, add 160 μl of 2-butanol (room temperature), vortex vigorously, and centrifuge at 3000 rpm for 30 sec.
Remove the upper butanol phase and discard.
Re-extract the aqueous phase with an additional 160 μl of 2-butanol as described previously and remove and discard the upper butanol phase.
Precipitate together with 10 μl sodium acetate (10 M) and 125 μl ethanol (-20C) at −80C for 20 min, centrifuge at 13,000 rpm, 4C for 15 min, remove the supernatant, wash in 70% ethanol, centrifuge again, and air-dry the pellet.
Dissolve the labeled DNA in 20 μl hybridization solution and perform a standard FISH procedure as previously described (Liehr et al. 1995).
In summary, we show for the first time the suitability of Photoprobe biotin-labeled probes for FISH approaches. Optimal FISH results could be obtained in metaphase (Figures 1A–1C) and interphase chromosomes (Figure 1D). The Photoprobe biotin-labeling system is simple to perform. Labeling can be done in less than 1 hr and the resulting FISH signals are more intense than labeling done with standard protocols. The latter might be due to the fact that in nick-translation and DOP-PCR, only one of the four nucleotides (mostly dUTP) is conjugated with biotin. Using the Photoprobe biotin labeling system a random and not base-specific chemical conjugation of the whole DNA strand is done, which seems to be more effective.
Footnotes
Acknowledgements
Supported by the DFG (PO284/6-1), the INTAS (2143), the BLE (99HS039), and the Wilhelm Sander-Stiftung (99.105.1). The continuous support of the Carl Zeiss GmbH (Jena, Germany) is gratefully acknowledged.
Dr M. Rocchi (Bari, Italy) is acknowledged for YAC probes of chromosome 2.
